Abstract
Keywords
Introduction
The extent of motor recovery following stroke is influenced by both direct loss of neurons and widespread neural network reorganization both local and distant to the site of the lesion.1,2 Poststroke motor recovery may be affected by atypical levels of interhemispheric inhibition between the lesioned and nonlesioned motor cortices resulting from a disruption of transcallosal neural network communication.3,4 However, our limited understanding of the complex neural mechanisms underlying stroke recovery may explain, in part, why poststroke rehabilitation approaches aimed at normalizing interhemispheric imbalance, such as those using therapeutic noninvasive brain stimulation, have been ineffective in reducing poststroke impairment. 5
Following stroke, changes in structural neuroanatomy and cortical activity patterns may occur in regions remote from the lesion in both the ipsilesional and contralesional hemisphere, implicating the role of expansive cortical networks in the recovery process.1,2 In contrast to unilateral hemisphere activation observed in healthy controls, bilateral activation of the ipsilesional primary motor cortex (iM1) and contralesional primary motor cortex (cM1) is often observed during paretic hand movement in stroke survivors and has been associated with poorer motor function.6-8 Stronger bilateral activation of motor cortical areas is correlated with the structural integrity of the corpus callosum and poor motor performance after stroke. 7 Findings from these neuroimaging studies support the notion that interhemispheric interactions between neural networks via transcallosal pathways play an important role in poststroke motor function.
Though interhemispheric interactions between neuronal networks in iM1 and cM1 have been implicated in poststroke motor recovery, measuring functional interhemispheric interactions directly is difficult. Previously, the primarily inhibitory 9 interhemispheric interactions between M1s have been assessed indirectly using electromyography (EMG) responses in arm and hand muscles elicited by transcranial magnetic stimulation (TMS) using either a paired-pulse paradigm (2 TMS coils overlying M1s) 10 or a single-pulse paradigm during sustained muscle contraction in the target muscle ipsilateral to the site of TMS. 11 In the single-pulse paradigm, TMS over the ipsilateral M1 generates a brief interruption in volitional muscle activity, the ipsilateral cortical silent period (iSP), that is thought to reflect interhemispheric inhibition from the ipsilateral to the contralateral M1 12 and mediated by inhibitory GABAA (type A γ-aminobutyric acid) and GABAB receptor activity. 13 After stroke, greater magnitude and duration of iSP in the paretic limb has been observed4,14,15 that was negatively correlated with motor function, suggesting greater interhemispheric inhibition from cM1 to iM1 in the most severely impaired individuals. 16 Increased interhemispheric inhibition from iM1 to cM1 is positively correlated with improved motor performance following bilateral upper limb rehabilitation. 17 Together, these findings suggest that excessive levels of inhibition from cM1 to iM1 coupled with reduced inhibition from iM1 to cM1 may impede poststroke motor recovery. 11 Despite the conflicting findings of other studies,16,18 the interhemispheric competition model of stroke recovery has been commonly used as a theoretical framework for the development of rehabilitation strategies, including the use of noninvasive brain stimulation in attempt to upregulate iM1 or downregulate cM1 in stroke survivors.19,20 However, the often conflicting findings of these therapeutic paradigms used in interventional studies are minimally effective and limited progress has been made to reduce upper limb motor disability for stroke survivors. 5
Noninvasive evaluation of cortical neuronal network reactivity to TMS using electroencephalography (EEG) recordings at the level of the scalp to measure evoked cortical responses is now possible. 21 Concurrent TMS-EEG permits a more direct probe into neural network connectivity at highly specific time-scales, thus overcoming some of the previous technological limitations of interhemispheric connectivity measures (e.g., EMG motor-evoked potentials and iSPs, functional magnetic resonance imaging, dynamic causal modeling) and potentially bridging the gap in our understanding of the functional role of poststroke interhemispheric interactions. Additionally, cortical recordings can be obtained from individuals with a limited or absent corticospinal tract and lacking peripheral motor responses to TMS. Because structural reserve within the lesioned hemisphere may be an important factor in the functionality of poststroke interhemispheric imbalance to recovery, 22 the use of direct cortical recordings may offer important insights into the neural recovery mechanisms of these individuals who are often the most severely impaired.
Recent findings from our group demonstrated the feasibility of using concurrent TMS-EEG in stroke 30 by showing that interhemispheric coherence between iM1 and cM1 was greater in a small sample of chronic stroke survivors compared to controls when the hand muscle was active. 31 However, whether TMS offers a unique contribution to assessments of interhemispheric coherence using resting state EEG alone has not been investigated. Additionally, the relationship between TMS-evoked interhemispheric coherence, motor function and iSP measures after stroke is unclear. The purposes of this study were to (1) test the effect of TMS on measures of interhemispheric coherence in stroke and age-matched neurologically intact controls, (2) investigate the relationship between interhemispheric coherence and paretic arm and hand motor behavior in stroke survivors, and (3) evaluate the relationship between interhemispheric coherence and EMG measures of iSP.
Methods
Nineteen individuals (age: 66 ± 11years, 8 females) with chronic stroke (>6 months) and 14 neurologically intact age-matched individuals (age: 53 ± 14 years, 6 females) participated in the single-session, cross-sectional study (Table 1). All participants were between the ages of 44 and 81 years. Participants with stroke had a single ischemic cortical or subcortical stroke confirmed by magnetic resonance imaging (MRI). Participants were excluded if they had hemorrhagic stroke, a stroke directly affecting the upper extremity region of the primary motor cortex or the corpus callosum, history of multiple strokes, neurodegenerative disorder or psychiatric diagnosis, or contraindications to TMS (Supplemental Figure S1). 32 All participants gave written informed consent and the experimental protocol was approved by the Emory University Institutional Review Board.
