Abstract
Introduction
Stroke is the leading cause of long-term disability. Cortical reorganization occurs with functional recovery, and physiological responsiveness is altered in the peri-infarct and ipsilateral areas after stroke. 1 These changes in the physiological responsiveness of the brain network accompany changes in neuronal excitability. 2 Consequently, inducing an excitatory condition in the peri-infarct cortex might play a crucial role in functional recovery after a stroke. Upregulating the peri-infarct cortical excitability using magnetic or electrical stimulation to enhance functional recovery after a stroke is being investigated in clinical trials. 3
Initially, stroke induces hypoexcitability in the peri-infarct motor cortex, stemming from increased tonic γ-aminobutyric acid (GABA) activity on neurons. 4 In an animal model, it was demonstrated that tonic neuronal inhibition mediated by extrasynaptic GABAA receptors is increased in the peri-infarct zone. 5 However, the changes in neuronal excitability over time mediated by GABA receptors, which correlate with functional recovery, are not well understood in human stroke patients. In cases with good functional recovery, there is initially reduced activation in the ipsilateral brain areas and transfer of activation to the contralateral cortex, which subsequently returns to the peri-infarct and ipsilateral networks. 1 In contrast, continuously increased cortical activity on the side contralateral to the stroke is correlated with reduced functional recovery. 6 To understand the mechanism of physiological plasticity during the recovery phase of ischemic stroke better, a longitudinal study to assess the changes in cerebral GABA activity is needed.
This study examined the mechanism of neuroplasticity during the recovery phase of ischemic stroke by assessing the changes in cerebral GABA activity in the cortex of patients with stroke using [18F]flumazenil ([18F]FMZ) positron emission tomography (PET). We hypothesized that functional recovery is related to a decrease in GABA activity in the ipsilateral cortex.
Methods
Participants
Patients with unilateral ischemic stroke were recruited from the Stroke Unit at Seoul National University Bundang Hospital in Korea. The inclusion criteria were patients with stroke within 1 month and with unilateral subcortical infarct confirmed by brain magnetic resonance imaging (MRI). We excluded patients who had multiple or bilateral lesions, cerebellar or pons lesions, recurrent stroke, or a history of seizure and neuropsychological deficit before stroke. In all, 10 patients (7 women, 3 men; age, mean ± standard deviation [SD], = 65.2 ± 10.5 years) participated in the study; 15 healthy participants (10 women, 5 men; age, mean ± SD, = 57.5 ± 5.7 years), whose MRI showed no structural lesion were recruited from the community as healthy controls (HCs). Table 1 lists the gender, age, time after stroke, and location and size of the infarction at the time of the study as determined by MRI. Lesion volumes were measured from diffusion-weighted imaging using Analyze, version 10.0 (Analyze Direct, Overland Park, KS).
Raw Patient Data on Paretic Hand Function.
