Abstract
Background. Individuals with chronic stroke often have long-lasting upper extremity impairments that impede function during activities of daily living. Rehabilitation robotics have shown promise in improving arm function, but current systems do not allow realistic training of activities of daily living. We have incorporated the ARMin III and HandSOME device into a novel robotic therapy modality that provides functional training of reach and grasp tasks. Objective. To compare the effects of equal doses of robotic and conventional therapy in individuals with chronic stroke. Methods. Subjects were randomized to 12 hours of robotic or conventional therapy and then crossed over to the other therapy type after a 1-month washout period. Twelve moderate to severely impaired individuals with chronic stroke were enrolled, and 10 completed the study. Results. Across the 3-month study period, subjects showed significant improvements in the Fugl-Meyer (P = .013) and Box and Blocks tests (P = .028). The robotic intervention produced significantly greater improvements in the Action Research Arm Test than conventional therapy (P = .033). Gains in the Box and Blocks test from conventional therapy were larger than from robotic therapy in subjects who received conventional therapy after robotic therapy (P = .044). Conclusions. Data suggest that robotic therapy can elicit improvements in arm function that are distinct from conventional therapy and supplements conventional methods to improve outcomes. Results from this pilot study should be confirmed in a larger study.
Introduction
Each year, more than 600 000 individuals in the United States have a first-ever stroke. 1 Fifty percent of stroke survivors older than 64 years have hemiparesis at 6 months poststroke and 26% are dependent in activities of daily living (ADL). 1 In the upper extremity, reaching and grasping movements are often impaired and a focus of rehabilitation. Impairments include decreased muscle activation and weakness,2,3 disrupted interjoint coordination,4-6 decreased smoothness of movement,7,8 and dyscoordination between reach and grasp movements. 9 Limitations in ADL ability and decreased quality of life can result from even mild impairments in upper limb function.10,11
Neurorehabilitation therapies have shifted toward task-specific training that incorporates context-specific repetitive practice of complex tasks involving many degrees of freedom (DOF). 12 Incorporating robotic devices into repetitive task practice may enrich the training experience via the application of forces to the limb, similar to when a human therapist assists movements. 13 The robot allows completion of movements throughout the range of motion and can also prevent inappropriate compensatory strategies. Seminal studies on proximal arm movement have found that robots can reduce impairments in stroke survivors.14-17 A recent meta-analysis of 19 studies found that robotic devices produced larger gains in arm function and ADL ability than comparison interventions. 18
Despite these positive results, many have rightly questioned if the gains from robotic training were clinically meaningful. Additionally, the optimal form of robotic interaction is still unknown, and a wide range of devices and assistance approaches are being investigated. A few devices provide practice of reach and grasp tasks. ADAPT 19 is a robotic task-practice system that presents tools that subjects manipulate, such as a door knob or jar, but no assistance or guidance is provided to the arm or hand. GENTLE/s 20 and ADLER 21 both use an end effector robot (Haptic Master) attached to the wrist to assist in functional task practice. ABLE 22 is a 4-DOF exoskeleton that guides users toward the desired joint coordination pattern but does not assist the hand and lacks supination/pronation. None of these devices combine assisted hand function with an exoskeleton that directly controls enough proximal arm joints to allow realistic practice of a range of functional tasks. To address this shortcoming, we have developed a novel robotic training protocol that combines the ARMin III 23 and HandSOME 24 exoskeletons, allowing coordinated whole limb training in reach and grasp tasks. Both real and virtual objects can be used, and multi-DOF tasks can be effectively practiced even in subjects with severe impairments. A novel joint-space training mode facilitates retraining of inter-joint coordination and prevents effective use of compensatory strategies that may limit functional recovery. 25 In this study, we performed a randomized clinical trial of robotic therapy compared with conventional therapy in chronic stroke subjects. It was hypothesized that individuals would have larger functional gains after robotic therapy compared with conventional therapy.
Methods
All study procedures were approved by the local institutional review board, and all subjects provided informed consent. Subjects were recruited through the MedStar National Rehabilitation Hospital (NRH) stroke database and by “word of mouth” with treating therapists at NRH clinics. The inclusion criteria were (a) adult with an ischemic or hemorrhagic stroke at least 6 months prior to entry into the study, (b) trace ability to move the wrist and fingers in extension, (c) trace ability for shoulder humeral elevation, and (d) persistent hemiparesis (score of 1 or 2 on the motor arm item of the National Institutes of Health [NIH] stroke scale 26 ). Exclusion criteria were (a) cognitive deficits that would limit their ability to complete the study protocol (a score of 24 or less on the Folstein Mini-Mental State Examination 27 ), (b) hemispatial neglect (>3 errors on the Star Cancellation Test 28 ), (c) severe sensory loss (a score of 2 on the sensory item of the NIH Stroke Scale), (d) excessive pain in any joint of the affected extremity, (e) an upper extremity injury prior to stroke that could limit participation.
All training occurred in an isolated laboratory at NRH. Subjects were randomized to start with conventional or robotic therapy and then crossed over to the other therapy following a washout period. Crossover study design is commonly used for pilot investigations to reduce the effects of between subject variability and thus reduces the necessary subject numbers. 29 Individuals serve as their own controls allowing for better comparison between interventions, especially if no change is expected independent of intervention, as is the case with chronic stroke subjects. A wash out period allows for stabilization of gains in the first treatment period and minimizes the potential of carryover effects.