Stroke Participant Characteristics. a
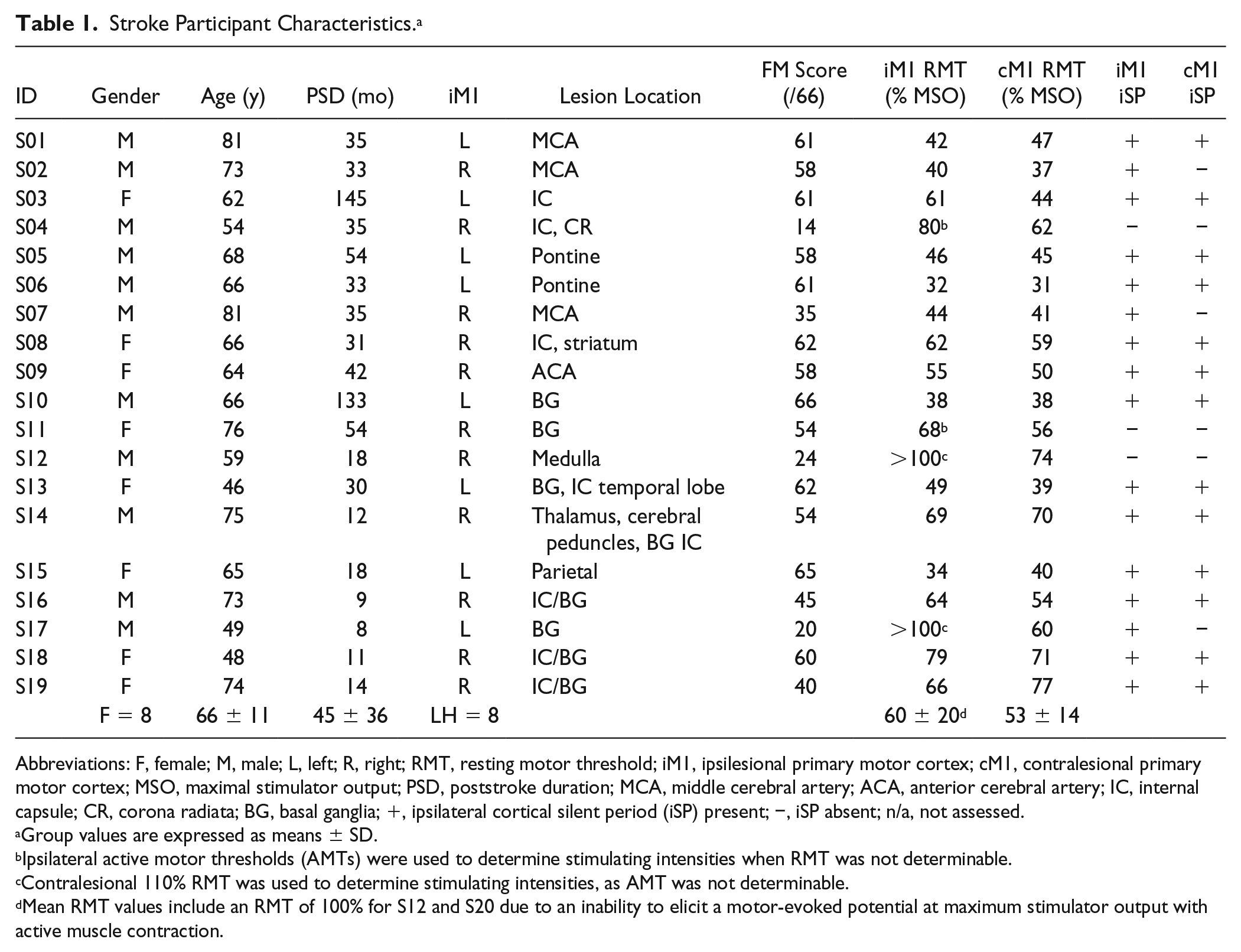
Abbreviations: F, female; M, male; L, left; R, right; RMT, resting motor threshold; iM1, ipsilesional primary motor cortex; cM1, contralesional primary motor cortex; MSO, maximal stimulator output; PSD, poststroke duration; MCA, middle cerebral artery; ACA, anterior cerebral artery; IC, internal capsule; CR, corona radiata; BG, basal ganglia; +, ipsilateral cortical silent period (iSP) present; −, iSP absent; n/a, not assessed.
Group values are expressed as means ± SD.
Ipsilateral active motor thresholds (AMTs) were used to determine stimulating intensities when RMT was not determinable.
Contralesional 110% RMT was used to determine stimulating intensities, as AMT was not determinable.
Mean RMT values include an RMT of 100% for S12 and S20 due to an inability to elicit a motor-evoked potential at maximum stimulator output with active muscle contraction.
Assessment of Motor Behavior
Upper extremity motor behavior was assessed using the Fugl-Meyer Assessment (UE-FM) and the Wolf Motor Function Test (WMFT). 33 Additionally, hand grip strength was assessed using a hand grip dynamometer. Hand motor dexterity was assessed using the Nine Hole Peg Test (NHPT). The WMFT and NHPT were both converted to a rate indicating how many times the participant could complete the task in 60 seconds. The stroke group underwent clinical assessments at the beginning of the session by a licensed physical therapist.
TMS Experimental Procedures
Single monophasic TMS pulses (Magstim 2002, MagStim, Wales, UK) were delivered though a figure-of-eight coil (70 mm). Participants were fitted with earplugs and seated upright in an armchair with both hands resting on a table. Stereotactic neuronavigation software (BrainSight®, Rogue Research Inc, Montreal, Quebec, Canada) was used to identify the target location of the abductor pollicis brevis (APB) muscle representation within the ipsilesional/nondominant (i/nd) and contralesional/dominant (c/d)M1. A high-resolution T1 anatomical MRI image (repetition time [TR] = 7.4 ms, echo thime [TE] = 3.7 ms, flip angle θ = 6°, field of view [FOV] = 256 mm, 160 slices, 1 mm thickness) was collected prior to the testing session and used to guide TMS delivery. At the beginning of the session, the optimal site for eliciting a motor-evoked potential in the contralateral APB muscle was determined using standard procedures and used for navigated TMS during the session. 34 Resting motor threshold (RMT) was determined bilaterally using standard procedures. 34
TMS-evoked EEG responses were assessed during resting and active motor states. During the resting assessment, participants kept both hands at rest while the experimenters monitored real-time bilateral APB EMG activity. During the active assessment, participants maintained a volitional contraction at 50% of their maximal force output using a handheld dynamometer in the hand ipsilateral to the site of TMS. 14 At each assessment, 50 TMS pulses were delivered over the M1 APB location at a jittered rate of 0.1 to 0.25 Hz. TMS intensities were set at 120% and 150% RMT for rest and active conditions, respectively. 14
Assessment of TMS-Evoked Cortical Responses
During each assessment, EEG signals (sampling rate 5kHZ, filter range DC to 1000 Hz) were continuously recorded using a 32-channel TMS-compatible electrode cap (Easy Cap, Brain Products GmbH, Gilching, Germany) connected to a BrainAmp DC amplifier (Brain Products GmbH). Signals were recorded using Recorder software (Brain Products GmbH) and saved for offline analysis. The reference channel location was chosen as the FCz electrode position and the ground was in the AFz position. An additional electrode was attached to the infraorbital area of the right eye and used to record eye blinks. Prior to assessments, impedance levels were lowered to ≤10 kohm for all channels and checked periodically to maintain this impedance level.
