Abstract
Background. Phase III trials of rehabilitation of paresis after stroke have proven the effectiveness of intensive and extended task practice, but they have also shown that many patients do not qualify, because of severity of impairment, and that many of those who are treated are left with clinically significant deficits. Objective. To test the value of 2 potential adjuvants to normal learning processes engaged in constraint-induced movement therapy (CIMT): greater distribution of treatment over time and the coadministration of d-cycloserine, a competitive agonist at the glycine site of the N-methyl-D-aspartate glutamate receptor. Methods. A prospective randomized single-blind parallel-group trial of more versus less condensed therapy (2 vs 10 weeks) and d-cycloserine (50 mg) each treatment day versus placebo (in a 2 × 2 design), as potential adjuvants to 60 hours of CIMT. Results. Twenty-four participants entered the study, and 22 completed it and were assessed at the completion of treatment and 3 months later. Neither greater distribution of treatment nor treatment with d-cycloserine significantly augmented retention of gains achieved with CIMT. Conclusions. Greater distribution of practice and treatment with d-cycloserine do not appear to augment retention of gains achieved with CIMT. However, concentration of CIMT over 2 weeks (“massed practice”) appears to confer no advantage either.
Keywords
Phase III trials of rehabilitation of paresis following stroke1-4 have demonstrated the effectiveness of particular rehabilitation strategies and their impact on long-term outcome. However, they have also shown that despite the intensity and dose of the treatments used, impact was modest, and many patients were left with significant impairment. Long-term impairment is even greater in patients who did not have sufficient baseline motor function to qualify for these trials. Thus, any adjuvant to therapies leveraging experience-dependent neuroplasticity, even if it had a modest effect, could be of clinically significant value. In this article, we report the impact of 2 potential adjuvants: 1 pharmacologic (d-cycloserine) and 1 consisting in the potential benefits of greater distribution of treatment over time. Their adjuvant value was tested in a trial employing constraint-induced movement therapy (CIMT): a thoroughly vetted rehabilitation treatment for upper extremity paresis that reliably yields, on average, a clinically significant benefit, thereby qualifying as a behavioral engine for testing adjuvant treatments. 5 It is the reliable benefit of CIMT that can potentially be amplified by an adjuvant treatment.
Distributed Practice
Massed practice is traditionally defined as continuous practice conducted over a single day in which the stimuli or skills to be retained are presented without any intervening irrelevant stimuli or activities. Distributed practice is traditionally defined as practice that is broken up into 2 or more sessions, each separated by an interlude of minutes, hours, or days, or as single-session practice in which repetitions of target stimuli are separated by 1 or more nontarget stimuli. In neurorehabilitation, target stimulus repetitions are typically separated by many intervening nontarget stimuli, and limits of human endurance dictate that sessions be conducted over multiple days. Thus, no rehabilitation therapy conforms to traditional definitions of massed practice, and our research focused rather on whether therapeutic efficacy is optimized by more or less condensed treatment. Studies of CIMT for upper extremity paresis following stroke may have cultivated the notion that the case for large amounts of highly condensed practice has largely been proven. However, this hypothesis has never been tested.
Studies contrasting the relative impact of massed practice and distributed practice on learning rate and knowledge retention date back to 1885.6-10 Literally hundreds of investigations have been conducted involving a range of knowledge and skill types. With very few exceptions, distributed practice has been found to be superior to massed practice in its effects on knowledge retention, whether the training involved factual knowledge, simple motor skills, or more complex tasks involving acquisition of factual knowledge, motor skills, and the development of strategies. This superiority has come to be known as the spacing effect. A small number of studies have tested the relative value of distributing learning in normal subjects over long intervals, such as days, weeks, or months—epochs that might be germane to neurorehabilitation.11-17 In all, superior retention was achieved with more distributed practice. Several studies have demonstrated that delayed recall is superior with distributed practice in populations of brain-injured participants, including patients with the amnestic syndrome, 18 traumatic brain injury,19-21 and multiple sclerosis. 22 However, in all these studies, distributed practice involved repetition intervals of minutes, not days, and the learning tasks were constituted as tests of the principle, not part of a rehabilitation program. The testing of the distributed practice effect employing distribution over weeks in the context of a rehabilitation program, as described in this report, appears to be novel.
A number of psychological theories have been offered to explain the spacing effect,10,23-27 and there is good evidence of molecular mechanisms as well. 28 One group of theories (contextual variability theories) has a clear bearing on training distributed over days to weeks. One particular contextual variability theory—the component levels theory of Glenberg—has stood the test of time and received extensive empirical validation.25,26 Glenberg posited that at times of learning, 3 levels of information are encoded: substantive, structural context, and general context. Substantive information might consist of the semantic features of the stimulus or the procedural components of the skill. Structural context refers to the substance and arrangement of other stimuli in the training set (eg, other words in a word list to be memorized or other stimulus pairs in a native word–foreign word learning paradigm). It might also consist of parametric variations of a skill being trained (eg, shooting basketballs from different distances and different positions). General context refers to attributes of the training environment (eg, room, trainer, training equipment), affective state of the subject, travel entailed to the training test site, the subject’s familial situation at the time, people accompanying the subject to the training site, and so on. Glenberg posited that recall is aided to the extent that there are contextual commonalities between the time of training and the time of recall. Training multiple times at substantial intervals (less condensed practice) aids recall by increasing the probability that there will be elements of commonality, structural but particularly general, between the contexts at one or more training sessions and the context at time of retrieval—elements that will aid retrieval.
Greater distribution of training also allows for more intervening nights of sleep, during which both declarative and procedural memories are consolidated. CIMT likely engages both declarative and procedural memory mechanisms. Sleep is emerging as a major engine of neuroplasticity in general and memory consolidation in particular.29-31 Bell et al 32 found that intervening sleep and spacing appeared to make independent contributions to long-term retention.
