Abstract
Background. Many individuals with moderate and severe stroke are unable to use their paretic hand. Currently, the effect of conventional therapy on regaining meaningful hand function in this population is limited. Efforts have been made to use brain-machine interfaces (BMIs) to control hand function. To date, almost all BMI classification algorithms are designed for detecting hand movements with a resting arm. However, many functional movements require simultaneous movements of the arm and hand. Arm movement will possibly affect the detection of intended hand movements, specifically for individuals with chronic stroke who have muscle synergies. The most prevalent upper-extremity synergy—flexor synergy—is expressed as an abnormal coupling between shoulder abductors and elbow/wrist/finger flexors. Objective. We hypothesized that because of flexor synergy, shoulder abductor activity would affect the detection of the hand-opening (a movement inhibited by flexion synergy) but not the hand-closing task (a movement facilitated by the flexion synergy). Methods. We evaluated the accuracy of a BMI classification algorithm in detecting hand-opening versus closing after reaching a target with 2 different shoulder-abduction loads in 6 individuals with stroke. Results. We found a decreased accuracy in detecting hand opening when an individual with stroke intends to open the hand while activating shoulder abductors. However, such decreased accuracy with increased shoulder loading was not shown while detecting a hand-closing task. Conclusions. This study supports the idea that one should consider the effect of shoulder abduction activity when designing BMI classification algorithms for the purpose of restoring hand function in individuals with moderate to severe stroke.
Keywords
Introduction
Stroke is the leading cause of disability in the United States. About 40% of all individuals are not able to use their paretic hand after a stroke. 1 Currently, for individuals with severely affected upper extremity poststroke, the evidence that conventional therapy can restore meaningful hand function is limited.2-6 Therefore, exploring the use of a brain-machine interface (BMI) to control a neuroprosthesis that provides an artificial means to regain basic hand function merits further investigation.
Since 2008, the effect of BMI-mediated detection of activities, such as grasping or finger extension, has been tested in individuals with stroke.7-17 However, these BMI studies have detected the intention of hand movements in isolation without consideration of the effect of arm activity on the hand. In reality, arm activity is often required to implement functional motor tasks, such as reaching and grasping an object. This clear gap between the current BMI classification algorithm design and real-world requirements could significantly affect the effectiveness of the targeted neuroprosthetics and negatively influence a stroke user’s willingness to use it.
The importance of exploring the effect of proximal arm activity on detecting a hand task in individuals with stroke comes from the loss of independent joint control in the paretic arm because of the abnormal muscle synergies. The most prevalent muscle synergy,18,19 the so-called flexor synergy, is expressed as an abnormal coupling between shoulder abductors and elbow/wrist/finger flexors in the paretic upper limb. Recent research in our lab provided evidence demonstrating that because of the flexor synergy, lifting the paretic arm resulted in synergy-related finger/wrist electromyography (EMG) activity, thus, reducing the ability of a more severely impaired individual with stroke to generate volitional wrist/finger extension. 20 Furthermore, we found that synergy-induced abnormal muscle coactivation patterns were associated with changes in cortical activity. 21 Therefore, it is likely that synergic activities also affect the accuracy of a BMI classification algorithm. Because previous studies have already shown that abduction affected the elbow/wrist/finger extension more than flexion, we hypothesized that shoulder abduction would affect the detection of a hand-opening (ie, a task inhibited by the synergy) but not a hand-closing task (ie, a hand movement that is less affected by the synergy as compared with opening).
Method
Participants
A total of 6 individuals with chronic hemiparetic stroke were recruited for this study (see Table 1 for participant information). All the participants sustained a unilateral lesion at least 1 year before participating in the study. The following inclusion criteria were applied to the participants: (1) no motor impairment in the nonparetic limb, (2) no brainstem and/or cerebellar lesions, (3) no severe concurrent medical problems (eg, cardiorespiratory impairment), (4) no acute or chronic pain in the upper extremities or spine, (5) able to provide informed consent, (6) able to elevate the paretic limb against gravity up to horizontal and generate some active elbow extension, and (7) able to understand the required tasks. The level of impairment severity was evaluated using the upper-extremity portion of the Fugl-Meyer Motor Assessment. 22 Participants scoring 10 to 60 out of 66 on the Fugl-Meyer Motor Assessment were admitted in the study. All participants provided written consent that was approved by the institutional review board of Northwestern University prior to participating in this study.
