Abstract
Introduction
Evaluating cognition, probing consciousness and predicting recovery are critical clinical issues in order to take care of patients with disorders of consciousness (DOC), that is, patients in a comatose, vegetative (or unresponsive wakefulness syndrome) or minimally conscious state. Sophisticated protocols have demonstrated conscious capacities in some of these patients, such as understanding and following verbal instructions.1,2 For example, it has been shown that a patient who appeared to be in a vegetative state (VS) was still able to understand complex verbal commands and produce mental imagery tasks such as “imagine you are playing tennis,” although clinical evaluation failed to detect overt signs of consciousness. 2 Even though this result is very remarkable, one major drawback of this kind of protocol is its lack of sensitivity. 3 It cannot detect patients who are in the process of recovering awareness, but who are unable to understand complex verbal commands and/or to produce the mental effort required by sustained mental imagery tasks. Increasing the sensitivity of clinical tests, notably by reducing the number of false negatives, is an important issue to evaluate the residual cognitive functions in this patient population.
Sensitivity of protocols may be enhanced by using experimental material that boosts expression of cognition. Indeed, some studies have suggested that the use of salient and emotional stimuli increases the probability to observe a cerebral response in patients with DOC. For example, it has been shown that the event-related P3 potential is obtained in 36% of patients with DOC in a conventional oddball paradigm using tones, while it is elicited in 52% of the patients in a paradigm that associates tones to emotional verbal stimuli (ie, a short phrase spoken by a member of the family). 4 In the same way, the P3 is elicited more frequently when the patient’s own name is used as the deviant stimulus in an oddball paradigm rather than a rare tone in the classical implementation of the oddball paradigm. 5
One of the most emotional and alerting stimulus is probably music. Most individuals state that their investment in listening to music derives from the ability of music to convey emotions. 6 Furthermore, numerous research has demonstrated that music listening conveys beneficial effects on cognitive processes, both for normal 7 and pathologic cerebral functioning.8,9 For example, patients with visual neglect show enhanced visual attention when the tasks are performed while listening to their preferred music relative to listening to unpreferred music. 10 After middle cerebral artery stroke, an increase of cognitive recovery and mood, as well as long-term plastic changes in early sensory processing (as indexed by the mismatch negativity), have been observed after having listened to music daily over a 2-month period.11,12 Recently, in conscious patients from intensive care units, it has also been shown that music exposure results in greater reduction of sedation frequency, in comparison with usual care or noise-canceling conditions. 13 In patients with DOC, only a few single-case behavioral studies have investigated the potential effect of music on cognition. For example, following exposure to live music and to familiar songs, a patient in a VS showed purposeful behavioral responses. 14 Similar effects have been described in other behavioral single-case studies, but it is difficult to draw firm conclusions as these studies did not use quantified measures and/or control conditions/groups.15,16
The aim of the present study was to evaluate the effect of music on cerebral functioning in patients with DOC, using bedside electroencephalographic recording. We hypothesized that exposure to preferred music would enhance the event-related response to a highly significant stimulus, the patient’s own name, as compared to a control condition (exposure to a continuous sound).
Methods
Subjects
The study was conducted in 13 patients with DOC (3 women; mean age ± SEM, 41.5 ± 16 years) in the Department of Intensive Care or in the Clinical Neurophysiology Unit of the Neurological Hospital, Lyon. One additional patient was excluded because of excessive electroencephalographic artifacts. Inclusion criteria were the following: no centrally acting drugs, no neuromuscular function blockers, and no sedation within the prior 24 hours. The etiology of brain injury was traumatic (n = 7), anoxic (n = 4), metabolic (n = 1), or tumorous (n = 1). All patients were in a chronic stage (from 1.5 months to 3 years postinsult), except one (20 days). Seven patients met the diagnostic criteria defining the VS 17 or unresponsive wakefulness syndrome 18 (5 traumatic; 1 anoxic and 1 tumorous), and 6 patients met the diagnostic criteria defining the MCS 19 (2 traumatic; 3 anoxic and 1 metabolic; see clinical details in Table 1).
Patients’ Characteristics and Outcomes.