The first hypothesis motivating this study was that, based on the theoretical rationale for the superiority of more distributed (less condensed) practice and the extensive empirical evidence from studies in normal subjects, more distributed practice would be more effective in brain injured subjects.
D-cycloserine
It is likely that 6 or more months postinjury, normal learning mechanisms (experience-dependent plasticity) are primarily responsible for recovery. Learning, whether declarative or procedural, represents alteration of neural connection strengths. This occurs through the processes underlying long-term potentiation (increase in neural connection strengths) and long-term depression (decrease in neural connection strengths). 33 The N-methyl-D-aspartate (NMDA) glutamate receptor, which gates a sodium-calcium channel, has been identified as a key component of the mechanisms underlying long-term potentiation. Normal learning processes mediated via the NMDA glutamate receptor can potentially be amplified by potentiation of NMDA glutamate receptor–mediated processes. D-cycloserine, a drug long used as an antibiotic in the treatment of tuberculosis, is a high-affinity partial agonist with moderate specificity at the glycine-B (strychnine insensitive) receptor of the NMDA-glutamate receptor complex.34,35 It is therefore capable of potentiating NMDA receptor channel activity without activating glycine-A (strychnine sensitive) receptors in the brainstem and spinal cord, which have inhibitory effects on neuronal firing. Extensive studies in normal animals (primarily rodents but also nonhuman primates) and animals with various lesions do suggest that d-cycloserine can enhance learning in a large variety of tasks. In studies of normal human subjects, d-cycloserine has been shown to potentiate declarative 36 and procedural memory formation 37 and to improve memory function in normal subjects treated with the anticholinergic drug scopolamine. 38
D-cycloserine has never been tested as an adjuvant to rehabilitation of stroke-related impairment. Our second hypothesis in the present trial was that d-cycloserine would be an effective adjuvant to CIMT for upper extremity paresis after stroke—by virtue of its mechanism of action, the extensive evidence of adjuvant effects on learning in animal studies, and its reported effects on declarative and procedural memory formation in human subjects.
We tested these 2 hypotheses in a prospective parallel-group randomized controlled trial employing a crossed design—d-cycloserine/placebo (double blinded) × more versus less condensed practice (single blinded)—in which all subjects received 60 hours of CIMT but those in the more condensed group received therapy 6 hours a day, 5 days a week, for 2 weeks while those in the less condensed group received therapy 2 hours a day, 3 days a week, for 10 weeks. This randomized controlled trial is best characterized as a phase IIa study: it was designed to determine the presence of an effect of the 2 interventions and to establish parameters for a power analysis for a subsequent larger trial. It was not expected that the results of this trial would achieve statistical significance.
Methods
Participants
Participants were recruited from inpatient and outpatient populations at the Malcom Randall VA Medical Center (Gainesville, Florida) and the Brooks Rehabilitation Hospital (Jacksonville, Florida), through newspaper articles and advertisements, and via contact with stroke support groups. The study was conducted at the VA Rehabilitation Research and Development Brain Rehabilitation Research Center at Malcom Randall VA and at outpatient clinics of the Brooks Rehabilitation Hospital. Twenty-four participants with moderate upper extremity paresis due to a stroke that had occurred at least 6 months prior were recruited to the study. Participants who had experienced 1 or more strokes on the same side of the brain were included unless there was a history of a clinical ischemic or hemorrhagic event affecting the other hemisphere or there was imaging evidence of more than a lacune or minor ischemic demyelination affecting the other hemisphere. Participants had to be between the ages of 18 and 80 years. There were no restrictions related to sex, ethnicity, or handedness.
Participants had to meet the following motor and functional criteria to enter the study:
Active motions of the wrist and hand: 20° of wrist extension from a relaxed flexed position, 10° of extension of any 2 digits at any joint, and 10° of thumb extension at either joint. All active motions had to be repeated 3 times within 1 minute.
Passive range of motion: 90° of flexion and abduction and 45° external and internal rotation at the shoulder, 45° elbow supination and pronation, elbow extension limited by no more than 30°, wrist extension to at least neutral position, and digit extension limited by no more than 30°. Participants were not required to exhibit any active shoulder or elbow motion.
Ability to sit independently for at least 2 minutes. Ability to ambulate was not required.
Motor Activity Log 39 score of 3 or less.
Exclusionary criteria included a history of a learning disorder, mental retardation, drug or alcohol abuse, more than very minor head trauma, subarachnoid hemorrhage, lobar cerebral hemorrhage, severe chronic obstructive pulmonary disease, cardiac dysrhythmias requiring medical treatment, serious medical illness, dementia or other neurodegenerative disease, multiple sclerosis, epilepsy, medically resistant depression prior to stroke, schizophrenia, severe visual impairment, pregnancy, breast feeding, and participation in CIMT within the prior 12 months. Serum creatinine documented within the prior 6 months had to be ≤1.5 mg/dL (d-cycloserine is renally cleared). Aphasia and other cognitive deficits did not preclude inclusion so long as subjects were sufficiently sentient to be able to understand the potential risks and benefits of the study, personally provide informed consent, and understand and cooperate with the treatment.
This study was approved and monitored by the Investigational Review Board of the University of Florida Health Science Center. All participants provided informed consent.
Randomization and Drug Treatment
This was a prospective randomized parallel-group study employing a 2 × 2 design: d-cycloserine/placebo × more/less condensed therapy. Subjects were randomized to 1 of the 4 treatment groups by the research pharmacist. Double blinding of drug treatment was maintained until all participants had undergone their final outcome evaluation. Because therapists could not be blinded to distribution of treatment, all outcome measurements were conducted by blinded evaluators who had no other role in the trial.
Participants took d-cycloserine (50 mg) by mouth on therapy days only, 4 hours before beginning therapy, to ensure maximal blood and brain levels of the drug over the treatment session. D-cycloserine and identical placebo containing capsules were prepared by a compounding pharmacy. The drug was used under an Food and Drug Administration investigatory drug exemption. Compliance was ensured through pill counts conducted by study therapists.