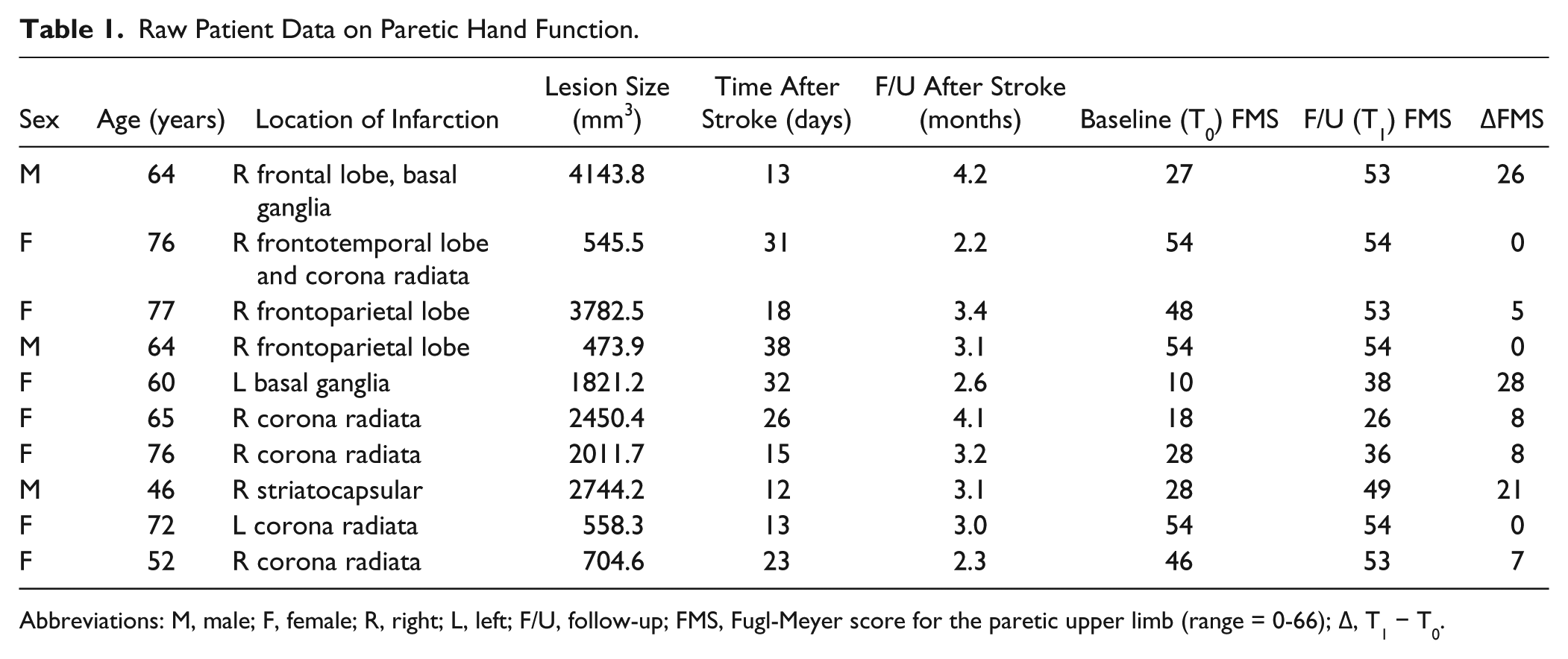
Abbreviations: M, male; F, female; R, right; L, left; F/U, follow-up; FMS, Fugl-Meyer score for the paretic upper limb (range = 0-66); Δ, T1 − T0.
Patients underwent PET within 1 month of a first-ever subcortical stroke and again at 3 months. This study complied with the Declaration of Helsinki, and the protocol was approved by the institutional review board of Seoul National University Bundang Hospital.
Measurements
This was a longitudinal study with observations made at 1 (T0) and 3 (T1) months after stroke onset. Regional GABAergic activity and upper-extremity motor function were assessed longitudinally, and the values were compared with those of age-matched controls (Figure 1).
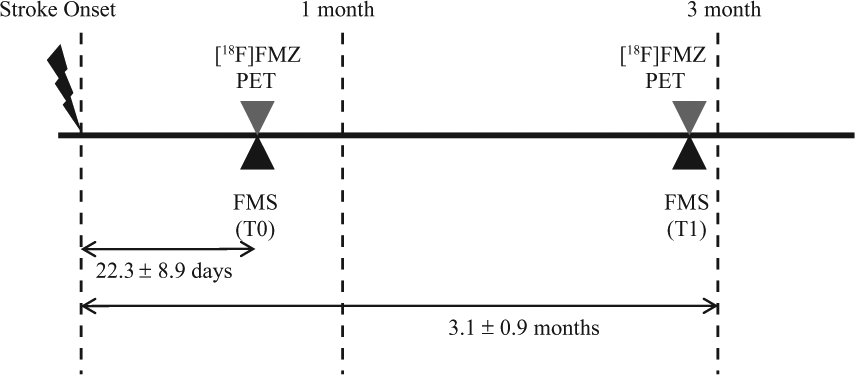
Experimental design.
