Abstract
Background. Mild traumatic brain injury (mTBI) and residual postconcussion syndrome (PCS) are common among combatants of the recent military conflicts in Iraq and Afghanistan. Hyperbaric oxygen (HBO2) is a proposed treatment but has not been rigorously studied for this condition. Objectives. In a secondary analysis, examine for possible effects on psychomotor (balance and fine motor) and cognitive performance 1 week after an HBO2 intervention in service members with PCS after mTBI. Methods. A randomized, double-blind, sham control, feasibility trial comparing pretreatment and posttreatment was conducted in 60 male active-duty marines with combat-related mTBI and PCS persisting for 3 to 36 months. Participants were randomized to 1 of 3 preassigned oxygen fractions (10.5%, 75%, or 100%) at 2.0 atmospheres absolute (ATA), resulting in respective groups with an oxygen exposure equivalent to (1) breathing surface air (Sham Air), (2) 100% oxygen at 1.5 ATA (1.5 ATAO2), and (3) 100% oxygen at 2.0 ATA (2.0 ATAO2). Over a 10-week period, participants received 40 hyperbaric chamber sessions of 60 minutes each. Outcome measures, including computerized posturography (balance), grooved pegboard (fine motor speed/dexterity), and multiple neuropsychological tests of cognitive performance, were collected preintervention and 1-week postintervention. Results. Despite the multiple sensitive cognitive and psychomotor measures analyzed at an unadjusted 5% significance level, this study demonstrated no immediate postintervention beneficial effect of exposure to either 1.5 ATAO2 or 2.0 ATAO2 compared with the Sham Air intervention. Conclusions. These results do not support the use of HBO2 to treat cognitive, balance, or fine motor deficits associated with mTBI and PCS.
Keywords
Introduction
An estimated 19% of US military personnel have sustained a traumatic brain injury (TBI) during deployment in Operations Enduring Freedom (OEF), Iraqi Freedom (OIF), and/or New Dawn. More than 266 000 TBI casualties have been reported by the Department of Defense between 2000 and 2013, more than 80% of which are mild. 1 Additionally, many in the military have had repetitive blast exposures, often with associated head trauma and cumulative brain injury that may further complicate recovery. 2 There is considerable evidence that many of these individuals have chronic dysfunction in the form of postconcussion syndrome (PCS). Several studies have reported PCS-like symptoms among veterans many years after mTBI, whether blast induced or non–blast induced.2,3-6 Symptoms commonly reported by veterans undergoing TBI evaluation include irritability, sleep disturbance, forgetfulness, anxiety, headaches, poor concentration, and pain. 3 Psychological distress is also highly associated with PCS in this population.6-8 An examination of national Veterans Health Administration data revealed that mental health comorbidities were present in 89% of OEF/OIF veterans diagnosed with TBI, with the most commonly reported being posttraumatic stress disorder and depression. 9 Chronic pain is also frequently associated with mTBI.9-12 The presence of multiple co-occurring symptom clusters has been termed postdeployment multisymptom disorder or more commonly post-deployment syndrome 13 and may be one of the reasons for the higher rate of persistent PCS in military personnel compared with civilians. 9
In addition to bothersome symptoms of pain and distress, objective neurological deficits are common after mTBI and have been documented with PCS. Neurocognitive impairment is the most pervasive chronic impairment after all severities of TBI.14-17 Cognitive impairment is well documented acutely after mTBI, and there is evidence of persisting cognitive performance deficits after mTBI, especially in attention and working memory.18-21 Objective cognitive impairment is included among the criteria for PCS in the Diagnostic and Statistical Manual of Mental Disorders, 4th edition (DSM-IV). Other neurological impairments known to occur after mTBI include fine motor and balance deficits. Balance disorders are well documented to occur acutely after mTBI,22,23 and postural instability after mTBI has also been described in select military samples.23-26 Among the National Institute of Neurological Disorders and Stroke (NINDS)–recommended outcome measures for TBI are tests of fine motor speed and dexterity. 27
The large number of veterans and service members (SMs) suffering from chronic PCS-like symptoms has led to an emphasis on mTBI treatment, spurring the creation of Military Heath System’s TBI clinics, the National Intrepid Center of Excellence (NICoE) satellite TBI clinics, and the VA Polytrauma System of Care. Unfortunately, to date, there are few empirically based treatments to offer. The standard of care is a simple approach, addressing the symptoms such as pain, depression, and insomnia. There are no proven medications or other interventions to treat the underlying brain injury.