All participants underwent 60 hours of CIMT. More condensed treatment consisted of 6 hours per day for 10 consecutive weekdays. Less condensed treatment consisted of 2 hours per day, 3 days a week (Monday, Wednesday, Friday), for 10 consecutive weeks.
CIMT40,41 was conducted by an occupational therapist, physical therapist, or physical therapy assistant. Short breaks in treatment were provided in all groups when needed. The actual time in therapy was recorded for each session. Total time in therapy, through the course of the trial, was documented to be similar for all groups: more condensed, placebo, 59.2 ± 1.8 hours (range, 55.5-60); more condensed, d-cycloserine, 60.5 ± 0.1 hours (range, 60-60.05); less condensed, placebo, 58.6 ± 2.7 hours (range, 54-61); less condensed, d-cycloserine, 59.3 ± 1.9 hours (range, 55.5-60.33). Participants contracted to wear the mitt on the unaffected hand 90% of their total waking hours throughout the course of treatment, regardless of whether it was a treatment day, except when use of that hand was required for considerations of safety, hygiene, and specifically agreed on activities, such as driving to and from the lab. A home diary was used to document daily compliance in wearing the mitt. Unfortunately, on examination, the diaries were insufficiently complete to enable quantitative assessment of compliance. All subjects, regardless of group assignment, went home following treatment. Following completion of CIMT, subjects were contacted by research therapists only to schedule follow-up testing.
CIMT included massed repetition, task practice with shaping (approach of a desired motor or behavioral objective by small steps and successive approximations), and intensive timed activities. The daily interventions were designed around a menu of functional activities that incorporated variations of strength, endurance, coordination, dexterity, and range of motion. Interest inventories and role checklists were used to incorporate the participant’s unique personality traits and activity interests into the menu. The participants gave daily input regarding the activities that they performed in the laboratory and when they returned home. In this way, a variety of purposeful and meaningful activities were incorporated with attention to unique limitations. Encouragement was provided continuously. Therapists sought to prevent participants from failing by providing assistance as necessary or changing the task. If an activity was strongly disliked or appeared to be too difficult, it was eliminated from the menu for the next day.
Outcome Measures
The primary outcome measure was time on the Wolf Motor Function Test (WMFT) for the affected limb.39,42 Secondary outcome measures included grip strength, the Fugl-Meyer Motor Scale–Upper Extremity, 43 the Motor Activity Log–quality, 39 the Stroke Impact Scale (version 2.0; participation—ability to participate in activities that are meaningful and give purpose to life; recovery—analog scale rating of degree of recovery from stroke), 44 Caregiver Strain Index, 45 and the Geriatric Depression Scale. 46 Grip strength was measured 3 times during each administration of the WMFT and the 3 values averaged. The WMFT, Motor Activity Log, and Fugl-Meyer were obtained at baseline, end of treatment, and 3 months after completion of treatment. At each assessment, the WMFT was conducted twice and the times averaged. The Stroke Impact Scale and the Caregiver Strain Index were obtained at baseline and 3 months after completion of treatment. At the end of treatment, participants were asked to guess whether they had been receiving drug or placebo.
Statistical Analysis
Statistical analyses were conducted with SAS 9.2 (SAS Institute, Cary, North Carolina). Means ± SDs were computed for continuous baseline variables, and outcomes and percentages were computed for binary variables for the 4 treatment groups. The balance of baseline characteristics for the 2 group comparisons (d-cycloserine vs placebo; more vs less condensed) was compared with a t test for continuous variables and χ2 or Fisher’s exact tests for categorical variables.
A general linear model was used to assess the impact of drug (d-cycloserine vs placebo) and distribution of practice group (more versus less condensed) and their interaction on WMFT score at 3 months after the end of treatment, controlling for baseline WMFT score. A paired t test was used to compare baseline with 3-month posttreatment WMFT scores for the entire group. Three months was chosen as the outcome point of interest because the impact of the distribution of practice effect theoretically affects retention of knowledge and skill, not acquisition of knowledge and skill. An intention-to-treat analysis was employed. Missing values were handled through last observation carried forward. Effect sizes, denoted as the semipartial statistic
Results
Of the 24 participants who entered the trial, 22 completed it (Figure 1). Baseline values for the comparison groups are detailed in Tables 1 and 2. No between-group differences were statistically significant. The participants in the more condensed group were, on average. somewhat more impaired (WMFT, 40.0 ± 33.1) than those in the less condensed group (25.2 ± 31.1), but the difference failed to achieve significance. Likewise, the participants in the placebo group were, on average, more severely impaired (WMFT, 38.6 ± 32.2) than those in the d-cycloserine group (26.7 ± 32.7), but again the difference was not significant.