Participant Information. a
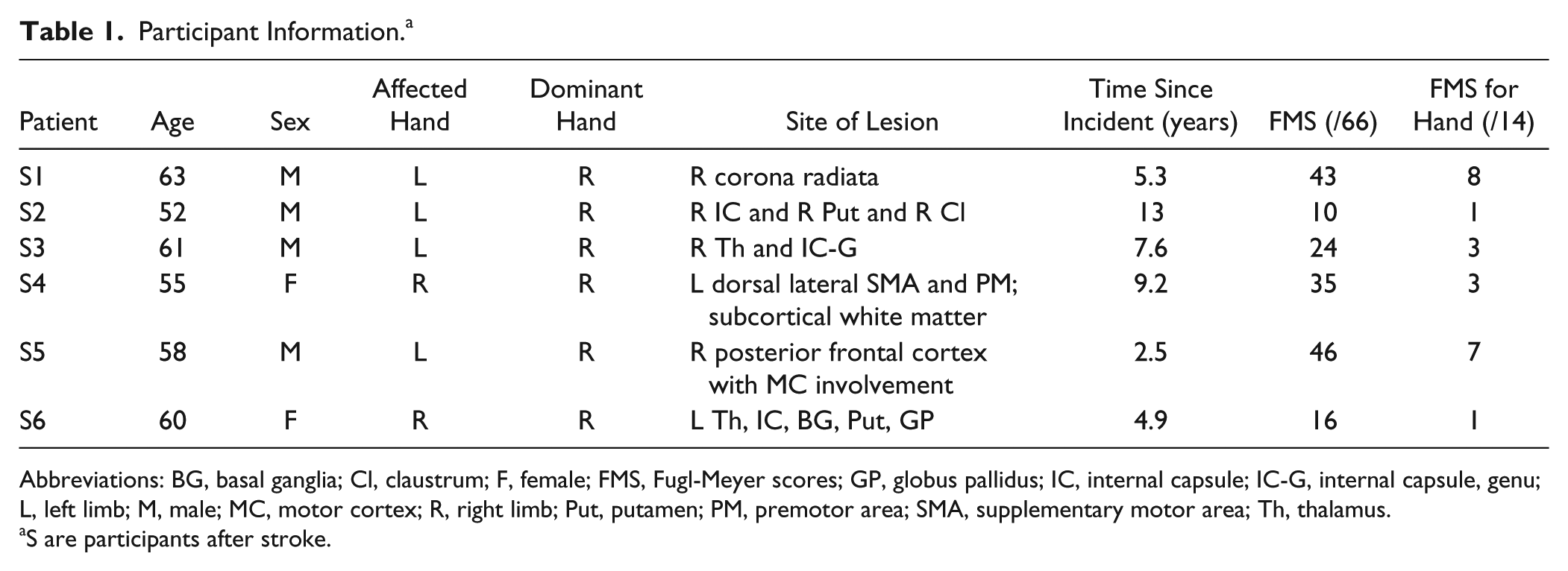
Abbreviations: BG, basal ganglia; Cl, claustrum; F, female; FMS, Fugl-Meyer scores; GP, globus pallidus; IC, internal capsule; IC-G, internal capsule, genu; L, left limb; M, male; MC, motor cortex; R, right limb; Put, putamen; PM, premotor area; SMA, supplementary motor area; Th, thalamus.
S are participants after stroke.
Experimental Setup
The Arm Coordination Training (ACT3D) robot was used to modulate the shoulder load during motor tasks. This admittance controlled robot can generate constant shoulder abduction loads during reaching tasks, as shown by both kinetic and kinematic data, and can thus precisely modulate the amount of flexion synergy expressed at more distal joints of the paretic upper limb.23-26
During all the experiments, participants sat in a Biodex chair that restrained the trunk and pelvis with straps to prevent motion during the experiment. Each participant’s forearm was strapped into a forearm-hand orthosis attached to the end effector of the ACT3D robot. At the start of the experiment, the participant’s limb lengths were measured and entered into the computer to scale the OpenGL-rendered graphical representation of the limb (avatar). Then, the tested arm was positioned at a home position of a 45° shoulder flexion angle, 75° shoulder abduction angle, and 90° elbow flexion angle (see Figure 1). The target position was set as far as the tip of the hand could reach, based on segment lengths, with the configuration of a 95° shoulder flexion angle, 75° shoulder abduction angle, and 0° elbow flexion angle.
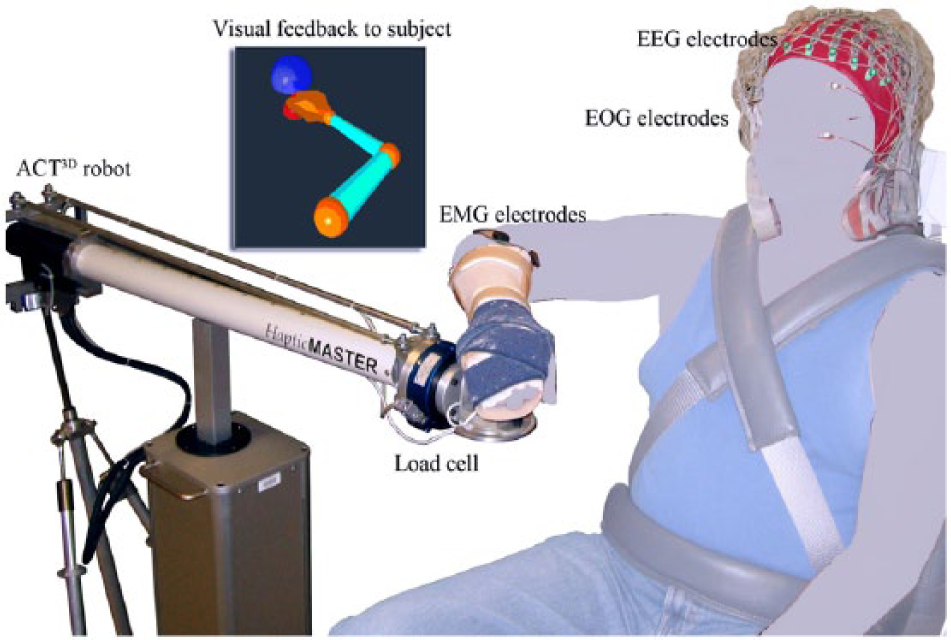
Experimental setup: the tested arm is required to move from the home position (red dot) to the target position (blue dot) at a comfortable speed, at which point the participant either opens or closes the hand.