A potential intervention that has increasingly been advocated is hyperbaric oxygen (HBO2). HBO2 administration involves breathing high levels of oxygen, usually 100% oxygen, at a pressure at least 1.4 times greater than the atmospheric absolute pressure at sea level (1 atmosphere absolute or ATA, which is equivalent to 760 mm Hg partial pressure of oxygen). 28 The intent of HBO2 therapy is to increase the oxygenation of the patient’s blood and tissues to supraphysiological levels. The partial pressure of oxygen will increase proportionally with an increase in the hyperbaric chamber compression pressure. One theory proposed by advocates to support its use in TBI patients is that exposure to HBO2 allows functionally retrievable neurons adjacent to severely damaged or dead neurons to return to more normal function by reactivating metabolic or electrical pathways. 29
In an effort to evaluate the efficacy and utility of HBO2 for the PCS seen in US combatants, the Departments of Defense and Veterans Affairs collaboratively supported 3 independent, randomized, blinded trials in this population. 30 In the first study, a randomized, controlled trial using 2.4 ATA HBO2 exposure compared with sham (room air at 1.3 ATA), Wolf et al 31 found no benefit in symptoms among SMs with PCS. The second phase of the VA/DoD research initiative focused on the effect of 1.5 and 2.0 ATA HBO2 equivalent exposures. In this recently completed 3-arm, randomized, blinded, sham-controlled feasibility trial in SMs with mTBI and PCS, Cifu et al 32 found no immediate (1-week postexposure) benefit on the primary outcome measure for PCS symptoms—the Rivermead Post-Concussion Questionnaire. Here, we report findings at 1 week postexposure among the secondary outcome measures from this trial. The objective was to determine the immediate postintervention effects of HBO2 in SMs with PCS after mTBI on psychomotor (balance and fine motor) and cognitive performance in order to explore potential benefits that might inform future larger studies. We hypothesized that HBO2 would lead to immediate postcompression improvements in cognitive and psychomotor performance as demonstrated by group difference between treated and untreated SMs.
Materials and Methods
The Defense Advanced Research Projects Agency and US Navy Bureau of Medicine and Surgery (as part of the DoD-VA collaborative research program) sponsored this single-center, 3-arm, randomized, blinded, sham-controlled trial of HBO2 exposure on symptomatic mTBI. All appropriate institutional review board and governmental approvals were obtained. The sample size estimates (n = 60) were calculated for a 10% difference to be found among the 3 groups on the primary outcome—postconcussive symptom severity as measured by the Rivermead Post-Concussion Questionnaire. 33 Inclusion criteria were TBI-specialist-confirmed diagnosis of mTBI based on the DoD definition of TBI, 34 postconcussive symptoms from mTBI for at least 3 months, injury occurrence in the past 3 years, stable psychiatric status for 2 months, stable psychotropic medication history for at least 1 month, and ability to undergo computerized testing. The diagnosis of TBI was confirmed by the study physiatrist’s history, physical examination, and a review of all the acute medical records. The only exclusion criteria were the presence of a disorder that contraindicated hyperbaric exposure or previous exposure to HBO2. Participants were active-duty US Marines recruited primarily from Marine Base Camp Lejeune, North Carolina.
Using computer-generated random numbers, participants were randomized as blocks of 12 participants to 1 of 3 conditions in the hyperbaric chamber, with each group receiving 40 total exposures. Once assigned to a particular exposure group, the participants’ experimental condition did not vary over the 40-exposure course. Exposures in this study were delivered using modifications of established protocols developed by the Navy’s Bureau of Medicine and Surgery Undersea Medicine department.31,35 The intervention dosing used in this study was chosen based on the consensus opinion of the DoD and VA. 30 To ensure participant and investigator blinding to the specific treatment exposure being received, all participants were pressurized inside the chamber to 2.0 ATA, which is equivalent to the atmospheric pressure attained during underwater diving to 33 feet of seawater. Participants breathed 1 of 3 oxygen-nitrogen gas mixtures blended to produce the oxygen exposure condition to which they were assigned. These were as follows: (1) the sham air group breathed 10.5% oxygen (balance 89.5% nitrogen) at 2.0 ATA; (2) the 1.5-ATA oxygen group breathed 75% oxygen (balance 25% nitrogen) at 2.0 ATA; and (3) the 2.0-ATA oxygen group breathed pure oxygen (0% nitrogen) at 2.0 ATA. These groups are hereafter referred to as Sham Air, 1.5 ATAO2, and 2.0 ATAO2, respectively. Chamber compression to 2.0 ATA generally required less than 3 minutes to attain. Once at 2.0 ATA of pressure, each participant breathed the assigned gas mixture for a period of 60 minutes (±1 minute). Exposures were conducted in a multiplace chamber with the breathing medium delivered via oxygen treatment hoods at gas flow rates of ≥20 L/min to ensure a consistent oxygen level exposure (Amron International Inc, Vista, CA). Chamber decompression to 1.0 ATA (ie, an average room air pressure of 759 mm Hg) similarly required less than 3 minutes to attain. During compression to and decompression from 2.0 ATA, all participants breathed ambient chamber air. The oxygen content of chamber air was closely regulated to remain between 19% and 23.5% surface equivalent. Any participant unable to complete a scheduled treatment because of transient contraindications to hyperbaric chamber exposure (ie, fevers, congestion, and inability to equalize sinus or ear pressure) was allowed to make up the missed treatment at the next available opportunity (eg, later the same day or on weekends, when exposures were not normally scheduled). No participant received more than 1 hyperbaric exposure per day.
