Abstract
Background. Although a mainstay of clinical sensory examination after damage in the spinal cord, pinprick sensation represents only one afferent modality conveyed in the spinothalamic tract. As an objective outcome, complementary information regarding spinothalamic tract conduction may be elucidated by measuring contact heat evoked potentials (CHEPs). Objective. To assess the value of CHEPs to measure spinothalamic tract function in spinal cord disorders compared with pinprick scoring. Methods. CHEPs were examined using a standard (35°C) and increased baseline (42°C) contact heat temperature. Pinprick sensation was rated as absent, impaired, or normal according to the International Standards for the Neurological Classification of Spinal Cord Injury. Results. Fifty-nine dermatomes above, at, and below the sensory level of impairment were analyzed in 37 patients with defined spinal cord disorder. In dermatomes with absent or impaired pinprick sensation, CHEPs using a standard baseline temperature were mainly abolished (3/16 and 8/35, respectively). However, when applying an increased baseline temperature, CHEPs became recordable (absent: 11/16; impaired: 31/35). Furthermore, CHEPs with increased baseline temperature allowed discerning between dermatomes with absent, impaired, and normal pinprick sensation when using an objective measure (ie, N2P2 amplitude). In contrast, the pain perception to contact heat stimulation was independent of pinprick scores. Conclusion. Applying pinprick testing is of limited sensitivity to assess spinothalamic tract function in spinal cord disorders. The application of CHEPs (using standard and increased baseline temperatures) as an objective readout provides complementary information of spinothalamic tract functional integrity beyond pinprick testing.
Introduction
Based on the somatotopic distribution of epicritic and protopathic sensations into anatomically distinct pathways (ie, dorsal columns and spinothalamic tract), sensory testing can be used to obtain considerable detail regarding the extent and location of damage in the spinal cord. 1 As a surrogate marker of functional integrity in these pathways, sensation is routinely examined by testing light touch and pinprick perception.2-4 These bedside sensory testing methods provide valuable insight about the function of the sensory system, yet the ordinal scale used to classify sensation does not account for the spectrum of deficits occurring after spinal cord disorders. Hence, there is emerging interest in developing tools that provide complementary information regarding the pathophysiology underlying sensory deficits. 5
As a complementary approach to light touch and pinprick scores, objective and quantitative information concerning the integrity of conduction in afferent fibers can be achieved by recording sensory evoked potentials using electroencephalography. The adoption of sensory evoked potentials to characterize spinal cord injury (SCI) in neurophysiological terms has primarily focused on measuring deficits in large diameter fibers conveying light touch sensation in the dorsal columns—so termed somatosensory evoked potentials.6,7
To examine conduction deficits in small diameter afferents ascending in the spinothalamic tract, noxious laser and contact heat pulses represent the most viable stimulation modalities for the acquisition of sensory evoked potentials.8-10 A major obstacle for SCI applications involving nociceptive evoked potentials (ie, laser evoked potentials and contact heat evoked potential [CHEPs]) is the need to balance high stimulation energies sufficient for the detection of afferent sparing in areas with reduced sensation, with the requirement for the duration of the event potential stimuli to be brief (ie, high rate of temperature change to induce a phase-locked cortical response) and restrictions on the absolute peak temperature to avoid skin burns. One recent technique shown to be effective in improving the acquisition of CHEPs has been increasing the baseline stimulation temperature from 35°C to 42-45°C. 11 In SCI, increasing the baseline temperature of contact heat stimulation has shown to be effective in demonstrating preserved cortical responses in dermatomes with “impaired” clinical pinprick sensation.11,12
The primary objective of this study was to assess the value of CHEPs to improve the examination of spinothalamic tract function in individuals with sensory deficits due to spinal cord disorders. More specifically, we aimed to examine if a novel stimulating protocol (ie, increasing the baseline temperature to 42°C) would improve the overall interpretation of spinothalamic tract deficits. A secondary objective was to determine the relationship between clinical pinprick scores (ie, absent, impaired, normal) and outcomes from CHEP stimulation.
Methods
Subjects
A retrospective cross-sectional analysis of 37 patients with a spinal cord disorder was conducted. All study procedures conformed to the standards set by the Declaration of Helsinki and was approved by the institutional research ethics board of the University of Zurich.