Assessment of EMG Corticomotor Excitability Measures
EMG activity was recorded from bilateral APB muscles during each assessment. Two disposable conductive adhesive hydrogel electrodes were attached over each APB muscle and a ground electrode was placed over the dorsum of each hand following standard preparation procedures. BrainSight software (v. 2.2.14) was used to visualize and record EMG signals using a 2-channel EMG device (Rogue Research Inc).
Data Reduction and Analysis
Quantification of Interhemispheric Coherence
All EEG data were preprocessed in EEGLAB. 35 This software was used to epoch (−1000 to 4000 ms relative to TMS) and re-reference (linked to FCz electrode position) all data. Interhemispheric coherence was calculated as the imaginary part of coherency (IPC) 36 value between electrodes overlying the scalp location of M1 of each hemisphere (left: C3, right: C4) within the beta frequency range (15-30 Hz) pre-TMS (−1000 to 0 ms) and post-TMS (0-300 ms). While coherence measures the linear dependency of 2 signals at a specific frequency, IPC requires a time-lag in the evaluation of these signals and reflects true interaction. 36 Utilizing IPC minimizes overestimation biases that artificially influence coherence from volume conduction and other artifacts (stimulation, movement, etc). IPC is a particularly useful approach to evaluate TMS perturbation-evoked EEG signals due to the insensitivity to instantaneous sources of artifact, such as that from the TMS. Perturbation-evoked interhemispheric beta IPC values were calculated during rest and active conditions and for each hemisphere of TMS delivery for all participants. IPC analyses were performed using custom MATLAB routines. 36
Quantification of the iSP
The iSP was determined using EMG data from each participant during the active iM1 and cM1 conditions. Ipsilateral APB muscle activity was rectified and averaged over 50 trials. The mean prestimulus EMG activity over 50 ms prior to TMS (mean pre-TMS EMG) was calculated and used to normalize the EMG data and in defining the iSP duration. iSP onset was defined as the poststimulus point at which EMG activity dropped lower than 2 standard deviations below the mean pre-TMS EMG and remained so for at least 2 ms. 37 iSP offset was defined as the time at which the post stimulus EMG activity resumed its mean pre-TMS EMG activity for at least 2 ms; iSP duration was then calculated as the time point of the iSP offset minus that of the iSP onset. 37 Study team members were blinded to group and lesion location during data processing and analysis.
Statistical Analysis
All clinical motor behavior, coherence, iSP, and data were tested for normality and homogeneity of variance using Kolmogorov-Smirnov and Levene’s tests, respectively. Parametric testing procedures were used if the data met assumptions of normality and homogeneity of variance as indicated by a nonsignificant test result. All analyses were performed using Statistical Package for Social Sciences version 24 (IBM Corp, Armonk, NY) with a critical α level set to .05. For all significant interactions and main effects, Bonferroni post hoc testing was performed. Groups were matched for stimulation hemisphere (ipsilesional M1/nondominant M1 and contralesional M1/dominant M1). To test the effect of TMS on interhemispheric coherence, 2 separate 2 × 2 (group-by-time) mixed-design analyses of variance (ANOVAs) were performed during i/ndM1 and c/dM1 TMS in both the active and rest conditions. Two-way mixed ANOVAs were performed to test if interhemispheric coherence and iSP duration for each group differed between hemisphere (i/ndM1 vs c/dM1) during the active and rest conditions. The relationships between interhemispheric coherence and iSP duration versus paretic arm motor behavior and relationships between interhemispheric coherence and iSP for each limb and corresponding hemisphere of stimulation were tested using Pearson product-moment correlation coefficients.
Results
Complete datasets of EEG coherence measures were collected for all participants in the control group. During the active condition in the stroke group, complete data sets were obtained for 18 of 19 participants; 1 participant trial (S11 cM1 TMS) had excessive (>50 ms duration) TMS artifact in the EEG recording (C3 and C4 electrodes) and was subsequently discarded from all active condition coherence analyses. During the resting condition in the stroke group, complete data sets were obtained from 16 of 19 participants; 2 participants (S01 and S03) did not complete testing during the resting condition due to length of the testing session; 3 participants in the stroke group were discarded from all resting IPC analyses due to prolonged TMS artifact resolution (>50 ms duration) in the EEG signal during iM1 (S04) and cM1 (S08 and S11) TMS.
Complete datasets of EMG iSP measures were calculated in 12 of 14 total control participants; a suppression of EMG activity was not present during ndM1 stimulation (C01) and dM1 stimulation (C02), and these participants were removed from the iSP analysis. Bilateral iSP measures were present in 13 of 19 stroke participants; the presence of an iSP could not be detected for 6 participants in the paretic hand (cM1 TMS) (S02, S04, S07, S11, S12, S20) and 3 participants in the nonparetic hand (iM1 TMS) (S04, S11, S12). Participants for whom iSP could not be calculated were discarded from iSP analyses. All data met assumptions of normality and homogeneity of variance.