This analysis focuses on the effects of these exposures on cognitive and psychomotor performance measures at 1 week following the last exposure. The administration time for the entire battery of cognitive and motor testing was about 2½ hours. Fine-motor speed and control were measured using the Grooved Pegboard. 36 Postural stability and balance were measured with computerized posturography (CPT) on dual-plate force platform, the NeuroCom Smart Balance Master(r) (NeuroCom; NeuroCom International, Inc, Clackamas, OR). The specific CPT test given was the Sensory Organization Test (SOT). The SOT is a composite index that defines abnormalities across somatosensory, visual, and vestibular systems. 37 In this, 6 equilibrium scores corresponding to the 6 sensory condition tasks are calculated from the patients’ performances on 3 trials per condition. The sensory conditions are as follows: (1) eyes open with a fixed surface and visual surroundings, (2) eyes closed with a fixed surface, (3) eyes open with a fixed surface and sway-referenced visual surroundings, (4) eyes open with a sway-referenced surface and fixed visual field, (5) eyes closed with a sway-referenced surface, and (6) eyes open with a sway-referenced surface and visual surroundings. SOT scores range from 0 (touching a support surface, shifting feet, or falling) to 100 (little or no sway). The composite equilibrium score is a weighted average of the 6 equilibrium scores and is an index of overall performance on the SOT (0-100).
Neuropsychological tests of cognitive performance were chosen for high sensitivity to attention, memory, and processing speed deficits and efficiency of administration. All tests are standardized and validated with proven reliability: Wechsler Test of Adult Reading 38 ; Conners Continuous Performance Test-II 39 ; Paced Auditory Serial Addition Test (PASAT) 40 ; Halsted-Reitan Trail Making Test A & B 41 ; Stroop color-word interference test 42 ; California Verbal Learning Test-II (CVLT-II) 43 ; Wechsler Adult Intelligence Scale III (WAIS-III) select items: digit symbol coding, digit span, letter-number sequencing, symbol search, and arithmetic 44 ; Delis-Kaplan Executive Function Systems (DKEFS) version of the Controlled Oral Word Association Test (COWAT) single-letter and category items 45 ; and Benton Visual Memory Test–Revised (BVMT-R). 46 Additionally, the Test of Memory Malingering (ToMM) was administered to assess validity of participant effort on cognitive performance testing. 47 All selected tests are recommended by the National Institute of Neurological Disorders (NINDS) TBI Comprehensive Evaluation Common Data Elements, and details on the psychometric properties with references for each test can be found at that Web site. 27
Given the multiple subtest results generated on most cognitive tests, prespecified secondary measures were chosen for each outcome domain of interest as follows: verbal fluency using COWAT Letter Fluency, executive function using Trails B, working memory using WAIS III Working Memory Index, 48 visual attention using Stroop Color-Word Interference, sustained visual attention using CPT-II Detectability Index, auditory attention using PASAT 2.0 pacing, delayed verbal memory using CVLT Total Long Delay Free Recall and Recognition Discriminability Index, 49 and delayed visual memory using BVMT-R Delayed Recall Total and Recognition Discrimination Index. Similarly, the prespecified balance measure was the Composite Equilibrium Score. Given the feasibility designation of this study, exploratory analysis was also performed on all cognitive subtests as well as balance SOT conditions 1 to 6. For each secondary and exploratory measure, any participant having an invalid prescore or postscore was excluded from that particular analysis. Cognitive testing performance validity was measured for each participant at both testing sessions using the ToMM. ANOVA was used for all between-group comparisons. Raw scores were utilized for all neuropsychological tests, excepting index scores where indicated. Multiple significance test correction was not applied to these analyses because the study was not specifically powered for the secondary outcomes. Instead, an unadjusted α = .05 was used for defining significance on each measure; trends (P < .15) were sought, and all P values are reported in full rather than dichotomously. Finally, all significant findings (P < .05) were examined with post hoc independent-sample t tests to identify which simple effects were significant.
Results
Participant Characteristics
A total of 128 SMs met preliminary study eligibility and consented for evaluation. Of 128 candidates, 61 met the full study criteria, consented to the experimental intervention, and were randomly assigned into the Sham Air control or 1 of the 2 HBO2 exposure groups. The primary reasons for exclusion were the inability to confirm the diagnosis of mTBI, active medication changes, and schedule conflicts. One participant was unavailable for the 1 week postintervention assessment, leaving a total of 60 participants for the present analysis. Following drop out of this single participant, the final groups consisted of 21 participants in the Sham Air compression group, 18 in the 1.5 ATAO2 equivalent group, and 21 in the 2.0 ATAO2 equivalent group. The 60 participants were all male and had a mean age of 23.2 years (standard deviation [SD] = 2.95). Two (3.0%) were African American, 47 (78.3%) were Caucasian, 10 (16.6%) were Hispanic, and 1 (1.6%) was Native American. Of the 60, 19 were married, 3 were divorced, and 38 were single. Pay grades E1 (Enlisted, Marine Private) to E6 (Enlisted, Marine Staff Sergeant) comprised 97% of the sample. ANOVA and χ2 analysis revealed no between-group differences with respect to age, pay grade, marital status, or race/ethnicity. Slightly more than a quarter of the participants had self-reported concussions (mean = 2.1; SD = 0.95; range = 1-4) prior to the most recent injury, with the only or most recent TBI occurring a mean of 8.5 months (SD = 6.58 months; range = 3-39 months) prior to the baseline assessments. Etiology of concussion included improvised explosive device blast (85.3%), rocket propelled grenades (3.0%), and mortar attacks (1.7%). The remaining 10% were uncategorized blasts. There were no precompression between-group differences on any participant demographic or historical characteristics.