Before data collection, a short training/preparation session was conducted to help participants get familiar with the motor tasks and associated feedback. At the beginning of a trial, the participant was instructed to move the paretic arm to the home position and relax there for 3 s. During this phase, the target position appeared as a blue or green sphere, with the color indicating a hand opening (blue) or closing (green) trial. The home target then disappeared, indicating to the participant that she or he should reach at a comfortable speed for the target within 2 s (ie, the reaching phase). The average duration of the reaching phase was about 1 to 1.5 s. A monitor continuously displayed an avatar of the true position of the tested arm/hand during the reaching phase. In some instances, participants were unable to reach the target. In this case, they were instructed to perform the task to the best of their ability. After reaching or attempting to reach the target for more than 2 s, the target position changed to yellow, and the position of the avatar then went to the target position, where it was frozen. On seeing the yellow target, participants were instructed to concentrate on either opening or closing the hand for 1 to 2 s while holding their arm in the same posture.
The hand-opening/-closing tasks were performed with 2 different shoulder loads: the supported condition, where participants were supported by a frictionless haptic table generated by the robot and the unsupported condition, during which the virtual table was lowered by 5 cm, requiring the participant to actively elevate and hold the paretic arm against gravity above the haptic table. When performing the hand task during the unsupported condition, a beeping sound was given to the participant when the paretic arm touched the table. All participants were able to elevate the paretic arm once they heard the beeping sound. If during a block of 20 to 30 trials, there were 3 consecutive trials with a short beeping signal, the data collection was paused, and a longer resting period was incorporated before starting the next block. Overall, 4 different tasks (opening/closing tasks in supported or unsupported conditions) were performed by each participant. Participants finished the opening and closing tasks under the supported condition on the first day and the unsupported condition 2 or 3 days later. In each condition, opening and closing tasks were performed randomly in several blocks of 20 to 30 trials for a total of 120 to 200 trials for each task. The experiment for each session lasted approximately 4 hours, including 2 hours for setup time and 2 hours for data collection. To minimize fatigue, a resting period of 8 to 20 s between trials was implemented as well as a 10- to 20-minute period between blocks.
We simultaneously collected EMG signals from the wrist/finger extensors (extensor carpi radialis longus and brevis and the extensor digitorum) and flexors (flexor carpi radialis and the flexor digitorum superficialis) of the tested arm as well as 160-channel electroencephalography (EEG) from the scalp and 2-channel electro-oculography signals from above and beneath the eye (Biosemi, Inc, Active II, Amsterdam, Netherlands) during each session (see Figure 1). All signals were collected with the reference placed at both ears and sampled at 1024 Hz.
EEG Data Analysis
The 160-channel EEG signals were first visually inspected to identify and remove channels constantly contaminated by noise (ie, channels with bad contact or movement artifacts). On average, about 8 to 10 peripheral channels were eliminated. The remaining EEG signals were then aligned to the onset of wrist/finger flexor (for the closing task, or for the opening task when the activity of wrist/finger extensors was absent) or wrist/finger extensor (for opening task). Subsequently, they were segmented from −1 to 1 s, with 0 representing the onset of EMG activity. The segmented EEG signals were then baseline corrected by removing the average voltage level during the baseline phase (−1 to −0.8 s) from all data points in the segment. Subsequently, a finite-difference surface Laplacian 27 transformation was applied to each EEG channel as a spatial high-pass filter to reduce the smearing effects caused by the head volume conductor and to increase the signal-to-noise ratio. 28 This resulted in the elimination of the outermost electrodes (about 29 channels), and thus, a spatial pattern of activation was generated by a total of about 120 electrodes throughout all sessions. Additionally, visual inspection of the processed signals was performed to remove trials contaminated by ocular or movement artifacts. Within each shoulder load condition, 80 to 150 trials remained for the 2 hand tasks combined. These remaining trials were then downsampled to 256 Hz and exported to MATLAB for further analysis.
In the MATLAB environment, a classification algorithm utilizing time-frequency synthesized spatial patterns (TFSP)29-31 with a newly added rejection scheme was implemented in the following manner. First, for a single trial of EEG data, the signal from one channel was decomposed into 13 frequency bands using constant Q value (Q = 4) band-pass filters, with their center frequencies ranging from 5 to 34 Hz. Subsequently, the Hilbert transform was used to extract profiles of the oscillatory activities, which were then divided into 72 equal-length time intervals (55 ms of each interval) with a 50% overlap. Next, the instantaneous power (ie, integration of the profile) was calculated in each of the time-frequency bins (t-f bins), and thus, the EEG signal from a single channel was represented by a 72 × 13 matrix of coefficients. The above processing was repeated in all channels. As a result of the above decomposition process, a single trial EEG signal was characterized by a series of spatial features represented in each of the 72 × 13 t-f bins.
