Abstract
Background. A significant factor in impaired movement caused by stroke is the inability to activate muscles independently. Although the pathophysiology behind this abnormal coactivation is not clear, reducing the coactivation could improve overall arm function. A myoelectric computer interface (MCI), which maps electromyographic signals to cursor movement, could be used as a treatment to help retrain muscle activation patterns. Objective. To investigate the use of MCI training to reduce abnormal muscle coactivation in chronic stroke survivors. Methods. A total of 5 healthy participants and 5 stroke survivors with hemiparesis participated in multiple sessions of MCI training. The level of arm impairment in stroke survivors was assessed using the upper-extremity portion of the Fugl-Meyer Motor Assessment (FMA-UE). Participants performed isometric activations of up to 5 muscles. Activation of each muscle was mapped to different directions of cursor movement. The MCI specifically targeted 1 pair of muscles in each participant for reduction of coactivation. Results. Both healthy participants and stroke survivors learned to reduce abnormal coactivation of the targeted muscles with MCI training. Out of 5 stroke survivors, 3 exhibited objective reduction in arm impairment as well (improvement in FMA-UE of 3 points in each of these patients). Conclusions. These results suggest that the MCI was an effective tool in directly retraining muscle activation patterns following stroke.
Introduction
More than 3.2 million people in the United States suffer chronically impaired upper-limb function because of a stroke. 1 Impairment of voluntary arm movement can be a result of not only weakness and loss of sensation but also abnormal coactivation.2-5 In contrast to spasticity, which is increased tone during externally imposed limb movement, abnormal coactivation, sometimes called “abnormal muscle synergy,”6,7 consists of increased tone during active or attempted voluntary movement by the patient. Stroke survivors often experience coactivation of anterior deltoid with biceps (flexor synergy) and posterior deltoid with triceps (extensor synergy). This constrains their movement to stereotypical patterns. 7 By reducing abnormal coactivation and restoring more normal patterns of activation, it may be possible to improve function.
Whereas some studies questioned the amount of impairment caused by abnormal coactivation,8,9 other evidence strongly suggests that its role is significant. 10 Moreover, the clearest way to determine the amount of significance is to prospectively treat the coactivation and assess the effects on movement. 10 Abnormal coactivation can also be defined as abnormal coupling between joint torques. 3 Ellis et al 11 recently demonstrated that abnormal arm joint torque couplings in stroke survivors could be reduced by training the participants to isolate individual joint torques with the use of visual feedback. This intervention also led to an increase in strength, demonstrating the significant role of abnormal coactivation in impaired function. However, this paradigm is not suitable for widespread use outside specialized clinics because of the size and expense of the necessary robotic equipment.
This study attempted to reduce coactivation directly by using a myoelectric computer interface (MCI). In an MCI, surface electromyographic (EMG) signals are mapped to different directions of cursor movement on a monitor. 12 Here, we tested whether an MCI specifically designed to target muscle coactivation could allow stroke patients to reduce abnormal muscle coactivation.
We first tested the MCI on healthy participants to determine whether they could learn to decouple 2 normally coactivating muscles (biceps and brachioradialis). Then, we tested the extent to which stroke survivors could learn to decouple 2 abnormally coactivating muscles (biceps and anterior deltoid). Our results suggest that MCI training did allow stroke survivors to reduce abnormal coactivation and improve upper-limb function.
Methods
Participants
A total of 5 right-handed participants (4 men and 1 woman, ages 23-27 years) free from neurological and musculoskeletal disorders and 5 individuals (1 man and 4 women, ages 50-58 years) whose stroke occurred 1.5 to 25 years prior to enrollment gave informed consent to participate in this study, which was approved by the Northwestern University Institutional Review Board. We included stroke survivors who (1) had hemiparesis with moderate to severe impairment of the affected arm (score of 12-40 on the upper-extremity portion of the Fugl-Meyer Motor Assessment [FMA-UE] 13 ), (2) exhibited coactivation of the biceps and anterior deltoid muscles determined by clinical observations during FMA-UE and by participants’ performance using the MCI during the initial screening process, and (3) had a single, unilateral, ischemic stroke at least 1 year prior to enrollment. We excluded those who had (1) significant pain in the upper limbs or spine, (2) substantial sensory loss in the affected upper arm, (3) significant vision loss, (4) cognitive impairment severe enough to affect digit span and understanding of task-related instructions, or (5) participated in another arm-motor study within 30 days of the start of this study. Participants were allowed to continue their usual exercise or physical therapy regimens but not allowed to start a new regimen during the study. All stroke patients had right-sided hemiparesis (FMA-UE score = 19 ± 3, mean ± standard deviation).
Behavioral Task
Participants sat in a chair with their right arm in an armrest. Healthy participants’ arms were held in a semipronated position (to make it more difficult to decouple biceps and brachioradialis) and immobilized with cushioned restraints at the hand, wrist, and upper forearm. Stroke participants’ impaired arms were held in a prone position.
Participants performed isometric contractions of multiple muscles to move the cursor (circle of radius 1.5 cm, Figure 1A) from a square target in the center of a monitor to a randomly presented target near the outer edge of the monitor—a modified center-out task 14 (Figure 1B). Activation of each muscle was mapped to 1 of 4 directions within the 2D cursor space (see Muscle Mapping). The center target corresponded to zero net muscle activation (resting position). Each trial started after holding the cursor in the center target for 512 ms, when an outer target appeared and the center target disappeared. Outer targets were located at a distance of 12 cm from the center target in all sessions except the first 5 and first 2 sessions for participants 1 and 2, respectively; in these first few sessions, they were located 7 cm from the center. (Distance was increased to make the task more challenging after it was noted to be too easy for these participants.) When the cursor reached the outer target it changed color, and participants were required to hold the cursor there for at least 33 ms (and up to 512 ms) to achieve success.