Psychomotor Performance Measures
Because of scheduling conflicts, 2 participants were missing Grooved Pegboard scores and 3 participants were missing balance testing scores at either precompression or postcompression. Psychomotor performance results for the remaining 58 and 57 participants, respectively, are displayed in Table 1. There were no between-group differences for either the dominant or nondominant hand on the Grooved Pegboard and no differences on SOT Composite Equilibrium scores at either preintervention or postintervention.
Psychomotor Outcomes.
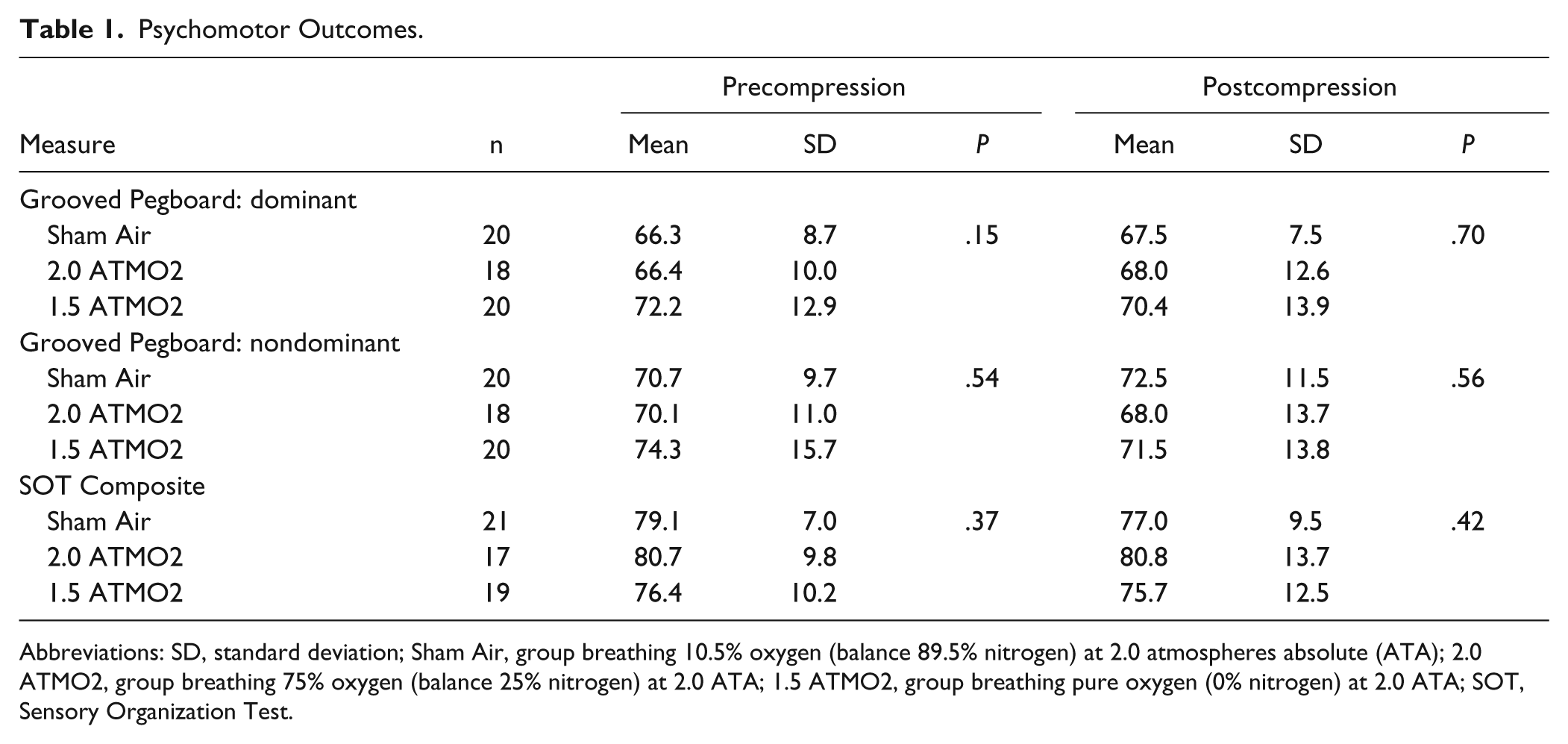
Abbreviations: SD, standard deviation; Sham Air, group breathing 10.5% oxygen (balance 89.5% nitrogen) at 2.0 atmospheres absolute (ATA); 2.0 ATMO2, group breathing 75% oxygen (balance 25% nitrogen) at 2.0 ATA; 1.5 ATMO2, group breathing pure oxygen (0% nitrogen) at 2.0 ATA; SOT, Sensory Organization Test.
Cognitive Performance Measures
Using traditional cutoffs, 10 participants failed performance validity testing on the ToMM and were excluded from the cognitive performance analyses. 50 Executive function and attention performance results for the remaining participants are displayed in Table 2. There were no preintervention or postintervention between-group differences in DKEFS (COWAT) Letter Fluency, Trails B, Stroop Color-Word Interference, CPT-II Detectability Index, or PASAT 2.0 pacing.