Randomization was done using a random number generator function in Matlab (MathWorks Inc, Natick, MA) that generated a list of numbers (1-10) randomly ordered. The first 5 listed subject numbers received conventional therapy first and the second set received robot therapy first. Additional subjects were added into the study to replace individuals who dropped out. Each therapy block was 1-month long (12 hours of therapy), with a 1-month washout period between blocks. Clinical tests were performed immediately before and after each therapy block. These tests were performed by an occupational therapist (OT) trained in performing these tests. The OT performing recruitment and clinical evaluations was not aware of the randomization order, so was blinded to group assignment. Standard precautions were taken to avoid potential unblinding. All treatments (robotic and conventional) were done in the same room and the OT did not enter the room during treatments. The OT did not discuss the nature or content of ongoing treatments with the subjects. Clinical tests included the Fugl-Meyer Test of Motor Function, 30 Action Research Arm Test, 31 and Box and Blocks Test. 32 The Fugl-Meyer (FM) assesses motor impairments at the shoulder, elbow, wrist, and fingers on a 3-point scale. The Action Research Arm Test (ARAT) assesses upper extremity functional limitations and includes 19 items divided into 4 subscales: Grasp, Grip, Pinch, and Gross movement. The Box and Blocks Test (B&B) is a time-based measure of gross functional ability. Subjects are asked to move as many 2.5 cm square blocks as possible from one side of a partitioned box to the other in 1 minute.
Conventional Therapy
Subjects received 12 hours of therapy from a neurologic clinical specialist physical therapist with 30+ years of experience. Therapy was consistent with the standard of care provided at NRH. Individualized programs were established based on assessment and patient goals. In weak muscles, manual therapy techniques were used to obtain isometric contractions in the shortened range. Subjects received mobilization to restricted joints as needed and stretching exercises to increase range of motion. Home stretching was recommended to subjects who had less than full passive range of motion in the fingers. Treatments focused on practice of specific tasks. These included reach and grasp of various objects, isolated hand movements (typing, “playing the piano,” molding putty), and whole body activities (swinging a tennis racquet, basketball handling skills, etc).
Robotic Training
Subjects received 12 hours of training with the ARMin III robot 23 and the HandSOME 24 device (Figure 1). The ARMin III provided active actuation of shoulder elevation, shoulder horizontal adduction/abduction, shoulder external/internal rotation, elbow flexion/extension, and forearm supination/pronation. The ARMin was controlled with custom software (MATLAB xPC Target, MathWorks Inc, Natick, MA) that minimized the effects of gravity and friction at the robot joints. 33 The HandSOME uses adjustable elastic elements to increase active range of motion, enabling coordinated finger and thumb movement in pinch-pad grasp (finger metacarpophalangeal joints and thumb carpometacarpal joint). The combination of the ARMin and HandSOME devices allowed for simultaneous reach and grasp of virtual and real-world objects. A technician received basic patient interaction training from the study therapists and oversaw the robotic therapy.
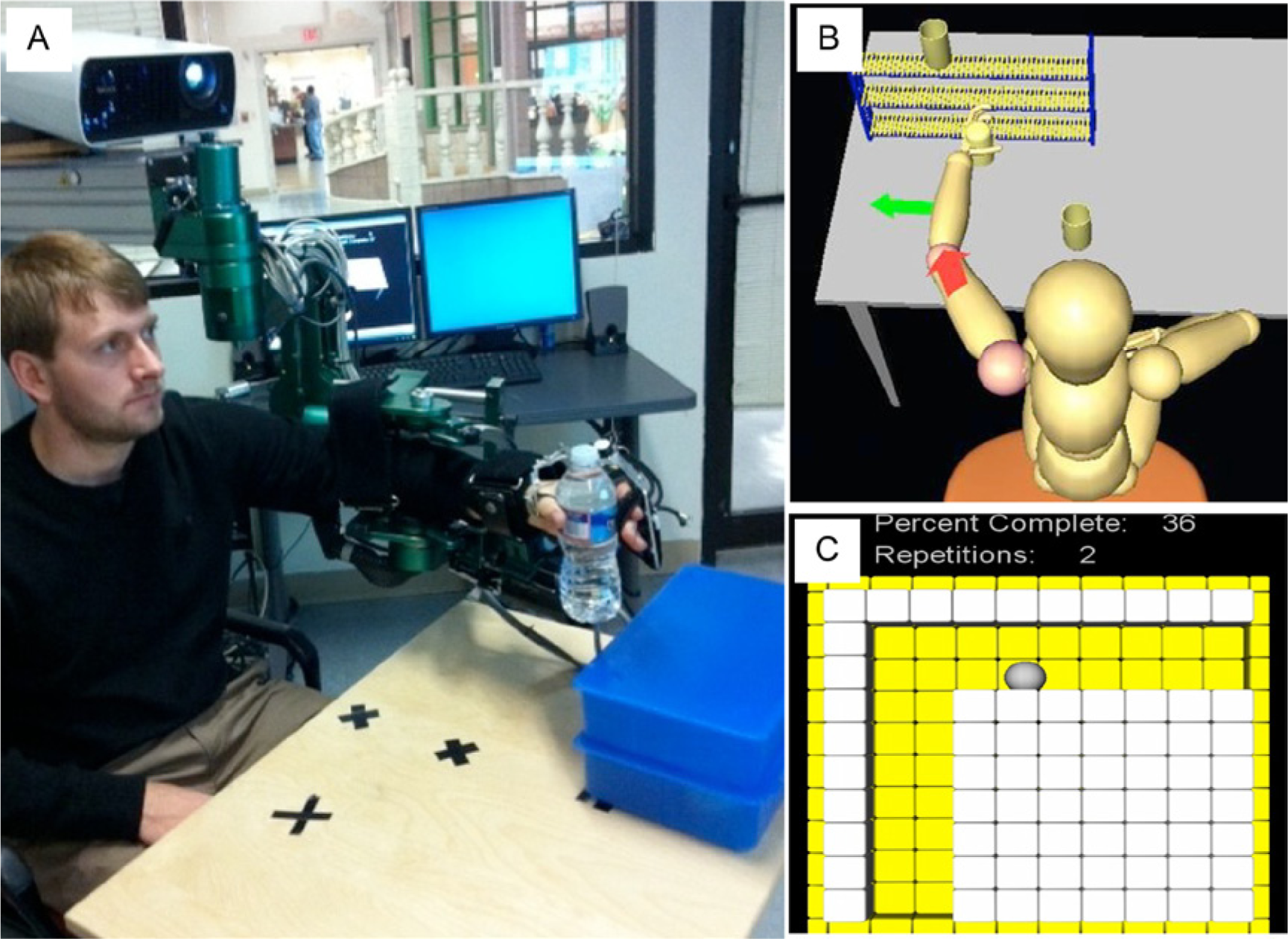
(A) ARMin and HandSOME devices. (B) Visual user interface during shelf task. Arrows indicate the direction of joint rotation needed to complete the task. The arrow colors indicate if a joint is not rotating in the correct direction and preventing advancement in the trajectory. (C) L-trajectory task visual interface.