Effect of TMS on Interhemispheric Coherence
Comparison of pre- and post-TMS coherence during the active i/ndM1 TMS condition showed a group-by-time interaction (
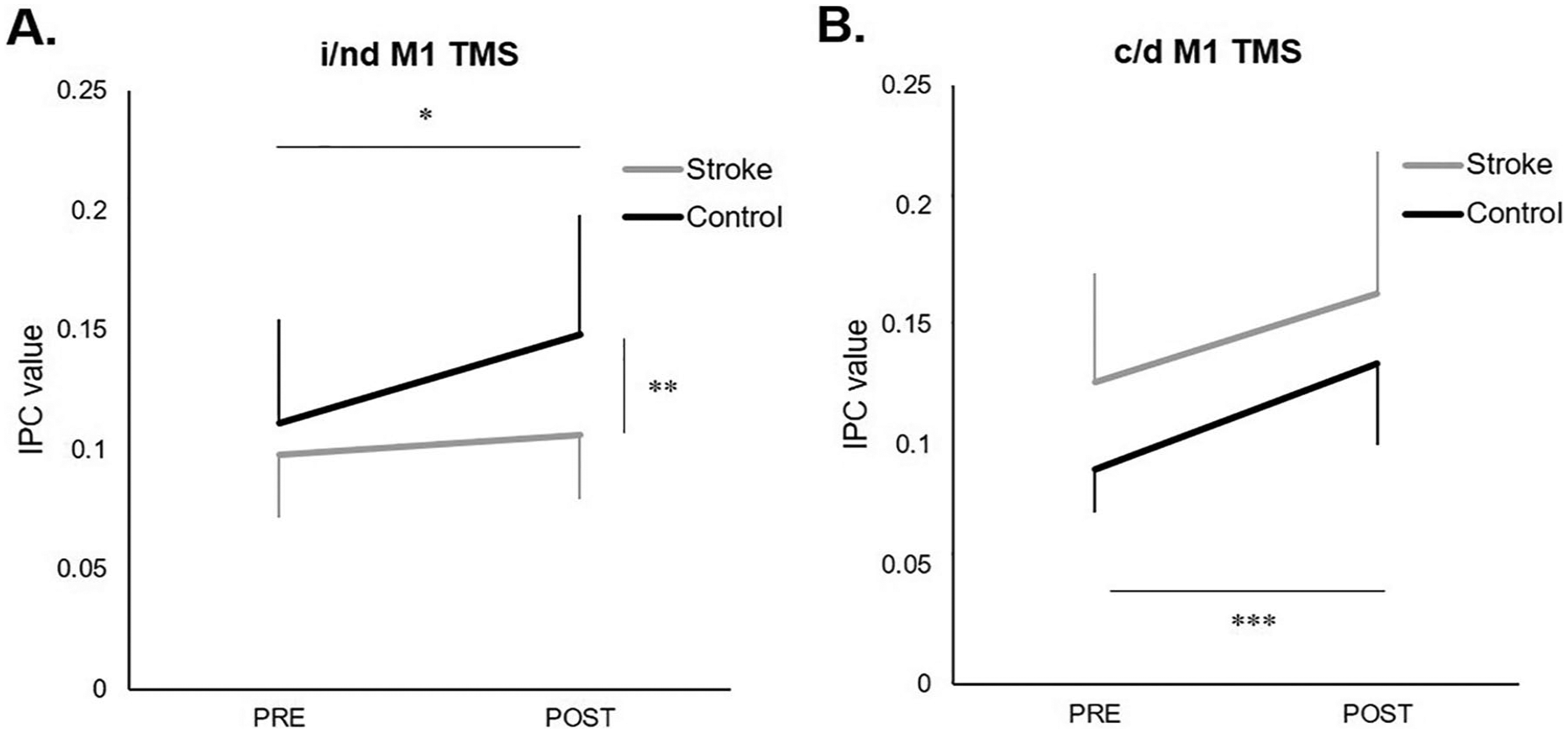
Coherence values (mean ± SD) during the active condition (A) ipsilesional (i)/nondominant (nd) primary motor cortex (M1) and (B) contralesional (c)/dominant (d) M1 transcranial magnetic stimulation (TMS) at pre and post time points relative to TMS. (A) During ndM1 TMS in controls, coherence increased immediately following TMS (*
Between-Group and Hemisphere Comparisons of Interhemispheric Coherence During Muscle Contraction
When testing the effects of group and hemisphere of stimulation during the active condition, we observed a significant group-by-hemisphere interaction (
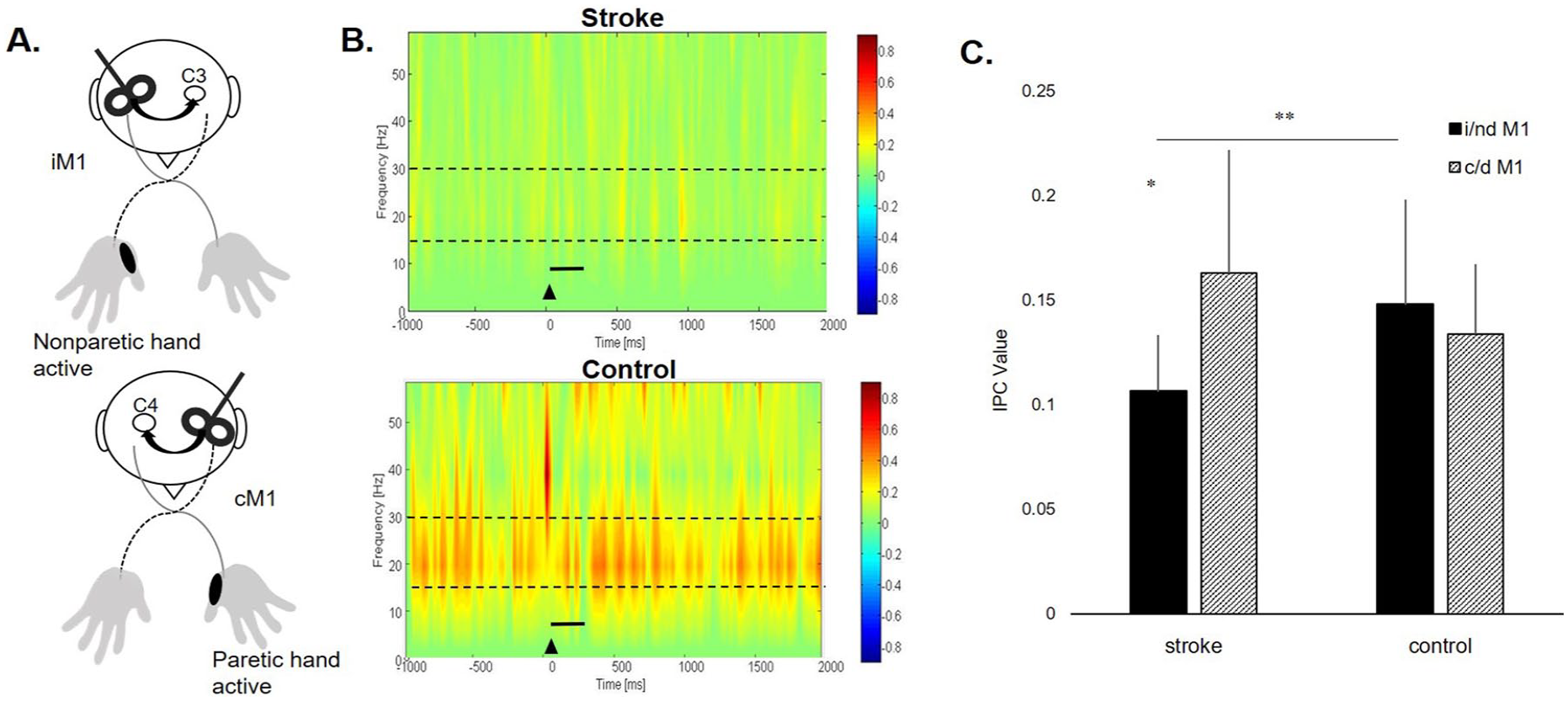
(A) Experimental paradigm for the active condition during ipsilesional (i) primary motor cortex (M1) and contralesional (c) M1 transcranial magnetic stimulation (TMS). TMS was delivered to the motor hotspot of the abductor pollicis brevis (APB) muscle contralateral to the site of stimulation while the APB muscle ipsilateral to the site of stimulation maintained a consistent contraction at 50% maximal volitional isometric contraction. iM1 and cM1 TMS were matched to nondominant M1 and dominant M1 TMS in the control group. (B) Time-frequency plot during the active iM1 TMS condition showing greater TMS-evoked interhemispheric beta imaginary part of coherency (IPC) values (warmer colors) in a representative participant poststroke (top) and a control participant (bottom). Black triangle denotes TMS onset. Broken lines indicate the frequency range (15-30 Hz) of interest. Solid black line represents time bin of data (0-300 ms) used for analysis. (C) Coherence values (mean ± SD) for i/nd M1 and c/d M1 TMS in stroke and control groups during the active condition. There was a significant group-by-hemisphere interaction (
Associations Between Interhemispheric Coherence and Motor Behavior
During the active iM1 TMS condition, we observed a negative relationship between coherence and UE-FM score (
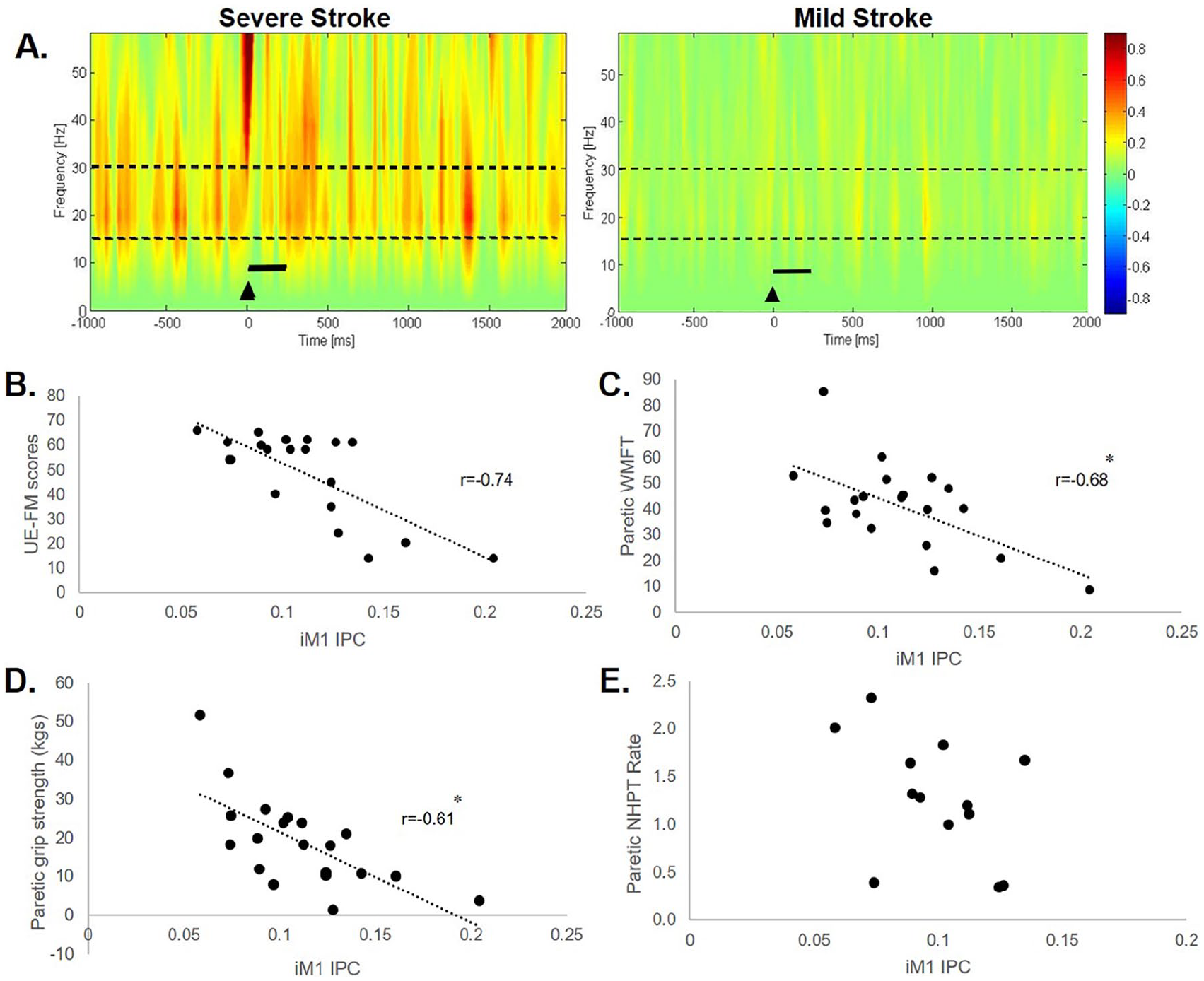