Cognitive Outcomes: Attention and Executive Function.
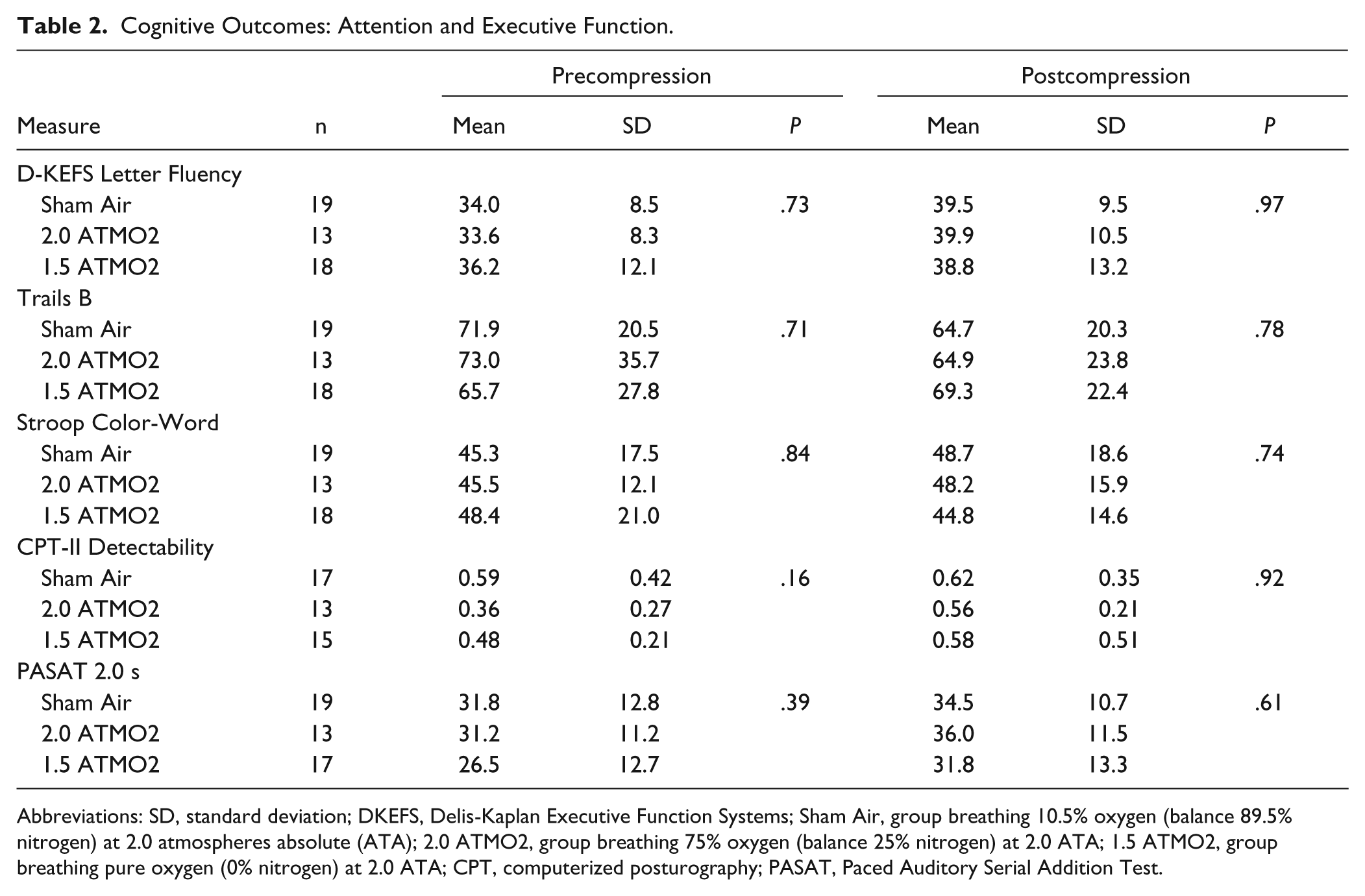
Abbreviations: SD, standard deviation; DKEFS, Delis-Kaplan Executive Function Systems; Sham Air, group breathing 10.5% oxygen (balance 89.5% nitrogen) at 2.0 atmospheres absolute (ATA); 2.0 ATMO2, group breathing 75% oxygen (balance 25% nitrogen) at 2.0 ATA; 1.5 ATMO2, group breathing pure oxygen (0% nitrogen) at 2.0 ATA; CPT, computerized posturography; PASAT, Paced Auditory Serial Addition Test.
Working memory and delayed memory performance results are displayed in Table 3. There were no preintervention or postintervention between-group differences in WAIS III Working Memory Index, Stroop, BVMT-R Delayed Recall total, or BVMT-R Recognition Discrimination Index. The 2 CVLT scores showed group differences at preintervention but not postcompression. Post hoc t test showed that the 1.5 ATAO2 group was significantly different from the 2.0 ATAO2 group at baseline on both the CVLT Long Delay Free Recall (P = .012) and CVLT Index d’ for Recognition (P = .021), and neither treatment group was significantly different from Sham Air.