The primary training tasks were the Shelf, Pouring, and Sorting tasks. Each task was practiced for 25 minutes in each session. In the Shelf task, subjects placed objects on a shelf using simultaneous shoulder elevation and elbow extension. This task practices movement out of the common flexor synergy. In the Pouring task, subjects grasped and poured from a pitcher using shoulder internal/external rotation and simultaneous wrist pronation/supination. In Sorting, subjects moved objects from one location to another using shoulder elevation and simultaneous shoulder horizontal abduction.
The robot was controlled using the Time Independent Functional Training (TIFT) method previously developed.34,35 Joint space walls limited movement unless the arm joints are moved with a coordination that is appropriate for the task. The goal of TIFT is to retrain proper interjoint coordination within the context of functional multijoint reaching movements. Difficulty was graded through changes in range of motion and the amount of arm gravity compensation. Subjects practiced first with virtual objects and then physical objects were added to increase difficulty. Real object shape, fragility, and weight were varied. Coordinated reach and grasp was practiced; subjects planned their trajectory to the object, opening their hand as they reached for the object. At the beginning of each task, subjects attempted movements with zero gravity compensation to assess changing ability across training sessions. For 15 minutes each session, game-like activities were played that focused on isolated joint movement. These included navigating mazes (L-Traj) and playing ping-pong against the computer.
Data Analysis
Baseline differences between subject groups were tested with unpaired t tests. Performance gains during the robot training were assessed by the percentage completion in the trials where no gravity compensation assistance was provided. Change over sessions was tested with a repeated-measures analysis of variance (RM-ANOVA) using session number as the within-subject factor. Change in the clinical test scores over the 4 evaluation time points was examined using a RM-ANOVA with time point as the within-subject factor and treatment order as the between-subject factor. Comparisons between treatments were performed by first calculating the pre–post gain for each therapy block. A RM-ANOVA was performed on these gains using within-subject factor of treatment type and between-subject factor of treatment order. Significant effects in the RM-ANOVA were investigated with subsequent Bonferoni-corrected t tests. Outliers were defined as scores greater than the upper quartile + 1.5 × interquartile range, or less than the lower quartile − 1.5 × interquartile range.
Results
The consort flow diagram is shown in Figure 2. Fourteen individuals were recruited. One subject was excluded because of a prior orthopedic injury and another subject did not meet the Folstein Mini-Mental State Examination criterion. The remaining 12 subjects ranged from severe to moderate impairment with baseline FM scores ranging from 13 to 34 (Table 1). Two subjects completed the protocol up through evaluation time point 3 but did not return for the second therapy block (conventional therapy). These 2 subjects were removed for between-treatment analysis but were included in the within-treatment analysis. The remaining 10 subjects completed the entire protocol and were randomly grouped into 5 subjects who received the robotic training first and 5 subjects who received conventional training first. These 2 groups of subjects were not different in age (P = .75), time poststroke (P = .90), and all of the baseline clinical measures (FM, P = .093; ARAT, P = .41; B&B, P = .24). We looked for outliers by calculating the difference between the treatment gains in each subject, but found no outliers in any of the outcomes.
