Abstract
Background. Creating the visual illusion of touch can improve tactile perception in healthy subjects. Objective. We were interested in seeing if creating the illusion of touch in an insensate area could improve sensation in that area. Methods. Fourteen people with chronic numbness participated in a randomized crossover experiment. The 4 conditions were the following: (a) stimulation over the unaffected limb with mirror visual feedback (experimental condition), (b) stimulation over the affected limb with mirror visual feedback, (c) stimulation over the unaffected limb without mirror visual feedback, and (d) stimulation over the affected limb without mirror visual feedback. Participants were assessed before and after each condition using the Ten-Test and mechanical detection thresholds. Data were analyzed using linear mixed models. Result. Only the experimental condition produced a change in the Ten-Test (mean difference = −1.1; 95% confidence interval = −1.8 to −0.4; P = .003), corresponding to a 24% improvement in sensation. No differences were observed for any condition in mechanical detection thresholds. Conclusion. Creating the illusion of touch may improve sensory function in areas of chronic numbness. This preliminary finding adds to the growing body of evidence supporting the use of techniques that directly target cortical function in people with peripheral nerve injury.
Peripheral nerve injury (PNI) is associated with changes along the neuraxis, including peripheral nerve degeneration and regeneration as well as structural and functional changes within the spinal cord and brain.1,2 While ongoing tactile deficit after PNI is clearly attributable to incomplete peripheral repair, 3 cortical changes have also been shown to correlate with tactile function and may contribute to poor recovery. 3 Consistent with this, treatments that explicitly target the brain appear to reduce tactile deficit after PNI. 1
One way to facilitate cortical aspects of tactile function is to exploit the cross-modal interaction between vision and touch. 1 Similar cortical areas are activated with touch of the hand and with watching a video of the hand being touched. 4 Furthermore, corresponding visual input enhances tactile sensitivity in healthy volunteers, 5 as does creating the illusion of touch using mirrors. 6 Here we report a randomized repeated-measures crossover experiment in which we assessed tactile function in people with sensory loss before and after creating the illusion of touch in the insensate area via the manipulation of visuotactile input.
Fourteen people with persistent sensory loss after PNI participated. Most injuries were unforeseen complications from surgery and no nerve repair had been undertaken (see Supplemental Table 1, available online at http://nnr.sagepub.com/content/by/supplemental-data). After signing consent, participants completed 4 different conditions, in random order, separated by at least 2 days (mean = 3.2 ± 1.7 days). Randomization was concealed and counterbalanced. All procedures were approved by the institutional ethics committee.
Participants wore clothes that exposed the testing area and removed all jewelry. The area of sensory loss was mapped and the border and central point marked with a pen. The corresponding area on the unaffected limb was identically marked. Distances from bony landmarks were noted for standardization, and participants were marked-up in an identical fashion before each session based on these measured distances.
The experimental condition involved placing a mobile mirror (lower-limb) or a mirror-box (upper-limb) in line with the participant’s parasagittal axis. The participants’ limbs were placed either side of the mirror with the reflective surface facing the unaffected side. The affected limb was therefore hidden from view, and the reflection of the unaffected limb appeared spatially aligned with the location of the affected one (Figure 1A). The marked area on the unaffected side was then stroked using a cotton swab, brushed with a paintbrush, and gently pricked with a medi-pin, with each type of stimulation lasting 2 minutes. Participants watched the stimulation in the mirror, such that they had the visual illusion of being touched in the numb area while simultaneously receiving tactile input from an area of normal sensitivity.