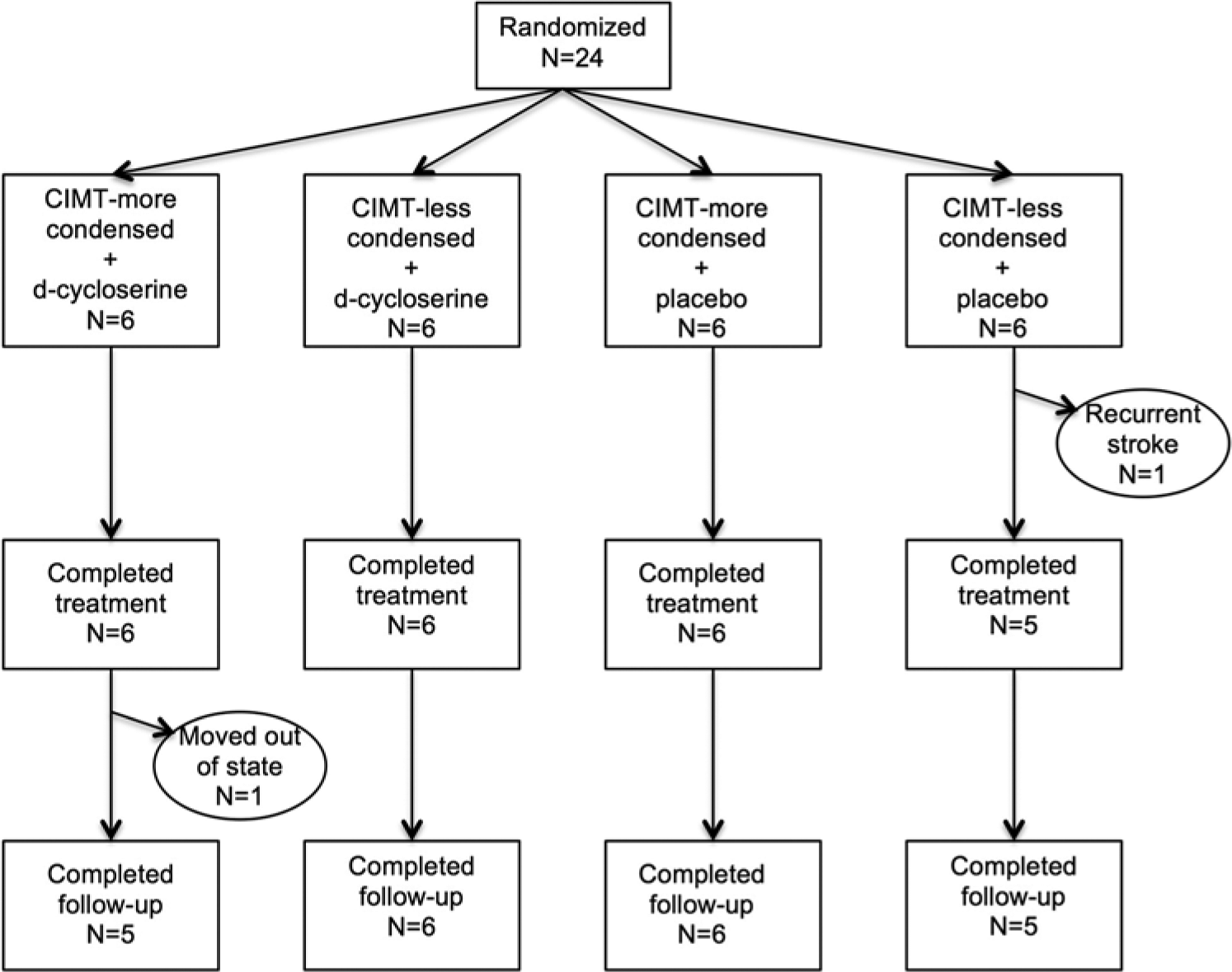
CONSORT diagram.
Baseline Values: More Versus Less Condensed a .
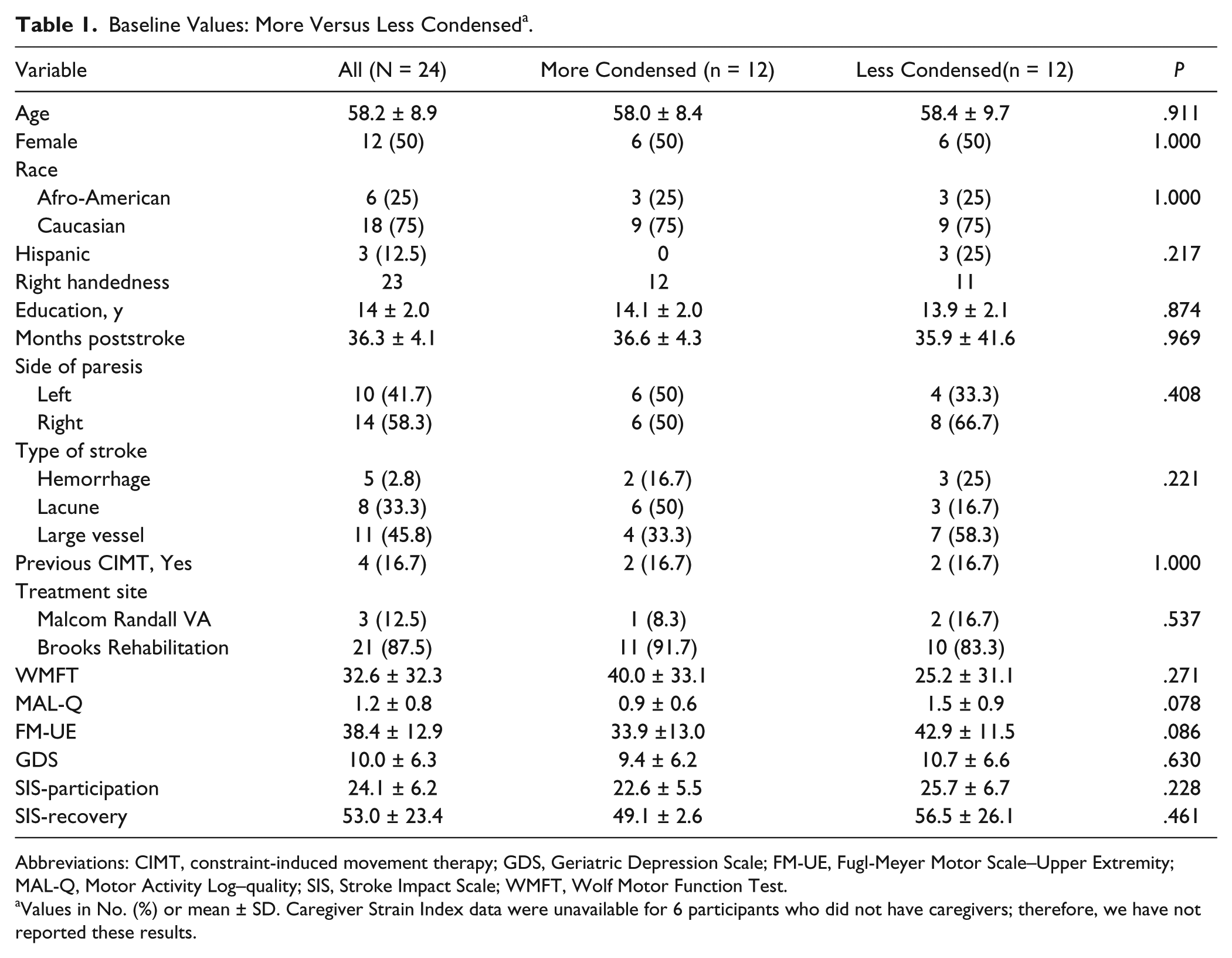
Abbreviations: CIMT, constraint-induced movement therapy; GDS, Geriatric Depression Scale; FM-UE, Fugl-Meyer Motor Scale–Upper Extremity; MAL-Q, Motor Activity Log–quality; SIS, Stroke Impact Scale; WMFT, Wolf Motor Function Test.