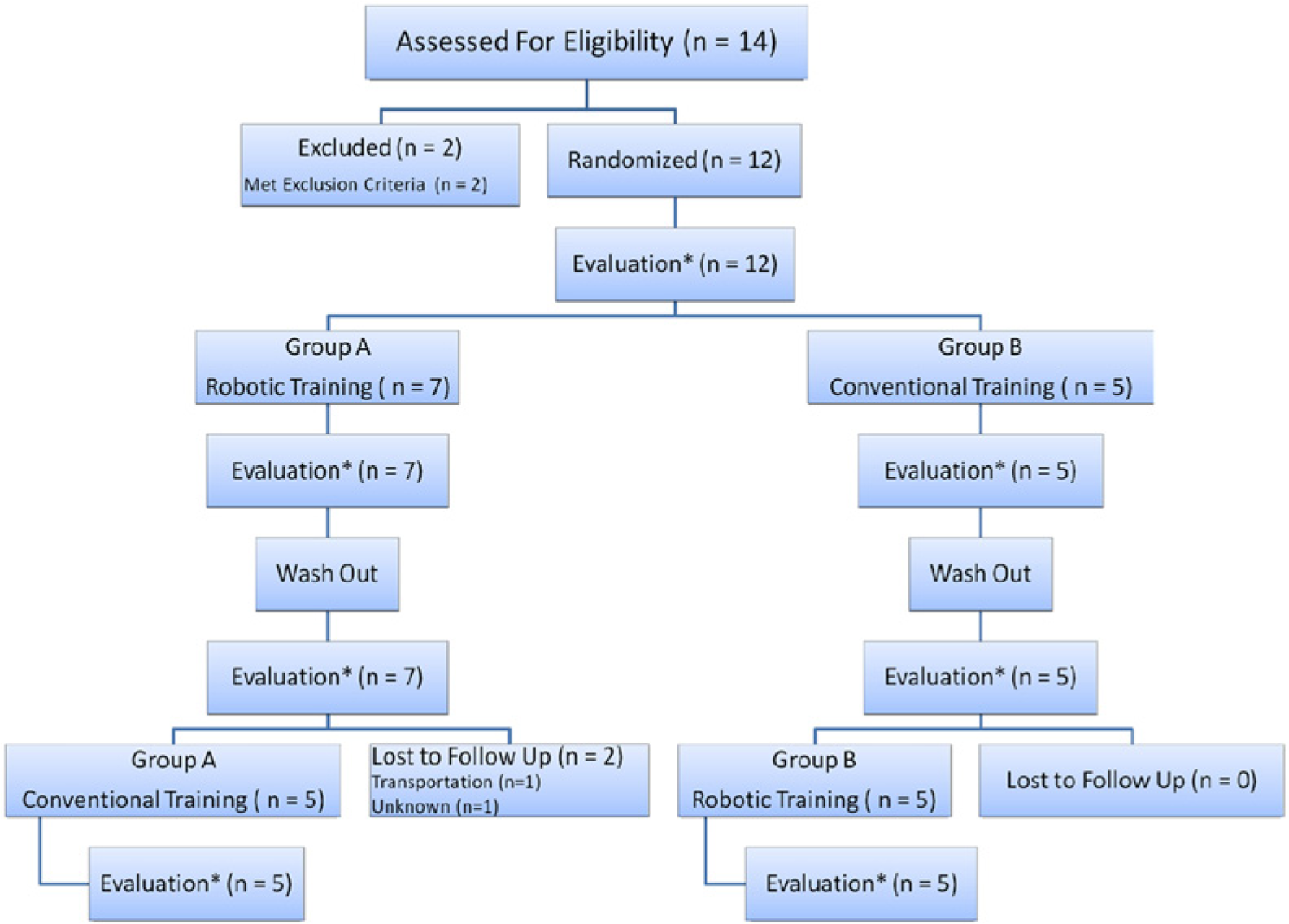
Consort flow diagram. Each training and wash out period was approximately 1 month in duration. Each evaluation was done by the same blinded occupational therapist.
Baseline Characteristics.

Subjects showed steady progress across robotic training sessions in trials where arm weight compensation was turned off (unassisted trials; Figure 3). Averaged across all subjects, the percentage completed at the end of training had increased 30%, 23%, 4.5%, and 11% in the L-Traj, Shelf, Pouring, and Sorting tasks, respectively. These gains were significant in all tasks (P < .042), except for Sorting (P = .19). Many of the subjects could do the Sorting task unassisted at the start of the training.