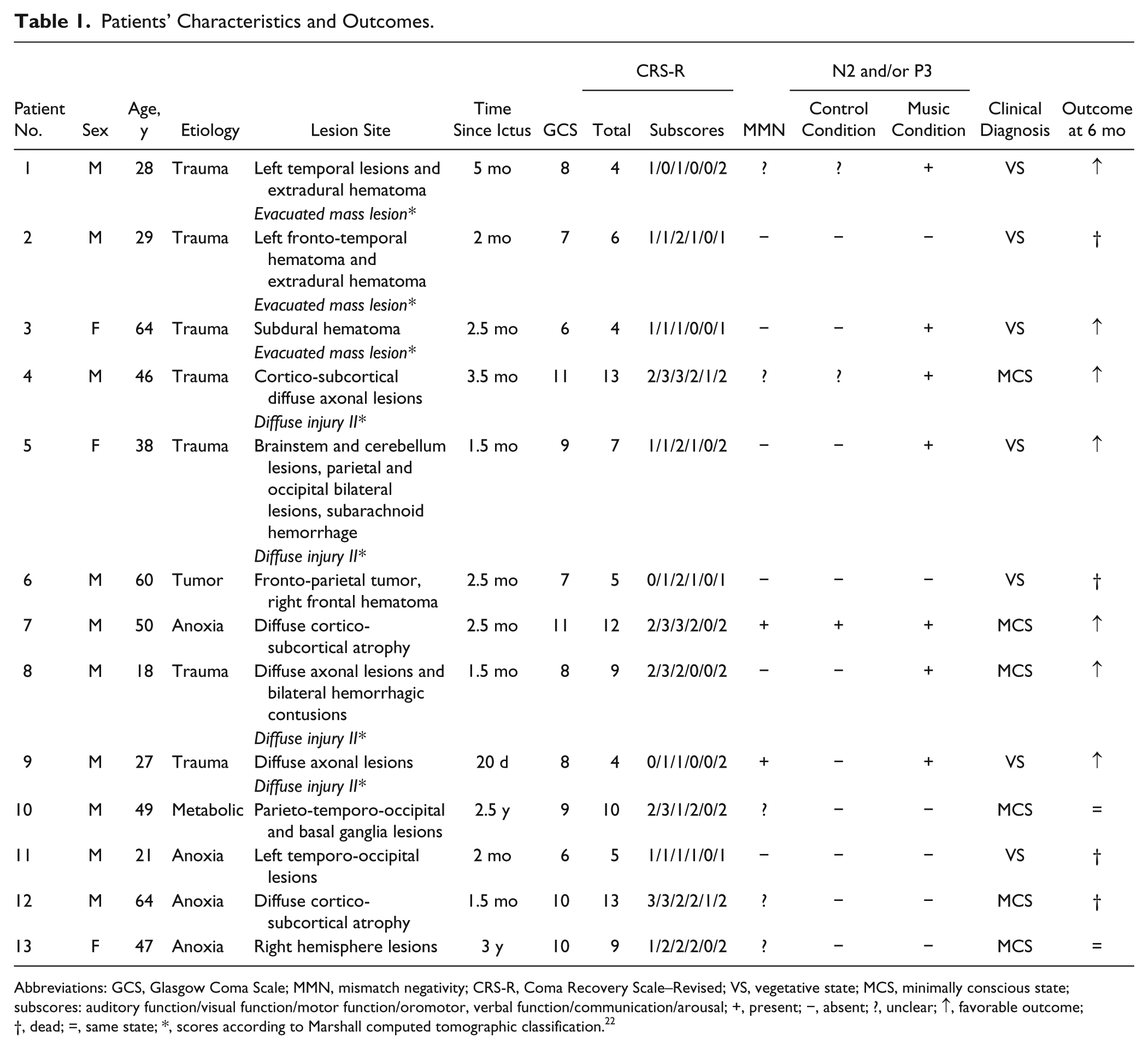
Abbreviations: GCS, Glasgow Coma Scale; MMN, mismatch negativity; CRS-R, Coma Recovery Scale–Revised; VS, vegetative state; MCS, minimally conscious state; subscores: auditory function/visual function/motor function/oromotor, verbal function/communication/arousal; +, present; −, absent; ?, unclear; ↑, favorable outcome; †, dead; =, same state; *, scores according to Marshall computed tomographic classification. 22
The Glasgow Coma Scale (GCS) 20 and the Coma Recovery Scale–Revised (CRS-R) 21 were completed by the medical staff before the present experiment. A scan or magnetic resonance imaging was performed 1.5 months in average before the experiment, except for patients 10 and 13 who were in a stabilized state (2.5 and 3 years after injury, respectively). For traumatic patients, the Marshall computed tomographic classification 22 scores are reported in Table 1.