Cognitive Outcomes: Memory and Working Memory.
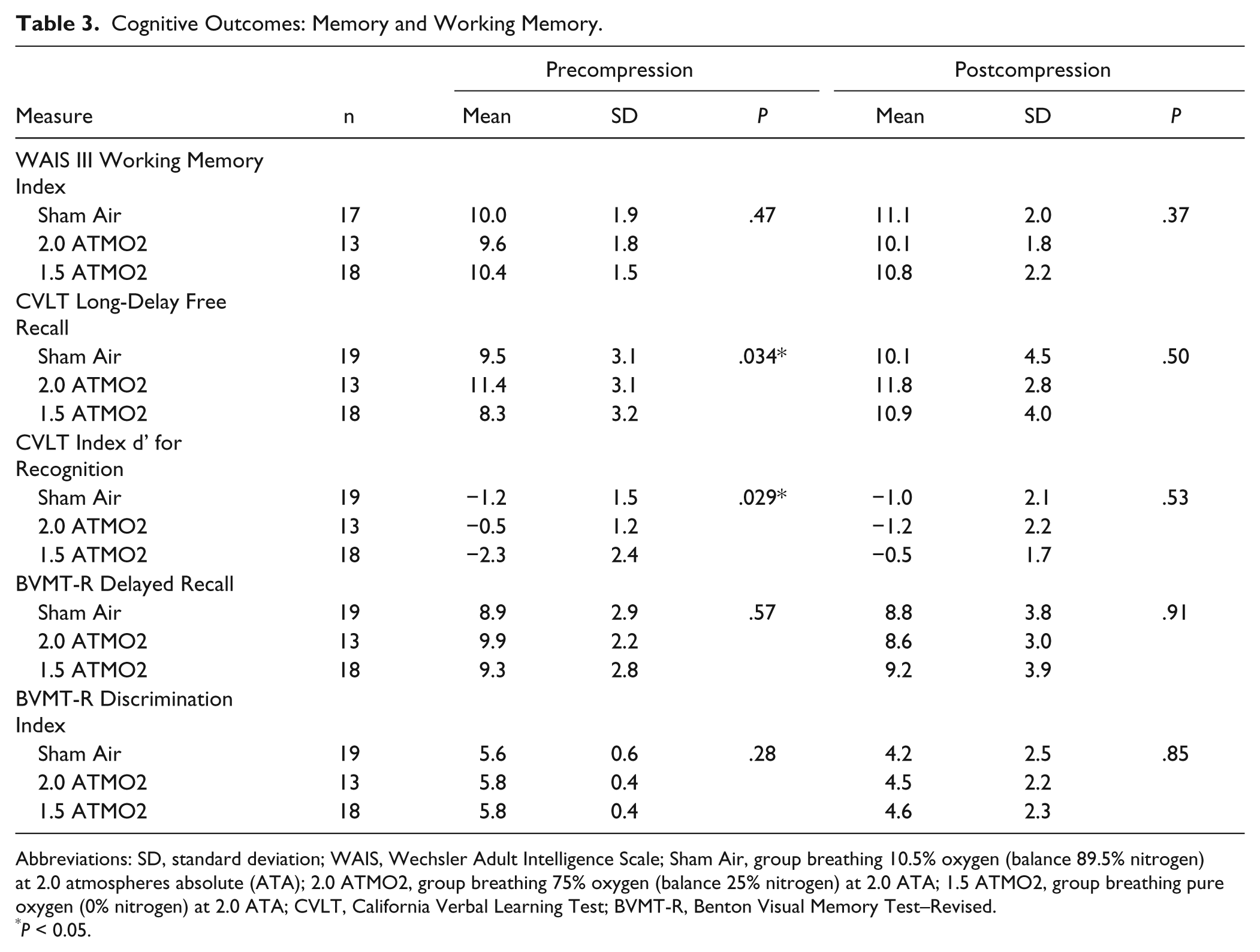
Abbreviations: SD, standard deviation; WAIS, Wechsler Adult Intelligence Scale; Sham Air, group breathing 10.5% oxygen (balance 89.5% nitrogen) at 2.0 atmospheres absolute (ATA); 2.0 ATMO2, group breathing 75% oxygen (balance 25% nitrogen) at 2.0 ATA; 1.5 ATMO2, group breathing pure oxygen (0% nitrogen) at 2.0 ATA; CVLT, California Verbal Learning Test; BVMT-R, Benton Visual Memory Test–Revised.
P < 0.05.
Results of the additional exploratory analyses are shown in the appendix (Tables A.1 to A.4). Once again, there were no significant between-group differences at postcompression on any of the balance or cognitive measures. Two tests showed significant differences at precompression: CVLT Short Delay Cued Recall (P = .037) and CVLT Recognition Total Hits (P = .005). Post hoc t tests showed that the differences for baseline CVLT Short Delay Recall were significant (P = .014) between the 1.5 ATAO2 and the 2.0 ATAO2 groups only, whereas the Recognition Total Hits showed differences between the 1.5 ATAO2 and 2.0 ATAO2 exposure groups (P = .006) and also between the 1.5 ATAO2 and Sham Air groups (P = .035).