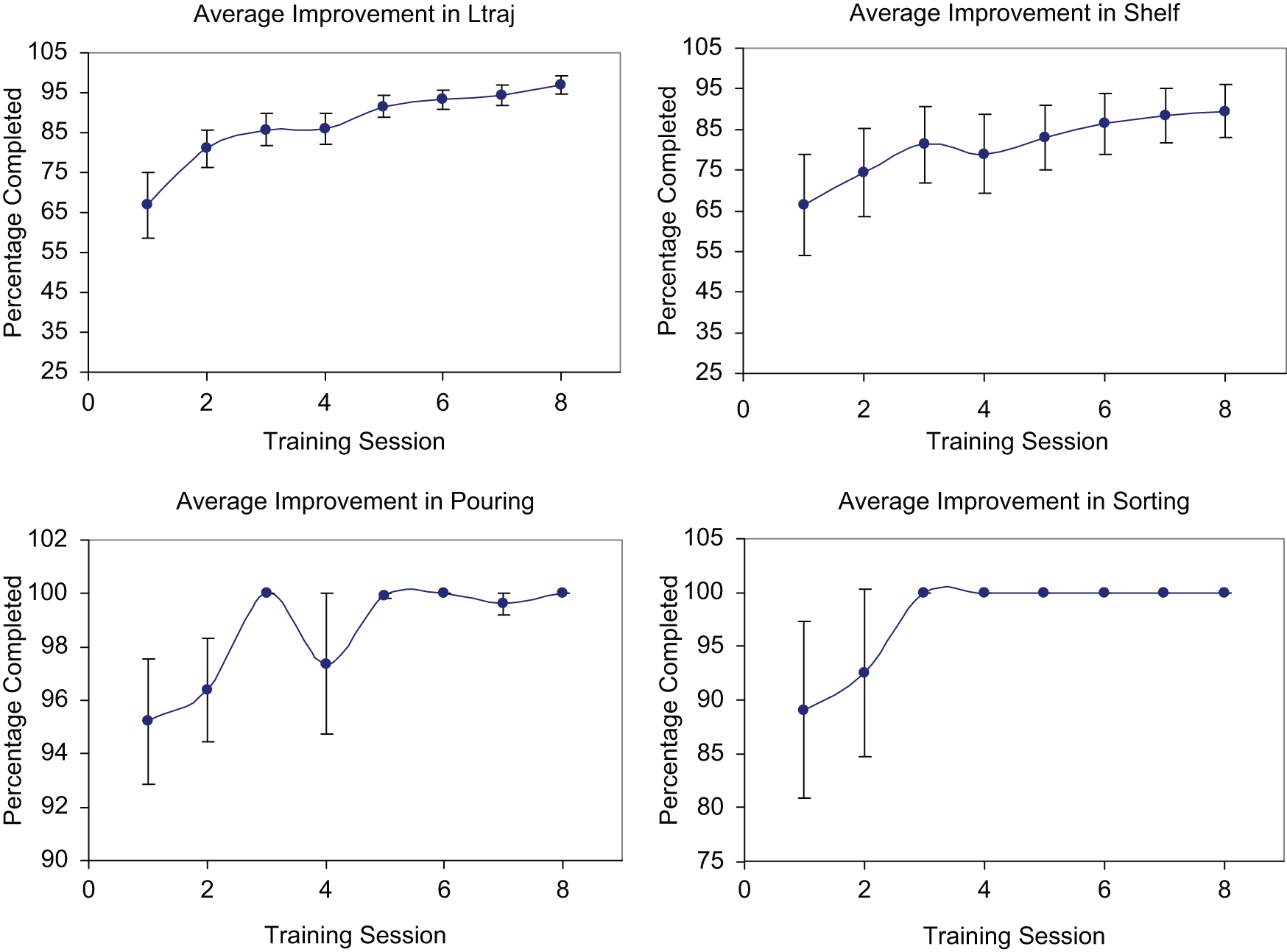
Average improvement over the 8 robotic therapy sessions with standard error bars. The gravity compensation was set to 0% for these trials.
Subjects improved significantly on clinical scales over the course of the entire treatment protocol (Figure 4; Table 2). RM-ANOVAs found that scores increased significantly in the FM (P = .013) and B&B scales (P = .028). The interaction between group and time point was not significant in any of these measures (FM, P = .53; ARAT, P = .073; B&B, P = .08). Mean gains between the first and final evaluation time point were 5.1 ± 1.7 for the FM, 2.4 ± 1.2 for the ARAT, and 4.3 ± 1.9 for the B&B. Five of 10 subjects had gains of 7 points or higher on the FM (>10% of full scale). RM-ANOVA found that there were no significant changes during the washout period in any of the outcomes (FM, P = .42; ARAT, P = .77; B&B, P = .53). Additionally, the group × time interactions were not significant for any of the outcomes (FM, P = .85; ARAT, P = .77; B&B, P = .92), indicating that changes over the washout period were similar in the 2 groups.
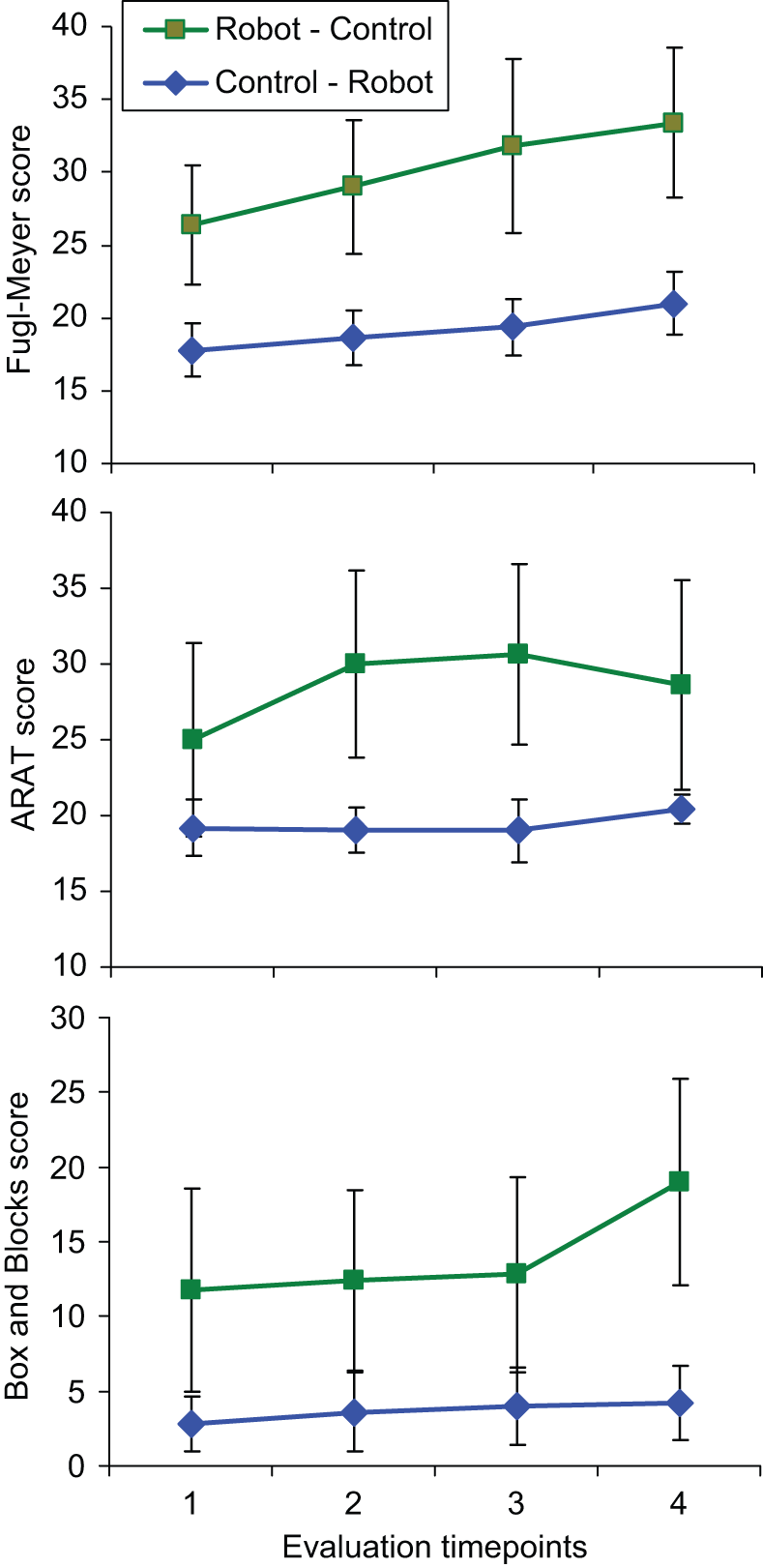
Average scores for all 10 subjects that completed the protocol, separated into the group that received robot therapy first and the group that received control therapy first. The 1-2 and 3-4 periods are the training periods (robotic or control), and the 2-3 period is the washout period.
Outcome Measures for Each Subject. a
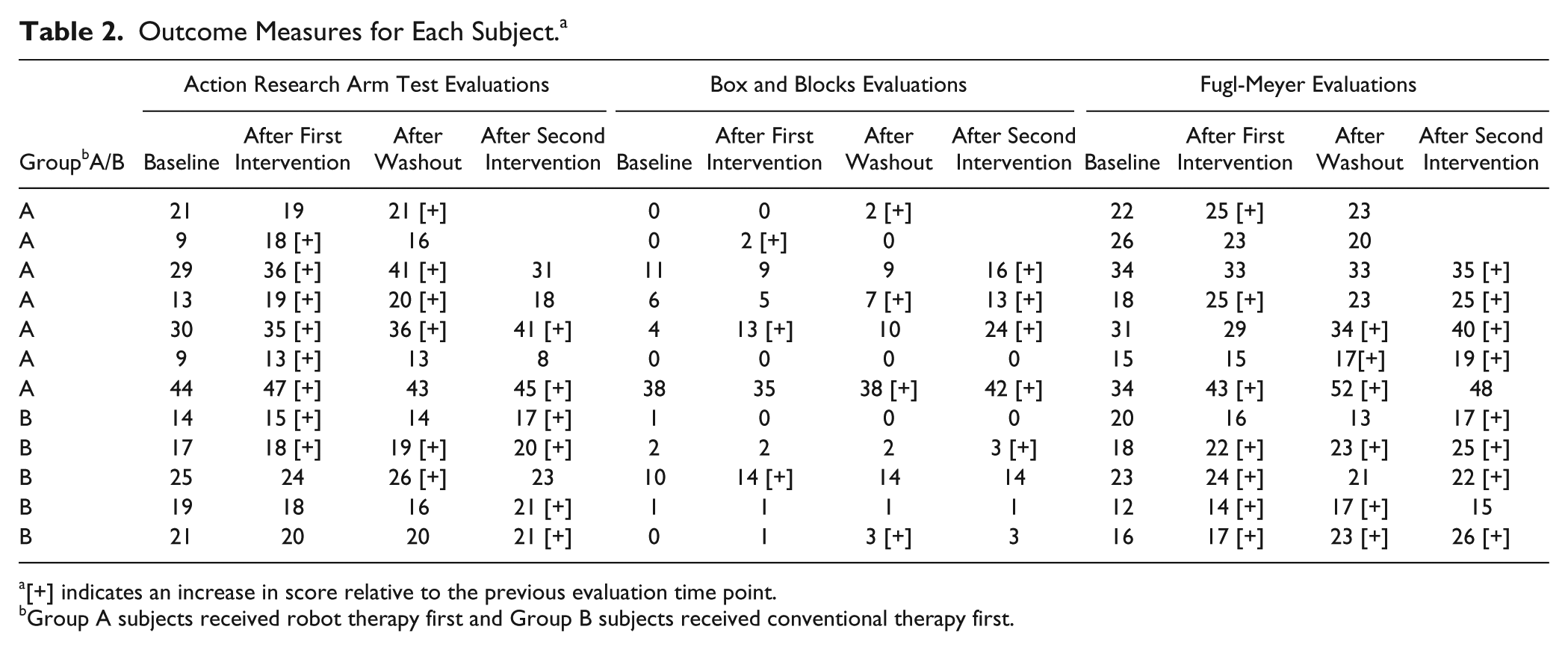
[+] indicates an increase in score relative to the previous evaluation time point.