All patients were assessed for clinical stroke severity and underwent physical and neurological examinations in the acute poststroke phase. Stroke-related neurological disability was evaluated using the National Institutes of Health Stroke Scale. The degree of handicap was assessed with the modified Rankin scale, and upper-limb motor function was evaluated using the Fugl-Meyer score (FMS), 7 which examines the presence of synergistic and isolated movement patterns and grasping ability of the upper limb (0 = lowest score; 66 = highest score). 7 Global function was evaluated using the modified Barthel index, which assesses independence in activities associated with daily living (0 = lowest score; 100 = highest score). 8 Two evaluators were trained and tested to ensure the reliability of outcome measures.
[18F]FMZ PET Acquisition
[18F]FMZ studies were conducted 1 (T0) and 3 (T1) months post–stroke onset to assess cortical GABAA receptor availability in patients with unilateral ischemic stroke. The mean interval between the onset of infarction and the first [18F]FMZ PET was 22.3 ± 8.9 days. All participants were free of any medication that might affect the central nervous system, and no specific preprocess was requested. [18F]-FMZ was synthesized using our reported method, 9 with high radiochemical purity (>99%) and specific radioactivity. [18F]FMZ PET images were obtained with a Discovery VCT PET/computed tomography (CT) scanner (GE Medical Systems, Milwaukee, WI). At 20 minutes after injection of 259 to 370 MBq of [18F]FMZ intravenously, a 20-minute emission image was obtained in static mode. The optimal static time was based on work by Mishina et al. 10 PET images were reconstructed with the built-in-advance software using an ordered-subset expectation-maximization algorithm with 47 continuous, 256 × 256, 3.75-mm-thick slices. Reconstructed images were corrected for attenuation, based on CT, and for scatter.
Data Analysis
All scan data and parametric images were processed using statistical parametric mapping software (SPM2; The Wellcome Department of Cognitive Neurology, Institute of Neurology, University College London, UK) implemented in MATLAB 7.6 (MathWorks, Natick, MA). First, all images from the participants were flipped, so that the affected hemispheres appeared on the left side and were spatially normalized to the study-specific template (voxel size = 2 × 2 × 2 mm3). For quantitative assessments, receptor availability on [18F]FMZ PET was calculated as the regional uptake ratio, in which the regional uptake is relative to the activity of the pons, where receptor sites are negligible. The uptake in the pons was extracted from each [18F]FMZ PET image by applying the standard region of interest for the pons after spatial normalization. Finally, [18F]FMZ ratio images were smoothed with a 12-mm (full-width at half-maximum) isotropic Gaussian kernel. 11
First, significant differences in receptor availability in the 1- and 3-month PET were compared with those of HCs using general linear models in a voxel-based manner. To exclude any potential effect of age on receptor binding, this variable was treated as a covariate of no interest. Second, the relationship between receptor availability at 1 month and functional performance was evaluated.
Results
Regional [18F]-FMZ Binding
The receptor availability in patients seen on [18F]FMZ PET at T0 was decreased significantly in the ipsilesional thalamus and bilateral subcortical white matter in the parietal cortex in comparison with HCs, whereas the receptor availability was increased in the ipsilesional cortical area (supramarginal cortex, BA 40) and contralateral cerebellum (Figure 2A). The findings were similar at T1, with a more widespread reduction in GABAA receptors in the cortical and subcortical white matter in the contralateral cerebellum in patients. The greater receptor availability in the contralateral cerebellum persisted, but to a lesser extent, and the higher perilesional receptor availability was no longer significant at T1 (Figure 2B).