Data Source
The data for this study were retrieved from the inpatient and outpatient clinic of the University Hospital Balgrist (Zurich, Switzerland). The clinical examination of spinothalamic tract function was performed in dermatomes defined by the International Standards for the Neurological Classification of Spinal Cord Injury (ISNCSCI). 2
Assessments
In accordance with the ISNCSCI, sensory level was defined as the most caudal intact dermatome for both light touch and pinprick sensation. 2 Pinprick examination serves as a surrogate marker for the spinothalamic tract function, based on the ability to discriminate sharp and dull sensation. As a reference, pinprick testing is first performed on the face. Pinprick sensation is scored according to a 3-point ordinal scale (absent, impaired, and normal). Absent sensation indicates a lack of ability to discriminate sharp–dull sensation (pinprick score = 0). Comparatively, impaired sensation implies sharp–dull discrimination remains intact (ie, patient reliably reports sharp and dull stimulation), but that sensation is considerably affected (pinprick score = 1). Importantly, sensation can be either decreased or increased compared with the reference area. If pinprick stimuli are perceived as similar to the reference area, sensation is defined as normal (pinprick score = 2). Light touch sensation was classified as normal (light touch score = 2), impaired (light touch score = 1), and absent (light touch score = 0) by comparing the skin area to be examined with an area in the face. The ISNCSCI assessment was performed by a trained examiner.
Contact Heat Evoked Potentials
Contact heat stimuli were delivered using a contact heat stimulator (PATHWAY Pain & Sensory Evaluation System, Medoc, Ramat Yishai, Israel). The device has been described previously.8,13 Briefly, the device is capable of heating from a controlled baseline temperature (30-45°C) at a rate of 70°C/s up to a maximum of 55°C. After reaching a peak stimulation temperature, the skin is rapidly cooled (40°C/s). About 15 to 20 contact heat stimuli were applied with an interstimulus interval of 8 to 12 seconds. The stimulation was applied in an area (approximately 16 cm2) surrounding the dermatomal sensory keypoints according to the ISNCSCI. 2 The thermode (27 mm diameter) was variably repositioned after each stimulus, within the boundaries of the respective dermatome. In response to an audio cue, perceived intensity was rated according to a 0 to 10 visual analog scale (VAS) 4 seconds after each stimulus. Subjects were instructed to keep their eyes open and were asked to blink, only after hearing the audio cue.
Contact heat stimulation was applied according to the patients’ level of lesion in dermatomes above, at, and below the level of injury. Stimuli were delivered from standard baseline temperature (35°C) to a peak temperature of 52°C. Dermatomes (absent, impaired, normal) that demonstrated small, unreliable CHEPs (amplitudes <10 µV) with standard stimulation were further assessed with increased baseline stimulation (42°C) to a peak temperature of 52°C. The threshold of 10 µV was chosen based on experience allowing to visually distinguish CHEPs from ongoing background activity. To specifically address the primary aim of this study, only dermatomes with both standard and increased baseline stimulation were included in the analysis. Standard baseline temperature stimulation always preceded increased baseline stimulation. The stimulations used in the present study are illustrated in Figure 1.
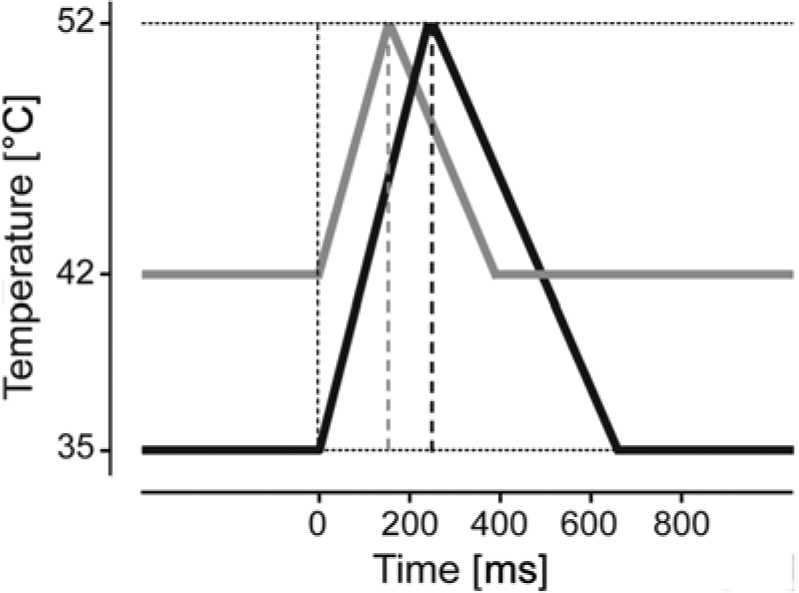
Contact heat stimulation parameters.
For the recording of CHEPs, scalp recording sites were prepared with Nuprep (D.O. Weaver & Co, Aurora, CO) and alcohol. Silver–silver disc recording electrodes were positioned according to the 10-20 electrode configuration system with the active electrode at the Cz position and referenced to the linked earlobes (A1-A2). A wet ground strap was attached to the subjects forearm. The impedance of scalp electrodes was kept below 5 kΩ. CHEPs were sampled at a sampling rate of 2000 Hz using a preamplifier (20 000×, bandpass filter 0.25-300 Hz, ALEA Solutions, Zurich, Switzerland). Data were recorded in a LabView-based program (V1.43 CHEP, ALEA Solutions, Zurich, Switzerland) with a 100-millisecond period pretrigger and 1 second posttrigger. Electroencephalography raw data were bandpass filtered offline from 0.5 to 30 Hz. After filtering, the single trial waveforms were averaged (15-20 trials) for each location and subject. To confirm the N2P2 response, odd/even trial averaging was visually analyzed within 200 to 600 ms poststimulus.14,15
Data Analysis
A q-q plot of the residuals was examined to test for normal distribution of the N2P2 amplitude and pain rating to contact heat stimulation. Based on pinprick classification, the proportion of dermatomes with recordable CHEPs (standard and increased baseline stimulation) was examined with Fischer’s exact test. Furthermore, a linear mixed model was used to assess the main effect of stimulation (ie, standard stimulation and increased baseline stimulation) and pinprick score, and the interaction of these 2 factors on the N2P2 amplitude and the rating.