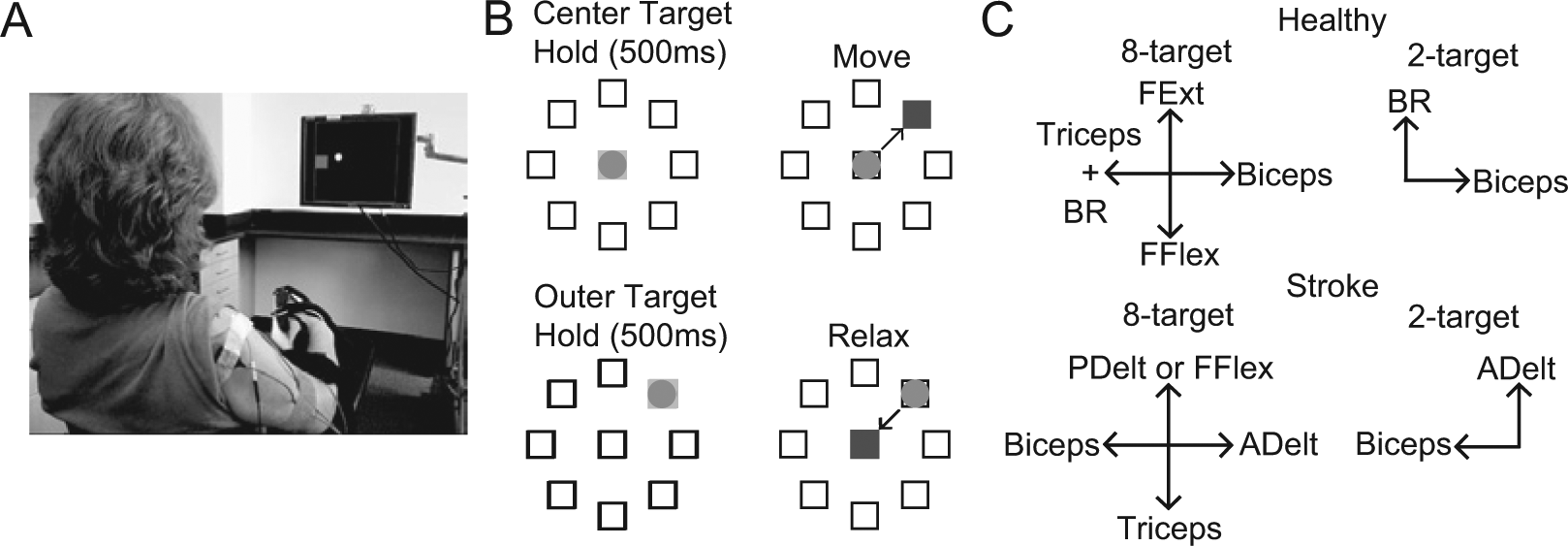
MCI task overview: A. Experimental setup with a stroke participant viewing a circular cursor and square target on the monitor. B. Schematic of MCI task and muscle mapping directions. Participants moved the cursor to 1 of either 2 or 8 (shown here) outer targets. Relaxing all muscles moved the cursor to the center. C. Muscle mapping directions in both the 8-target task (left) and 2-target task (right) for both healthy (top) and stroke (bottom) participant groups.
Cursor Control Signal
We derived the cursor position in real time using EMGs from multiple arm muscles. Surface EMG was recorded with bipolar electrodes spaced 1 cm apart, amplified with a gain of 1000 (Delsys Bagnoli), digitally sampled at 1 kHz (National Instruments USB-6229) and collected using a customized program in BCI2000. 15 The electrode positions were marked at the end of the session with a henna marker to ensure the same location in the next session.
The control signals in each direction were derived from EMGs by low-pass filtering at 500 Hz, rectifying, high-pass filtering at 20 Hz, and then convolving with a 400-ms rectangular window. The vector sum of the control signals determined the 2D cursor position.
At the start of each session, participants were instructed to produce 2 maximum voluntary contractions (MVCs) of each muscle independently. To avoid fatigue, the control signals were scaled by a factor that allowed cursor movement to the targets at a relatively comfortable level of contraction (~42% of the MVC). Because EMG activation levels sometimes varied because of slight changes in electrode placement or skin impedance, we sometimes altered the gains across sessions to make the effort level similar across sessions. However, the gains remained the same across all trials within each session.
Muscle Mapping
Each participant performed 2 tasks in each session: a training task that used 2 outer targets and a “generalization” task that used 8 outer targets. In the training task, the 2 coactivating muscles (biceps brachii and brachioradialis for healthy participants and anterior deltoid and biceps brachii for stroke survivors) were mapped to orthogonal directions, and only these 2 muscle signals were summed to form the control signal (Figure 1C). Thus, participants could only succeed in the task if they learned to decouple the coactivating muscles. We also designed an 8-target task to attempt to assess the extent to which participants could generalize the learned decoupling to a different task. In the 8-target task, the 2 coactivating muscles were mapped to opposite horizontal directions. In healthy participants, an independent muscle (triceps) was added in the direction of the brachioradialis to make the task more challenging. Two other muscles not typically coactivated with these muscles (flexor and extensor digitorum superficialis for healthy participants and triceps and either posterior deltoid or flexor digitorum superficialis in stroke participants) were mapped in opposite vertical directions. Participants were informed of the specific directions corresponding to activation of each recorded muscle (mapping direction, Figure 1C).