(A) Time-frequency plots during the active ipsilesional primary motor cortex (iM1) transcranial magnetic stimulation (TMS) condition showing greater TMS-evoked interhemispheric beta imaginary part of coherency (IPC) values (warmer colors) in a participant poststroke with severe arm impairment (S12, upper extremity Fugl-Meyer Assessment [UE-FM] score = 24) compared with a participant with mild arm impairment (S14, UE-FM score = 54). Black triangle indicates TMS onset. Broken lines indicate the frequency band range (15-30 Hz) of interest. Solid black line represents bin of data (0-300 ms) used for analysis. During the active iM1 condition, relationships between IPC versus (B) UE-FM score, (C) paretic Wolf Motor Function Test (WMFT), (D) paretic grip strength, and (E) paretic Nine Hole peg Test (NHPT) rate in the stroke group. There was a negative relationship between IPC values versus UE-FM (
Between-Group and Limb Comparisons of iSP Duration and Magnitude
When testing the effects of group (stroke vs control) and limb (paretic/nondominant vs nonparetic/dominant) during the active condition on iSP measures, there was a main effect of group; individuals with stroke showed longer iSP duration compared with controls (
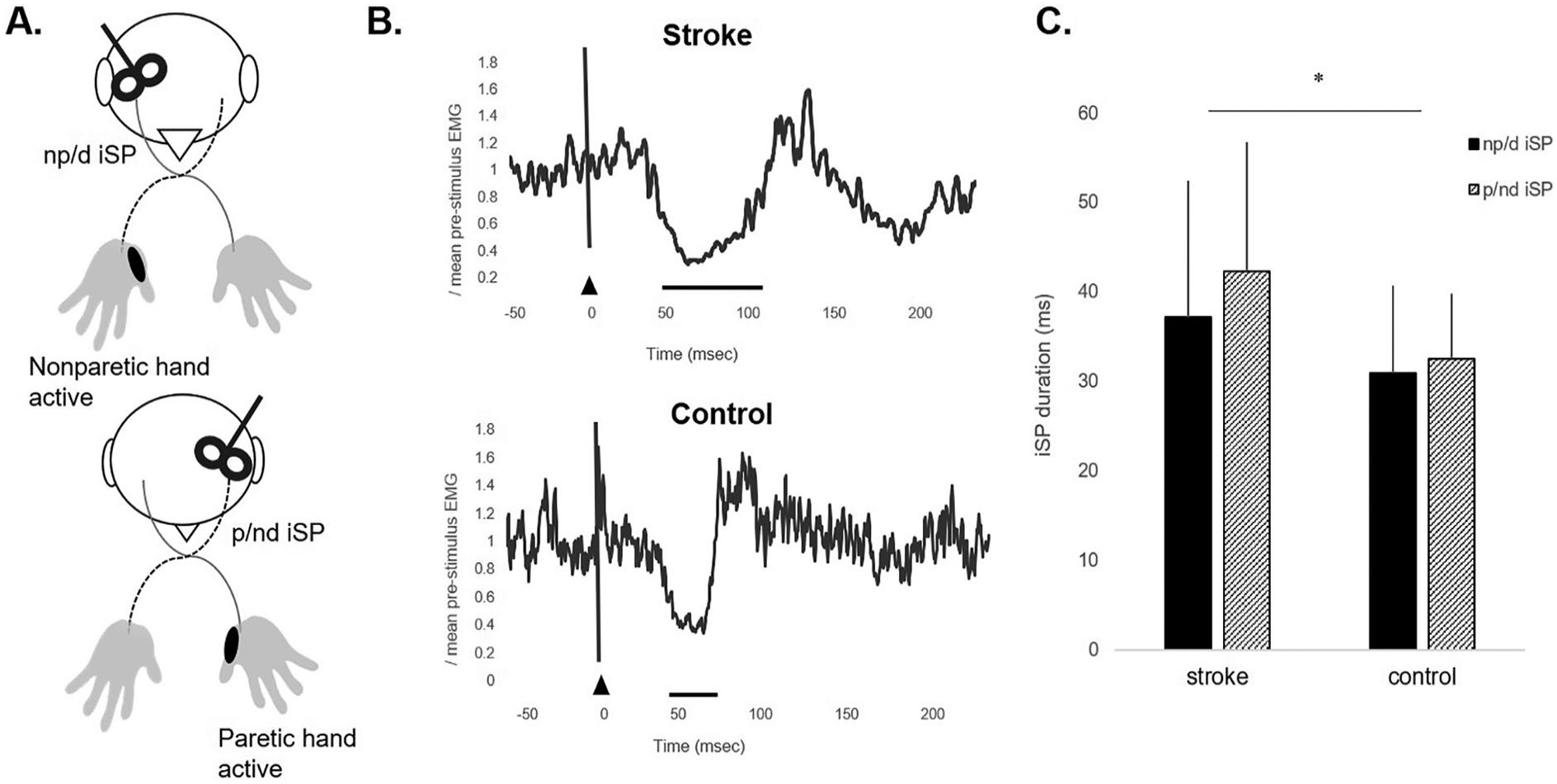
(A) Experimental paradigm during ipsilateral cortical silent period (iSP) data acquisition for the nonparetic (np)/dominant (d) hand and paretic (p)/nondominant (nd) hand. Transcranial magnetic stimulation (TMS) was delivered to the motor hotspot of the abductor pollicis brevis (APB) muscle contralateral to the site of stimulation while the APB muscle ipsilateral to the site of stimulation maintained a consistent contraction at 50% maximal volitional isometric contraction. (B) Electromyographic (EMG) activity representing the paretic iSP of a participant in the stroke group (S13, iSP duration = 62.3 ms) and the nondominant iSP of a participant in the control group (iSP duration = 28.97 ms). Black triangle indicates TMS onset. Solid black line represents iSP duration. (C) Mean ± SD iSP duration for the np/d and p/nd hand in stroke and control groups. Individuals poststroke showed longer iSP durations compared with controls (*
Associations Between iSP Duration and Motor Behavior
There were no significant relationships between paretic iSP duration and UE-FM score (
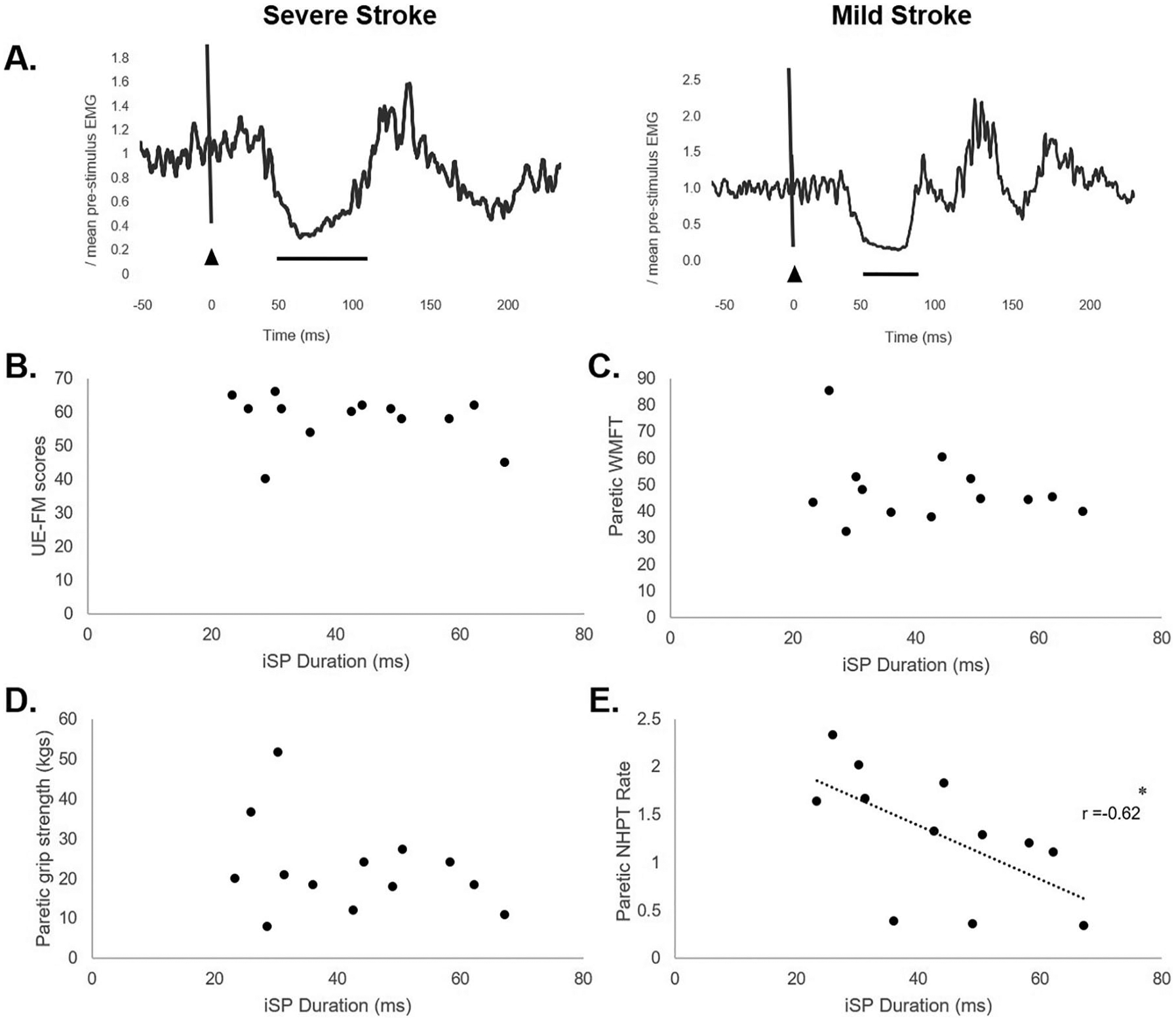