All multiple comparisons were Bonferroni corrected. Statistical significance was set at α = .05. All statistical analyses were performed in SPSS v.19 (SPSS Inc, Chicago, IL).
Results
Patient Characteristics
Patient characteristics are listed in Table 1. The average age of subjects with a spinal cord disorder was 48.8 ± 14.6 years. In total, 59 dermatomes were included in this examination. Sixteen, 35, and 8 dermatomes were classified according to ISNCSCI as absent, impaired, and normal pinprick sensation, respectively. Of the 16 dermatomes with absent pinprick, only 2 demonstrated complete loss of light touch sensation, with the remaining 14 either impaired (n = 12) or normal (n = 2). Impaired pinprick was associated with impaired light touch 74% of the time (n = 26); the remaining dermatomes with impaired pinprick having normal light touch sensation (n = 9). In 100% of dermatomes with normal pinprick sensation (n = 8), light touch was perceived normally.
Clinical Characteristics of Patients With a Spinal Cord Disorder.
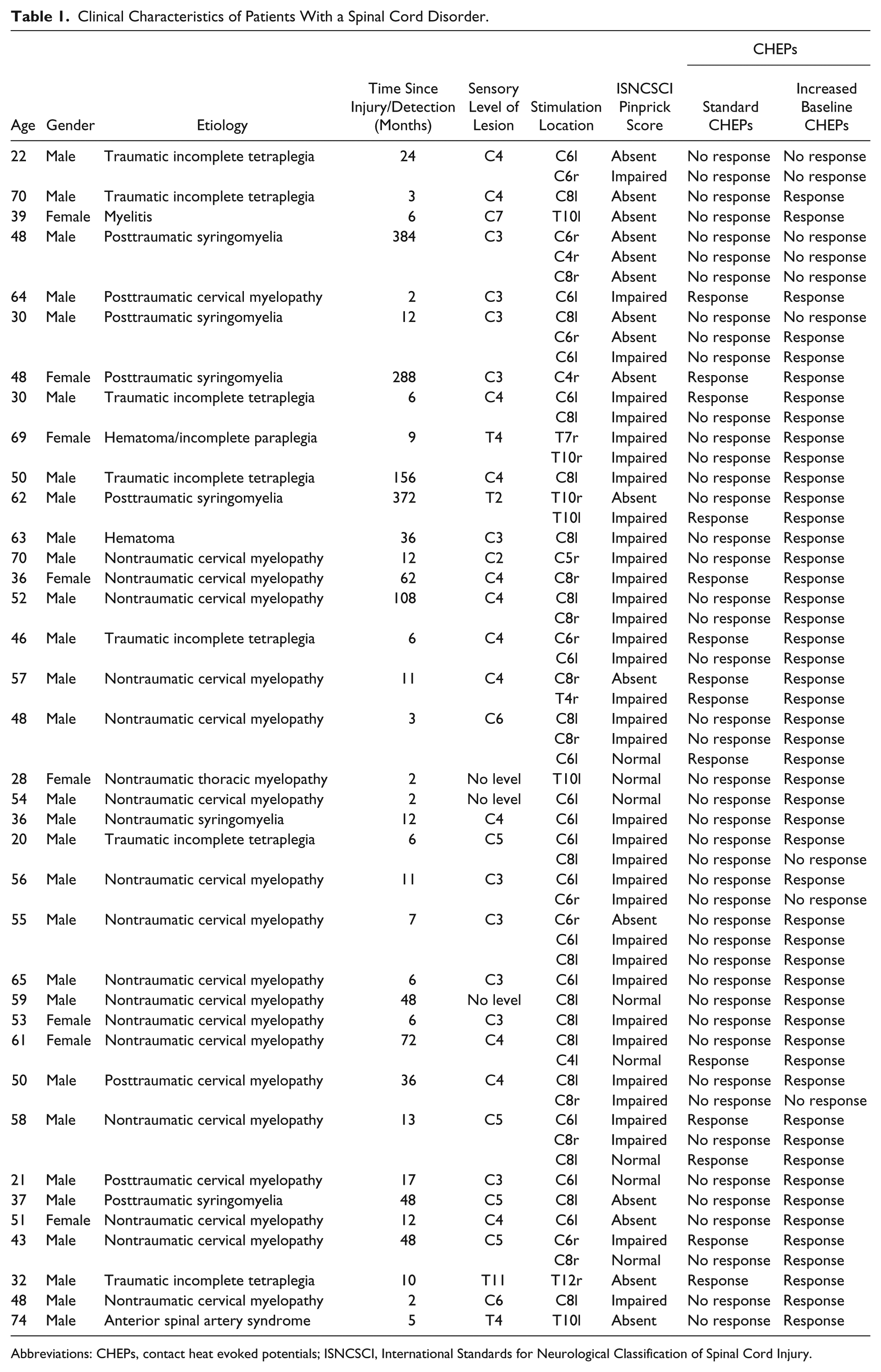
Abbreviations: CHEPs, contact heat evoked potentials; ISNCSCI, International Standards for Neurological Classification of Spinal Cord Injury.