In some stroke participants, the affected muscles retained some spontaneous muscle activity (mainly in the biceps and flexor digitorum) when trying to relax,16,17 which prevented the cursor from reaching the center target. Therefore, we subtracted the baseline activity of each muscle (0.5-s window starting 1 s after the completion of a trial) from the corresponding control signal for the next trial.
Experimental Paradigm
Healthy participants participated in 3 sessions separated by up to 1 week. In each session, they performed 10 minutes of the 8-target task (pretraining), 20 minutes of the training task, and 10 minutes of the 8-target task (posttraining). Stroke participants participated in 3 sessions per week for 6 weeks. In each session, they performed 10 minutes of the pretraining task, 30 minutes of the training task, and another 10 minutes of the posttraining task. For analysis, the first and last 2.5 minutes (for healthy) or 5 minutes (for stroke) of training were denoted early and late training, respectively.
Performance and Outcome Measures
We quantified participants’ performance in controlling the MCI via 3 different metrics: success rate (percentage of targets successfully acquired), time to target (TT; time between the outer target appearance and the cursor entering the target), and path length (PL; cumulative distance the cursor traveled in each trial, normalized by the distance between center and outer targets).
We defined the level of coactivation between muscles as the Pearson correlation coefficient (R) between the filtered EMGs (control signals) during the period from outer target appearance to the end of the outer target hold time on consecutive trials that were concatenated. Statistical significance was assessed using paired t tests.
To investigate the contribution of each muscle to movement to each target in more detail, we computed muscle tuning curves. 12 These were computed from the average control signal during the outer target hold period for each trial during the posttraining task and aligned to the mapping direction of each muscle.
Finally, we evaluated FMA-UEs before the first session and at the end of the last session. We surveyed participants after the study using the following questions:
Did you notice any improvement in your arm function during the study? If so, what improved?