None of the patients had a history of impaired auditory acuity. All patients showed normal or slightly delayed middle latency auditory-evoked potentials. Somatosensory evoked potentials were observed in all patients (except patients 5 and 13). Mismatch negativity (MMN) responses were observed, with a classical oddball paradigm involving tones, in only 2 patients (patients 7 and 9); in 5 additional patients, the MMN response was unclear.
Six months after the electroencephalography (EEG) experiment, a complete behavioral description was performed, for each patient, by the medical staff in rehabilitation units. Seven patients have shown a favorable outcome, that is, they gained supplementary behavioral responses (Table 1). Five of them (patients 1, 4, 7, 8, and 9), who were in a VS or a minimally conscious state (MCS) at test, have developed an accurate communication that denotes an emergence from MCS. For the other 2 patients (patient 3 and 5), who were in a VS at test, a visual fixation was observed; the presence of such behavioral reaction indicates a MCS. Of the other 6 patients, 2 patients (patients 10 and 13) remained in the same state and 4 patients (patients 2, 6, 11, and 12) died (Table 1).
Thirteen age-matched healthy controls (8 women; mean age ± SEM, 40.8 ± 15 years) also participated in the experiment. None of them had a history of audiological or neurological disease.
The experiment was conducted in agreement with the guidelines of the Declaration of Helsinki and approved by the local ethics committee.
Auditory Stimulation
Eight first names were selected for the testing of each subject (patient or control subject): the subject’s own first name (SON) and seven other first names (OFNs). Before the experiment, the seven OFNs were selected from a list of 18 disyllabic first names (nine masculine and nine feminine) of similar high frequency of occurrence in France (higher than 150 000 since 1900). The full list of first names was presented to the patient’s family, or to the control subjects, in order to exclude familiar first names (ie, close relative’s name). All first names (SON and OFNs) were uttered by the same female speaker with a neutral intonation, recorded (16 bits, 44 100 Hz), equalized to the same dB-A-weighting level, and presented binaurally during the experiment at approximatively 80 dB-A sound pressure level intensity. The mean duration of the first names was 614 ± 137 ms.
For each subject, 10 sequences of 64 first names were created. In each sequence, the SON and the seven OFNs were presented 8 times in a pseudo-random order (with no repetition of a same name and with a homogeneous temporal distribution of the first names); all first names were presented with the same probability (12.5% for each name). The mean stimulus onset asynchrony was 1414 ± 137 ms. The mean duration of a sequence of first names was 1 minute 26 ± 1.8 seconds.
Five excerpts of music were selected for each subject from a questionnaire on musical preferences completed by close relatives of the patients or by the control subjects themselves. The chosen musical excerpts were dynamic, musically coherent, and representative of the whole musical piece. Fading in and fading out were added to avoid a rough end or start of the excerpt. The mean duration of the musical excerpts was 1 minute ± 0.5 seconds.
A continuous music-like noise stimulus was created for the control condition: its spectral characteristics were close to that of music, that is, with a band-pass filter from 20 Hz to 1 kHz and a 10 dB/oct slope from 1 to 20 kHz. This music-like noise stimulus consisted of a spectral approximation of music established from different styles of music, but did not share other acoustic characteristics (eg, pitch, rhythm, envelope, or timbre). Fading in and fading out were added. The duration of the music-like noise stimulus was 1 minute.
All stimuli were delivered using Presentation software (version 14.9). Music (M) and control (C) conditions were presented 5 times in an alternating order, which was counterbalanced between patients and between control subjects (ie, MCMCMCMCMC or CMCMCMCMCM). Each sequence of the first names was preceded by 1 of the 5 preferred music pieces in the music condition and by the music-like noise stimulus in the control condition (Figure 1). The interval between the end of the music (or music-like noise stimulus) and the beginning of the sequence of first names was 2000 ms, and the interval between the end of a sequence of first names and the beginning of the music (or music-like noise stimulus) was 1500 ms.