Values in No. (%) or mean ± SD. Caregiver Strain Index data were unavailable for 6 participants who did not have caregivers; therefore, we have not reported these results.
Baseline Values: Placebo Versus D-cycloserine a .
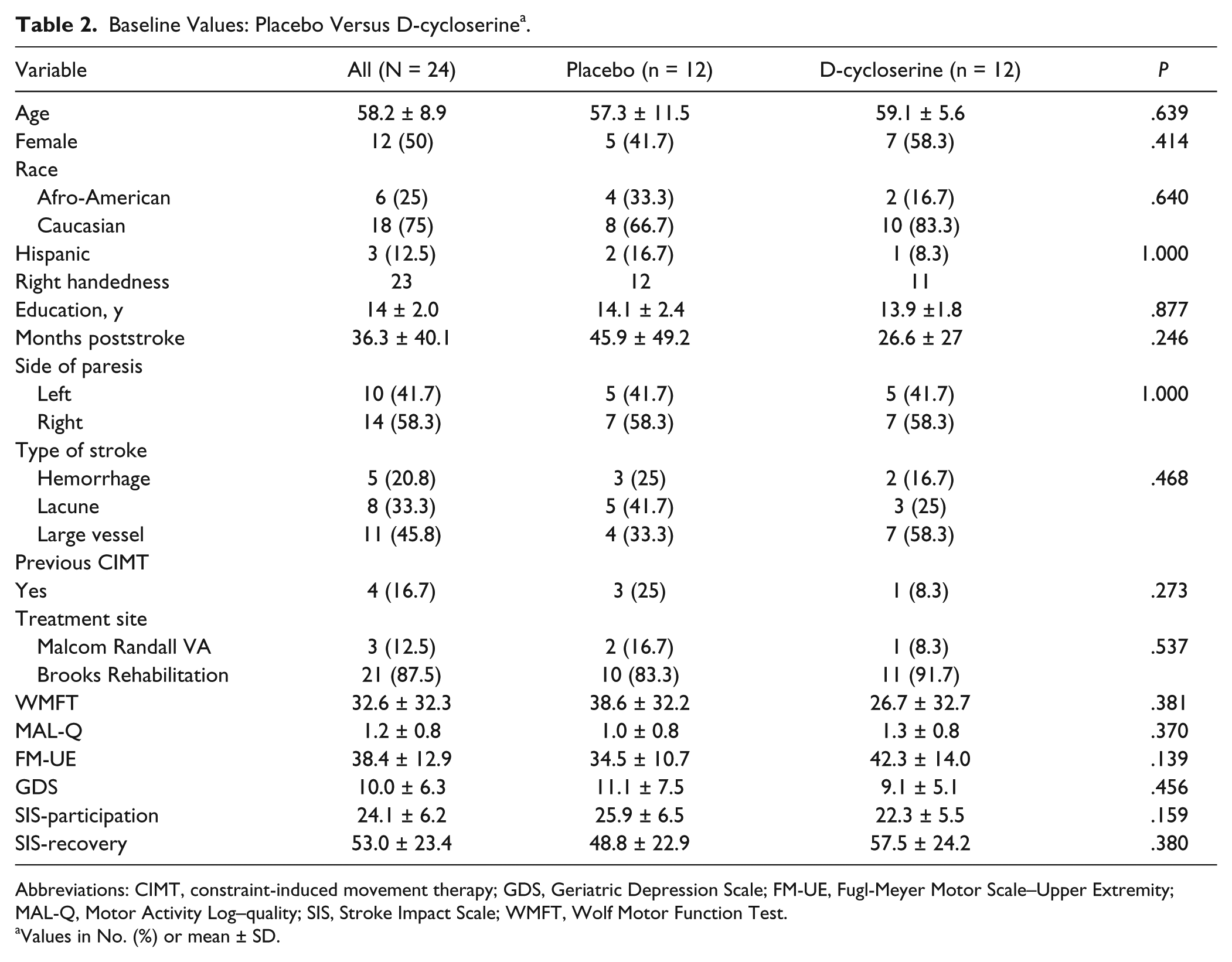
Abbreviations: CIMT, constraint-induced movement therapy; GDS, Geriatric Depression Scale; FM-UE, Fugl-Meyer Motor Scale–Upper Extremity; MAL-Q, Motor Activity Log–quality; SIS, Stroke Impact Scale; WMFT, Wolf Motor Function Test.
Values in No. (%) or mean ± SD.
The results of the analysis of treatment effects for primary and secondary outcome measures are detailed in Tables 3 and 4. The general linear model demonstrated no significant effects of training distribution group, d-cycloserine, or their interaction on WMFT score at 3 months posttreatment, controlling for baseline WMFT (Table 5). There was a robust benefit from CIMT, as reflected in substantial gains in all subgroups shown in Table 3 and 4, and a decrease in WMFT scores of 11.7 ± 15.7 (P = .0013;
Outcomes: More Versus Less Condensed.