Was the amount of training too little, too much, or just right?
Did you enjoy participating in the study?
How would you recommend changing the MCI training paradigm to make it more enjoyable?
Results
Healthy Participants: Task Performance and Reduction of Coactivation
We first examined the healthy participants’ ability to learn to control the MCI in both tasks (Figure 2). Participants learned the training task quickly, as evinced by the rapid improvements in PL (Figure 2A, solid lines) and TT (Figure 2B, solid lines) from session 1 to 2. PL improved by 69% (ΔPL = −1.3 ± 0.8, P = .17, paired t test), and TT improved by 62% (ΔTT = −1.59 ± 0.9 s, P = .16) from early training in session 1 to late training in session 3. They learned the 8-target task quickly as well (Figure 2, dashed lines). Performance improvement persisted for 1 week.
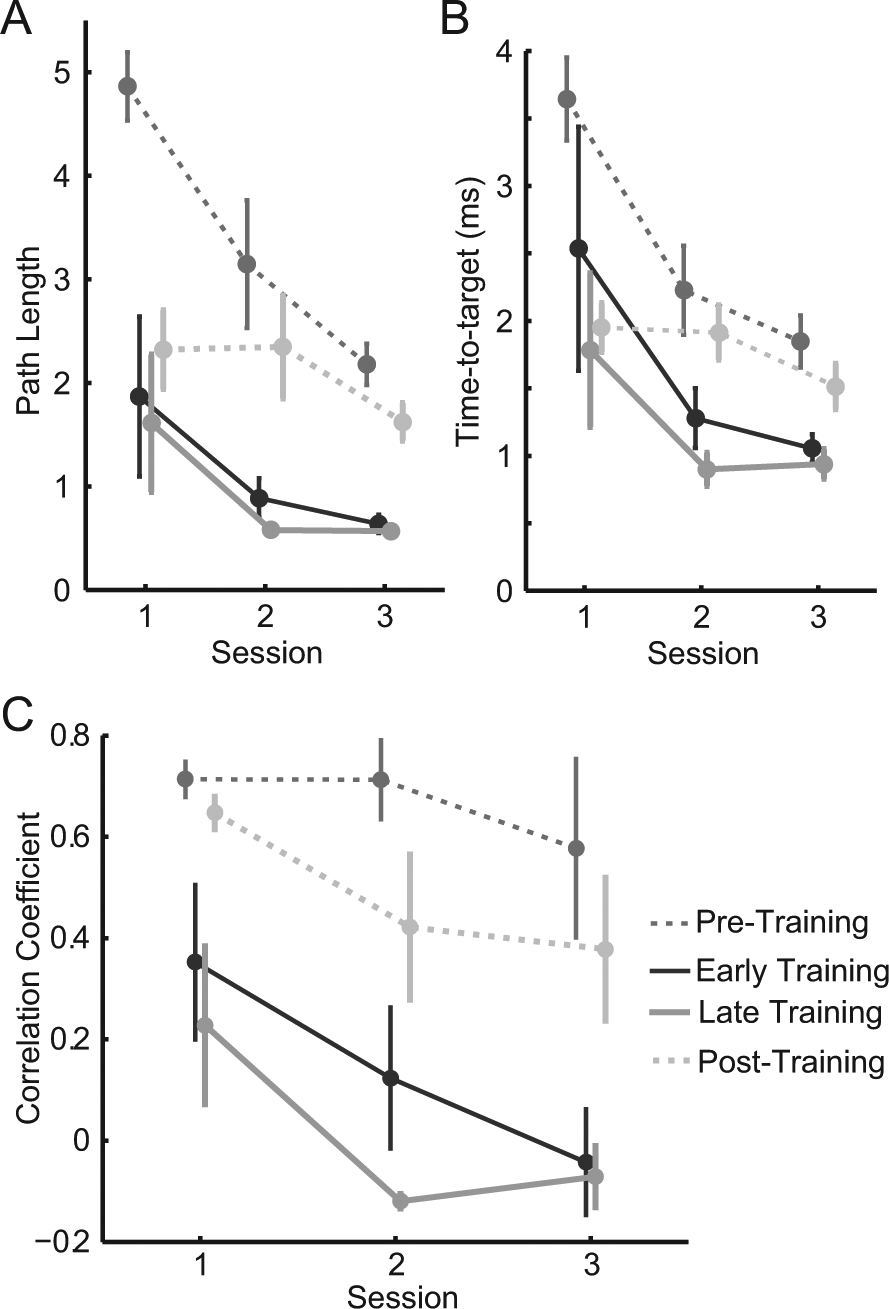
Healthy participants’ task performance and coactivation changes: A. Path length and B. time to target measures. C. Mean R between biceps and brachioradialis activity averaged across all participants for pretraining and posttraining (dashed lines) and early and late training (solid lines). Error bars represent standard error (SE).
Healthy participants readily learned the task of decoupling biceps and brachioradialis muscles within 2 sessions on consecutive days (Figure 2C). During the training task, correlation between these muscles decreased by an average (±SE) of 0.42 ± 0.18 from early training in session 1 to late training in session 3, though not quite significantly (P = .055). Correlation decreased significantly from the pretraining phase of session 1 to the posttraining phase of session 3 (ΔR = −0.33 ± 0.12, P = .046).
Stroke Participants: Task Performance
Despite being told the mapping direction of each muscle prior to starting the 8-target task, stroke participants initially displayed poor control of the cursor (Figure 3A, left). By the final session, the movement trajectories more closely approached straight lines (Figure 3A, right) to each target. In the training task, trajectories tended to move along the diagonal between the 2 targets (Figure 3B, thin lines) in the first session, but over time, the trajectories became straighter (Figure 3B, thick lines). Representative EMG traces during movement in the anterior deltoid (Figure 3C) and biceps (Figure 3D) directions reveal a decrease in muscle coupling from the first session (left) to the last session (right), consistent with the cursor’s path to the intended target.