Group A subjects received robot therapy first and Group B subjects received conventional therapy first.
Treatment differences were analyzed with RM-ANOVA on clinical score gains from pretreatment to immediate posttreatment (Figure 5). A significant treatment effect was found in the ARAT, with gains after robotic training significantly larger than after control therapy (P = .033). The interaction between treatment type and group was not significant (P = .15), indicating the advantage of robotic training over control therapy was present independent of the order of the treatments. Subsequent within treatment analysis showed significant gains in the ARAT immediately after robotic training (P = .018, gain = 3.25 ± 1.0) but not after conventional therapy (P = .84, gain = −1.1 ± 1.3). To examine if the gains immediately after training were diminished one month later, we examined ARAT gains from the first to the third evaluation time point, which includes the first training period and the 1-month washout period. Mean gains over this period were larger in the robot-trained subjects (gain = 5.0 ± 1.7) than the control subjects (gain = −0.2 ± 0.86). Additionally, between the first and third time point, 4 of 7 subjects achieved gains after robot training of 6 points or higher (>10% of full scale), while the largest gain was 2 points in the 5 control subjects at this time point.
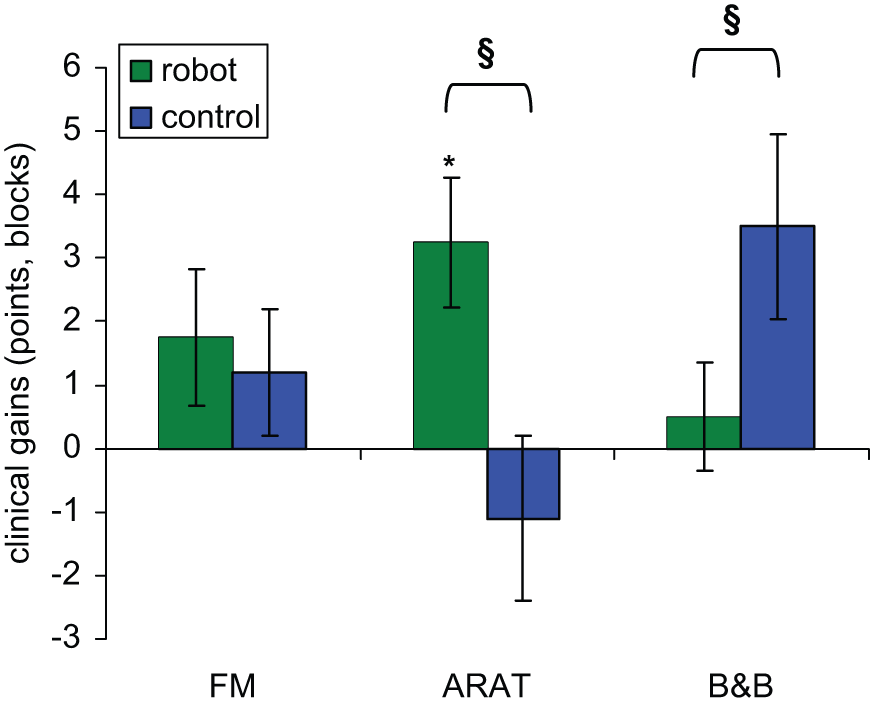
Average gains from pretreatment to immediate posttreatment. Error bars are standard error of the mean. *Significant treatment gains (P < .05). §Significant difference between treatment types (P < .05).
Similar analysis for the FM found no significant differences between treatments (P = .68), and no interaction between treatment type and order (P = .96). Despite an overall significant increase in the FM between the first and last evaluation time points, gains from each treatment type did not reach significance (robot, P = .26; control, P = .51). We also examined the subscores of the FM that were the most directly related to the novel aspects of the robotic training. The hand function FM subscore (14 points) is related to the HandSOME and increased significantly after robotic training (P = .02, gain = 0.75 ± 0.3). The TIFT is related to the movement out of synergy and movement combining synergy subscores. These 2 subscores were summed into a composite score (12 points) and was also found to increase significantly after robotic training (P = .04, gain = 0.92 ± 0.4).