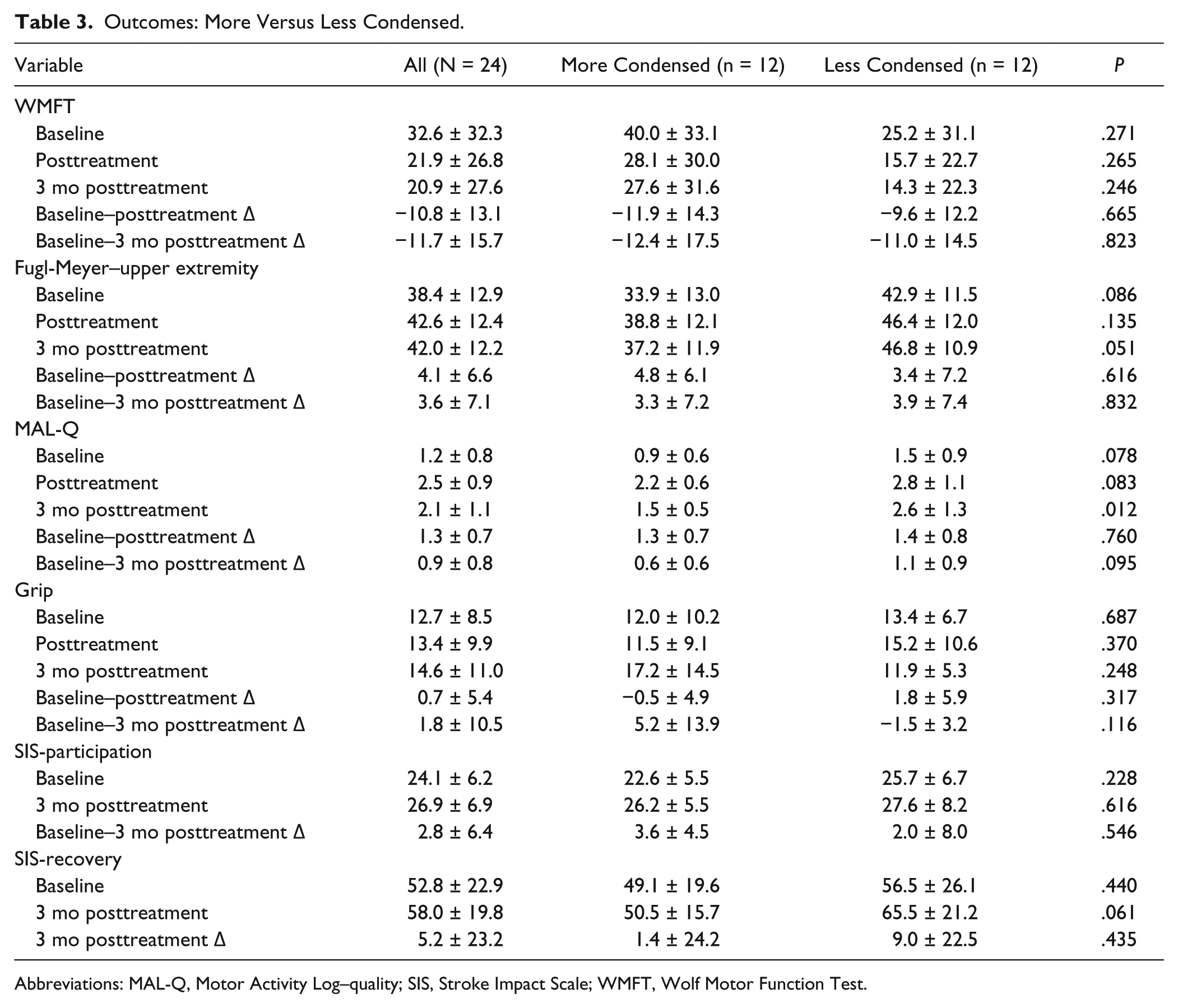
Abbreviations: MAL-Q, Motor Activity Log–quality; SIS, Stroke Impact Scale; WMFT, Wolf Motor Function Test.
Outcomes: Placebo Versus D-cycloserine.