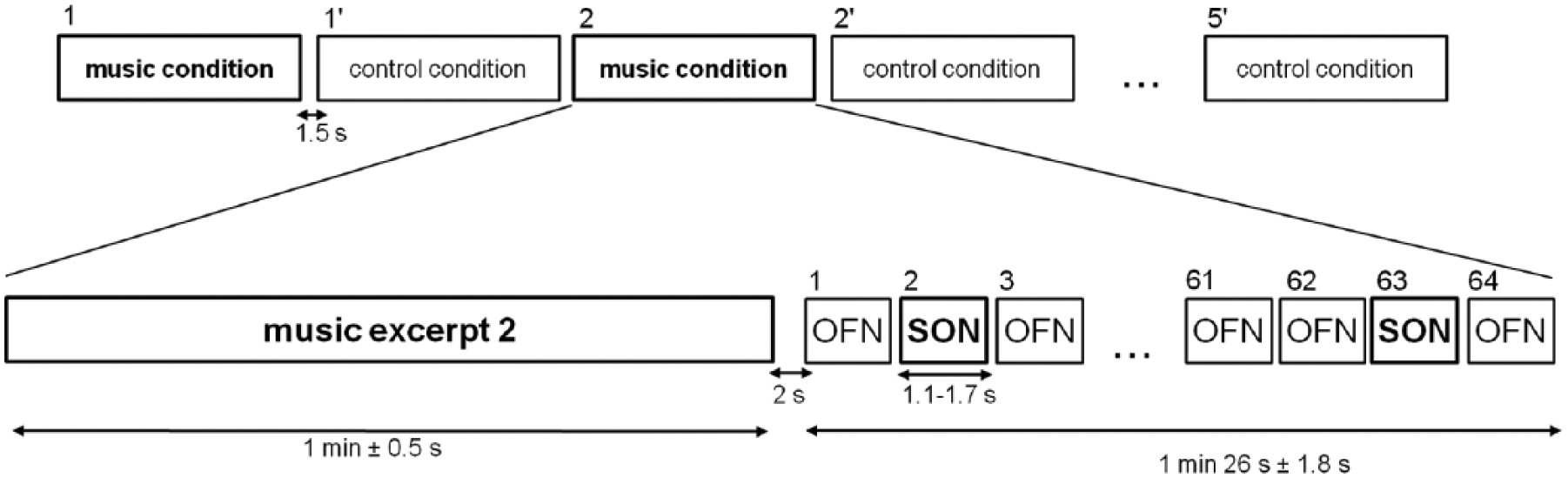
Auditory paradigm. The experimental design consists of 5 blocks of the music condition and 5 blocks of the control condition. Music and control conditions were presented in an alternating order, which was counterbalanced between patients or between control subjects. Each condition was composed of an excerpt of music or a music-like noise stimulus followed by a sequence of first names. In each sequence of first names, 64 first names were presented in a pseudo-random order.
Electroencephalogram Acquisition
For patients, the data were acquired at their bedside. EEG signals from 13 Ag/AgCl electrodes (F3, Fz, F4, T3, C3, Cz, C4, T4, P3, Pz, P4, O1, O2; arranged following the international 10-20 system) referenced to the nose, were amplified using SystemPlus EEG amplifier (Micromed) and sampled at 256 Hz (16 bits) using an analog band-pass filter of 0.1 to 100 Hz. Horizontal electro-oculogram (EOG) was monitored via bipolar electrodes placed laterally to the external canthi, and vertical EOG via bipolar electrodes placed diagonally above and below the right eye. A ground electrode was placed near FPz and impedances were kept below 5 kohm.
Control subjects were seated in a sound-attenuated testing room. EEG signals from 95 Ag/AgCl actives electrodes (Acticap) referenced to the nose, were amplified using BrainAmp EEG amplifier (BrainProducts) and sampled at 500 Hz (32 bits) using an analog band-pass filter of 0.1 to 200 Hz. Vertical EOG was recorded from one electrode mounted beneath the right eye and referenced to the frontal electrode (FP2) directly above the eyes and the horizontal EOG was recorded as the difference in activity between 2 frontal lateral electrodes (F9 and F10). A ground electrode was placed near Fz and impedances were kept below 30 kohm.