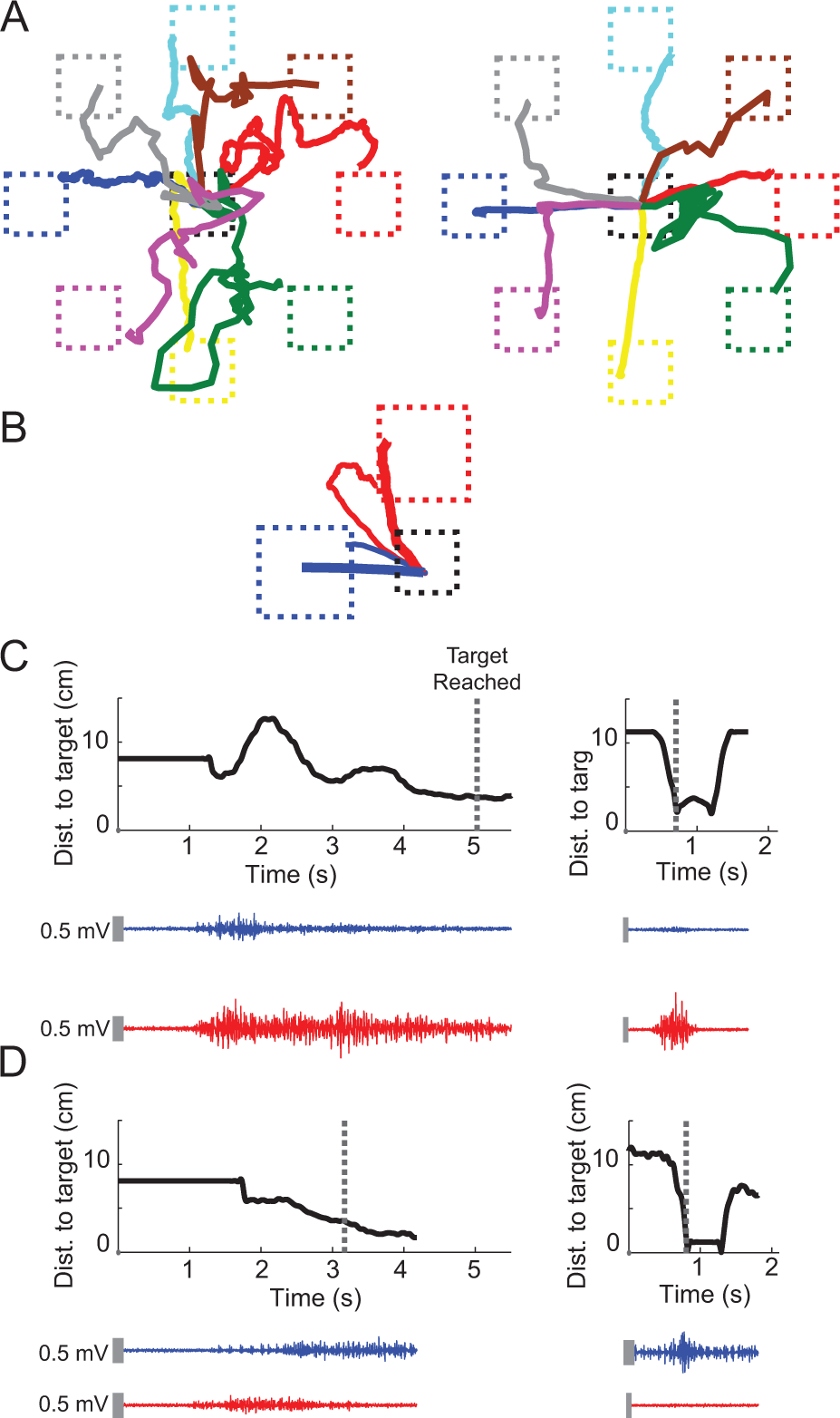
Mean movement trajectories to each target and representative EMG traces from a stroke participant during (A) pretraining in the first session (left) and posttraining in the last session (right) and (B) early training in the first session (thin lines) and late training in the last session (thick lines). Blue and red trajectories represent cursor movement in the biceps direction and anterior deltoid direction, respectively. Representative EMG traces of biceps (blue) and anterior deltoid (red) for single trials during movement in the (C) anterior deltoid target direction and (D) biceps target direction show reduction of coactivation from early training in the first session (left) to late training in the last session (right). Black curves represent the cursor’s distance to the intended target (dashed boxes in B) over time. Dashed lines in (C) and (D) represent the time at which target was reached. Vertical gray bars represent the electromyographic scale (0.5 mV).
Participants’ task performance gradually improved in the 8-target task over time, as evinced by the changes in success rate (23 ± 5%, P = .008, Figure 4A), TT (−2.5 ± 0.5 s, P = .008, Figure 4B) and PL (ΔPL = −2.8 ± 1.0, P = .05, Figure 4C) compared with the first session. Substantial improvement was also seen between early training in the first session and late training in the final session (Figure 4, ΔSR = 15 ± 5%, P = .03; ΔTT = −1.9 ± 0.5 s, P = .01; ΔPL = −1.3 ± 0.5, P = .056).
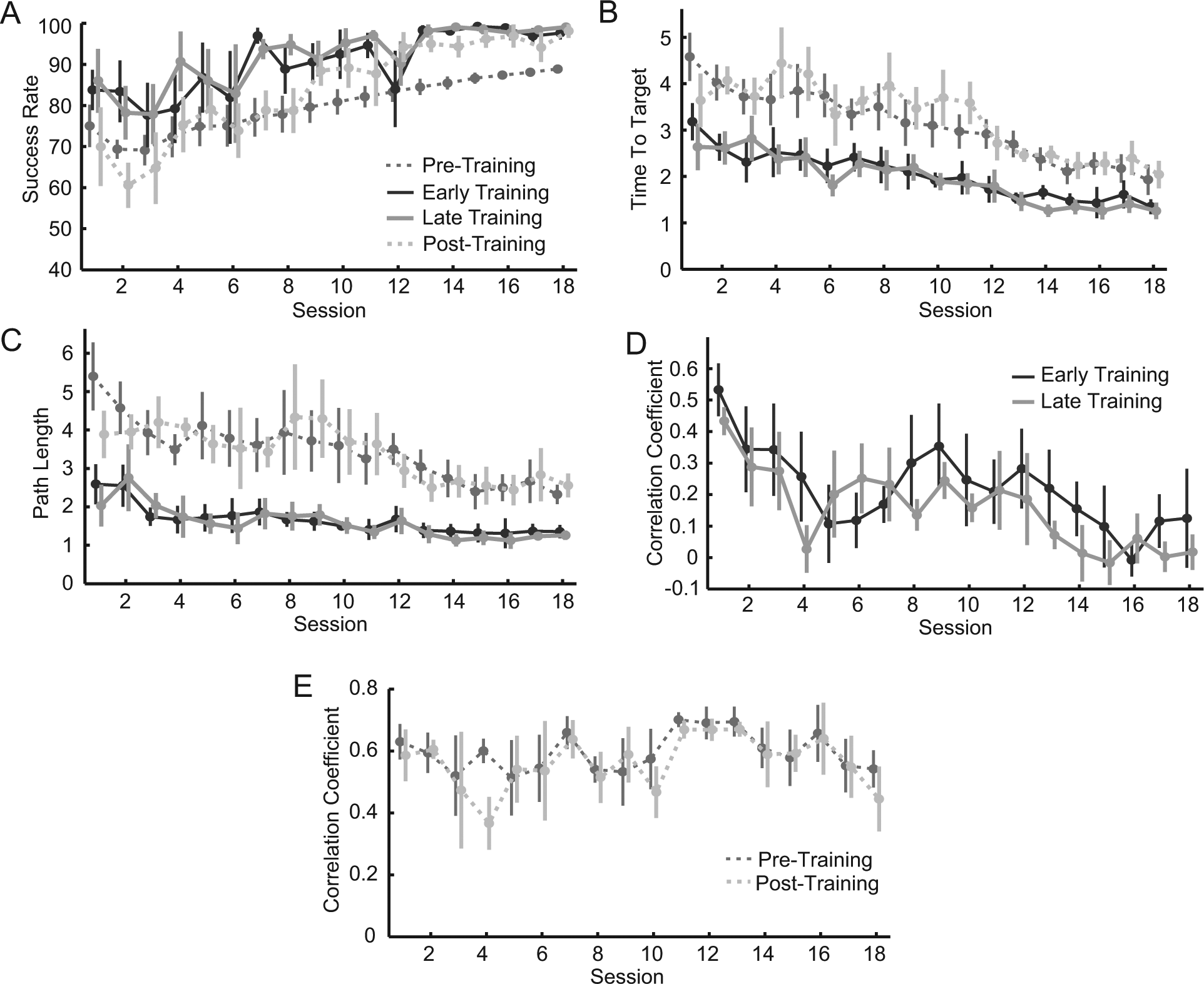
Stroke participants’ task performance and coactivation changes: Performance measures, including (A) time-to-target, (B) path length, and (C) success rate averaged across all participants show steady improvement during both training (solid lines) and pretraining and posttraining (dashed lines) tasks. Mean R between biceps and anterior deltoid during the (D) training and (E) pretraining and posttraining tasks. Correlations steadily and significantly decreased during the training task.