(A) Electromyographic activity representing the paretic ipsilateral cortical silent period (iSP) of a severely affected participant (S13, Nine Hole Peg Test [NHPT] rate = 1.11, iSP duration = 62.3 ms) and a mildly affected participant (S10, NHPT rate = 2.01, iSP duration = 30.3ms) in the stroke group. Black triangle indicates transcranial magnetic stimulation (TMS) onset. Solid black line represents iSP duration. Relationships between paretic iSP duration versus (B) upper extremity Fugl-Myer Assessment (UE-FM) score, (C) paretic Wolf Motor Function Test (WMFT), (D) paretic grip strength, and (E) paretic NHPT rate in the stroke group. There was a negative relationship between iSP duration paretic NHPT rate (
Associations Between Interhemispheric Coherence and iSP
During the active nd/iM1 TMS condition, there was no relationship between coherence and iSP duration in the nonparetic/dominant or in the paretic/nondominant hand for either group (stroke nonparetic
Discussion
Here we provide novel evidence that TMS-evoked measures of interhemispheric interactions between ipsilesional and contralesional motor cortices during motor activity are atypical and negatively associated with paretic hand motor behavior. Our findings show that the typical increase in interhemispheric coherence occurring immediately following TMS in neurologically intact controls and the cM1 in stroke was not observed following iM1 stimulation in the stroke group. This observation supports the addition of TMS to assessments of cortical connectivity as a unique contribution to enhance our understanding of neural connectivity poststroke. Interestingly, interhemispheric coherence measures were not associated with transcallosal inhibition assessed with the EMG-based iSP during the same task. This finding suggests there may be distinct neuromechanistic contributions to interhemispheric interactions indexed by interhemispheric coherence and iSP. Further supporting this concept, each measure was associated with different clinical measures of paretic arm motor behavior. Thus, TMS-evoked EEG and EMG measures of interhemispheric interactions may reflect unique neural mechanisms contributing to different aspects of abnormal poststroke paretic arm and hand motor behavior.
Interhemispheric interactions were dependent on motor activity, as differences between groups and relationships to poststroke clinical function were observed only during muscle contraction but not at rest. Beta frequency oscillations have cortical generators in M1, appear to be directly controlled by inhibitory GABAergic neuronal activity, 38 and are modulated by motor state.39,40 In our previous work, differences in amplitudes and latencies of cortical TMS-evoked-potentials between stroke and controls were observed during both active and rest conditions, 30 whereas, here we observed group differences in interhemispheric connectivity and relationships to clinical function measures only during an active motor state. Additionally, we observed interactions between stroke and control groups before and immediately following TMS during the active condition but not at rest. Here, higher interhemispheric beta coherence in the most severely impaired stroke survivors may suggest increased communication between iM1 and cM1 during sustained motor activity38,41-43 likely reflecting poststroke neural network reorganization. 21 This finding is also consistent with previous studies demonstrating atypical modulation of interhemispheric inhibition during volitional motor activity after stroke3,31 and the presence of bilateral cortical activation patterns during motor activity in the most severely affected stroke survivors.6-8 Although causality is difficult to determine, bilateral cortical activation patterns in these individuals may reflect stronger interhemispheric GABAergic interactions engaged during muscle activation triggered by lesion-induced neural dysfunction within the lesioned hemisphere.44,45 Together, these results inform future studies that the state of the motor system represents an important consideration when assessing measures of functional cortical connectivity and may even influence neuromodulatory effects of noninvasive brain stimulation paradigms. 46
Our findings, for the first time, revealed that higher TMS-evoked interhemispheric beta coherence during sustained ipsilateral nonparetic hand muscle contraction was associated with greater impairment of paretic arm motor behavior in stroke survivors. While decreased interhemispheric inhibition from cM1 to iM1 may occur with recovery of function following rehabilitation,
47
others have found no relationship between change in interhemispheric inhibition and poststroke motor function48,49 or even showed greater cM1 activity with the recovery of motor function after stroke.