N2P2 Amplitudes and Pain Rating in Relation to Pinprick Scores
The results of Fisher’s exact test of the proportion of recordable CHEPs (yes/no) using standard and increased baseline stimulation are summarized in Table 2. In 16 dermatomes with absent pinprick sensation (n = 13), contact heat stimulation with standard baseline temperature (35°C) evoked a cortical response in 3 sites (3/16). In contrast, an evoked potential was recorded with an increased baseline temperature (42°C) in 11 dermatomes with “absent” pinprick sensation (11/16). Four representative examples of CHEPs from standard and increased baseline stimulation in dermatomes with absent pinprick sensation are shown in Figure 2. In 35 dermatomes with impaired pinprick sensation (n = 25), normal baseline temperature stimulation evoked a response in 8 sites (8/35). Using increased baseline stimulation, CHEPs were recordable in 31 dermatomes (31/35). Out of 8 dermatomes with normal pinprick sensation (n = 8), a CHEP was recordable in 3 sites. Comparatively, a CHEP could be recorded from all normal dermatomes using increased baseline stimulation.
Summary of Number of Recordable Contact Heat Evoked Potentials.
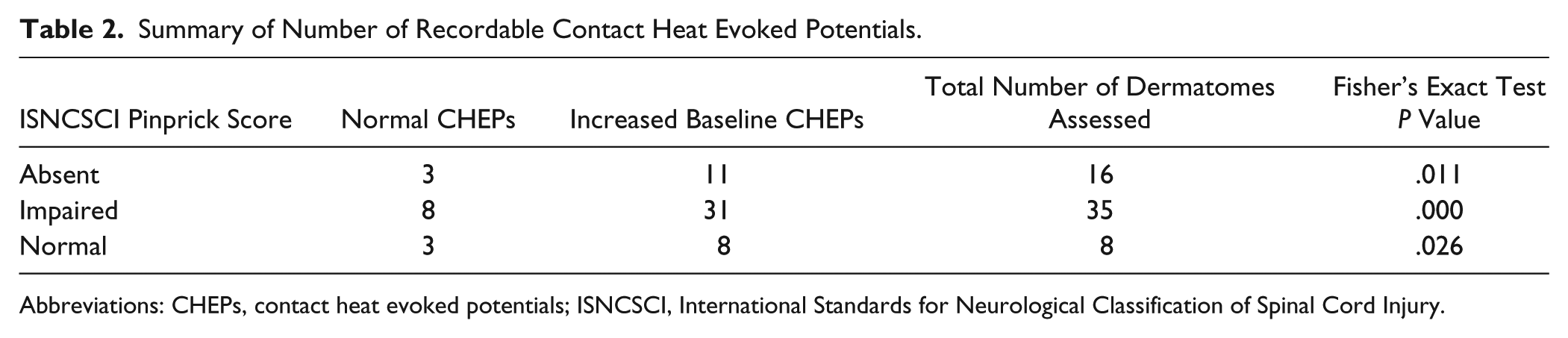
Abbreviations: CHEPs, contact heat evoked potentials; ISNCSCI, International Standards for Neurological Classification of Spinal Cord Injury.
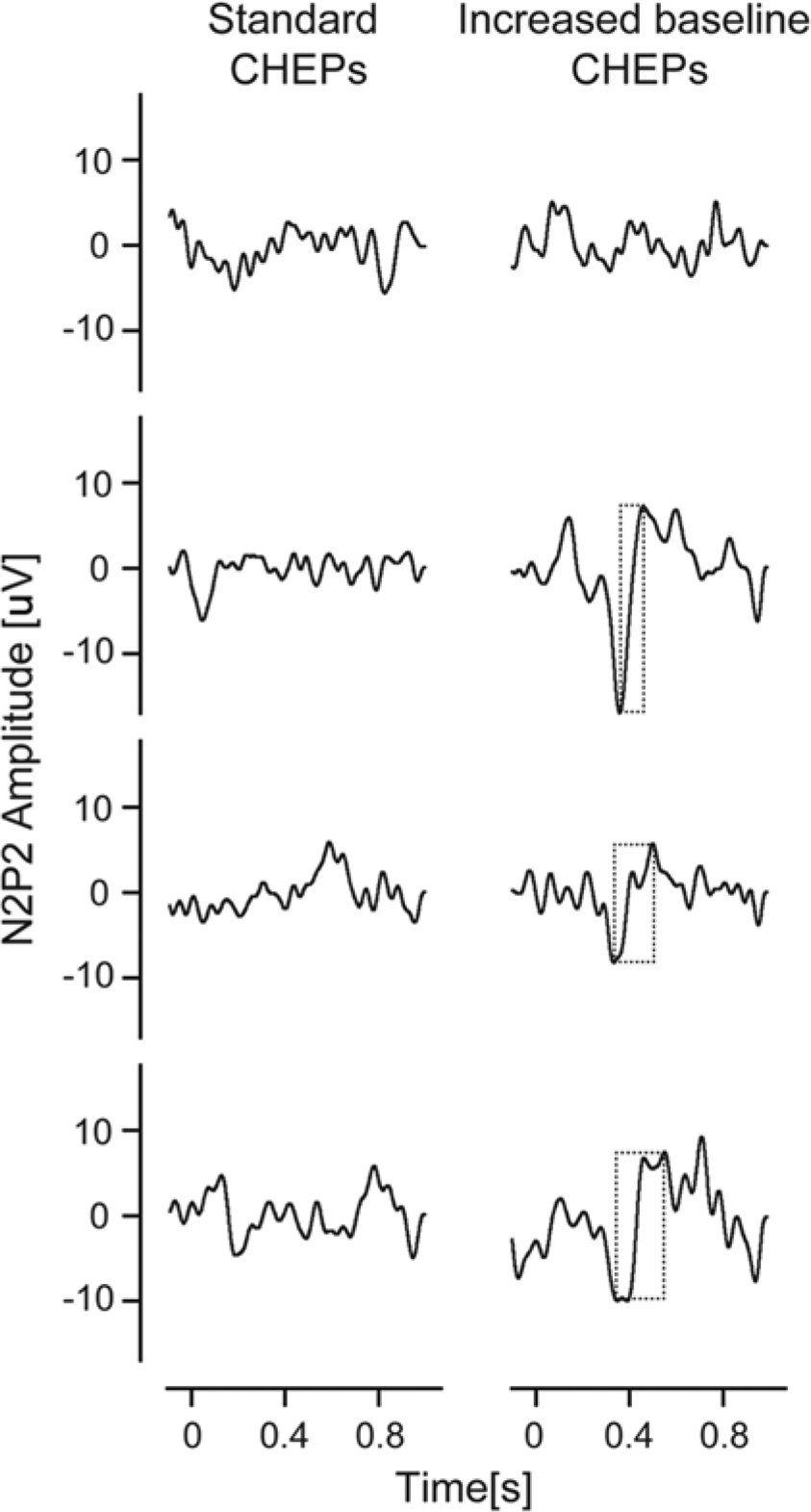
CHEPs with pinprick score zero.
In Figure 3, CHEPs to both standard and increased baseline stimulation according to the extent of pinprick preservation (ie, absent, impaired, and normal) are displayed. Compared with standard baseline, N2P2 amplitudes were significantly higher when the baseline temperature was increased to 42°C (F = 74.669, P = .000). Changes in the amplitudes from standard to increased baseline stimulation significantly depended on the scoring of the dermatomes (F = 4.348, P = .018). Pair-wise comparisons between the pinprick scores of the increased baseline stimulation showed significant differences between normal and absent pinprick score (normal > absent, P = .006), meaning that N2P2 amplitude was significantly higher in the dermatomes with normal than in the dermatomes with absent pinprick score. Comparison to impaired pinprick score did not reach significance (normal > impaired, P = .150; impaired > absent = .137). None of the pair-wise comparisons were significant for the standard stimulation temperature (normal > absent, P = .915; normal > impaired, P = .868; impaired > absent, P = 1.000). The increased baseline stimulation caused higher pain ratings than the standard stimulation (F = 28.653, P = .000). However, the increase in VAS rating from standard to increased baseline stimulation did not significantly depend on the pinprick score (F = 2.526, P = .089).