Electroencephalogram Analysis
EEG signals were analyzed using custom scripts written in Matlab R2008b (MathWorks) and BrainVision (BrainProducts) software. For 4 patients, signals containing eye blinks were corrected with an Infomax independent component analysis. 23 Raw data were 1 to 30 Hz band-pass filtered and an additional 50 Hz notch filter was applied. Data were segmented into epochs of 1200 ms (including 200 ms before the stimulus) and baseline corrected within 200 ms before SON presentation and before the presentation of OFNs preceding each SON presentation (thus allowing us to compare averages with the same signal to noise ratio). Epochs with amplitudes exceeding ±100 µV for patients and ±50 µV for control subjects were excluded automatically from further analyses. Epochs containing muscle artifacts were excluded using visual inspection. For each control subject and patient, an average of 4 trials has been removed for each condition (leaving a mean of 36 trials for statistical analyses in each condition). Epochs were averaged as a function of electrodes, stimulus (ie, SON or OFN) and condition (ie, music or control condition). The event-related potential analyses were done blinded to the patients’ clinical diagnosis and outcome.
Statistical Analysis
Statistical analysis was conducted similarly to a previous study that had used a similar event-related potentials technique in the same type of patient population.
24
Individual statistical analyses compared the amplitude of the responses to SON and to OFNs as a function of condition (ie, music or control condition). For each patient or control subject, paired
We further checked the statistical significance of the
Results
In healthy controls, the SON evoked the classical N2 and P3 components (see 1 example in Figure 2A), as it has been previously reported.
24
For each of the control subjects, N2 and/or P3 amplitude were significantly higher (
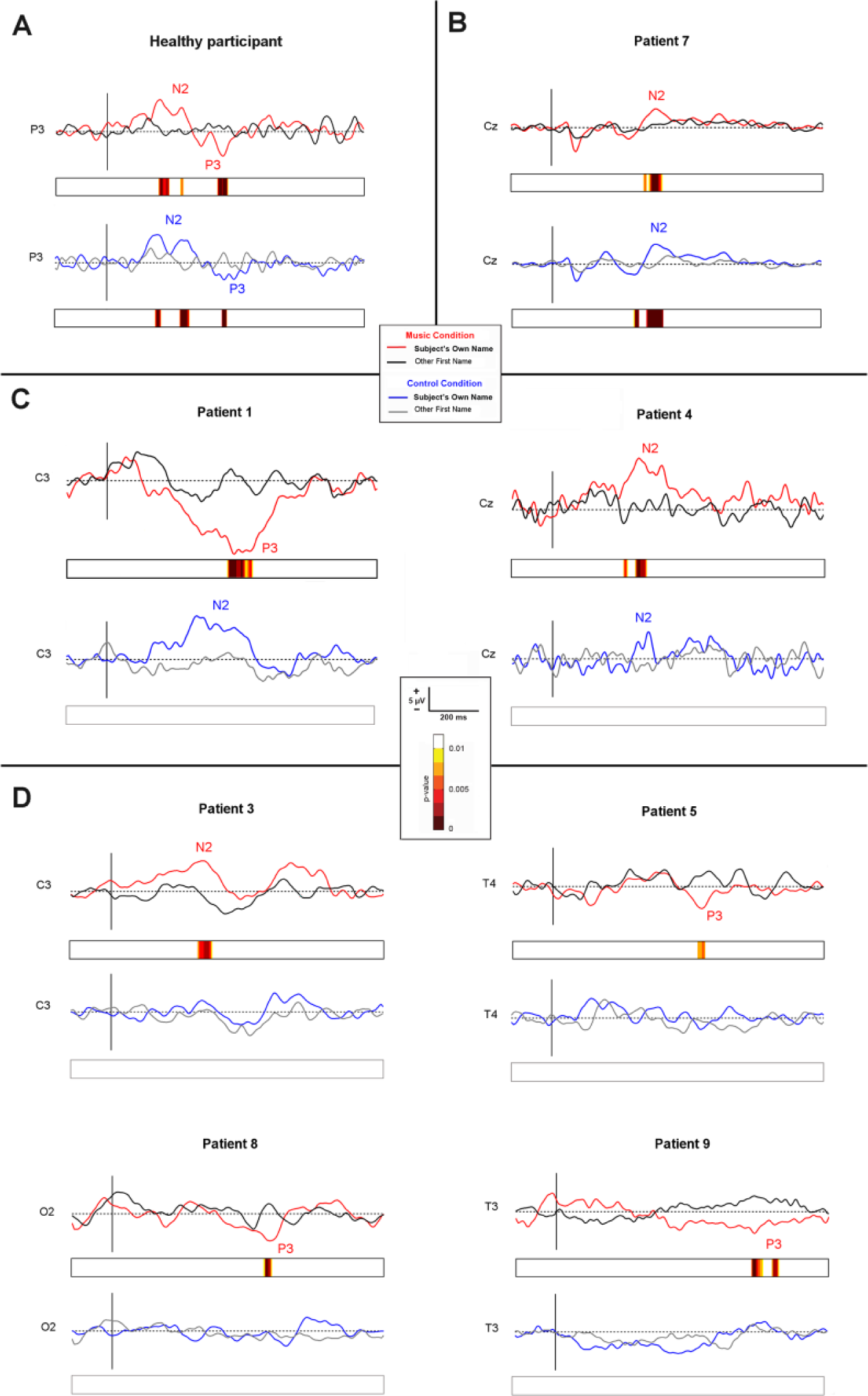
Event-related potentials. Individual event-related potentials in music and control conditions to the subject’s own name (SON) and to other first names (OFNs), in 1 representative control subject (A) and in the 7 patients who showed significant discriminative responses in the music condition (the first group presented in the main text) (B-D): (B) in a patient (patient 7) who showed a significant discriminative response to the SON in music and control conditions, (C) in 2 patients (patients 1 and 4) who developed a discriminative, but nonsignificant, response to the SON in