Repeated-measures analysis of variance found a significant treatment effect in B&B gains (P = .009) as well as a significant interaction between treatment type and order (P = .024). Within treatment analysis found that the treatment effect was because of larger gains in B&B scores after conventional training (P = .08, gain = 3.5 ± 1.5) than after robotic training (P = 1.0, gain = 0.5 ± 0.9). Analysis of the significant interaction between treatment and order was done by comparing treatment differences in each subject group separately. There was no difference between treatment gains in subjects who received conventional therapy first (P = 1.0); however, in subjects who received robotic therapy first, gains in B&B after conventional therapy were larger than after robotic therapy (P = .044).
Discussion
The combined treatment protocol was effective, with significant gains in the FM and B&B over the 4 evaluation time points. Five of 10 subjects had gains of 7 points or larger on the FM by the end of the study. Within-subject analysis on FM scores showed no difference between treatment type, indicating that the 2 treatments were equally effective in promoting gains on the FM. In contrast, gains on the ARAT from robotic training were significantly larger than from control treatment. This advantage in the robotic training was also present when examining gains from the first to the third time point, which included a 1-month washout period. Over this period, 4 of 7 robot-trained subjects had gains of 6 points or higher on the ARAT, while the largest gain in control subjects was 2 points. Gains in B&B from control therapy were larger than from robotic therapy in subjects who received the robotic therapy first.
Our results support the premise that robotic and conventional therapies are complementary. The robotic treatment produced greater gains in the ARAT and the control therapy showed an advantage in the B&B. Key differences between the ARAT and B&B may explain these contradictory results. Both ARAT and B&B involve grasp and transport of objects; however compensatory strategies are marked down in the ARAT while they are not penalized in the B&B. The robots advantage in the ARAT may be related to its focus on promoting normal movement coordination instead of compensatory strategies during functional task practice. Several studies have shown that preventing the use of compensatory trunk movement can facilitate improved reaching movements.36,37 At the shoulder and elbow, the TIFT algorithm forces the subject to produce normal coordination patterns before the hand can advance to the target. In a previous study we found that in the Shelf task, stroke subjects spontaneously performed the targeted pattern of synchronous shoulder abduction and elbow extension when guided by TIFT, while these same subjects used compensatory strategies involving the flexor or extensor synergies when guided by an end point tunnel. 38 Another potential advantage of the robot therapy is the HandSOME device. Although only a simple grasp pattern is practiced in HandSOME, the fingers are positioned appropriately by the device allowing practice with large and small objects even in subjects with limited range-of-motion. The novelty of the TIFT and HandSOME make this robotic therapy a potentially efficient supplement to conventional therapy. It would be difficult for a human therapist to provide therapy comparable to TIFT and HandSOME. Even if it were possible, the physical effort required on the part of the human therapist would limit the number repetitions compared to the robotic intervention.
A second key difference between the B&B and the ARAT is that speed is a major component of the B&B, and only a secondary factor in the ARAT. The robot has a very large inertia which does not lend itself to effective practice of rapid movements. Speed was not trained during the majority of robot sessions and was only a factor in the games, which were a minor portion of the protocol. This artificial, high-inertia training environment is minimized when movements are slow but does not allow realistic training of fast movements, which are needed for the B&B. In contrast, the majority of the repetitive practice in the conventional therapy involved “free” movements which allowed more emphasis on speed of movement. Gains from control therapy were larger than from robot therapy only if the control therapy was preceded by robotic therapy. It is possible that the initial robot therapy improved motor coordination and range-of-motion, and allowed more productive practice in the free environment during subsequent control therapy.
A recent large multisite clinical trial reported no differences between equal doses of robotic and conventional therapy. 17 Our results suggest several differences, perhaps because our robotic training protocol is unique compared to other robotic approaches under investigation. First, while many approaches offer practice of components of functional tasks, 17 the ARMin and HandSOME system allow realistic practice of reach and grasp of real objects. Thus, coordinated action of many muscles is trained, such as reaching for an object while opening the hand and transporting the object without dropping or crushing it. Second, while other systems apply assistance to task completion when the subject’s errors exceed thresholds in spatial or temporal metrics, our approach relies on graded gravity compensation for limb weight and passive constraints that do not allow movement unless there is proper coordination between different muscle groups. While the former strategy often results in the robot applying forces to the arm that drag it toward the target, our approach eliminates this possibility while also preventing use of compensatory strategies. Finally, while many arm protocols ignore the hand, or only involve isometric gripping, the HandSOME allows highly repetitive practice of grasp of real and virtual objects, with minimal reduction in grasp force. This is made possible by an extension assistance profile that minimizes the assistance level at each point in the range of motion.
Three previous studies have tested mechanically assisted reach and grasp training in stroke subjects. FM gains were 3.7 points using the GENTLE/s robot, 20 3.3 points using the T-WREX, 39 and 3 points using the Pneu-WREX. 40 We had smaller FM gains of 1.75 points after robot training, but hours of treatment were ½ of that in the T-WREX and Pneu-WREX studies, and the GENTLE/s and Pneu-WREX studies included subjects in the subacute recovery stage, when larger gains are more likely. In our study, total FM gain after receiving both treatments was 5.1 points, suggesting available treatment time might be most effective if divided between robotic and conventional therapies. