Abstract
Background. Brain lesions may disturb binocular fusion and stereopsis, leading to blurred vision, diplopia, and reduced binocular depth perception for which no evaluated treatment is currently available. Objective. The study evaluated the effects of a novel binocular vision treatment designed to improve convergent fusional amplitude and stereoacuity in patients with stroke or traumatic brain injury (TBI). Methods. Patients (20 in all: 11 with stroke, 9 with TBI) were tested in fusional convergence, stereoacuity, near/far visual acuity, accommodation, and subjective binocular reading time until diplopia emerged at 6 different time points. All participants were treated in a single subject baseline design, with 3 baseline assessments before treatment (pretherapy), an assessment immediately after a 6-week treatment period (posttherapy), and 2 follow-up tests 3 and 6 months after treatment. Patients received a novel fusion and dichoptic training using 3 different devices to slowly increase fusional and disparity angles. Results. At pretherapy, the stroke and TBI groups showed severe impairments in convergent fusional range, stereoacuity, subjective reading duration, and partially in accommodation (only TBI group). After treatment, both groups showed considerable improvements in all these variables as well as slightly increased near visual acuity. No significant changes were observed during the pretherapy and follow-up periods, ruling out spontaneous recovery and demonstrating long-term stability of binocular treatment effects. Conclusions. This proof-of-principle study indicates a substantial treatment-induced plasticity of the lesioned brain in the relearning of binocular fusion and stereovision, thus providing new, effective rehabilitation strategies to treat binocular vision deficits resulting from permanent visual cortical damage.
Keywords
Introduction
We perceive our visual world with 2 eyes providing us 2 slightly disparate visual images, which require continuous unification into a binocular, “cyclopean” percept. This unification mechanism is termed binocular fusion and comprises a motor component (motor fusion) as well as a sensory, nonmotor component. Motor fusion relies on vergence eye movements (convergence/divergence) necessary for the proper alignment of both eyes when viewing an object at different distances. The sensory component is related to the merging of the disparate monocular images into a fused, stereoscopic representation containing spatial depth. 1 Both mechanisms are important for all binocular activities, particularly in the near space because of the greater disparity of the 2 visual images at this distance. With the advent of small visual displays on smart phones and other near-space hand-held devices, the requirements for fusion and stereopsis have continuously risen in the past decade. 2
Brain systems associated with motor fusion are based on a network that includes midbrain oculomotor areas and frontal eye fields. 2 The sensory component relies on the interplay of several visual cortical areas starting from V1, analyzing contour-based stereocues (local stereopsis), to extrastriate areas (V2-V8) for a more refined analysis of disparity information (global stereopsis). Studies with brain-injured individuals showed that the right hemisphere (in right-handers) is dominant for global but not for local stereopsis3,4 and that global stereopsis depends largely on the integrity of the left or right temporal cortex, whereas local stereopsis relies on more posterior, occipital areas.5,6 In patients with unilateral brain lesions, a high incidence of disturbed binocular depth perception (29% after left- and 69% after right-sided stroke) and a frequent, not strict, association with homonymous visual field defects have been reported. 7 Furthermore, a frequent relation between right extrastriate cortex lesions in patients with stroke and reduced stereoacuity has been noted,8,9 again highlighting the pivotal role of the right hemisphere in stereoscopic depth perception. Additionally, astereopsis is also frequent in traumatic brain injury (TBI; 41%). 10
Like astereopsis, motor fusion impairments are also a recurring sequel following brain damage (20% of patients after stroke, 30% after TBI11,12). They typically manifest as “blurred vision” because the perceived scenery of each eye can no longer be fused into a binocular percept. Inaccurate reading, severely reduced reading duration until diplopia emerges (often <15 minutes), deficits in almost all near-space activities (eg, PC work, using a smart phone, and reaching/grasping/manipulating objects), and asthenopic symptoms (eye pressure, fatigue, and headache) are typical consequences. 11 Finally, patients develop horizontal diplopia after a short period of binocular activity (10-15 minutes). 12 As a prerequisite of stereopsis, motor fusion impairment reduces or even impedes stereopsis, because no disparity information can be derived from the monocular input if the eyes are not properly aligned. This hampers many visual/visuomotor activities requiring depth perception: for example, 3D visual object recognition/grasping of 3D objects,13,14 walking on stairways, 15 or motion perception. 16
Thus, reduced motor fusion has a severe impact on a patient’s daily visual and visuomotor activities as well as on their vocational rehabilitation. Although some degree of spontaneous recovery has been noted,17,18 Hart 11 reported virtually no recovery in 5 out of 7 patients with TBI 1 year postlesion. Despite the frequent occurrence of fusion disturbances and astereopsis following brain damage and the intense research on stereovision in neurophysiology and neuroimaging, these issues have been totally neglected in neurorehabilitation.
To the best of our knowledge, there exists no evaluated treatment for these disorders. This is surprising because the neural correlates underlying fusion and stereovision are well explored (see above), and both can be measured by standard diagnostic procedures. 19 Furthermore, studies using perceptual learning paradigms in unimpaired controls, amblyopic patients, and neurological patients revealed promising results in visual/visuospatial tasks, partially along with generalization on related visual tasks after systematic practice.20-25 These findings indicate some potential for treatment-induced behavioral plasticity in (re-)learning of impaired visual capacities after brain damage. Additionally, studies on cats with bilateral visual cortex ablations showed complete reinstatement of binocular depth perception after 4 amphetamine doses, 26 suggesting that treatment-induced relearning of binocular vision is principally possible.
The current study, therefore, aimed to evaluate whether impaired motor fusion and stereopsis can be treated using prismatic and dichoptic devices in 2 consecutive samples of patients with either stroke or TBI. Based on the above-mentioned considerations, we hypothesized a priori that fusional amplitude and stereopsis improve selectively during treatment but not during treatment-free intervals (baseline and follow-up) and that this transfers to related visual tasks such as binocular reading duration. In contrast, we expected no changes in accommodation because this includes monocular brain mechanisms devoted to the near response, 27 which are different from the binocular mechanisms involved in fusion and stereopsis. Moreover, because etiologies of stroke and TBI differ substantially in their mechanisms and neuropathology of brain damage (for an overview on TBI pathology, see Gaetz, 28 and see Woodruff et al 29 and Durukan and Tatlisumak 30 for stroke), differential treatment effects of these etiological groups were also of interest.