the control condition and who showed a significant discriminative response to the SON in the music condition and (D) in 4 patients (patients 3, 5, 8, and 9) who showed a discriminative response to SON only in the music condition. Note that event-related potentials for patients of the second group were not depicted because of the absence of significant discriminative response to the SON.
Two groups of patients could be distinguished according to the presence or absence of a discriminative response to the SON. The first group of patients (n = 7) showed a discriminative response (N2 and/or P3) to the SON, as compared with the OFNs. For all of them, the discriminative response was significant in the music condition (
A second group of patients (n = 6) did not show any discriminative response to the SON, neither in the music condition nor in the control condition. None of them developed a MMN to tones (Table 1).
The presence/absence of an event-related discriminative response to the SON seemed to be linked to the patient’s outcome. Indeed, all patients (7/7) developing a significant discriminative response to the SON following the music (the first group presented above) have shown a favorable outcome, that is, gained supplementary behavioral responses (such as visual fixation or accurate communication) 6 months after the present experiment. No such link could be established between a favorable outcome and the presence of a discriminative response in the control condition, or the presence of a MMN to tones. Furthermore, all patients (6/6) without a discriminative response (second group), in both music and control conditions, remained in the same state or died 6 months after the experiment.
Discussion
The aim of the present study was to determine the potential effect of music on the cognitive functioning of patients with DOC. We evaluated with the event-related potential method whether the patient’s ability to discriminate his or her own first name (relative to unfamiliar first names) was enhanced after having listened to 1 minute of his or her preferred music (music condition) as compared with a meaningless sound (control condition). We showed that the discriminative cerebral response (N2 and/or P3 components) to the patient’s first name was more often observed in the music condition, than in the control condition. Furthermore, we showed that the presence or absence of a discriminative response in the music condition seemed to be associated with a favorable or unfavorable outcome, respectively, for the patient.