Stroke Participants: Reduction of Coactivation
Stroke participants steadily learned to decouple biceps and anterior deltoid activity during the training task (Figure 4D). Correlations between the 2 muscles steadily decreased, with an average of 99% (ΔR = −0.52 ± 0.08, P = .005) reduction between early training of the first session and late training of the last session (Table 1). Correlations during pretraining and posttraining decreased by 23% (ΔR = −0.19 ± 0.15, P = .30, Figure 4E).
Details of Performance Changes. a
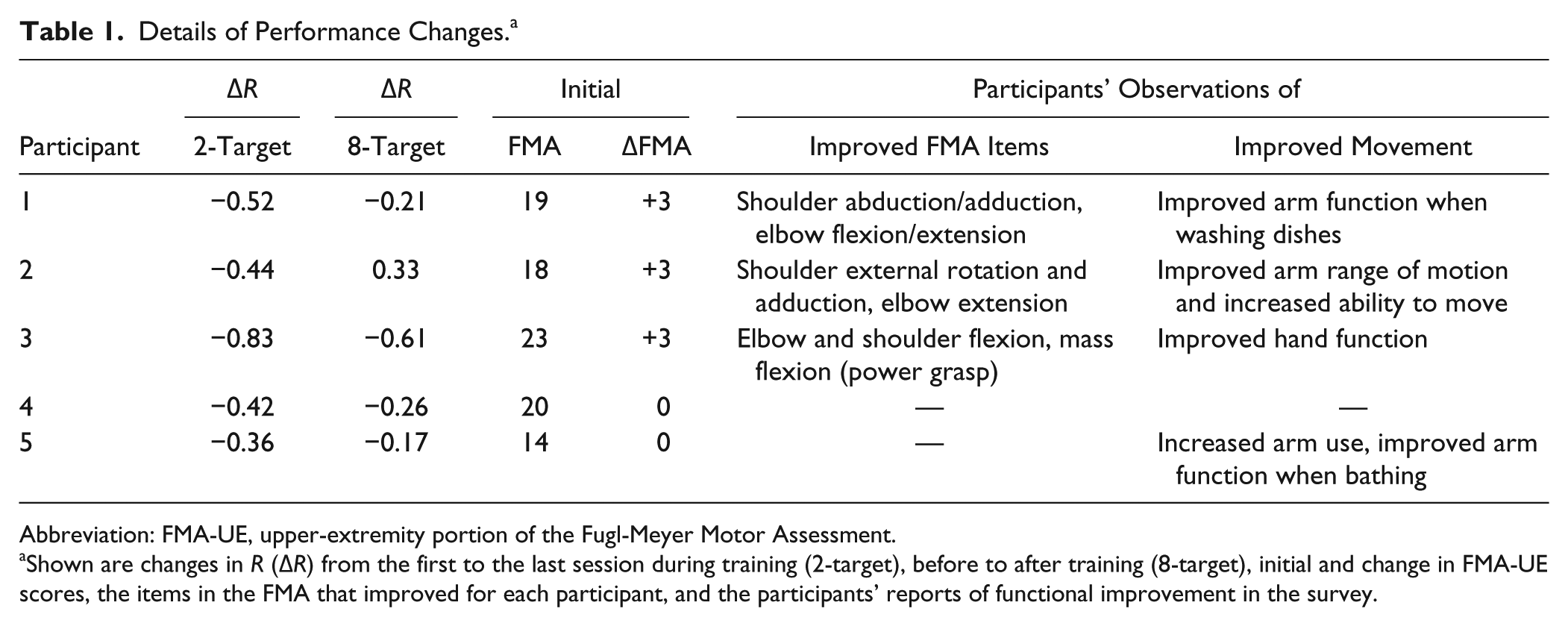
Abbreviation: FMA-UE, upper-extremity portion of the Fugl-Meyer Motor Assessment.
Shown are changes in R (ΔR) from the first to the last session during training (2-target), before to after training (8-target), initial and change in FMA-UE scores, the items in the FMA that improved for each participant, and the participants’ reports of functional improvement in the survey.
Stroke Participants: Muscle Tuning Curves
The average tuning curves across all stroke participants (Figure 5A) demonstrate that the biceps showed increased tuning toward the appropriate mapping direction and adjacent targets over time. The anterior deltoid was tuned predominantly in the appropriate direction from the start and, therefore, did not change greatly. When averaged over all muscles and participants, a gradual increase in tuning depth (peak EMG amplitude) in the appropriate mapping direction was seen over time (Figure 5B). This also suggests better control of the muscles over time.
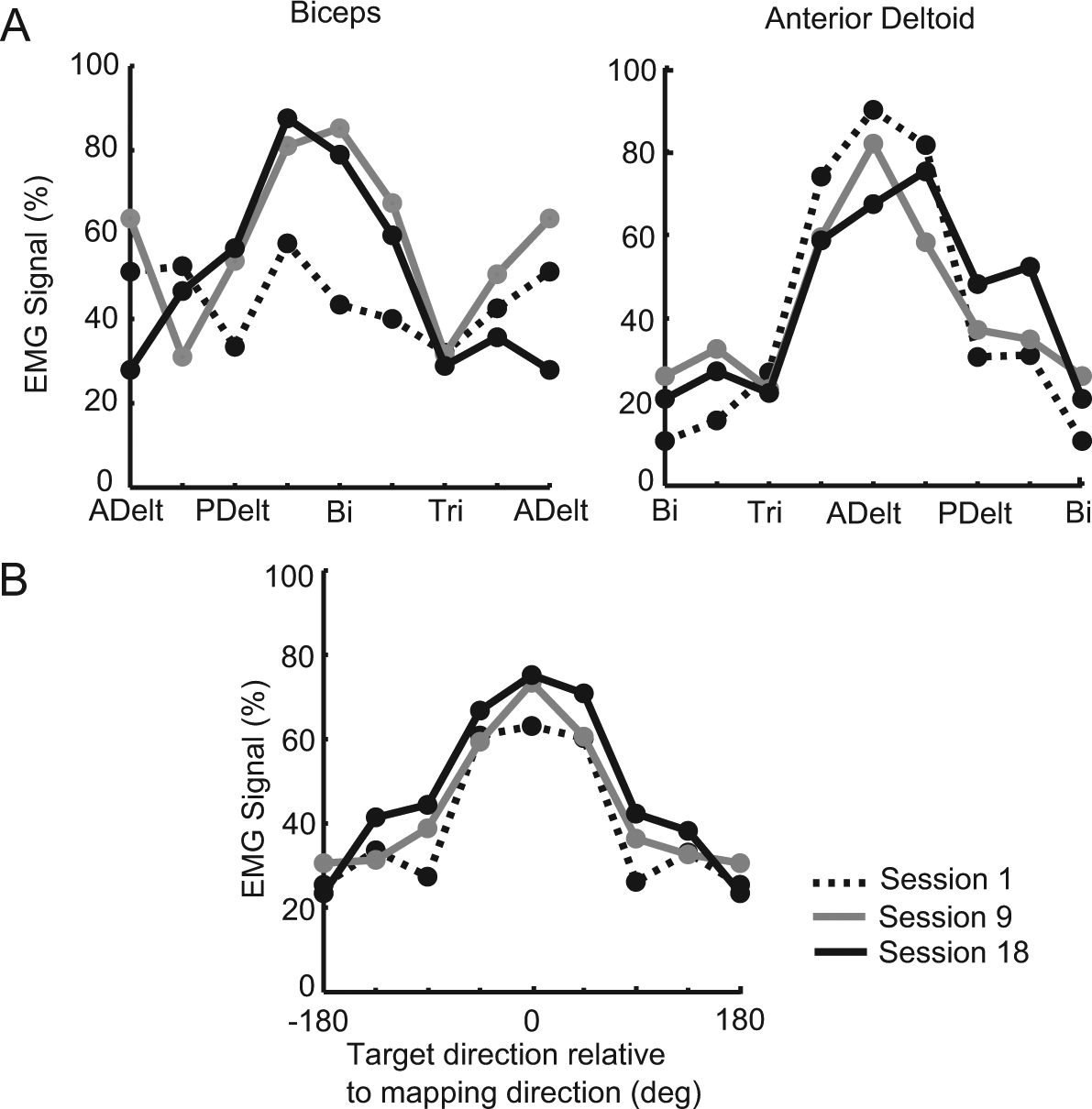
Evolution of muscle tuning curves in stroke participants: A. Tuning curves of the biceps and anterior deltoid averaged across participants from posttraining tasks of the first (dashed line), ninth (gray line), and last (black line) sessions. Control signals are normalized to the maximum level in both sessions and averaged across trials. B. Tuning curves averaged across all 4 muscles and participants over sessions. Tuning depth in all muscles gradually increased over time.
Effects of Training on Arm Function
In all, 4 stroke participants reported subjective improvement of arm or hand function in activities of daily living, including bathing, dressing, and washing dishes, during the study period (Table 1). All stroke participants enjoyed performing the MCI training but said that it could be made even more enjoyable by integrating with a video game. Four stroke participatnts asked for a larger dose of training, and the remaining participant said the dose was just about right; 3 participants displayed improvement in FMA-UE scores by 3 points each; the other 2 participants did not improve their scores. Reduced impairment correlated with reduced coactivation during training: ΔR = −0.52 ± 0.11 versus −0.34 ± 0.09 for participants whose FMA-UE scores did and did not improve, respectively.