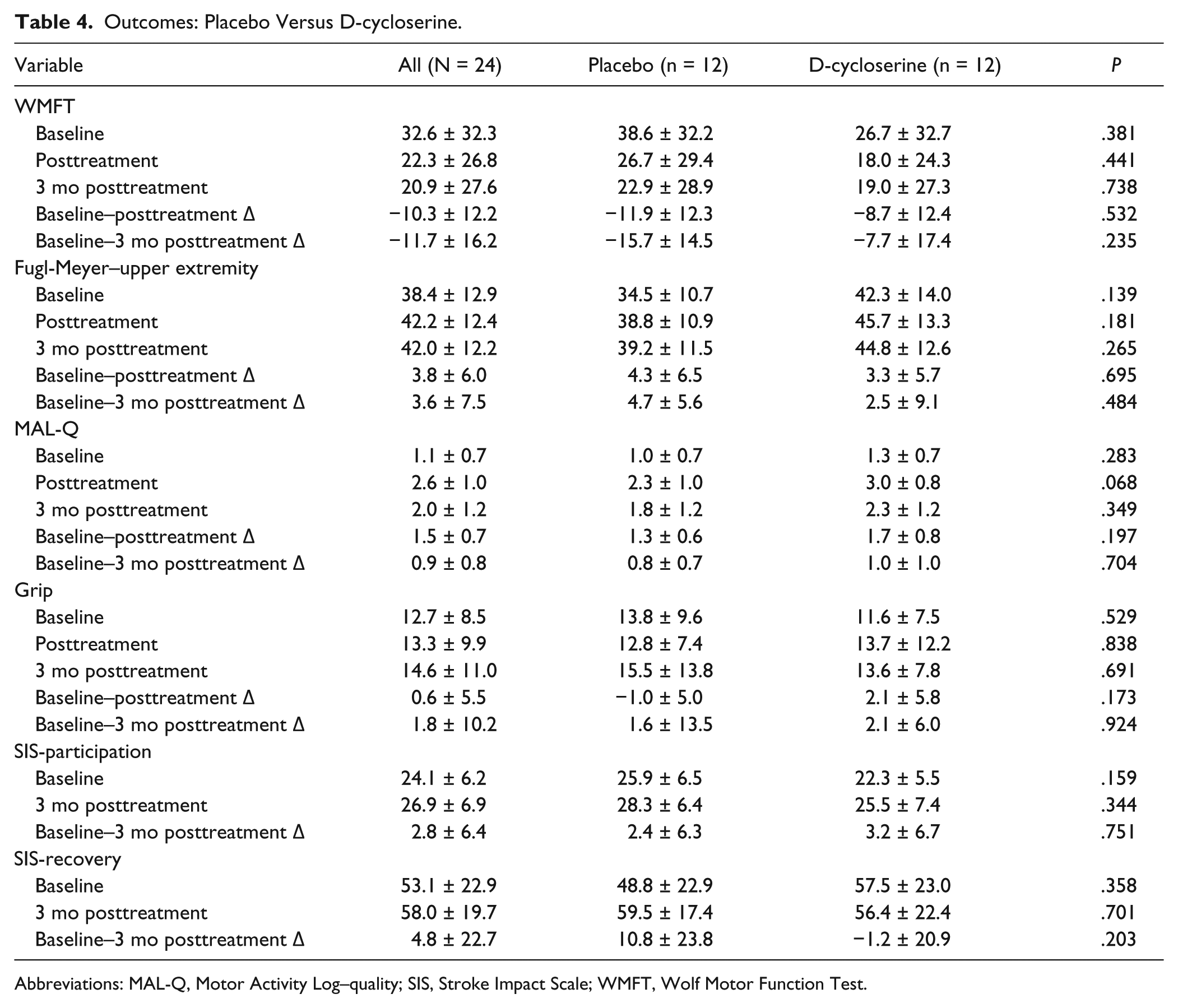
Abbreviations: MAL-Q, Motor Activity Log–quality; SIS, Stroke Impact Scale; WMFT, Wolf Motor Function Test.
Regression Analysis: Wolf Motor Function Test Results at 3 Months Posttreatment.
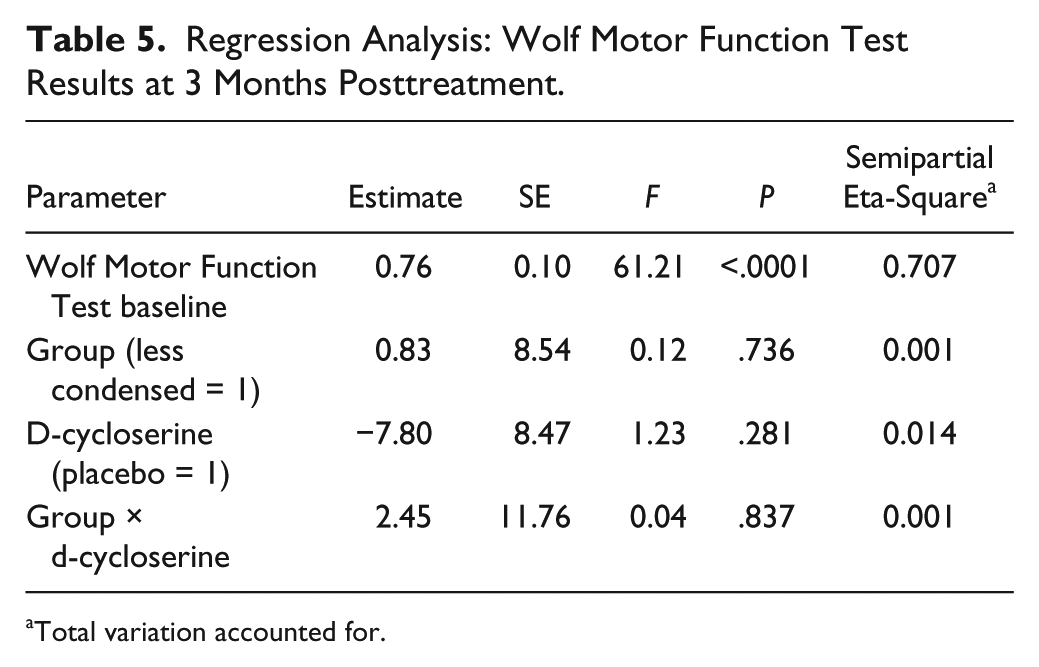
Total variation accounted for.
At the end of treatment, participants were asked whether they thought they had been receiving drug or placebo. Of the 12 participants taking d-cycloserine, 10 thought that they were receiving placebo, and all 12 participants taking placebo thought that they were receiving placebo.
Safety
One participant experienced a stroke during the trial (not during actual treatment). There were no adverse events associated with d-cycloserine. One participant in the more condensed training group experienced a small skin tear and a sore elbow.
Discussion
We posited that less condensed training would yield greater retention of results of treatment 3 months later because of the greater likelihood of commonality between treatment conditions and test conditions and the greater number of intervening nights of sleep. We also posited that d-cycloserine would increase learning because of its potentiation of sodium-calcium influx through NMDA-glutamate voltage-gated sodium-calcium channels, which are crucial to learning. Neither hypothesis was supported by the study results. Yet, our results provide no support for the concept that CIMT must be concentrated into 2 weeks of training. The results of distributing the 60 hours of treatment over 10 weeks were the same as when the therapy was provided over 2 weeks. D-cycloserine proved to be safe.
Potential Reasons for Treatment Failure
Both the adjuvants to CIMT employed in this trial could have failed if participants failed to improve with CIMT because both adjuvants potentially affect only the neuroplastic response of the central nervous system. However, our results demonstrate that there was a robust benefit from CIMT, as reflected in substantial gains on all measures and a substantial, statistically significant decline in WMFT score (effect size, 0.75). Thus, there was a substantial neuroplastic response susceptible to influence by the adjuvants.
It is possible that the adjuvants tested here could achieve a small but significant impact in a less severely impaired population. Mean baseline WMFT score in this study was 32.6 ± 32.3, and baseline upper extremity Fugl-Meyer score was 38.4 ± 12.9, reflecting moderate to severe impairment for a CIMT-eligible population.
Both adjuvants could have failed if the neuroplastic response elicited by CIMT was approaching ceiling. There is increasing evidence that paresis caused by stroke is, in nearly all cases, primarily due to a white matter lesion. 47 The loss of most corticospinal and corticobulbar fibers projecting from the hemisphere to the brainstem may impose strong limits on just how much recovery can be achieved by behavioral treatments, however intensive and extended, as perhaps best reflected in the results of recent phase III trials of intensive therapy for upper extremity paresis and gait impairment following stroke.1-4
The apparent failure of both adjuvants to augment acquisition and retention of functional gains achieved during therapy could reflect inadequate statistical power. This goes to the heart of a methodological issue that poses a major challenge for neurorehabilitation science. The actual cost of delivering treatment over a less condensed schedule or coadministering a drug such as d-cycloserine would be very small and could readily be defended on the basis of small marginal gains. However, to achieve 80% power to detect the observed gains in WMFT attained by administration of d-cycloserine (−11.9 vs −8.7) and less condensed training (−11.9 vs −9.6), a randomized controlled trial identical in design to ours would require 474 and 1038 participants, respectively.
Greater distribution of treatment over time could have failed to yield a greater retention of benefit for a number of reasons. Our interval from posttreatment to retention test (3 months) may have been too short to allow for differential attenuation of gains in the more condensed treatment group. The effects of more distributed learning may have been too small in a clinical population to be measurable, given all the other potential sources of variance. Less condensed treatment could have been associated with reduced concentration on therapy, loss of continuity, reduced involvement by caregivers, and loss of motivation. However, these effects should have been reflected in poorer acquisition by this group, something not apparent in our data. There is evidence that the benefits of distributed learning are relatively less in older adults than in young adults. 48 Gains in function achieved with CIMT substantially involve procedural memory acquisition. It is possible that contextual variability effects on procedural memory acquisition (in contrast to declarative memory acquisition) are too small to be detected in a small study such as ours or, conceivably, too small to be relevant to any rehabilitation that predominantly involves procedural memory acquisition. Finally, it is possible that distributing therapy over a longer period was not a sufficiently potent factor in establishing commonality between circumstances of treatment and retention. Conducting the treatment in the participants’ homes might be more effective in achieving this objective. This may, for example, have been an important factor in the LEAPS trial, in which the participants, though encouraged to walk outside formal therapy, actually received impairment-based therapy delivered entirely in their homes that included no walking. They did as well as those receiving functional task practice in the clinic, which included body weight–supported treadmill training and overground walking. 1
D-cycloserine treatment could have failed to yield greater learning and retention for several reasons. First, learning processes engaged by CIMT may already have been optimal at the molecular level and may not have been susceptible to improvement by a drug. Second, the dose of d-cycloserine that we used may not have been optimal, despite our efforts to pick the best dose based on extant animal studies and human subject trials. However, a recent study suggests that a higher dose (250 mg) may be required to facilitate declarative memory acquisition, 36 even as lower doses (eg, 100 mg) may suffice for enhancement of procedural memory formation. 37 Finally, repeated drug exposure may have led to tachyphylaxis, particularly in the more condensed treatment group, as has been reported in animal studies.49,50 However, given the 10-hour half-life of d-cycloserine (http://www.medicines.org.uk/emc/medicine/20415/SPC), one would expect that 80% of the drug would be eliminated by the next daily dose. Furthermore, we did not observe a differential response to the drug in the more and less condensed groups, even though those in the less condensed group also received more total drug.
Finally, because of the shortcomings of home diaries, our study could not quantify the potential effects of ambient therapy—that is, therapy implicit in activity occurring outside treatment sessions. Ambient therapy could have made a major contribution to outcome,5,51 particularly in less condensed therapy groups. It is likely that quantitative methods employing such devices as accelerometers will be needed to adequately measure ambient therapy in trials of upper extremity rehabilitation.
Trial Design
Because of the theoretical orthogonality of distribution of practice and d-cycloserine effects, we set up a 2 × 2 trial design in the interests of greater efficiency: we could perform 2 parallel-group randomized controlled trials at the same time, each involving 24 subjects. Our a priori assumption of orthogonality was borne out by the lack of a significant interaction effect. Notwithstanding the small cell size, the use of covariate adaptive randomization could have yielded a better balance of severity across groups.
Conclusions
In this pilot randomized controlled trial, we found no impact of greater distribution of CIMT over time or the administration of d-cycloserine on the retention of functional improvement achieved through therapy. However, we also found that there was no advantage to the massed practice of traditional CIMT programs.
Footnotes
Acknowledgements
We are grateful to Matt Morrow, the study pharmacist; Floris Singletary, our clinical coordinator at Brooks Rehabilitation Hospital; Carolyn Hanson, our clinical coordinator at the Malcom Randall VA Medical Center; and the therapists who made this study possible—Molly Dunn, Brooke Hoisington, and Margo Fitch.
Authors’ Note
Study data can be obtained by contacting the principal investigator.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Supported by Department of Veterans Affairs Rehabilitation R&D grant B6346R: Improving stroke rehabilitation: spacing effect and d-cycloserine (Clinicaltrials.gov NCT00711568).