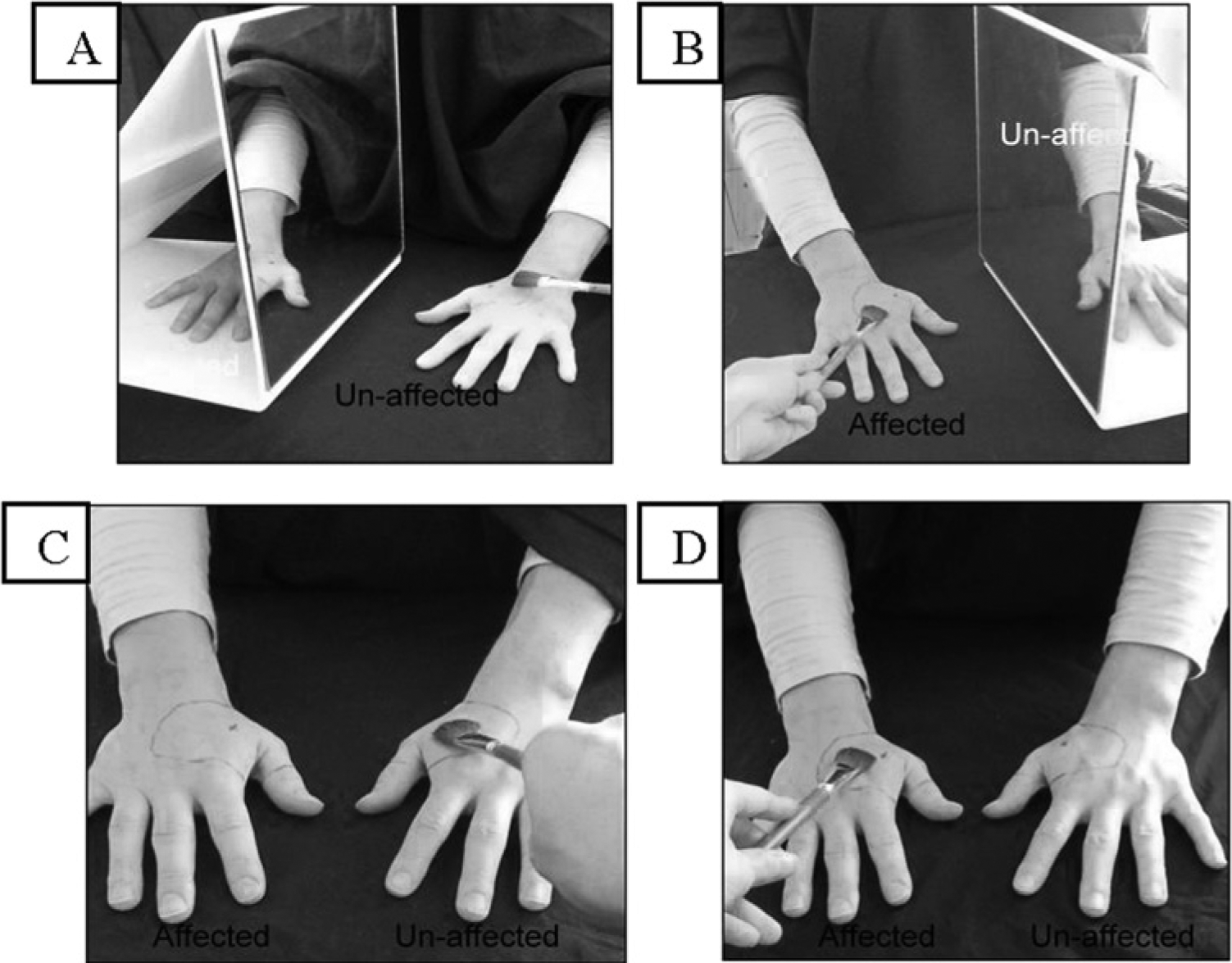
Experimental set up.
The 3 control conditions were the following: sensory stimulation over the affected area with mirror visual feedback while the subject attended to the reflected image, sensory stimulation over the unaffected area with no mirror, and sensory stimulation over the affected area with no mirror. In both nonmirror conditions, the participant attended directly to the area being stimulated. The stimulation protocol was standardized so the amount and type of stimulation was identical across all conditions.
Sensation in the numb area was assessed with the Ten-Test 7 and mechanical detection threshold (MDT; using Semmes-Weinstein Monofilaments). For the Ten-Test, both the numb area and the corresponding area on the unaffected limb were stroked with a cotton swab while vision was occluded. The participant rated the sensation on the numb area, in comparison to the unaffected side, on an 11-point numerical rating scale, where 0 = no sensation and 10 = same as the other side (normal). Both outcomes were assessed prior to and immediately after completion of each treatment condition. All measurement was undertaken by the same independent investigator who was blinded to condition. At the completion of each condition, participants were informed by the treating therapist not to indicate to the assessor the treatment received at that or any previous session.
One participant did not attend all sessions and the participant’s data were excluded. For the remaining 13 participants, a linear mixed-model analysis compared Ten-Test scores across Time (pre–post) and Condition. There was a significant interaction between Time and Condition (P = .015) and a pre–post difference for the experimental condition only (P = .003; mean difference = −1.1; 95% confidence interval = −1.8 to −0.4). This corresponded to a 24% increase in sensation (95% confidence interval = 40% to 8%). There was no pre–post difference for any other condition (P > .05). An equivalent analysis on the MDT data was not significant. No carryover or order effects were detected for either outcome measure (P > .05). Full details of the outcomes measures for each condition can be found in Table 1.
Mean Change Scores in Outcome for Each Condition (Pretreatment − Posttreatment).
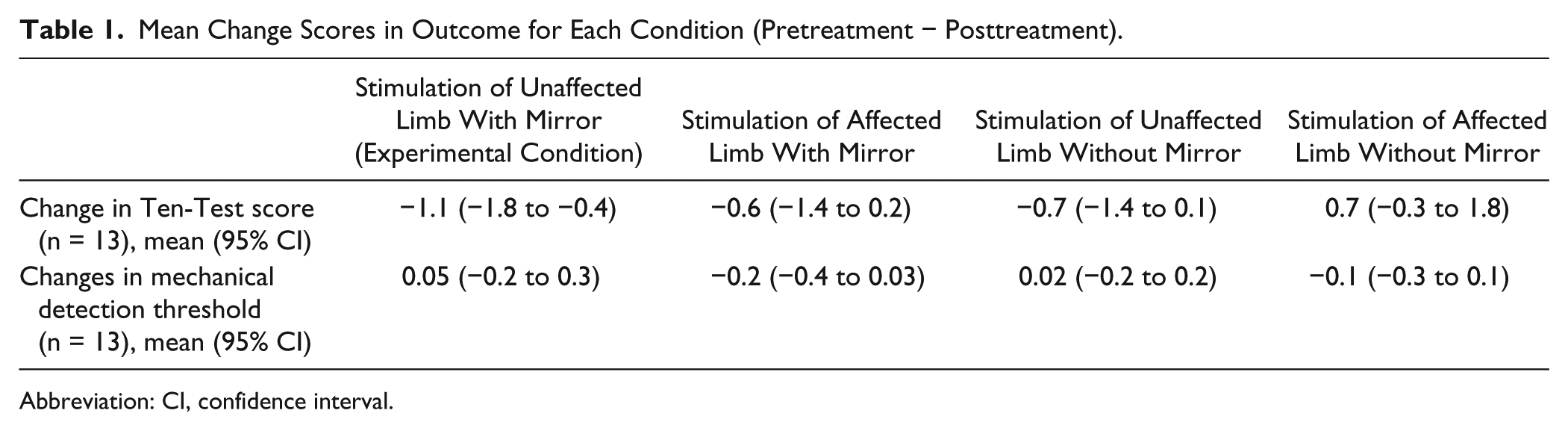
Abbreviation: CI, confidence interval.
Our results show that a single session of illusory touch improved sensation in an area of persistent tactile deficit secondary to PNI. That only the illusory condition improved sensation strongly suggests that the improvement is mediated in the central nervous system, most likely the brain. Given that prolonged sensory deprivation influences the cortical representation of the affected area,1,2 and that cortical reorganization can occur rapidly with sensory manipulation, 8 one might predict that the visuotactile illusion used here had a direct effect on the cortical representation of the affected area by enhancing the nontactile contributions to the perception of touch.
We assessed sensation immediately after the treatment and would not predict a sustained improvement from one session; additionally we only saw a modest improvement in sensory function. However, an accepted tenet of neuroplasticity is that repeated training has a cumulative effect on cortical function and that it is possible that repeated training might deliver larger and more sustainable benefits. Moreover, other innovative approaches to the management of cortical changes in people with PNI such as audio-tactile retraining 9 and temporary cutaneous anesthesia of adjacent areas 10 do show potentially sustainable benefits with long-term application. We contend that the current results suggest that the investigation of repeated training using illusory touch is warranted, both in the early period after nerve repair, before sensation has begun to return, as suggested by Lundborg and Rosén, 1 as well as in those with persistent sensory loss. Clearly, further research is needed to corroborate the current findings and explore whether repeated training leads to meaningful, long-term improvements.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