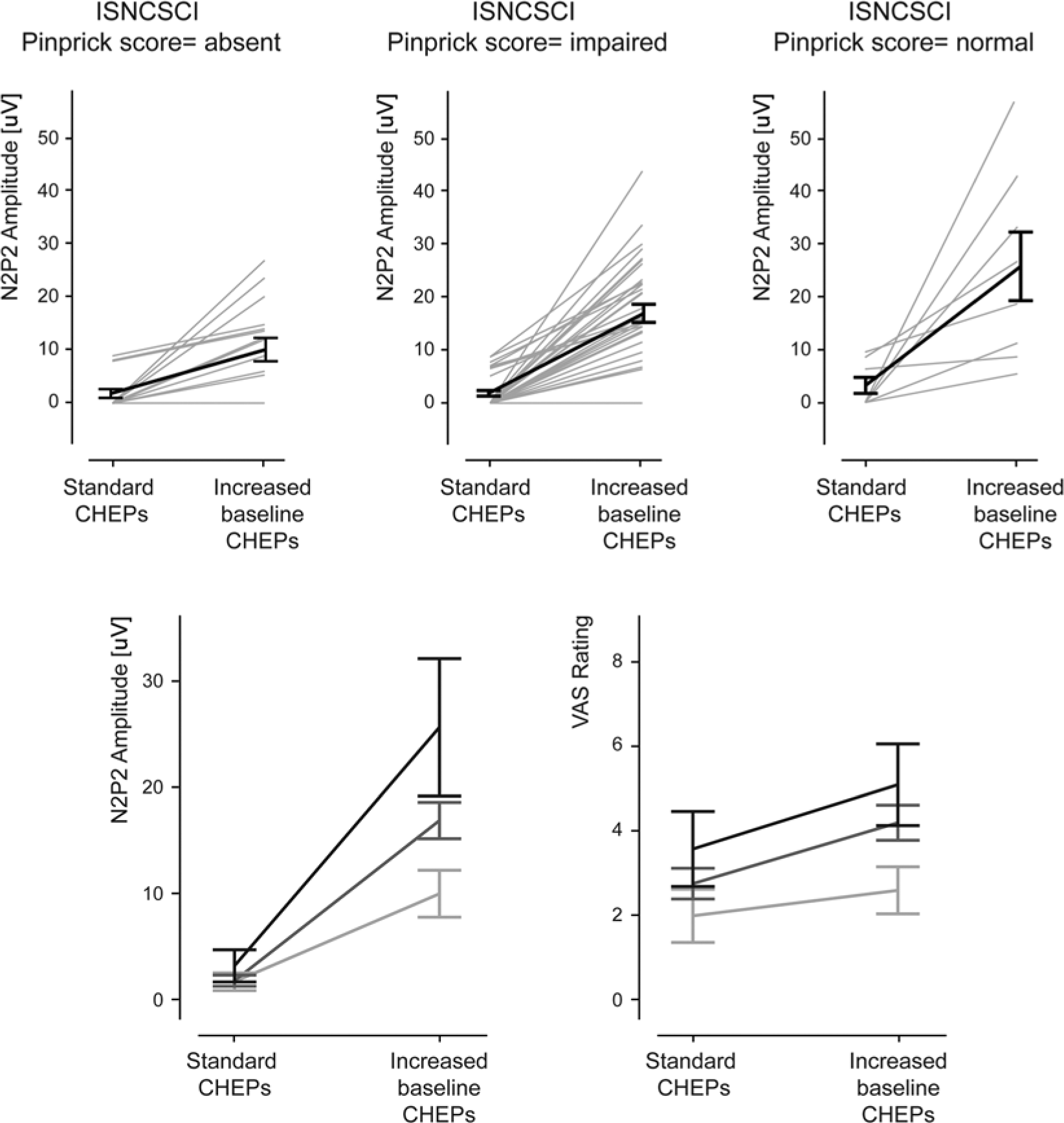
Overview of CHEPs amplitude divided into pinprick scores.
Discussion
Complementary to pinprick sensation, the present study has shown that CHEPs improve the assessment of spinothalamic tract function in patients with spinal cord disorders. The value of increasing the baseline temperature preceding CHEP stimulation was clearly demonstrated—most notably to reveal residual spinothalamic tract function in individuals with absent pinprick sensation and abolished CHEPs based on standard stimulation techniques. In comparison to pain rating, changes in N2P2 amplitudes induced by going from standard to increased baseline stimulation temperature was able to discriminate between dermatomes with normal and absent pinprick sensation. In general, these findings support that CHEPs provide information regarding the integrity of the spinothalamic tract beyond the examination of pinprick sensation.
Spinothalamic Tract Function in Spinal Disorders
In simple terms, the use of advanced sensory testing methods, including CHEPs, could play a complementary role to clinical techniques (eg, pinprick) in diagnosis and prognosis of spinal cord disorders. Indeed, an accurate assessment of sensation is important for determining neurological level of impairment and severity and predicting long-term neurological and functional outcomes.16-18 To fully characterize the extent of damage in spinothalamic pathways, examining different sensory modalities, such as heat, in conjunction with sharp–dull discrimination may be of clinical value. It should also be considered, by testing discrimination between sharp and dull (ie, ISNCSCI), not only is spinothalamic tract function examined (ie, sharp sensation) but also the dorsal column pathway (ie, detection of dull sensation). However, CHEPs represent a functional measure of spinothalamic tract integrity8,10; thus, CHEPs were only compared with pinprick sensation. Recent studies have also demonstrated the use of advanced sensory testing (eg, quantitative sensory testing) to elucidate mechanisms of neuropathic pain after SCI.19-22 While the underlying mechanism remains unknown, studies support that damage of the spinothalamic tract is requisite for the development of neuropathic pain.20-23 To confirm the presence of a lesion in the spinothalamic tract, as well as better assess the severity of damage, the methods presented here (ie, acquiring CHEPs from an increased baseline temperature) may also be useful for characterizing neuropathic pain.
Mechanism of Increased Baseline Contact Heat Evoked Potentials
The effect of increasing the baseline temperature on the acquisition of CHEPs is 2-fold. First, using a 42°C baseline reduces the discrepancy between the theoretical and actual peak temperature achieved at the level of the nociceptor.11,12 Thus, improved acquisition of CHEPs using an increased baseline temperature is akin to increasing the peak temperature, which in turn results in higher pain ratings and larger amplitude waveforms.24-26 In individuals with sensory deficits, adopting an increased baseline stimulation protocol may be important to increase the magnitude of stimulation and determine afferent sparing in areas with reduced sensation, while maintaining a safe peak temperature. Second, increasing the baseline temperature shortens the overall stimulation duration, thus yielding a more synchronized afferent volley and reduced trial-by-trial jitter. 12 Importantly, such an effect can be observed in absence of increasing the rating of perceived intensity if the peak temperature is adapted to balance the perception between standard and increased baseline stimulation. 12 In patients, improved synchronization of the afferent volley may also have attributed to a higher probability of detecting a CHEP using a 42°C baseline in dermatomes with diminished pinprick sensation (ie, impaired or absent). Indeed, in a subset of dermatomes (ie, 29 dermatomes), pain rating changed nominally (<1 point on the VAS) but was accompanied by large change in N2P2 amplitude in response to stimulation from an increased baseline temperature compared with standard baseline (Figure 4). In contrast, in healthy subjects going from standard to increased baseline stimulation, there is a clear increase in pain rating. 11 Interestingly, this finding would suggest that sensory deficits associated with spinal cord disorders may be related to a combination of reduced overall quality (ie, diminished sensation) and reduced synchrony of afferent volley along the neural axes.
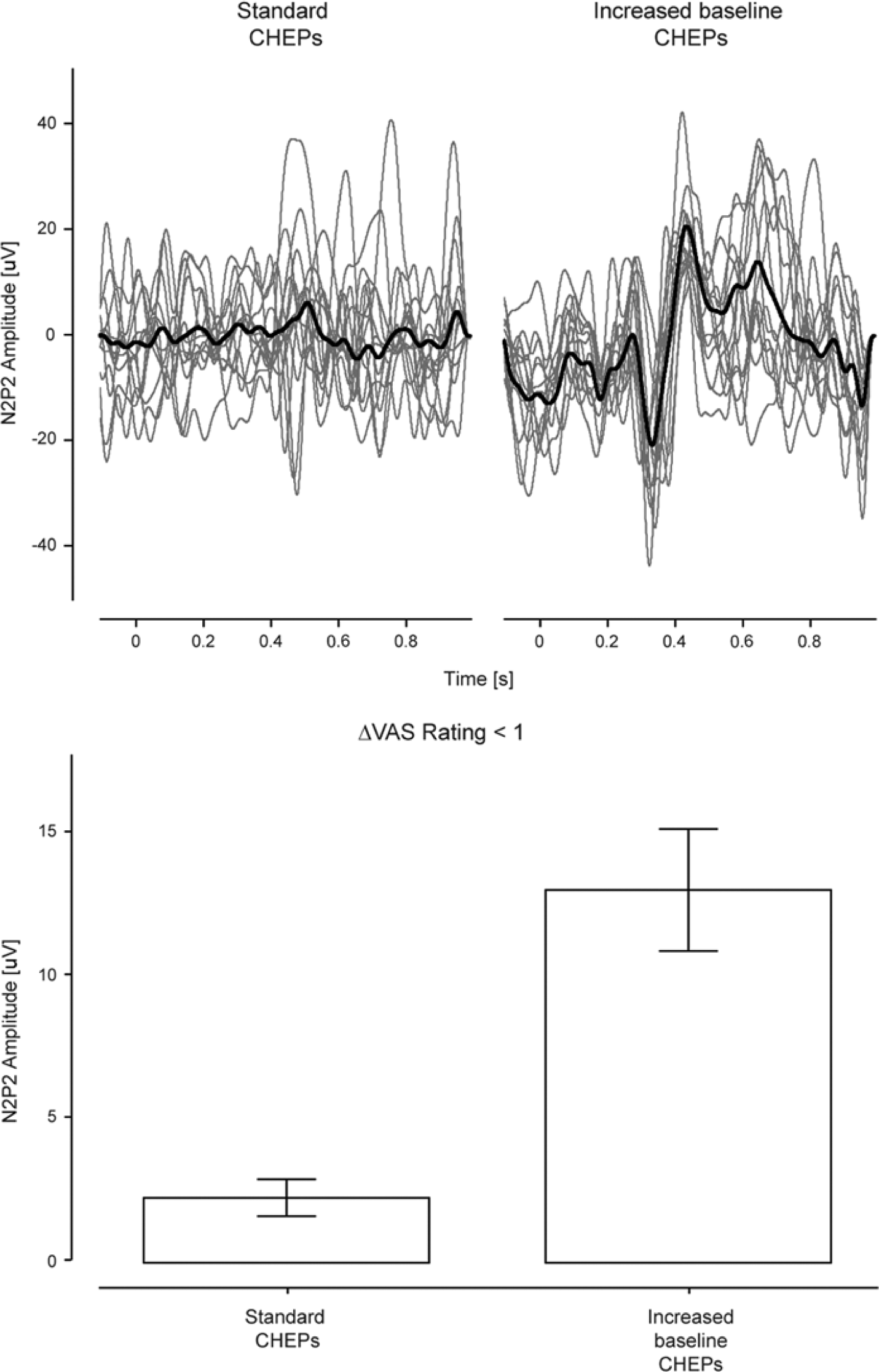
Dissociation of CHEPs and VAS rating.
Beyond Pinprick Sensation
The observation of small amplitude or nonrecordable CHEPs in dermatomes with “normal pinprick” sensation warrants consideration. On one hand, such an observation may indicate subclinical afferent deficits, that is, damage missed according to the clinical exam. Given the limitations of pinprick testing (eg, subjective), and that the majority of “normal” dermatomes were examined close to the level of spinal disorder, the presence of subclinical deficits would not be surprising. On the other hand, it is important to consider that difficulties acquiring CHEPs from conventional baseline temperatures (eg, 32°C to 35°C) have been reported in neurologically healthy subjects. 27 In such an event, evidence of impairment in normal dermatomes may be a function of testing error. Based on our experience,11,12 increasing the baseline stimulation to 42°C overcomes acquisition problems in neurologically healthy subjects. Indeed, none of the neurologically intact subjects examined in Kramer et al 12 demonstrate peak-to-peak amplitudes <10 µV. In the context of the present findings, dermatomes with normal pinprick sensation coupled with low-amplitude CHEPs (<10 µV) in response to both normal and increased baseline temperature stimulations could be interpreted as robust evidence of subclinical deficits in the spinothalamic tract.
In dermatomes with low-amplitude CHEPs and impaired pinprick sensation, applying increased baseline CHEP stimulation revealed a wide array of outcomes. In a subset of these dermatomes, N2P2 remained <10 µV or nonrecordable, while in others, amplitudes increased comparable to dermatomes with normal pinprick sensation. Furthermore, the vast majority of dermatomes with “absent” pinprick sensation, in individuals with incomplete sensory deficits, demonstrate recordable CHEPs after increased baseline stimulation.
These observations underscore 2 important limitations of the clinical sensory examination: (a) classification of impairment provides no indication as to varying levels of afferent deficit (ie, cannot distinguish severely and mildly impaired) and (b) the absence of sharp–dull discrimination does not reflect complete disruption of afferents in the spinothalamic tract.
Limitations
While we have highlighted an advantage of adopting CHEPs, interpretation of these findings beyond that increasing the baseline temperature improves the probability of detecting residual spinothalamic tract function in individuals with spinal cord disorders warrants caution. Specifically, our observations only apply to dermatomes that demonstrate reduced amplitude CHEPs using standard contact heat stimulation techniques, and in patients with incomplete lesions (ie, all subjects had sensation preserved according to light touch or pinprick below the level of lesion). Because of these limitations, we cannot determine the true sensitivity of CHEPs to measure deficits in the spinothalamic tract in dermatomes with absent, impaired, and normal pinprick sensation. Indeed, our findings likely underestimate the value of CHEPs recorded from standard stimulation to distinguish between classifications of impairment.
Here, we have shown in a subset of dermatomes dissociation between large changes in N2P2 amplitude after increased baseline stimulation accompanied by a small change in the perceived pain intensities (Figure 4). As in the current patient population no pain status was assessed, it remains to be defined if this specific dissociation of recorded nociceptive response and pain perception can be explained by underlying neuropathic pain states.
A general limitation of cortical evoked potentials in repose to nociceptive stimulation (including laser evoked potentials) is that the cortical response is not reflecting the early arrival of incoming neural activity but is rather a processed response; their generation, however, relies on the functional integrity of the spinothalamic tract.
Conclusion/Implications
In the present retrospective study, we have shown that using increased baseline CHEP allows assessing the spinothalamic tract beyond pinprick sensation, revealing subclinical afferent sparing. Increased baseline stimulation should be considered in future studies to complement standard pinprick testing to objectively evaluate the functional integrity of the nociceptive system.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Center of Competence in Research (NCCR) and the Swiss National Science Foundation (SNSF). JK was funded by the International Foundation for Research in Paraplegia, Zurich.