After decomposing spatial features onto 72 × 13 t-f bins, the classification of the hand-opening/-closing tasks was performed for the supported and unsupported conditions separately. Under a given shoulder load, we first calculated the characteristic spatial patterns for each hand task in each of the specific t-f bins by averaging all the spatial patterns within the specific t-f bin (ie, for a given shoulder load, the characteristic spatial pattern for hand opening is
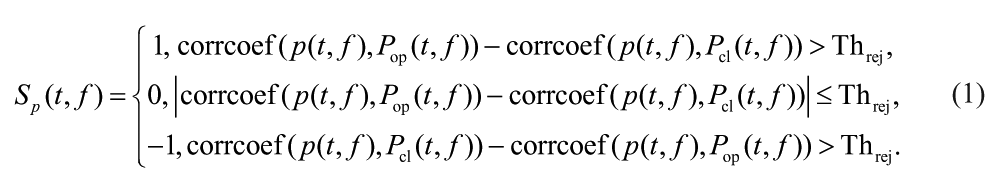
A Sp(t, f) equal to 1 or −1 indicated a detection result of hand-opening or a hand-closing trial, and 0 represented an uncertain judgment. As shown in Equation (1), at this first level, the correlation coefficient between the testing trial and the 2 characteristic patterns was used to quantify the distance between the testing trial and the characteristic pattern. The decision of one t-f bin went to the class whose characteristic pattern was closer to the testing trial. A rejection scheme was added to the original TFSP BMI algorithm at each of the t-f bins. When the difference in the similarity judgment was smaller than the rejection threshold (Threj), the corresponding grid did not contribute to the final decision making. The output of the first-level Sp(t, f) by each of the t-f bins was then used at the second level to determine a final decision. The second level synthesis worked akin to a voting system, in which each individual (equal to each of the t-f bins) voted, and the final judgment went to the decision that was agreed by most of the individuals. All the accepted grids shared the same voting rights, and the final judging decision R(p) regarding the class of a testing trial was made by summing the results over the entire 72 × 13 time-frequency grids as follows: R(p) = sign (sum(Sp(t, f))). A positive R(p) indicated that a majority of bins judge that the current trial was a hand-opening event, whereas a negative one indicated a hand-closing event. No trial would be rejected completely unless R(p) = sign (sum(Sp(t, f))) = 0, which never happened in any test trials to date.
Finally, we calculated the true positive rate (TPR, defined as the number of hand-opening trials that were correctly recognized, normalized by the total number of hand-opening trials) and true negative rate (TNR, defined as the number of hand-closing trials that were correctly recognized, normalized by the total number of hand-closing trials). The BMI algorithm was repeated with a rejection threshold of 0 to 0.95, with a step increase of 0.05. The leave-4-out cross-validation method 32 was used to test the overall performance under either the supported or unsupported conditions with each of the given rejection thresholds.
To understand the neural underpinnings of our results, we further identified the t-f bins that performed differently under “supported” and “unsupported” conditions, separately for each of the 2 hand tasks. To implement this, we first gave a score equal to 1, −1, or 0 to each of the t-f bins if it detected a single trial correctly, falsely, or refused to make a decision, respectively. The final recognition score of a t-f bin was then calculated by summing the scores cross all the trials with the rejection threshold set as 0.35 (ie, the overall optimal rejection threshold). Subsequently, we used a 1-way repeated-measures analysis of variance (RMANOVA) to identify t-f bins with a significant change in the recognition score when a different level of limb support was applied (dependent factor: recognition score; independent factor: supported versus unsupported condition; samples: different trials, P < .05). These t-f bins were defined as shoulder-load sensitive t-f bins (SS t-f bins). Finally, we counted the number of SS t-f bins in θ (4-7 Hz), α1 (8-10 Hz), α2 (10-13 Hz), β1 (13-20 Hz), and β2 (20-30 Hz) bands before and after the onset of EMG.
To explore the spatial features, we reconstructed the cortical activity of participant S2 from 0 to 50 ms while performing hand-opening and hand-closing tasks using his paretic left arm under the supported and unsupported conditions. Inverse source reconstruction was conducted using the LORETA method (Lp = 1),33,34 based on ensemble-averaged EEG signals and a participant-specific boundary element head model developed by participant-specific anatomical magnetic resonance imaging (MRI). Although the inverse procedure was performed over the whole cortex, only the activities in the region of interest, consisting of the bilateral premotor (PM), supplementary motor area (SMA, including both pre-SMA and SMA proper), and primary motor (M1) and primary somatosensory (S1) cortices, were shown. Because of the absence of MRI data in the remaining 5 participants, cortical activity reconstruction was not performed in the other 5 participants.
Statistical analyses were applied to detect the difference at a P < .05 significance level or a trend of reaching significance at a P between .05 and .1.
Results
Overall, equal numbers of hand-opening and -closing trials were used: A 2-way ANOVA (hand task, support level) analysis result showed no significant difference in the trial numbers between the hand-opening and -closing tasks (P = .78; F = 0.07) or between the unsupported versus supported conditions (P = .40; F = 0.75).