Given the limited sample size and feasibility designation of the study, nonsignificant postcompression between-group trends (P < .15) were also examined. No trends existed for any of the prespecified secondary measures. Within the exploratory measures, only the WAIS III Processing Speed Index showed a trend (P = .082) in between-group difference postcompression; however, the mean was nominally lowest (slower) in the 1.5 ATAO2, and a similar trend existed at precompression.
Discussion
This is the second DoD-VA collaborative, randomized, double-blinded, sham-controlled feasibility trial studying the effects of HBO2 on PCS. From analysis of the present clinical trial, Cifu et al 32 separately reported the immediate, 1-week postcompression effects on the primary outcome symptom measures. We now report the findings on the secondary cognitive and psychomotor performance measures. The goal of the current analyses was to evaluate the effects of HBO2 on the neurological impairments that are common after mild TBI and may accompany the nonspecific symptom complaints associated with PCS. Comprehensive testing was done and included measures known to be sensitive to the subtle impairments that are typical of PCS, such as indices of complex attentional control,51,52 delayed recognition memory,49,53 memory proactive interfence, 52 and CPT.
The only between-group differences in major outcomes were found at precompression on a subset of CVLT score—a cognitive test of auditory verbal memory. Despite the multitude of prespecified cognitive and psychomotor secondary outcome measures analyzed at an unadjusted α = .05 level, there were no significant immediate postcompression differences between the randomized groups. There was only 1 exploratory cognitive test that showed nonsignificant trends of group differences at 1 week postcompression, but it was confounded by the presence of a similar trend at pretreatment. We postulate the findings to be best explained by chance effects of randomization (type 1 error) because patterns of improvement postcompression suggest the normalization of these chance pretreatment group differences.
Our study findings are in contrast to a recent uncontrolled HBO2 trial of 16 participants with PCS after blast exposure during military service that showed improvement in not only symptoms but also cognitive and psychomotor characteristics. 54 One week after HBO2 at 1.5 ATA, Harch et al 54 reported significant improvement in full-scale IQ (+14.8 points; P < .001), Wechsler Memory Scale (WMS) IV Delayed Memory (P = .026), WMS-IV Working Memory (P = .003), Stroop Test (P < .001), Test of Variables of Attention (TOVA) Impulsivity (P = .041), TOVA Variability (P = .045), and Grooved Pegboard (P = .028). These divergent findings are likely accounted for by our study testing for between-group differences and not for single-group within-group change. The lack of a sham control restricted Harch et al 54 to assessment of within-group differences only. Given the higher level of scientific rigor in the current study, the best explanation is that the improvements found by Harch et al 54 are a result of practice effect, placebo effect, and/or the nonspecific effects of attention and care.
This trial represents the second randomized, double-blinded, sham-controlled, prospective feasibility study of HBO2 in the symptomatic chronic mTBI population that demonstrates no significant symptomatic improvements of HBO2 at either 1.5 or 2.0 ATAO2 equivalents over the Sham Air control. The strength of this study is that it incorporated many features lacking in prior studies, including randomization, blinding, and control groups. An additional strength is the use of a performance validity check on cognitive testing, with exclusion of those failing the test. Some of the participants could have been motivated by secondary gain to score poorly on testing, especially given the extended time in Pensacola with minimal responsibilities associated with study participation. These current findings, which parallel those of the primary symptom measures used by both Cifu et al 32 and Wolf et al, 31 are particularly important, in that this study utilized the typical treatment pressures advocated by hyperbaric clinicians.54,55
The multitude and breadth of cognitive performance testing included in this study represents both a strength and weakness. We sought to be expansive and include tests highly sensitive to the effects of mild TBI, especially both auditory and visual complex attention and delayed memory, so as to not miss any objective improvements. The main limitation of this approach was the increased chances of type 1 error, which is false-positive between-group differences. Given that the sample size was powered only for the primary outcome, we chose not to apply corrected α levels for the multitude of secondary outcome analyses. Nonetheless, no posttreatment between-group differences were found, nor was there any consistent pattern of performance improvement for 1 treatment group over the other or for treatment over sham. The study has a number of other inherent limitations. The sample was exclusively male, and the results may not generalize to women. The sample size was reduced for the analysis of cognitive performance because of the added exclusion of invalid performance testing, and the randomized group with the fewest participants also had disproportionately more cognitive performance validity failures in it. Because of the large number of outcomes, there were also some randomly missing data points. Overall, these missing data represented much less than 1% of the variables. Because of these issues, we chose to also identify nonsignificant trends (P < .15) at posttreatment. Only one of the many exploratory measures showed such a trend, and it was disfavoring the 1.5 ATAO2 group (slower processing speed). This along with the fact that the overwhelming majority of P values were closer to 1 than to 0 raises confidence in the overall findings of nonefficacy.
In conclusion, this study found no immediate postcompression beneficial effect of HBO2 on cognitive or psychomotor performance at either 1.5 or 2.0 ATAO2 compared to the Sham Air intervention. These results from this randomized controlled trial do not support the use of HBO2 to treat cognitive, balance, or fine-motor dysfunction associated with mTBI and PCS. Future trials of HBO2 for PCS should consider measuring outcomes at longer intervals postintervention and/or in combination with rehabilitation therapy to assess for a potential delayed and/or priming effect.
Footnotes
Appendix A
CPT-II Exploratory Outcomes.
| Precompression |
Postcompression |
||||||
|---|---|---|---|---|---|---|---|
| Measure | n | Mean | SD | P | Mean | SD | P |
| CPT-II Omissions | |||||||
| Sham Air | 17 | 10.8 | 16.2 | .26 | 7.5 | 10.7 | 1.00 |
| 2.0 ATMO2 | 13 | 3.8 | 3.5 | 7.3 | 19.8 | ||
| 1.5 ATMO2 | 15 | 6.6 | 9.5 | 7.8 | 13.6 | ||
| CPT-II Commissions | |||||||
| Sham Air | 17 | 16.4 | 7.9 | .56 | 16.8 | 7.7 | .81 |
| 2.0 ATMO2 | 13 | 19.0 | 5.6 | 17.3 | 5.7 | ||
| 1.5 ATMO2 | 15 | 17.9 | 6.1 | 15.7 | 7.0 | ||
| CPT-II Hit RT | |||||||
| Sham Air | 17 | 387.2 | 76.3 | .25 | 374.8 | 73.4 | .66 |
| 2.0 ATMO2 | 13 | 348.9 | 45.6 | 360.5 | 49.7 | ||
| 1.5 ATMO2 | 15 | 389.5 | 81.6 | 384.5 | 83.0 | ||
| CPT-II Hit RT Standard Error | |||||||
| Sham Air | 17 | 8.00 | 3.35 | .24 | 8.27 | 4.03 | .97 |
| 2.0 ATMO2 | 13 | 6.34 | 2.66 | 7.88 | 8.14 | ||
| 1.5 ATMO2 | 15 | 8.57 | 4.26 | 7.82 | 5.58 | ||
| CPT-II Variability | |||||||
| Sham Air | 17 | 15.2 | 13.8 | .63 | 16.6 | 12.8 | .86 |
| 2.0 ATMO2 | 13 | 11.2 | 9.3 | 17.9 | 37.1 | ||
| 1.5 ATMO2 | 15 | 13.2 | 9.2 | 13.5 | 19.8 | ||
| CPT-II Perseverations | |||||||
| Sham Air | 17 | 1.65 | 2.50 | .38 | 2.53 | 3.53 | .65 |
| 2.0 ATMO2 | 13 | 0.69 | 1.32 | 1.23 | 1.48 | ||
| 1.5 ATMO2 | 15 | 1.47 | 1.51 | 2.53 | 6.05 | ||
| CPT-II Hit RT Block Change | |||||||
| Sham Air | 17 | 0.019 | 0.035 | .66 | −0.003 | 0.040 | .53 |
| 2.0 ATMO2 | 13 | 0.008 | 0.033 | 0.018 | 0.075 | ||
| 1.5 ATMO2 | 15 | 0.017 | 0.035 | 0.007 | 0.039 | ||
| CPT-II Hit SE Block Change | |||||||
| Sham Air | 17 | 0.075 | 0.112 | .21 | −0.005 | 0.140 | .32 |
| 2.0 ATMO2 | 13 | 0.016 | 0.061 | 0.059 | 0.171 | ||
| 1.5 ATMO2 | 15 | 0.044 | 0.078 | 0.048 | 0.074 | ||
| CPT-II Hit RT ISI Change | |||||||
| Sham Air | 17 | 0.068 | .040 | .42 | 0.086 | 0.047 | .85 |
| 2.0 ATMO2 | 13 | 0.063 | 0.023 | 0.078 | 0.070 | ||
| 1.5 ATMO2 | 15 | 0.052 | 0.037 | 0.077 | 0.039 | ||
| CPT-II Hit SE ISI Change | |||||||
| Sham Air | 17 | 0.085 | 0.194 | .81 | 0.103 | 0.168 | .47 |
| 2.0 ATMO2 | 13 | 0.092 | 0.126 | 0.045 | 0.183 | ||
| 1.5 ATMO2 | 15 | 0.059 | 0.074 | 0.049 | 0.098 | ||

Abbreviations: CPT, computerized posturography; SD, standard deviation; RT, reaction time; SE, standard error; ISI, inter-stimulus interval; Sham Air, group breathing 10.5% oxygen (balance 89.5% nitrogen) at 2.0 atmospheres absolute (ATA); 2.0 ATMO2, group breathing 75% oxygen (balance 25% nitrogen) at 2.0 ATA; 1.5 ATMO2, group breathing pure oxygen (0% nitrogen) at 2.0 ATA.
Authors’ Note
The views expressed herein do not necessarily represent the views of the Department of Veterans Affairs, Department of Defense, or the US Government.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Funding was provided for the primary study by a Defense Advanced Research Projects Agency grant (N66001-09-2-206), US Navy Bureau of Medicine and Surgery for contract funding temporary duty requirements, and the US Army Medical Material Development Activity for end-of-study contract funding. Drs Franke’s and Walker’s efforts were additionally supported, in part, through contracts from the Defense and Veterans Brain Injury Center. The funding sources had no role in the study design, analysis, interpretation of the data, the writing of the article, or the decision to submit the paper for publication.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