Discussion
This study demonstrated that MCI training effectively enabled (1) healthy participants to decouple 2 normally coactivating muscles and (2) stroke participants to decouple 2 abnormally coactivating muscles. Moreover, the majority of stroke participants displayed both subjective and objective evidence of reduced arm impairment after the training. Although changes in FMA scores were modest, this was not unexpected because they were achieved with a small amount of training and only aimed to decouple 1 pair of muscles. These results suggest that MCI training could be developed as a therapy to directly retrain muscle activation patterns and improve overall arm function in chronic stroke participants.
The pathophysiology of abnormal coactivation is poorly understood, and there exists evidence, mostly indirect, to support origins both in increased bulbospinal outflow 18 and in the cortex.19-22 It is also unclear whether abnormal synergies are fixed and simply augmented by therapy 23 or can be changed. The results in our study suggest that they may be malleable. However, this cannot be firmly established without examining the responses of the other arm muscles to the training of these 2 muscles. That is, it is not clear whether the participants’ central nervous system learned to simply suppress a single muscle or changed the entire synergy. It is also not clear at what level of the central nervous system (cortex, brainstem, or spinal cord) these changes occurred. Future studies may address these questions.
The MCI provides specific and intuitive feedback to participants about their muscle activity. Although a prior study has shown the ability to decouple joint torques in stroke survivors, 11 to our knowledge, this is the first study to demonstrate the ability to directly decouple muscle activations in this patient population. Most studies using EMG biofeedback have attempted to increase strength in the target muscles, 24 whereas a few have sought to decrease muscle activity.25-27 One study did show this ability in children with generalized dystonia within a single day. 28 Another study did use EMG biofeedback of antagonist muscle to reduce cocontraction during agonist movement. 29 However, that study did not provide feedback in an intuitive fashion as in the MCI, nor about the relative activation of both muscles, and required the user to produce a high level of elbow flexion force with the contractions. In contrast, the MCI provides intuitive feedback about the coactivation that only allows success when the muscles are decoupled. This could allow users to concentrate solely on succeeding in the task without explicitly thinking about activating individual muscles, which could take advantage of implicit motor learning mechanisms. Implicit motor learning is necessary for behavioral improvement, more durable over time, and less attentionally demanding than explicit learning.30-32 Furthermore, because the MCI only requires EMG activity, not production of substantial forces, it could potentially be used in severely impaired stroke survivors. This is critical, because these patients are often excluded from rehabilitative trials 33 and are less likely to benefit from conventional therapy because of their extremely limited movement. 34
Stroke participants learned to reduce coactivation more slowly than did healthy participants. This comparison is far from ideal because healthy participants were younger and were learning to decouple muscles that often, but not always, coactivate, whereas stroke participants were learning to decouple abnormally coactivating muscles. Nevertheless, this result suggests that hemiparetic stroke survivors may have slower rates of motor learning in the paretic arm. This issue has not been well studied to date, 35 with 1 study showing possible impairment of learning in the paretic arm, which was attributed to weakness. 36 Although this is a difficult question to study, 35 further study seems to be warranted, and the MCI could provide a means of circumventing the confounding effects of impaired movement because only EMG activity, not movement, is required to control the MCI.
Although the reduction in coupling during the 8-target task did not reach significance, this could have been a result of the small sample size. In addition, the 8-target task was substantially more complex than the 2-target task because it sometimes required the participant to coactivate pairs of muscles. Moreover, because each control signal was opposed directly by another, participants could succeed in the task without decreasing coactivation simply by increasing activity in one muscle much more than in the opposing muscle; this would not be reflected in R values. Because of these complexities, in retrospect, we believe that the 8-target task was not optimal for testing generalization of the learned decoupling. Indeed, reduced coupling during the training task correlated with FMA-UE scores, whereas reduced coupling during the 8-target task did not. Therefore, we place more emphasis on the subjective reports and improved FMA-UE scores, which suggest that the training did transfer to activities of daily living in stroke participants.
Several issues remain to be addressed before the MCI can be tested as a clinical treatment. These include determining (1) appropriate dosage and duration of training, (2) the number of muscle pairs to decouple, (3) the use of isometric versus nonisometric activations, and (4) the duration for which beneficial effects will persist. The optimal amount of training needed to gain significant improvement in functional outcome is an outstanding question in many rehabilitation paradigms. All participants expressed interest in increasing the training intensity, which is strongly correlated with motor learning, cortical reorganization, and recovery after stroke.37-39 Because the only requirements are surface EMG recording and relatively simple software, the system could easily be implemented in the community setting and potentially in patients’ homes, which would allow more frequent training. Whereas participants uniformly enjoyed the task, the MCI could be integrated into gaming environments that would further enhance motivation and training intensity. Many activities of daily living require nonisometric activations, and therefore, it is possible that MCI training using unrestrained movements may translate to greater functional gains than the isometric training used here. We plan to investigate some of these issues in future studies.
Footnotes
Acknowledgements
We thank Robert Davisson Flint and Eric Lindberg for technical assistance and Mark Shapiro for the Delsys system.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Northwestern Memorial Foundation Dixon Translational Research Grants Initiative (supported in part by the NUCATS Institute NIH grant UL1RR025741).