The box plots of TPRs and TNRs under the supported and unsupported conditions with each of the rejection thresholds were plotted in Figure 2. Results from a 1-way MANOVA (independent factor: shoulder abduction load with the rejection threshold as the nested factor; dependent factors: TPR and TNR) showed that shoulder abduction load significantly influenced the accuracy rate for detecting hand opening (F = 28.737; P < .0001) but not the accuracy rate for detecting a hand closing (F = 0.0824; P > .1). Scheffe post hoc tests showed that the classification algorithm detected the hand opening with significantly higher accuracy during the supported condition as compared with the unsupported condition (mean difference = 12.8%; P < .0001).
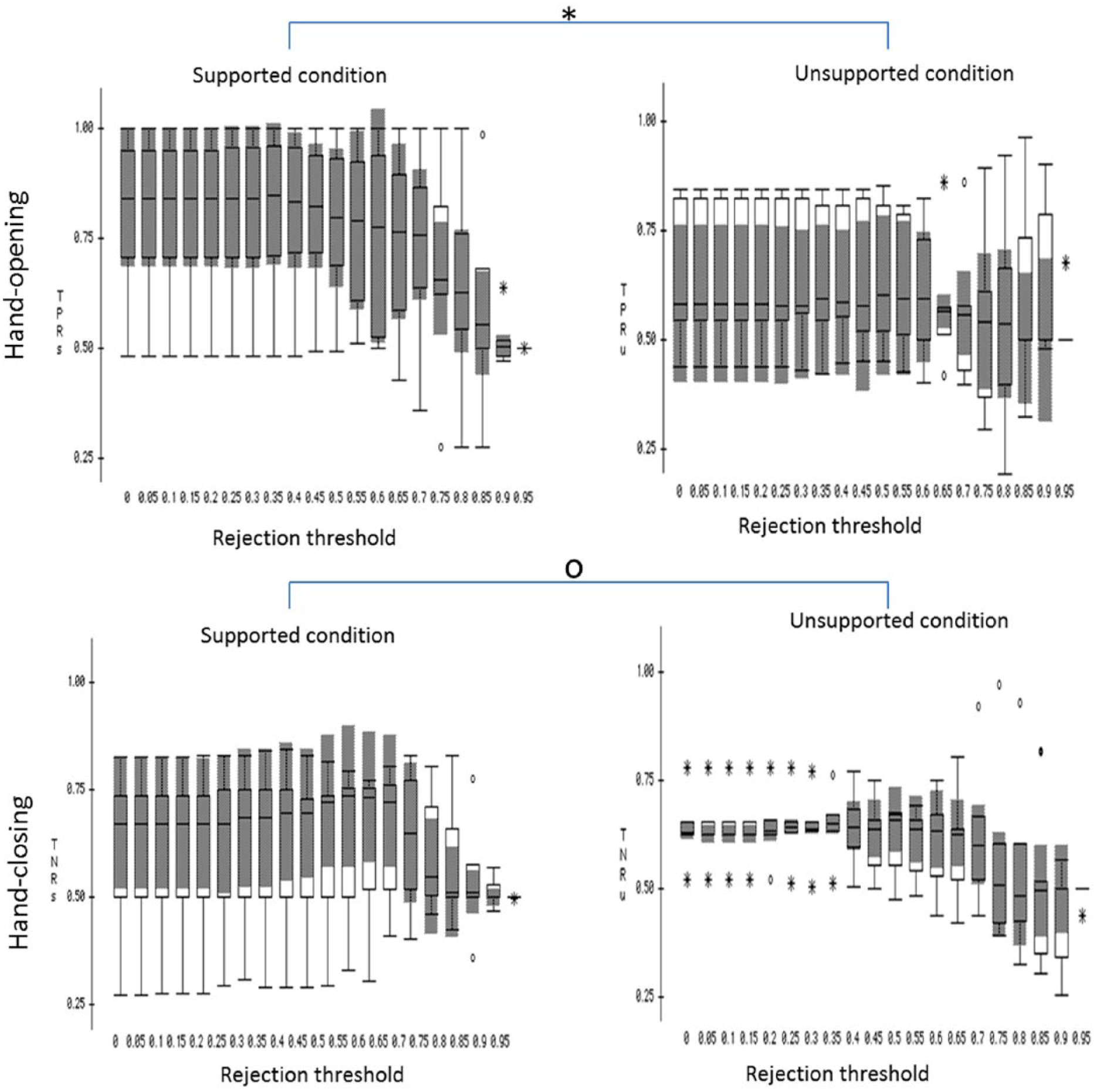
The box plot of the brain-machine interface performance over the 6 participants under supported (left column) and unsupported (right column) conditions, with different rejection thresholds applied.a
Across participants, the means and standard errors of the number of SS t-f bins are shown in Figure 3. As shown in Figure 3, overall more SS t-f bins were found for the hand-opening task (gray bars of Figure 3) than for the hand-closing task (white bars). A 3-way RMANOVA was used to test the effect of the time window (2 levels, before and after EMG onset), frequency band (5 levels), and hand task (2 levels, opening and closing tasks) on the number of SS t-f bins. Results of RMANOVA reported a significant effect of the hand task (F = 9.854; P = .0257) on the number of SS t-f bins but not on the other 2 factors (time window: F = 0.091, P = .7745; frequency band: F = 0.353, P = .839). There were no significant interactions between these 3 within-participant factors, either. A nonparametric Friedman post hoc test reported a significantly greater number of SS t-f bins in the low-β band (13-20 Hz) after EMG onset for the hand-opening task as compared with the closing task (P < .05). No other significant results were found by the post hoc analysis.
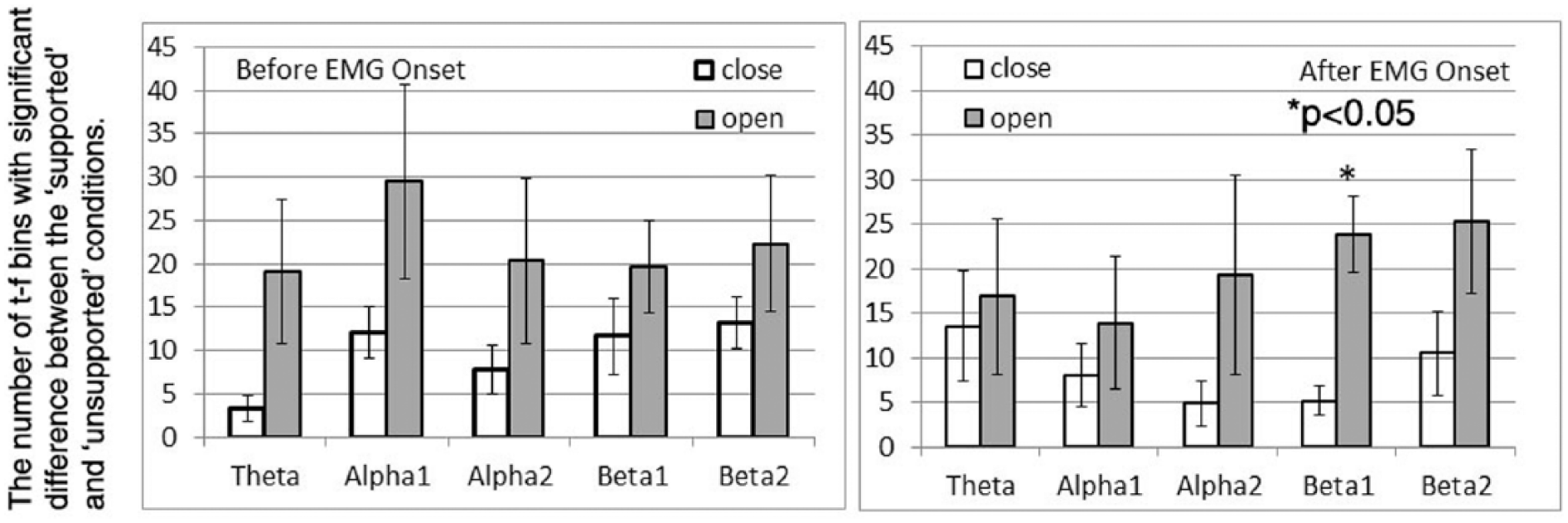
The number of time-frequency (t-f) bins with significant difference in recognition score between the supported and unsupported conditions at different frequency bands during the time window of before electromyography (EMG) onset (left) and after EMG onset (right).
Cortical activities of participant S2 from 0 to 50 ms while performing hand-opening and -closing tasks with the paretic left arm during supported and unsupported conditions are shown in Figure 4. As illustrated in Figure 4, during the supported condition, only the contralateral M1/S1 cortical regions were involved in the hand-opening task (Figure 4A); however, bilateral activity was detected during the hand-closing task (Figure 4C). Thus, the spatial patterns during the supported condition for the 2 hand tasks were quite different. However, during the unsupported condition, bilateral cortical activity was observed for both the hand-opening and -closing tasks (see Figure 4B and 4D). The similarity in cortical spatial patterns for the 2 hand tasks during the unsupported condition reduced the accuracy of the classification algorithm.
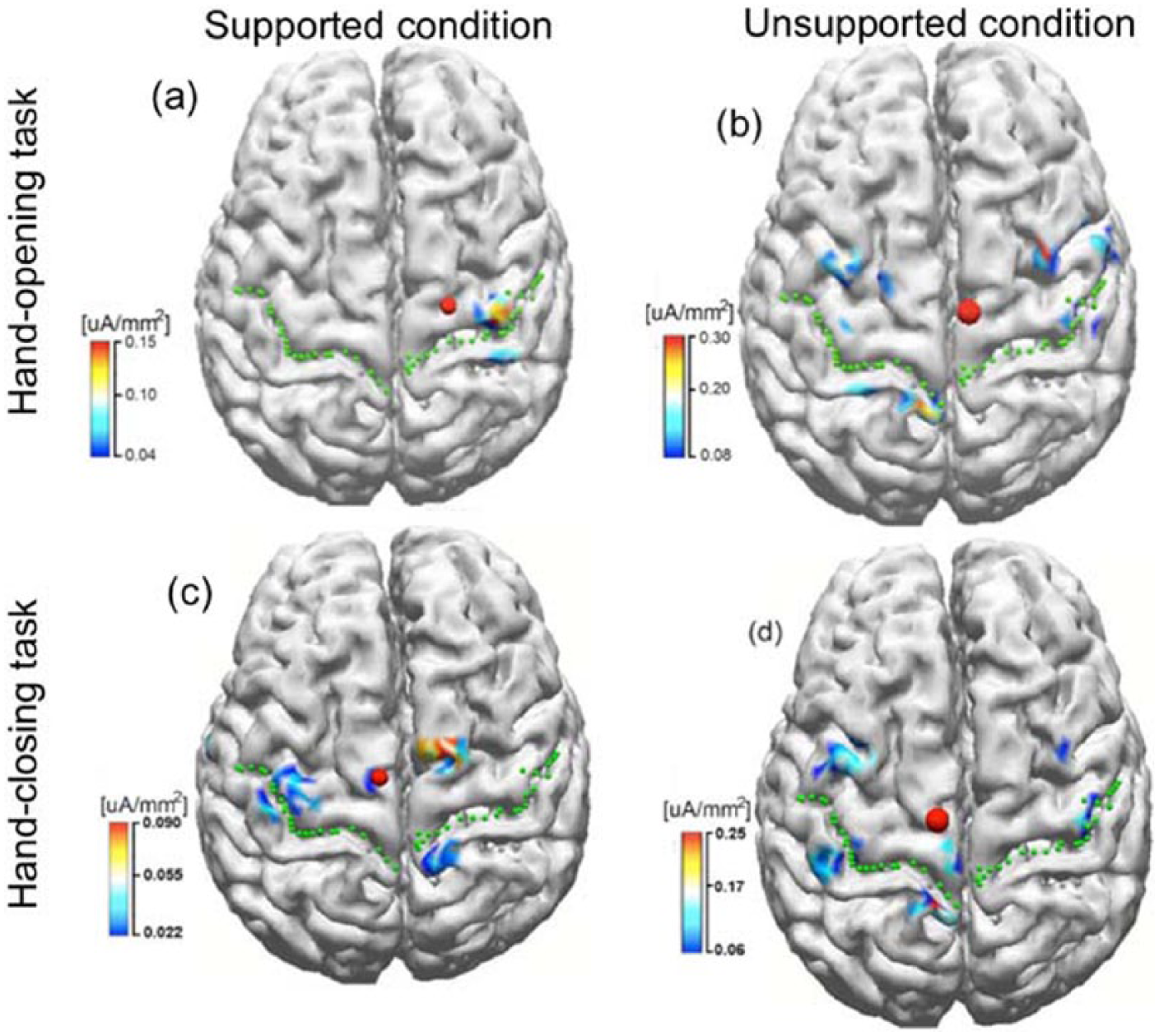
The inverse results in stroke participant S2 when performing tasks of hand opening (the upper row) and hand closing (the lower row) using his paretic arm (left arm) under supported (left column) and unsupported conditions (right column), respectively. The green dotted lines indicate the central sulcus, and the red dots represent the center of gravity of the cortical activity within the region of interest.
Discussion
Our results obtained from the paretic arm of 6 participants with chronic hemiparetic stroke showed a significant reduction in the accuracy of detecting the hand opening with an increased shoulder abduction load. However, such shoulder-abduction-induced reduction in the accuracy was not demonstrated in the hand-closing task.
The Loss of Independent Joint Control in Individuals With Stroke Affects the Accuracy in Detecting the Hand-Opening Task
We believe that the shoulder abduction-load-induced reduction in the accuracy for detecting the hand-opening task is a result of changes in brain activity that also results in a flexor synergy. Previous reports from our laboratory have shown that the flexion synergy decreases the ability to reach25,26 and open the hand 21 when increased shoulder abduction loading is applied. The main underlying neural mechanism is postulated to be an increased reliance on brainstem descending pathways, such as the corticoreticular spinal tract (CRST), because of the loss of corticospinal tract (CST) fibers from the lesioned hemisphere. It is well known that CST monosynaptically facilitates contralateral extensor and flexor muscles, 35 and ipsilateral CST activation of spinal motoneurons is absent or very weak. 36 In contrast, CRST is a bilaterally organized system. Previous studies have shown that CRST projects to both motoneurons and interneurons involved in digital control. 37 CRST tends to facilitate flexors and suppress extensors ipsilaterally, and the reverse contralaterally.38-40 Thus, it is possible that the ipsilateral CRST and remaining CST projections are both effective resources to drive hand closing regardless of the supporting conditions. The case of hand opening is more complex. As we already know, ipsilateral CRST suppresses extensors. Therefore, residual CRST from the nonlesioned ipsilateral side may not be a good backup for hand opening, and hand opening may be primarily dependent on contralateral residual CST. Such dependence on contralateral residual CST is also reflected as the greater paralysis of finger extensors regardless of shoulder abductor activity. 41 During the supported condition, using contralateral residual CST for the opening task versus using bilateral CRST and CST for the closing task might allow the detection of the 2 hand tasks. However, when additional shoulder abductor muscle was required, ipsilateral CRST was used for both hand tasks, and contralateral cortical activity related to hand opening might no longer be detectable. This explanation was supported by the cortical activity results from a single participant, as shown in Figure 4. It was also supported by the significant change in the number of SS t-f bins in detecting hand opening at the lower β band (13-20 Hz) after EMG onset when different SABD loading was required. As reported before, corticomuscular communication conducted via the CRST oscillates in a lower frequency band (10-15 Hz)42,43 than that conducted via CST, which is in a higher frequency band (15-40 Hz; see Soteropoulos and Baker 44 ). Our finding of a significant difference in the number of SS t-f bins for the hand-opening task in the low β band probably also reflects the fact that CST and CRST were used to drive hand opening during the supported as compared with the unsupported condition.
In short, our results show that shoulder loading has a significant impact on the accuracy of detecting a hand-opening task in individuals with stroke, presumably because hand opening is more exclusively dependent on activity in the contralateral lesioned hemisphere via the contralateral CST. Conversely, the impact of shoulder abduction loading on the detection of the hand-closing task is not significant, presumably because hand closing can be generated by both contralateral cortical activity via the CST and ispilateral cortical activity via the CRST.
Possible Limitations in the Interpretation of Our Data
Previous literature has shown that the human brain, even after a stroke, can still adapt to a BMI device to improve performance. 12 Because we always conducted experiments under the supported condition on the first day and experiments under the unsupported condition about 2 or 3 days later, it is reasonable to expect that the effect of learning could affect our findings. In case a participant learned from the first day’s experiment, any potential learning effects should favor the unsupported condition, and thus, imply an even lower detection rate under an unsupported condition if there is no effect of learning. Therefore, the order of the experiments should not invalidate our results. If anything, it should only strengthen them. Furthermore, there is no reason to believe that the effect of learning would affect the hand-opening and -closing tasks differently. Because of the above 2 reasons, we ruled out an effect of learning on our findings.
Results of conducting the experiments in a fixed sequence could also be affected by some random differences such as the participant’s overall mental state, noise from random sources, day-to-day impedance differences between electrodes and skin, and so on. These differences would be reflected as baseline differences and were removed from analysis. Furthermore, because of their random nature, there is no reason to believe that noise from random sources should bias one testing condition over another.
Other possible confounding factors that may affect our signals are muscle fatigue and EMG contamination of EEG activity. It is possible that the participant may be more fatigued on the second day than on the first day. To avoid this issue, we separated the 2 experiments by 2 or 3 days, and to limit the effects of muscle fatigue within a day, we included longer resting periods between trials and between blocks for experiments in the unsupported condition. No participant reported fatigue during or after the experiment. We also checked the frequency spectrum of EMG data. We did not find a significant difference in the EMG spectrum between the first 10 and last 10 trials. Therefore, no clear evidence of muscle fatigue was found. Consequently, we argued that muscle fatigue was not a primary contributor to the observed decrease in BMI performance when a greater shoulder abduction load was applied.
Additionally, higher muscle artifacts might be introduced to EEG signals under the unsupported condition as compared with the supported condition. The increased EMG activity mostly affected the outside EEG electrodes, which were removed from further analysis, thereby reducing their influence on EEG signals. Furthermore, we argued that if EMG activity indeed contaminated EEG to a greater extent when the arm was not supported, then it should have affected both the opening and closing tasks similarly. However, our results showed that different shoulder loads only significantly affected the accuracy in detection of the hand-opening task. To further confirm that EMG did not affect t-f bins differently for various conditions, a 2-way repeated-measures ANOVA (ie, supporting conditions and the hand tasks) was used to compare the spectrum in 2 control frequency bands: 30 to 40 Hz and 40 to 50 Hz. In these 2 bands, the spectrum should not be significantly modulated by a brain signal change, but could very easily be modulated by an EMG artifact signal. Repeated-measures MANOVA did not yield any significant effects (P > .1).
The difference in sensory input under the supported and unsupported conditions as well as additional motor output for shoulder abduction under the unsupported condition could also contribute to the changes in BMI performance. We argued that the sensory input and motor output caused by shoulder abductor activation were the same for both the hand-opening and hand-closing tasks. However, we only observed the changes in the BMI performance for the hand-opening task but not for the hand-closing task. Therefore, the common sensorimotor information caused by the shoulder activity cannot explain selective changes in the BMI performance for the hand-opening task only but not for the closing task.
Finally, our discussion of possible neural mechanisms that would explain our findings (ie, shoulder loading affects the BMI performance of the hand-opening task but not the closing task) was partially based on imaging results from a single participant. Imaging results on an increased number of participants are required for a more solid explanation of the neural mechanisms. In short, our results provide, for the first time, evidence that shoulder loadings affect the BMI performance differently for the hand-opening as opposed to the -closing task.
Clinical Implications and Future Directions
In this article, we provided evidence of a decreased accuracy of the classification algorithm in detecting hand opening when an individual after a stroke intended to activate the hand while also activating shoulder abductors. This conclusion is based on the off-line performance of the TFSP algorithm. However, this conclusion is likely generalizable to real-time applications as well. Our conclusion about shoulder activity affecting the accuracy of detecting hand tasks can probably also be generalized to other joints, such as the elbow and the wrist; and to other synergetic patterns, such as the impact of shoulder adduction on the detection of the hand-closing task. In short, our results demonstrate the necessity of considering the impact of multijoint activity when designing a BMI device such as a neuroprosthetic device for potential future clinical use in individuals with stroke and suggest that regaining more independent shoulder and elbow control may be vital to improve BMI performance for the purpose of recapturing basic hand function.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by NIH grants (UL1 RR025741, R01 HD047569-01A1 and 2R01HD039343-11A1) as well as by the NIDRR grant (H133G120287).