Methods
Participants
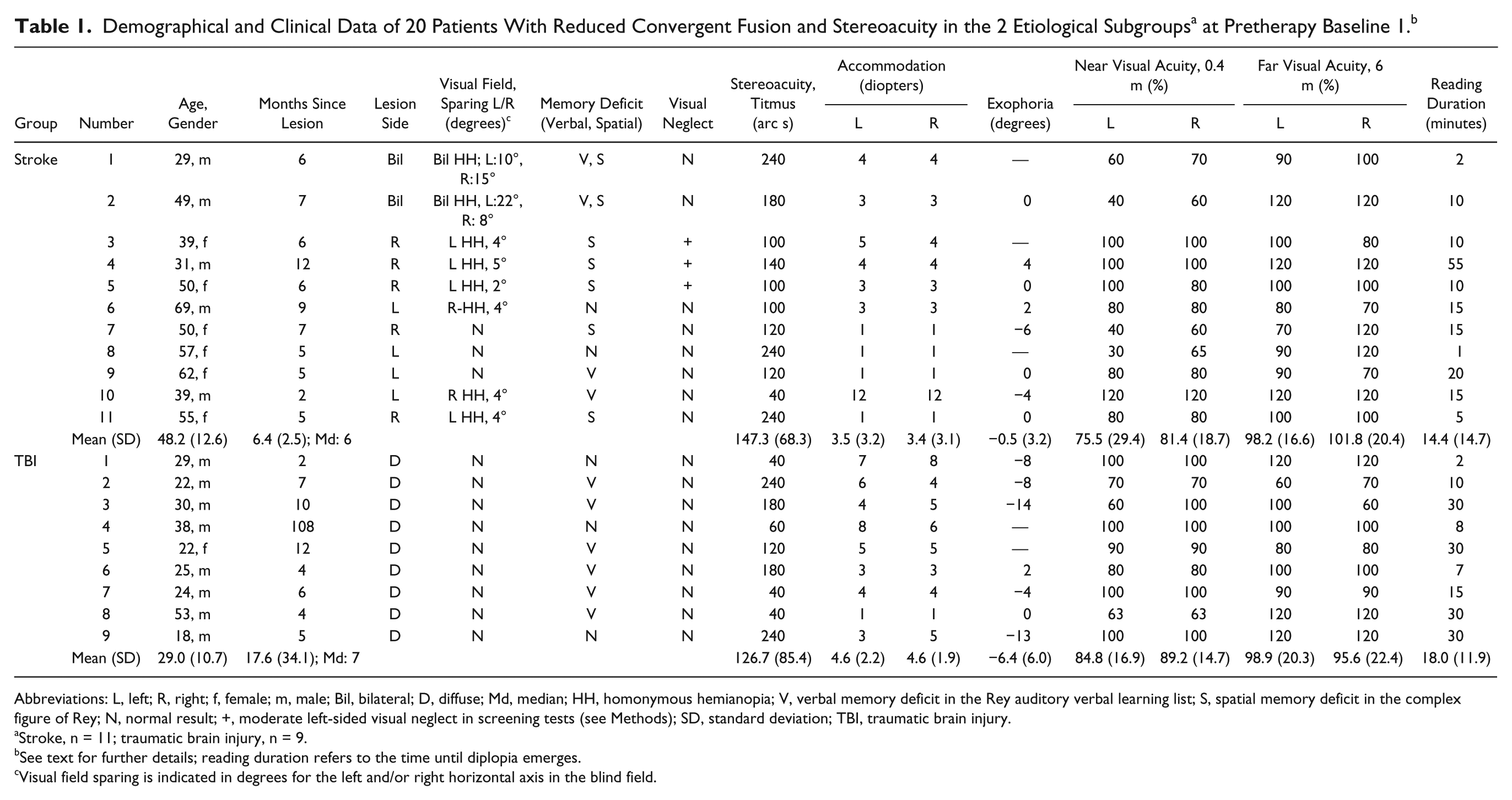
A total of 20 patients (13 men) with severely reduced convergent fusion resulting from unilateral or bilateral brain damage of cerebrovascular (n = 11) or traumatic (TBI; n = 9) origin participated. The participants were recruited from the neuropsychological outpatient department of Saarland University. All patients showing substantial problems with binocular vision (reduced fusion, stereoacuity, and subjective reading duration) that could not be explained by other neuropsychological or ophthalmological deficits were included. Participants with permanent diplopia and/or permanent blurred vision and patients with diseases of the anterior visual pathways were excluded. All participants were dominantly right-handed (mean laterality quotient = 85; range = 60-100 [German version of the Edinburgh handedness inventory] 31 ). Table 1 displays the demographic and clinical details for the 2 groups. The stroke group was significantly older than the TBI group: t(18) = 3.617; P = .002. There was no significant difference in lesion chronicity: t(8,073) = −0.984; P = .354.
Abbreviations: L, left; R, right; f, female; m, male; Bil, bilateral; D, diffuse; Md, median; HH, homonymous hemianopia; V, verbal memory deficit in the Rey auditory verbal learning list; S, spatial memory deficit in the complex figure of Rey; N, normal result; +, moderate left-sided visual neglect in screening tests (see Methods); SD, standard deviation; TBI, traumatic brain injury.
Stroke, n = 11; traumatic brain injury, n = 9.
See text for further details; reading duration refers to the time until diplopia emerges.
Visual field sparing is indicated in degrees for the left and/or right horizontal axis in the blind field.
Treatment Design
To control for spontaneous recovery on the patient’s outcome variables, a multiple baseline design was chosen 32 (see Figure 1). Three baseline measures of convergent motor fusion in the absence of any specific visual training were obtained in the pretherapy phase, 3 to 6 weeks before treatment onset (mean = 4 weeks). However, other relevant therapies (memory training and occupational therapy) were performed continuously during this period and the whole trial (on average 3 sessions at 50 min/wk).
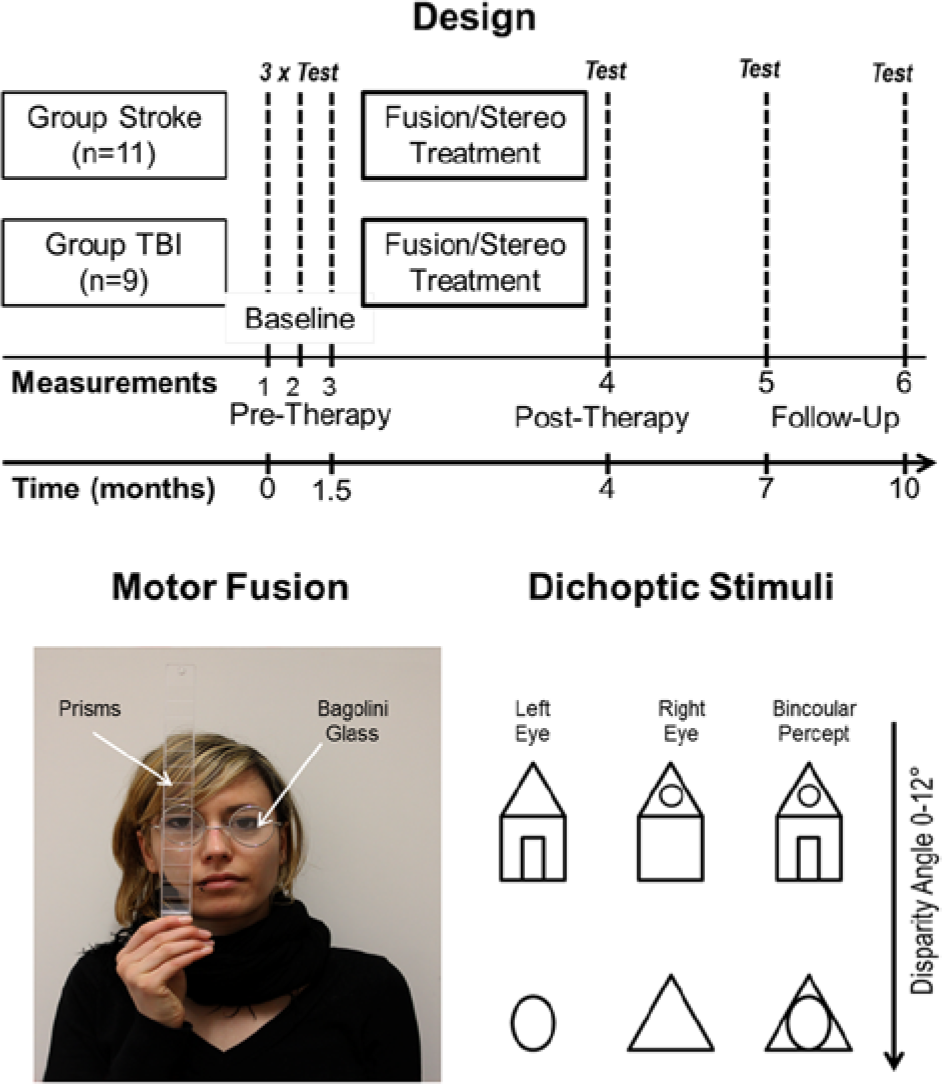
Top: Treatment design, treatment groups (stroke, n = 11; traumatic brain injury [TBI], n = 9), and time points of measurement (test) before fusional/stereotreatment (3 baselines, pretherapy), after treatment (posttherapy), and at 3 as well as 6 months after the end of treatment (follow-up). Bottom left: Measurement of convergent, horizontal fusional range by prisms with base out while the participant fixates a point light source at 5 m distance through a Bagolini glass for dichoptic viewing. Bottom right: Examples of stimuli used for dichoptic training. Different stimuli were viewed by the left and right eye, which resulted in a combined, “fused” binocular percept in the case of normal fusion. Disparity angles of the stimuli ranged from 0° to 12°, increasing with treatment progress.
After the third baseline test, fusion/stereotraining as described below was performed for 60 minutes, twice a week. On average, patients obtained 13.92 training sessions (range = 9-15; standard deviation [SD] = 1.53) within a period of 4 to 8 weeks. After the last training session, posttherapy assessment was performed. Finally, 2 follow-up assessments were conducted at 3 and 6 months after the end of training.
Oculomotor, Orthoptic, and Neuropsychological Assessments
All patients received a complete orthoptic assessment using their best correction during testing, including high-contrast letter visual acuity for each eye for the near (0.4 m) and far viewing distances (6 m, Visus GmbH, Stuttgart). Eye movements (saccades and pursuit eye movements), exophoria (see below), strabismus, and gaze palsies were assessed. No patient showed abnormalities of the anterior visual pathways, strabismus, any history of eye disease, or gaze palsies. Binocular visual fields were mapped using dynamic perimetry with a Tübingen bowl perimeter (see Lane et al 22 ; for a detailed description see Kuhn et al 15 ). To screen for visual neglect, 5 tests—comparable to the Behavioural Inattention Test 33 —were performed: horizontal line bisection, number cancellation, drawing a clock face from memory, figure copying (star, daisy, and face), and an indented reading test (180 words). Neglect was diagnosed when the midline deviated >5 mm ipsilesionally, 34 >1 target was omitted on one side in number cancellation, when numerals/picture parts were omitted/misplaced on the clock face’s/figure copy’s left side, or when >2 reading errors were committed. 35
Convergent Motor Fusion
Horizontal, convergent fusional range was assessed with prisms (range = 0-45 cm/m; Visus GmbH, Stuttgart).19,36 While 1 eye gazed at a light point (5 m distance) through a Bagolini glass separating the stimuli presented to each eye, prisms of increasing diopters (horizontal, base out, in steps of 2 diopters) were positioned in front of the other eye (see Figure 1). The value up to which both monocular images could be correctly fused, that is, seeing 1 single light point, is defined as convergent fusional range (expressed in cm/m, corresponding to prism diopter; 1 prism diopter = 0.5° of visual angle). A 2-fold seen light point indicates the convergent fusion threshold. To enhance reliability, 3 measurements were conducted during each session. Measurement variability was very small (2 cm/m), and the maximal value kept for at least 5 s without diplopia is reported.
To assess the patients’ values relatively to an unimpaired control sample, fusional range was also measured in 10 participants (3 men; age = 20-70 years) without any medical history of brain damage, strabismus, or anterior visual pathway diseases. The mean fusional range was 20 cm/m (range = 10-45 cm/m). The lower normative bound of convergent fusion was determined as 2 SDs below the mean value, resulting in a cutoff score of 15 cm/m (15 diopters), which is consistent with recent normative studies. 19
Additional Measures
To assess the transfer effects of fusion and stereotraining on other visual measures, the following assessments were performed.
Stereoacuity
Stereoacuity was assessed twice at pretherapy (2 measures), posttest, and follow-up 1 (3 months post training) in all participants by the Titmus test (Stereo Optical, Chicago, IL). It assesses local stereopsis, 37 using black, contoured circles viewed through Polaroid glasses to separate the stimuli presented to each eye. This contour-based measure of stereopsis was chosen because the majority of visual stimuli in our surrounding world contain visual contours. From a theoretical, perceptual learning-derived perspective, we realized later in the course of the study that it would be interesting to know whether global stereopsis could be improved as well by our contour-based treatment methods. This would indicate a transfer of training on higher stereoprocessing stages in the brain—that is, temporal-parietal cortices. Additionally, the first 3 (easiest) test items of the Titmus test allow correct results using monocular depth cues but not the items 4 to 9. Consequently, we assessed stereoacuity with the TNO test (Laméris Ootech BV, Ede, NL) once before and after fusion treatment in 5 patients (2 stroke and 3 TBI). It is important to note that the TNO test items cannot be solved by monocular cues. It tests global stereopsis without any monocular depth cues 37 by random-dot images with red-green glasses for image separation. 19
Subjective Reading Duration
Binocular reading duration was defined as the time (in minutes; recorded by stopwatch) until blurred vision/diplopia emerged while reading simple texts (Arial, point size 12, double-line spacing, black print on white paper). It was assessed twice during baseline at pretherapy, posttherapy, and follow-up 1.
Accommodation, Exophoria
Accommodation as a monocular control variable was measured with a standard accommodometer (Clement Clarke, London, UK). Normative, age-related values for each eye were available. 38 Exophoria was measured by a cover and prism cover test.
Treatment
Three treatment devices were used. First, patients practiced horizontal, convergent fusion with the prisms as described above using an amplitude they could just correctly fuse. The amplitude was increased when the patient was able to fuse both images for at least 2 minutes without diplopia. Second, a dichoptic device was applied 39 (see Figure 1, bottom). It displays 2 slightly different images to each eye, with increasing horizontal disparity (0°-12°), which have to be fused. Again, disparity angle was increased when the patient could fuse for at least 2 minutes. Third, a cheiroscope 40 was used. It laterally separates the visual image of the left eye by a mirror. With the left eye, the participant perceives a picture and with the other eye, a blank sheet of paper on which he or she has to retrace the projected picture. The basic principle is that the better the patient fuses both images, the more exact the copy appears. Patients practiced 5 minutes per picture, conducting 3 drawings in each therapy session. Short breaks of 1 to 2 minutes were given during treatment with every device. Each device was used for 20 minutes (including breaks) every session.
Data Analysis
To examine any impact of spontaneous recovery on convergent fusional range, stereovision, and reading duration during pretherapy, the baseline measures (convergent fusional range, stereoacuity, and subjective reading duration) in each group were analyzed by repeated-measures ANOVAs/t-Tests. Because pretherapy values did not differ significantly, averaged values were used for further analyses (see Results section, below). Analogously, a paired t test was performed for the 2 follow-up time points of assessment of fusional range. For the analysis of treatment effects on convergent fusion, stereovision (Titmus), and subjective reading time, 2 × 3 repeated-measures ANOVAs were conducted with the between-subjects factor etiology group (stroke, TBI) and the within-subjects factor treatment (averaged pretherapy, posttherapy, follow-up values, with averaged values for convergent fusional range). Post hoc Bonferroni-adjusted pairwise comparisons were computed for a more specific examination of significant results.
For accommodation and near and far visual acuity, a 2 × 2 mixed-design ANOVA was performed with the between-subjects factor etiology group (stroke, TBI) and the within-subjects factor treatment (baseline, immediately after training). Because exophoria was not evident in all patients, treatment effects were analyzed via paired t tests separately for the etiology groups. The α error was set at .05, 2-tailed.
Results
Convergent Motor Fusion
Because there was no significant change in fusional range during the baseline measures for either the stroke—F(1.362, 13.617) = 2.58, P = .125—or the TBI group—F(1.141, 9.127) = 0.675, P = .452—averaged pretherapy values were used for further statistical analysis. The same averaging procedure was conducted with the follow-up values: tstroke(10) = 1.170, P = .269; tTBI(8) = −0.555, P = .594.
There was a main effect of treatment: F(1.040, 18.718) = 35.146, P < .001, ηp2 = 0.661. Pairwise comparisons indicated a significant improvement between pretherapy (MStroke = 8.39, SDStroke = 4.54; MTBI = 8.37, SDTBI = 2.69) and posttherapy measurements (MStroke = 23.9, SDStroke = 10.55; MTBI = 14.00, SDTBI = 5.81), whereas there was no significant difference between the posttherapy and follow-up measures (MStroke = 22.50, SDStroke = 10.31; MTBI = 13.67, SDTBI = 6.82). Additionally, there was a significant interaction between treatment and etiology group—F(1.040, 18.718) = 7.109, P = .015, ηp2 = 0.283—showing that the stroke group had a higher training benefit than the TBI group, whereas the main effect of group barely failed statistical significance—F(1, 18) = 3.964, P = .062. Figure 2 illustrates the development of convergent fusional range for the 2 groups.
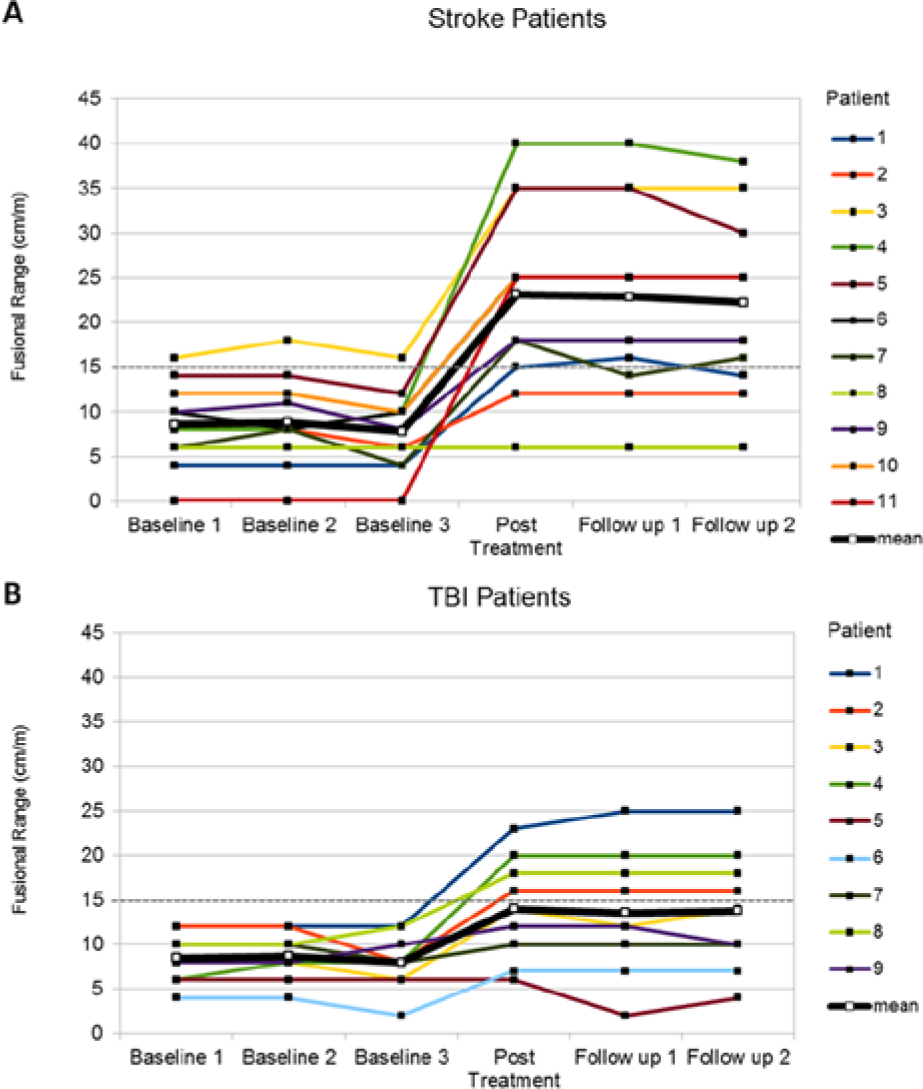
Mean values of convergent fusional range at pretherapy, posttherapy, and follow-up tests for the stroke (A) and traumatic brain injury (TBI) groups (B). The gray dotted line shows the lowest cutoff of the convergent fusional range in the healthy normative sample. Higher values indicate better motor fusion.
The different beneficial effects are also reflected in the total number of patients scoring below the a priori determined cutoff score of the normative sample: whereas 10 out of 11 stroke patients had a convergent fusional range <15 cm/m at pretherapy, at posttherapy and follow-up, the number diminished to 2. In contrast, in the TBI group, 5 (posttherapy) and 4 (follow-up) patients remained below the cutoff after training as compared with 9 of 9 participants scoring below cutoff at pretherapy.
Additional Measures
Stereoacuity
Titmus baseline values did not differ significantly (tStroke(10) = 1.491, P = .167; tTBI(8) = −0.359, P = .729). However, there was a main effect of treatment—F(2,36) = 24.734, P < .001, ηp2 = 0.579—across both groups. Post hoc pairwise comparisons displayed a significant improvement in stereoacuity between pretherapy (MStroke = 145.45, SDStroke = 69.77; MTBI = 127.78, SDTBI = 78.86) and posttherapy (MStroke = 76.36, SDStroke = 60.54; MTBI = 77.78, SDTBI = 66.67), but there was no further change at follow-up (MStroke = 76.36, SDStroke = 60.54; MTBI = 77.78, SDTBI = 66.67; see Figure 3). In the 5 patients who additionally performed the TNO test, global stereoacuity improved from 268 (range = 60-500; SD = 215.69) to 57 arc s (range = 15-120; SD = 40.25).
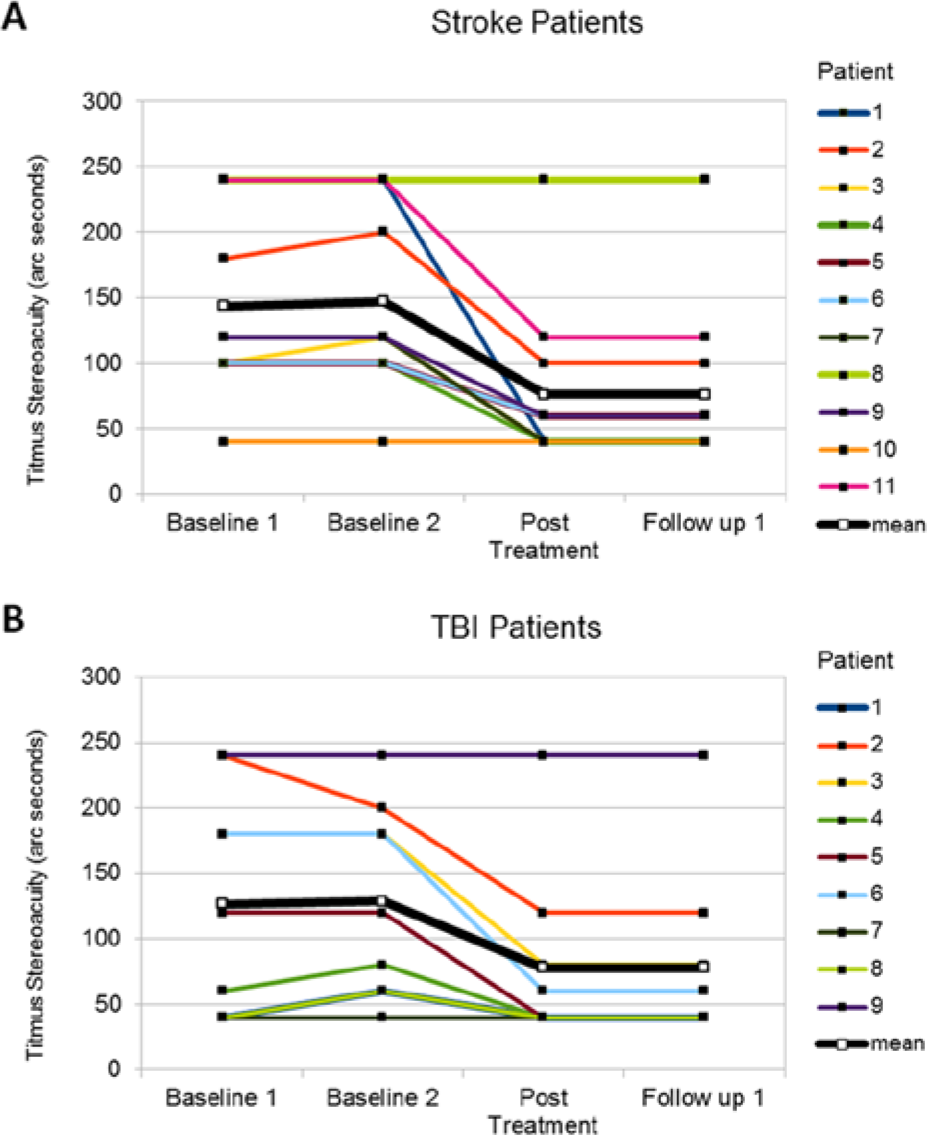
Mean values of the Titmus stereotest (arc s) at pretherapy, posttherapy, and follow-up for the stroke (A) and traumatic brain injury (TBI) groups (B). Lower values indicate better stereoacuity.
Subjective Reading Duration
There was no significant change in baseline measurements: tStroke(10) = −0.146, P = .887; tTBI(8) = 0.354, P = .733. Examination of treatment effects revealed a main effect of treatment: F(1.017, 18.308) = 56.590, P < .001, ηp2 = 0.759. Post hoc comparisons showed a significant increase in subjective reading duration between averaged pretherapy (MStroke = 14.41, SDStroke = 13.90; MTBI = 17.83, SDTBI = 11.73) and posttherapy measures (MStroke = 60.55, SDStroke = 44.05; MTBI = 103.33, SDTBI = 46.57); that effect remained stable until follow-up 1 (MStroke = 58.27, SDStroke = 42.89; MTBI = 102.22, SDTBI = 46.24). Besides this, the interaction between group and treatment was also significant—F(1.017, 18.308) = 5.348, P = .032, ηp2 = 0.229—indicating that the TBI group had a higher training benefit. Furthermore, the main effect—group—was marginally significant: F(1, 18) = 4.195, P = .055; see Figure 4.
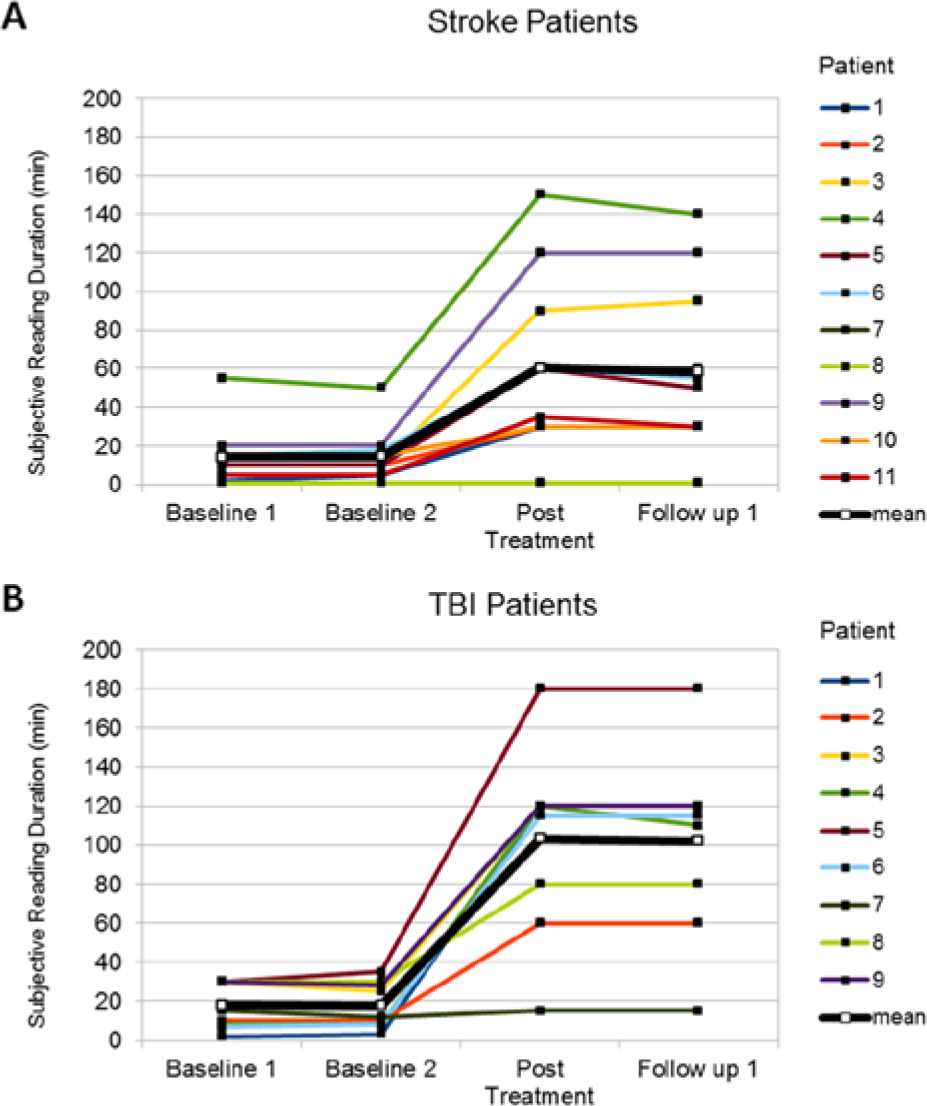
Mean values of subjective reading duration (minutes until diplopia occurred) at pretherapy, posttherapy, and follow-up for the stroke (A) and traumatic brain injury (TBI) group (B).
Accommodation
No significant changes were observed in accommodation before versus after training for either eye for the stroke and TBI group: FLeft eye(1,1 8) = 2.678, P = .113; FRight eye(1, 18) = 3.319, P = .085. According to Duane’s normative values, 38 accommodation was significantly reduced in the TBI group (lower cutoff: 6.8 diopters) but not in the stroke group (lower cutoff: 1.2 diopters). Because after the age of 45 the normal accommodative range is only 4 to 5 diopters, our stroke sample was not impaired in accommodation at pretherapy and did not change posttherapy.
Visual Acuity
Concerning near visual acuity, there was a main effect of treatment for both eyes (posttherapy values left eye: MStroke = 85.45, SDStroke = 24.23; MTBI = 94.44, SDTBI = 13.33; right eye: MStroke = 88.64, SDStroke = 15.181; MTBI = 100, SDTBI = 0.00), showing a slight improvement in both groups posttherapy (see Table 2): FLeft eye(1, 18) = 6.854, P = .017, ηp2 = 0.276; FRight eye(1, 18) = 8.687, P = .009, ηp2 = 0.326. Far visual acuity did not change significantly for either eye in any group (smallest P value: left eye, FLeft eye(1, 18) = 3.028, P = .099; see Table 2).
Mean (SD) of Near- and Far-Visual-Acuity Values (%) for the Stroke and TBI Groups Before and After Therapy.
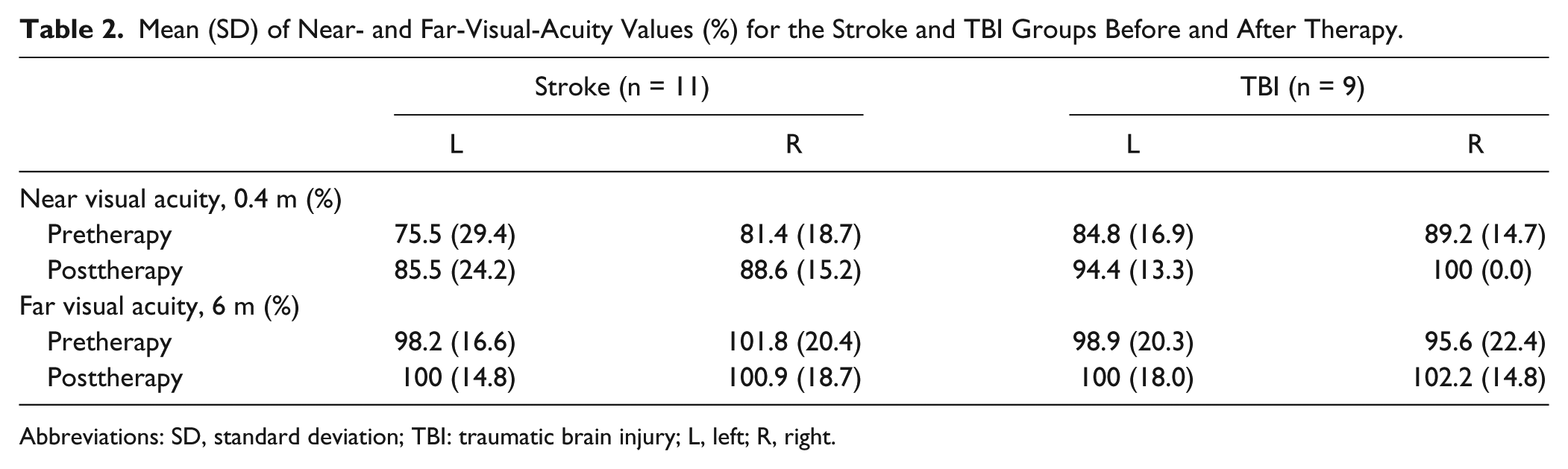
Abbreviations: SD, standard deviation; TBI: traumatic brain injury; L, left; R, right.
Exophoria
In all, 8 patients in the stroke group and 7 of the TBI sample (see Table 1) showed exophoria at pretherapy. There was no change in values for either patient in the stroke group. The TBI group showed a numerical improvement compared with pretherapy assessment (posttherapy values: MTBI = −0.31, SDTBI = 7.08), which failed statistical significance: t(6) = −1.566, P = .168.
Additional Analyses
Spearman rank correlations revealed a significant relationship between motor fusion and stereoacuity (Titmus; rsStroke = −0.775, P < .001; rsTBI = −0.331, P = .385) as well as between stereoacuity and reading duration (rsStroke = −0.702, P < .05; rsTBI = 0.108, P = .780) at pretherapy only for the stroke group. The average stereoacuity improvement (averaged baseline − posttest) correlated significantly with near-visual-acuity gain (posttherapy) for both eyes (left eye: rsStroke = −0.629, P < .05; rsTBI = −0.390, P = .299; right eye: rsStroke = −0.649, P < .05; rsTBI = −0.416, P = .265), again only for the stroke group. Lesion chronicity was not significantly correlated with average improvement of motor fusion, stereoacuity, or subjective reading duration in either sample (largest r = 0.353; smallest P = .287).
Because visual acuity may influence stereoacuity, we evaluated the impact of near-visual-acuity improvement on fusional and stereoacuity gain during treatment. Point-biserial correlations were computed between the change of binocular visual acuity (the higher monocular value served as the binocular value) and the average improvement in stereoacuity and motor fusion across both collapsed etiology groups. This revealed no significant results (r = −0.357, P = .08 for near-visual-acuity change and stereoacuity gain; r = −0.297, P = .150 for near-visual-acuity change and fusional gain).
Discussion
Motor Fusion
The present study investigated for the first time the effects of a novel perceptual retraining of binocular fusion and stereoacuity using dichoptic, graded treatments in patients with stroke or TBI. There was a large improvement of convergent motor fusion in both groups, resulting in a 3-fold increase in the stroke group and an almost doubled value in the TBI group after treatment. These results remained stable at 2 follow-up tests, revealing long-term stability of the treatment. Interestingly, the stroke group had a significantly higher training benefit than the TBI group, whereas the main effect of group was marginally significant. This differential outcome might be explained by the neural damage typically caused by TBI, which is usually not restricted to a circumscribed cortical area but causes extensive shearing injuries, often also affecting the brainstem.41,42 Because the midbrain is crucially involved in motor fusion, 2 the putative involvement of this region in the TBI sample is the most plausible explanation for the lower training benefit. Notwithstanding this difference, it is remarkable that 7 out of 11 patients with stroke and 4 out of 9 with TBI reached the normal range for motor fusion after treatment. Moreover, there was no improvement during the pretherapy phase despite triple measurements and despite continuous binocular visual input to the patients, thus ruling out artefacts caused by retesting, spontaneous recovery, or unspecific visual stimulation. Obviously, the treatment effect requires repetitive and specific binocular exercises with graded task difficulty individually adapted to the respective fusional and stereocapacity.
Stereoacuity
Apart from the treatment-related improvement in motor fusion, stereoacuity, which relies partly on binocular fusion, also improved significantly and permanently in both samples. This was seen in the Titmus test (local stereopsis) and also in all 5 patients performing the TNO test (global stereopsis). It is important to note that these improvements were not related to the observed, slight increases in near visual acuity but occurred as well in participants who showed no improvement in near visual acuity. This excludes the possibility that training-related increases in stereoacuity were merely induced by better visual acuity after training. Rather, the slightly improved near visual acuity was an additional effect of the visual training that was performed in the near distance when training with the cheiroscope/dichoptic stimuli.
One potential explanation for the treatment effect may reside in the multiplicity of brain regions dealing with disparity information. 43 This hypothesis is supported by imaging studies44,45 that reveal several cortical areas (early visual as well as dorsal and ventral stream areas) associated with disparity information coding. Accordingly, damage to some area may be partially compensated by recruitment of unlesioned brain areas involved in disparity coding. The pharmacologically induced recovery of binocular vision in cats with bilateral occipital lesions 26 is in agreement with this hypothesis.
Reading Duration, Accommodation, and Visual Acuity
Because reduced reading duration is one of the most typical complaints of patients with fusional disorders, 12 we hypothesized a priori that alongside with an improved motor fusion, reading duration should also increase. The time until diplopia emerged could be considerably prolonged in both samples by treatment (5-fold in the TBI group; 4-fold in the stroke group), an effect that was not observed during baseline measures. Interestingly, the TBI group showed a significantly higher gain on reading time than the stroke group, which is the opposite effect observed for fusional gain. Again, the main effect of group only barely failed statistical significance. The differential improvement may be partly related to the fact that the stroke group was approximately 20 years older than the TBI group. This age difference is congruent to epidemiological and incidental evidence, which report that TBIs occur more frequently in earlier decades than stroke.46-48 Furthermore, aging is accompanied by multiple changes in sensory and cognitive functions.49,50 Concerning reading, peripheral visual parameters showing age-related impairments such as changes in cornea curvature or thickening of the lens 51 might explain the lower gain of our stroke group.
The significant improvement in near, but not far, visual acuity might on the one hand be attributed to the fact that convergent fusion primarily has an impact on near-space vision. In addition, the majority of treatment devices (cheiroscope and dichoptic stimuli) were applied at the near distance. On the other hand, training by prisms was conducted at a 5-m distance. This might be related to the slight, though nonsignificant improvement in far visual acuity after training.
Finally, no improvements were seen in accommodation, which was only impaired in the TBI group. Hence, the improvements in motor fusion were highly specific for binocular fusion and stereovision, with no significant impact on monocular, accommodative responses. This is in agreement with the anatomy of the accommodative system showing only partial overlap with systems for convergent fusion/stereovision, especially where occipital and cerebellar structures are concerned. 27
Relation to Visual Field Defects
The frequent although not strict association of homonymous visual field defects with disturbed motor fusion and stereoacuity in our stroke sample is striking, replicating the findings of Danta et al 7 with respect to stereoacuity. In our study, 8 out of 11 patients with stroke (72.7%) showed scotomas. This correspondence may result from (a) neighboring lesion areas and/or (b) the fact that any scotoma may hamper the solution of the stereo-correspondence problem—that is, that one of the disparate visual images of an object may fall into the blind field. Because the visual fields of our patients remained unchanged after fusion/stereotraining, the brain is obviously capable of solving this problem despite persisting hemianopia.
In conclusion, we developed and successfully evaluated in this proof-of-principle study a novel treatment that significantly improved motor fusion, stereoacuity, and reading duration, thus reducing diplopia in stroke or TBI patients. Moreover, the rather small number of only 14 treatment sessions indicates an economical method. These results show a substantial treatment-induced plasticity of the lesioned brain in the perceptual relearning of fusion and stereovision and provide new, effective rehabilitation strategies to treat deficits of binocular vision after brain damage.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Georg Kerkhoff receives funding from the Deutsche Forschungsgemeinschaft (DFG), International Research Training Group IRTG 1457 Adaptive Minds.