The finding that the SON can evoke an event-related P3 potential in patients with DOC has been previously reported. 26 The presence of the P3 has been interpreted as an index of a discriminative processing to a very salient and emotional word.27,28 The P3 to the SON could be also seen as a marker of self-processing. Indeed, it has been shown, in healthy subjects, that its amplitude correlates with regional cerebral blood flow changes in medial cortical structures, 29 that is, structures that are known to be involved in self processing. 30 In agreement with this hypothesis, similar medial structures are activated in patients with DOC following SON presentation.31,32 Functionally, the P3 to the SON has been interpreted as reflecting top-down attentional and/or arousal mechanisms. 33 Indeed, the amplitude of the P3 is enhanced not only in active versus passive listening conditions for healthy subjects 25 and for patients with DOC 34 but also in waking relative to sleeping states for healthy subjects. 25 Even though the present study cannot bring definitive conclusions on the functional role of the P3 in the processing of the SON, our results show that not all patients developed the same cognitive processes. Indeed, some patients showed a discriminative N2 component, but not a P3 component. This suggests that not all processes associated to the processing of the SON were recovered in these patients. Further studies should be conducted to understand whether the posterior N2 reflects automatic mismatch functions and/or controlled processes in patients with DOC. 35
The present study shows that the discrimination of the SON was improved when its presentation was preceded by the patient’s preferred music, as compared with when it was preceded by a neutral and meaningless sound context. Previous studies have shown that music can have a positive effect on cognition (eg, language processing, visual attention, etc), both in healthy subjects and in brain-damaged patients,10,11 but it has never been shown in patients with DOC (except for some single-case studies, which are missing control conditions). In healthy subjects, beneficial effects of music have been explained in the context of an “arousal and mood hypothesis,” attributing the effect of music listening on cognitive abilities to changes in listeners’ arousal and mood.36,37 This hypothesis is concordant with neuroimaging studies that have shown that music is an emotional stimulus that activates limbic and paralimbic structures (such as the hippocampus), as well as structures involved in the reward circuit (such as the nucleus acumbens).38 -41 According to this hypothesis, the beneficial effects of music on the cognitive functions in patients with DOC could be explained by an overall cortical arousal, probably in the cerebral structures that have been reported to be involved in emotional and mood states. A second and complementary hypothesis to explain the boosting effects of music observed in the patients with DOC is that music might have autobiographically primed the processing of the SON. Indeed, it has been shown previously that hearing these types of autobiographical stimuli (either one’s preferred music or one’s own first name) was associated with the activation of medial prefrontal areas.29,42 Thus it could be hypothesized that in the present study, the preferred music has facilitated the discrimination of the SON by a preactivation of the medio-anterior part of the cortex.
The present study also suggests a possible link between the presence/absence of the discriminative response to the SON in the music condition and the favorable/unfavorable outcome of the patient. All patients (7/7) who had developed a significant N2 and/or P3 event-related potential in the music condition have shown a favorable outcome (appearance of a new major behavior, such as visual fixation or accurate communication, indicative of MCS and emergence from MCS, respectively). Furthermore, all patients (6/6) without a discriminative response to the SON in the music condition have not shown a favorable outcome (they died or no new behavior was observed). This link was observed neither for the control condition nor for the MMN paradigm using tones (Table 1). This result is in line with other studies that have suggested that the MMN has a low predictive value for a favorable outcome. 43 This is also consistent with our hypothesis suggesting that the emotional salience and the autobiographical context (SON, preferred music) are important for an accurate evaluation of the residual cognitive capacities in patients with DOC. Indeed, the link observed here might be due to the beneficial effect of music for a more accurate evaluation of the patient’s cognitive functioning.
This study provides, for the first time, evidence for a beneficial effect of music on cognition in patients with DOC. These results should be confirmed with a greater number of patients, especially to confirm the link between the presence of cerebral discriminative responses in the music condition and the outcome of patient. A larger cohort of patients will also allow for additional group analyses, for example to study the effect of music on cognition according to age, etiology, or level of consciousness of the patient. Moreover, it will be interesting to further investigate the characteristics of the musical material that created the beneficial effect on cognition. Indeed, future studies would need to disentangle the general effect of music (because of its acoustic features) from its autobiographical effect (because of its emotional and meaningful contents), for example by using another control condition (eg, unfamiliar music) and/or another autobiographical stimulus (eg, speech). Finally, together with other recent data investigating the potential beneficial effect of music on brain functioning and brain plasticity,7 -9 our data might further suggest a possible role of music in rehabilitation, that is, for a long-term effect of music on cognition and consciousness in patients with DOC. Thus, these data encourage future research testing the potential long-term role of music listening on cerebral plasticity in patients with DOC.
In conclusion, the present study suggests that a preferred music context is more favorable than a neutral context to the expression of residual cognitive functioning in patients with DOC. Thus, the use of autobiographical contexts should be encouraged in patients with DOC before clinical evaluations in order to enhance their sensitivity.
Footnotes
Acknowledgements
The authors thank the staff of the Department of Intensive Care (Neurological Hospital), the staff of the Clinical Neurophysiology and particularly Ramona Peverelli for her availability. We also thank Laurence Tell, Florent Gobert, Clotilde Cadart, Mélodie Faguet, Tatiana Selchenkova, and Nicolas Grimault for their contribution in the preparation of this protocol.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by institutional grants from the LabEx CeLyA (“Centre Lyonnais d’Acoustique,” ANR-10-LABX-60); by the grant ANR-09-BLAN-0310; by the grant ANR CogniComa; and the French Association for Cranial Traumatism (“France Traumatisme Crânien”).