50
In support of the latter finding, when the activity in cM1 is suppressed, stroke survivors with severe impairment show poorer reaction times
51
and animals with large lesions lose the ability to perform a reaching task.
52
Interestingly, when comparing coherence between groups and hemispheres, our results imply that
Findings from this study indicate TMS-evoked cortical responses may augment information available with stand-alone TMS and EEG and potentially index unique features of reactivity and connectivity in the corticomotor system not previously available. Our findings support findings of previous studies implicating the functional role of cortical connectivity in poststroke motor recovery.14,28,29 Additionally the concurrent TMS-EEG paradigm may also provide insight into the directionality of interhemispheric interactions and distinct hemispheric contributions to cortical connectivity strength. Coherence during iM1 but not cM1 TMS was negatively associated with poststroke motor behavior suggesting that neural excitability and connectivity of iM1 may be the primary contributor to persistent upper limb motor impairment after stroke. This notion is in agreement with previous literature. 53 Associations between interhemispheric coherence and motor behavior generally did not occur in the absence of TMS, which may further support iM1 connectivity as the origin for atypical interhemispheric interactions after stroke. Additionally, structural reserve of the lesioned hemisphere and corticospinal tract may influence the strength of interhemispheric connectivity 54 and whether interhemispheric inhibition plays a maladaptive or functional role over the course of poststroke motor recovery. 22 This possibility may be supported by the data distribution of interhemispheric coherence and motor impairment in Figure 3B; the negative correlation is driven primarily by individuals with UE-FM scores <50. Given that iSPs could not be elicited and quantified in participants primarily with more severe impairment (Table 1), using TMS-evoked cortical responses provided an opportunity to evaluate interhemispheric interactions across a wider range of poststroke motor disability. Categorizing patients according to impairment level may further clarify different functional roles of cortical reorganization patterns salient to motor recovery in individuals poststroke. Improved understanding of the mechanisms of cortical connectivity offered by multimodal neuroimaging approaches such as TMS-EEG could be important for the development of more effective poststroke treatments, particularly when developing therapeutic noninvasive brain stimulation paradigms. These paradigms include cortico-cortical paired associative stimulation to strengthen or weaken transcallosal pathway connectivity. 55
Interhemispheric coherence was not associated with iSP suggesting that distinct neural mechanisms contribute to each of these measures. Interestingly, our previous study found that latencies of early TMS-evoked potentials correlated with the contralateral silent period duration in the paretic hand of stroke survivors. 30 Differences between these findings and those of the present study could be because local TMS-evoked cortical responses and the contralateral SP reflect the excitability of similar intracortical neural circuits while interhemispheric coherence and iSP reflect different neural mechanisms of inhibition. Likewise, our findings show that interhemispheric coherence was associated with overall paretic arm motor behavior (UE-FM, grip strength, WMFT) but not with fine motor control (paretic NHPT rate) (Figure 3); however, the iSP was negatively associated with fine motor control but not with overall paretic arm motor behavior (Figure 5). This dichotomy supports the notion that distinct neural mechanisms of interhemispheric interactions are reflected in each of these measures and are uniquely associated with different clinical features of poststroke paretic arm and hand motor behavior. The iSP reflects neural activity of not only cortical but also subcortical sources, including rubrospinal projections involved in fine motor control of the hand. 56 The influence of such subcortical pathways could explain iSP as an index of fine motor skill function in stroke survivors. Given its lack of muscle specificity, interhemispheric coherence measured with EEG may serve as a better index of interhemispheric contributions to overall upper limb motor behavior after stroke. We propose that interhemispheric coherence and the ipsilateral silent period are complimentary measures of interhemispheric interactions that can be used in tandem to understand the contributions of typical and atypical interhemispheric communication to normal and dysfunctional motor control.
Limitations
There are several limitations to the present study that could influence the interpretation of the observed results. Given the limited sample size, we were unable to stratify the heterogeneous stroke cohort based on lesion location and demographic characteristics. Because iSP is associated with mid-callosal volume, 57 future studies could employ measures of transcallosal anatomy to further characterize brain structure-function relationships mediating stroke recovery. Limited spatial resolution of scalp-based EEG recordings restricts the interpretation of the cortical generators contributing to the interhemispheric coherence measures evaluated in the present study. Nonetheless, there is evidence to indicate the primary motor cortex as a main generator of cortical activity recorded in the electrodes of interest (C3/C4) and sensorimotor cortical activity is likely the primary source of beta oscillatory activity.58,59 TMS-evoked potentials possess cortical and peripheral-evoked components 60 ; thus we cannot rule out the influence of peripheral contributions (eg, somatosensory and auditory potentials) to coherence measures. In the present study design, the possibility that differences observed in the active condition were influenced by the higher stimulation intensities should also be considered.
Conclusions
TMS-evoked cortical coherence during hand muscle activation may advance findings from previous investigations of poststroke interhemispheric interactions and offer new insights into neural network connectivity that inform and extend our previous understanding of the neural mechanisms underlying poststroke arm and hand motor function. The findings of this study support future investigations exploring the effect of rehabilitation on TMS-evoked interhemispheric coherence and coincident changes in paretic limb motor function. Further research utilizing multimodal approaches should inform models of poststroke recovery that have implications for the clinical prognosis and rehabilitation of persistent upper limb motor impairment in stroke survivors.
Supplemental Material
NNR862552_Supplemental_Material_CLN – Supplemental material for Role of Interhemispheric Cortical Interactions in Poststroke Motor Function
Supplemental material, NNR862552_Supplemental_Material_CLN for Role of Interhemispheric Cortical Interactions in Poststroke Motor Function by Jacqueline A. Palmer, Lewis A. Wheaton, Whitney A. Gray, Mary Alice Saltão da Silva, Steven L. Wolf and Michael R. Borich in Neurorehabilitation and Neural Repair
Footnotes
Acknowledgements
We would like to acknowledge Kamal Shadi for assisting in data analyses.
Supplementary material for this article is available on the
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by the American Heart Association (AHA00035638) and the Eunice Kennedy Shriver National Institutes of Child Health & Human Development of the National Institutes of Health (K12HD055931 and 5R24HD050821).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