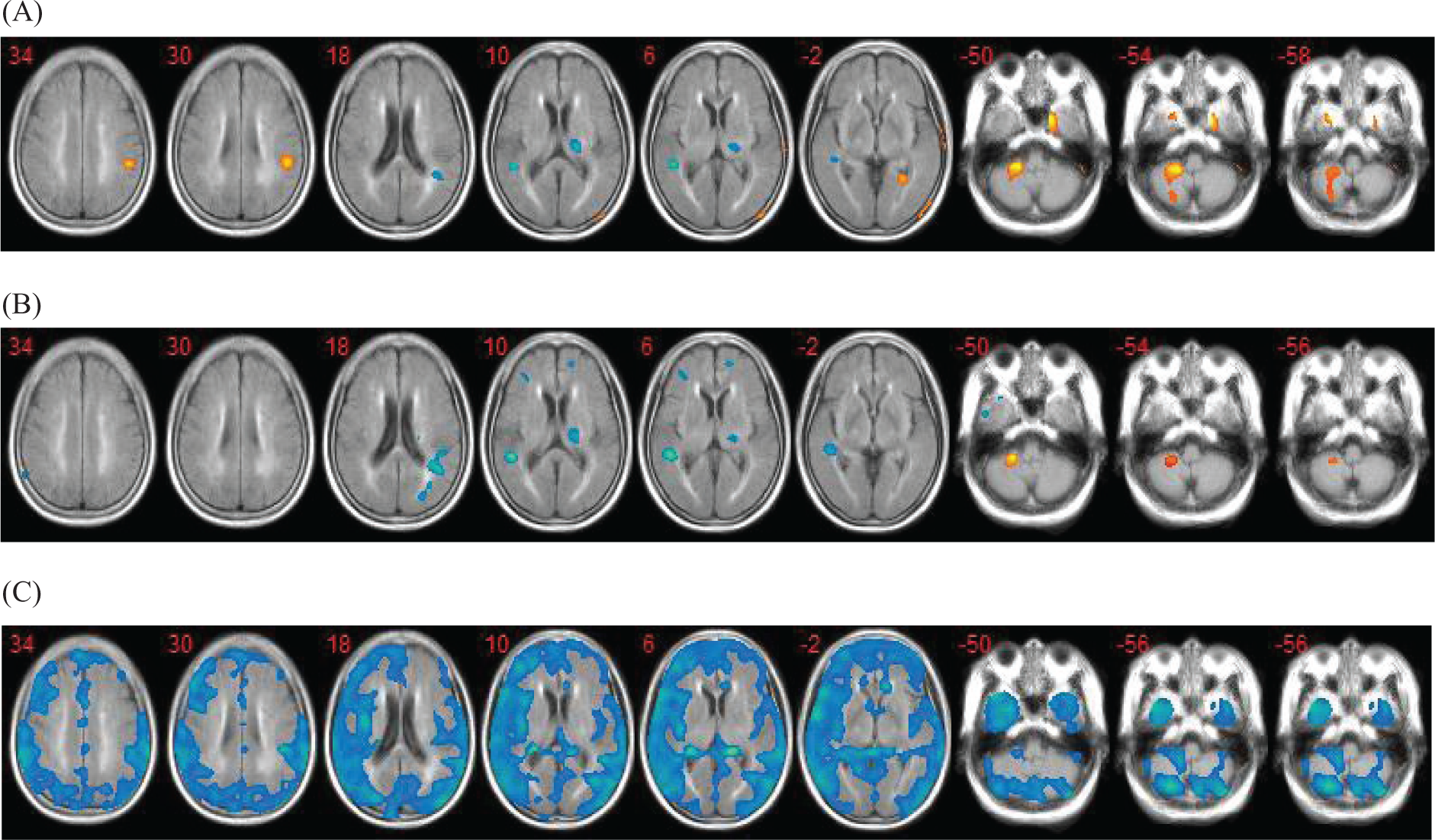
The GABAA receptor availability in patients seen with [18F]FMZ PET at (A) T0 and (B) T1. (C) Δ = T1 − T0.
A comparison of the T0 and T1 [18F]FMZ PET studies revealed that the GABAA receptor availability generally decreased throughout the cerebral cortex and cerebellum during the follow-up period, especially in the contralateral hemisphere (Figure 2C). The mean uptake ratio of [18F]FMZ PET in the brain was also decreased significantly at T1 (2.36 ± 0.14 vs 2.26 ± 0.14;
Correlation Between Motor Function and GABAA Receptor Availability
The initial and follow-up motor functions of the patients are summarized in Table 1. Stroke patients showed significant functional improvement; FMS scores were higher at follow-up than at baseline (paired
A positive correlation was found between baseline motor function and GABAA receptor availability at T0, with lower GABAA availability in the lesion-side frontal and parietal cortex, including the primary motor and sensory cortex (M1, S1), supplemental motor cortex (SMA), dorsolateral prefrontal cortex, and inferior parietal cortex. Lower GABAA availability in the globus pallidus (GP)/thalamus (mainly the ventrolateral and ventroposterolateral nuclei) on the lesion side was related to reduced motor function, as measured with the FMS. The receptor availability in the GP contralateral to the infarct side was positively correlated with the baseline motor function (Figure 3A). To further investigate the impact of initial local GABAergic activity on outcome, we examined the correlation between the initial GABAergic availability and FMS at follow-up and found that the availability of GABAA in the bilateral primary motor cortex and contralateral SMA and GP at T0 was positively correlated with the FMS score at T1 (Figure 3B).
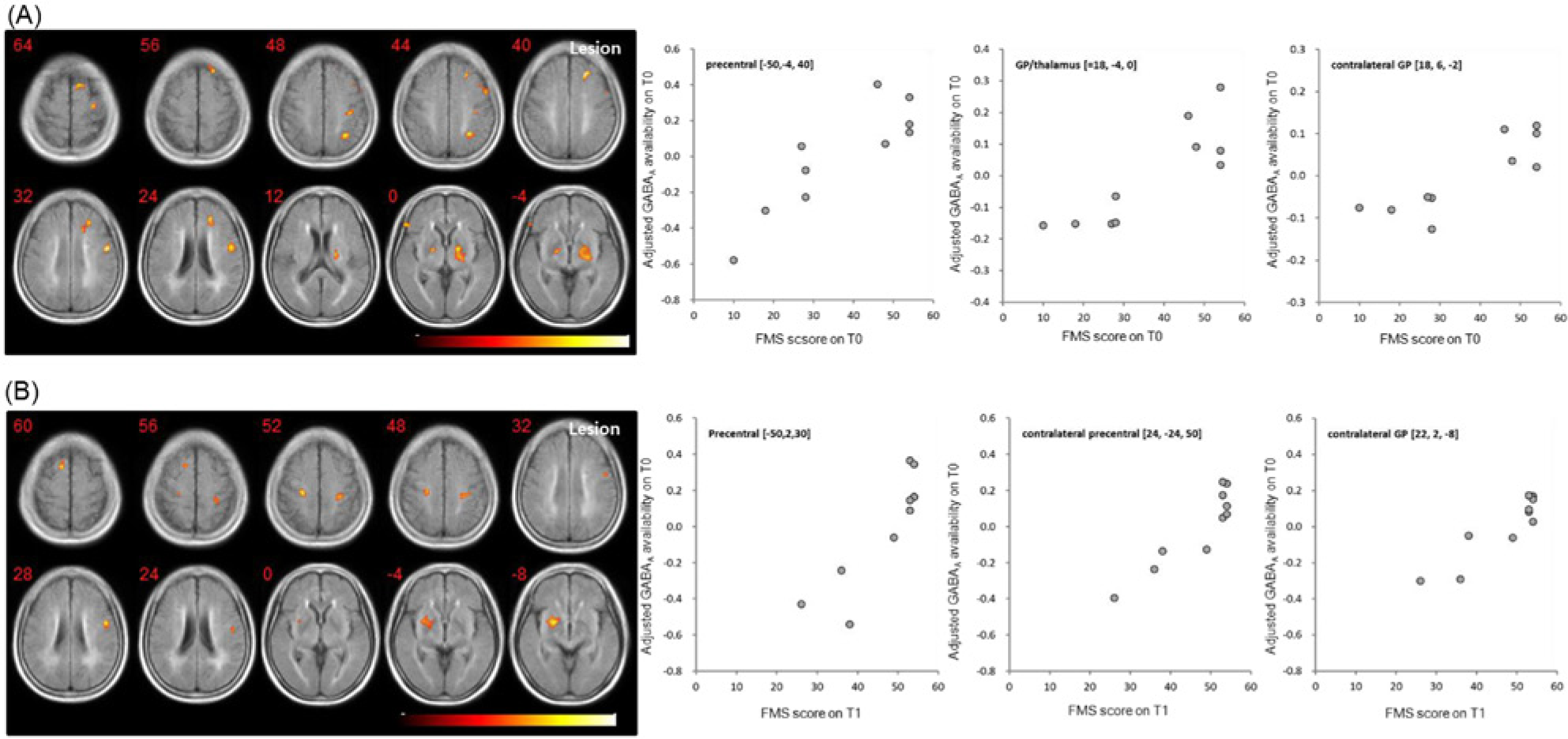
Correlation between the initial GABAA receptor availability and the T0 PET and motor function measured by the FMS score at T0 (A) and T1 (B).
Examining the correlation between the improvement in FMS (ΔFMS) and the changes in GABAA availability (ΔGABAA) during follow-up, we observed that a greater decrease in contralateral cortical and subcortical GABAA availability was correlated with more improvement in motor function (
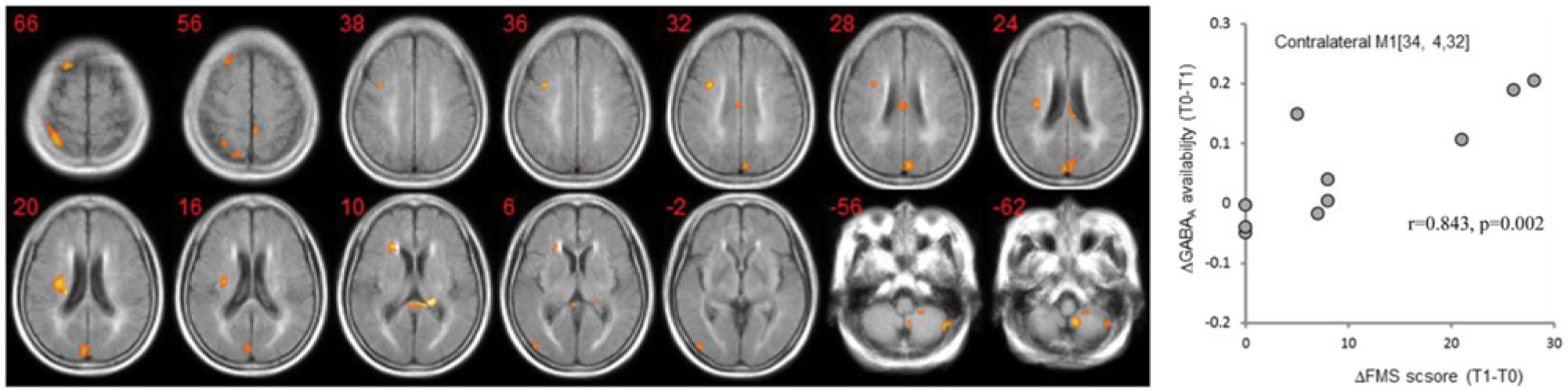
Correlation between the improvement in FMS (ΔFMS) and the changes in GABAA receptor availability (ΔGABAA) during follow-up.
Discussion
In this study, serial [18F]-FMZ PET imaging was used to evaluate GABAergic receptor availability and its relationship with motor recovery in patients with subcortical stroke. To our knowledge, this is the first prospective, controlled longitudinal study demonstrating changes in local GABAergic receptor availability in the ipsilesional and contralesional M1 and showing that the motor nuclei of the thalamus are significantly related to motor recovery after stroke.
FMZ is a specific, high-affinity neutral antagonist that binds reversibly to the benzodiazepine site of the GABAA receptor complex, which is localized on axodendritic synapses, with a cortical distribution closely following the local density of neurons. Therefore, it is considered a sensitive marker of neuronal density and integrity. Earlier imaging studies of GABAA receptors were used to indicate neuronal integrity in stroke because GABA-benzodiazepine receptors are expressed universally in the central nervous system.
Recently, [18F]-FMZ PET imaging studies demonstrated the plasticity of GABAA receptors. Regional FMZ binding was altered without a change in the volume of the gray matter and the affinity of the benzodiazepine site. It was affected by the functional state of GABAergic neurons (ie, the release and uptake of the transmitter during activity).12,13 The functional alteration of GABAergic neurons has been studied in several neuropsychiatric disorders using FMZ PET.14,15 We believe that the FMZ binding in our study reflected the functional alteration and neuronal density of GABA. The excellent reproducibility and reliability of FMZ PET quantification has been reported, in which the variability of receptor binding quantified with a reference tissue model was 8.08% to 9.08% (mean of all brain regions). 16
Previous studies have used [11C]FMZ as an in vivo marker of GABAA-receptor binding.17-19 The [18F]FMZ used in our study has an advantage over [11C]FMZ because it has a longer half-life and greater structural similarity to FMZ, which increases its affinity to the GABA-receptor complex containing α1, α2, α3, and α5. In our study, the global GABAergic activity was increased compared with the controls 1 month after the stroke and returned to the control level 3 months after the stroke. This pattern of brain activation in our patients corroborates the results of a previous study, 5 and these findings suggest that changes in the regional GABAA receptor binding are related to motor recovery in stroke patients.
GABA is the principal inhibitory neurotransmitter in the brain. GABAergic signaling in the motor cortex also plays an important role in the development of perilesional or use-dependent plasticity after brain damage. Decreased GABAA receptor density with increased extracellular GABA concentration after ischemia has been demonstrated in vivo and in vitro. 20 A decrease in GABA-related inhibition facilitates use-dependent plasticity, with the expansion of trained representations in the human motor cortex, 21 whereas the pharmacological administration of GABA agonists such as lorazepam, a GABAA receptor–positive allosteric modulator, can reduce use-dependent plasticity in the human motor cortex after brain damage. 22 Studies on monkeys have also demonstrated that activating GABAA receptors in the medial motor cortex with a GABA agonist increased error rates during motor sequence production. 23 Therefore, increased GABAA receptor binding in the motor cortex could be related to poor functional linking of the neurons for complex motor skills with less effective plasticity.
GABAergic signaling also plays important roles in the functional link between sensory and motor cortical points for motor movements. Initially, increased GABAergic inhibition in the motor cortex might be related to abnormal functional linkage of motor cortical points by limiting the sensory input to receptive fields. The motor system comprises a network of cortical and subcortical areas that interact via excitatory and inhibitory circuits, ultimately leading to motor output, and the balance within this network might be critically disturbed after stroke. 24
In our subcortical stroke patients, the GABAergic activity decreased throughout the cerebral cortex and cerebellum, especially in the contralateral hemisphere during the follow-up period. It might be related to decreased intracortical inhibition or decreased transcallosal inhibition of the ipsilesional hemisphere by the contralesional hemisphere during the motor recovery process. A transcranial magnetic stimulation study demonstrated that the strength of intracortical inhibition was normalized in patients who had had a subcortical stroke and had subsequently recovered normal or near-normal motor function 25 and that transcallosal inhibition of the ipsilesional hemisphere by the contralesional hemisphere was increased in patients with chronic subcortical stroke who have motor control deficits. 26
The role of the contralesional M1 during stroke recovery remains unclear. The original deficits caused by the first stroke reappeared in patients who recovered well from that stroke and had a new lesion causing contralateral hemiparesis. 27 Several neuroimaging studies have also demonstrated increased activity in the contralesional M1 during simple affected hand movements.28,29 The increased activity in the contralesional M1 appears to emerge consistently with coactivity in the ipsilesional M1, provided that damage to the ipsilesional M1 is limited.5,30 In our study, the greater GABAergic availability in the contralesional M1 in the initial study reflected good motor recovery in the follow-up study, in which we found no significant differences in GABAergic availability compared with normal controls. This change in the relative interhemispheric GABAergic activity might reflect a change in the strength of interhemispheric interactions, which are important for the normal coordination of hand movements and stroke motor recovery. 31
Although we observed a change in brain activation in patients with subcortical infarction, it is also expected that the GABAergic activity in M1 will increase initially and then decrease over time after cortical infarction, based on the report by Cramer, 32 which found that when a stroke injures the cortex, cortical function is reduced initially and then increases over time, and the behavioral outcome is correlated with the extent of this increase.
In stroke patients, the intrinsic neural coupling between the ipsilateral SMA and M1 was found to be reduced significantly, and these changes in the intrinsic motor networks were correlated with motor impairment.
24
We also found a correlation between the GABA activity of the ipsilesional SMA and M1 in our patients (from
Recent studies have shown that the motor improvement after stroke is accompanied by topographic shifts in the corticomotor representations within intact cortical regions. 6 The reorganization maps are present not only in the primary motor cortex 33 but also in the sensory, auditory, and visual cortices, 34 and even in the white matter and basal ganglia. 35 Our patients also showed a decrease in GABAA receptor binding in the temporal lobe contralateral to the lesions and a trend toward decreased receptor binding in the ipsilateral thalamus. Changes in the connectivity of M1 with the thalamus might be related to the cortico–basal ganglia–thalamus–cortical loop, which is engaged in processing information of the motor control and learning motor sequences. 36 In our study, motor recovery was correlated with changes in GABAergic activity in the thalamus. This change might reflect a reorganization of the cortico–basal ganglia–thalamo–cortical loop necessary for motor recovery.
We cannot explain exactly why GABAA receptor binding is changed in the parietal white matter and temporal lobe. This might be related to improvement in the processing of proprioceptive information or language function. Both the bilateral parietal and sensorimotor cortices are activated during the processing of proprioceptive information after training, 37 and the functional connectivity with the temporal lobe correlates with the functional outcome after an aphasic stroke. 38
In this study, the GABAergic activity increased in the contralateral cerebellum 1 month after the stroke and persisted 3 months after the stroke but to a lesser extent. The reason for this increased GABAergic activity in the contralateral cerebellum might be to increase contralateral cortical excitability to promote functional recovery. It has been demonstrated that corticomotor excitability was associated with level of inhibition from the contralateral cerebellum (cerebello-cerebral inhibition), 39 and the decrease in this cerebello-cerebral inhibition was associated with improvement of motor function. 40
Conclusions
In conclusion, a decrease in GABAergic activity throughout the cerebral cortex and cerebellum during motor recovery after stroke was identified. Future studies of a larger sample size correlating GABAergic activation patterns with motor recovery are needed to identify the neuroanatomical substrates that subserve motor recovery. Such research might also guide the development of more effective rehabilitative interventions for motor improvement after a stroke. This work supports the rationale for a novel strategy to promote motor recovery after stroke.
Footnotes
Authors’ Note
Yu Kyeong Kim and Eun Joo Yang contributed equally to this work.
Declaration of Conflicting Interests
The authors have no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by the Basic Science Research Program through a National Research Foundation of Korea (NRF) grant funded by the Ministry of Education, Science and Technology (MEST; Grant Numbers 2009-0076321 and 2011-0030815).
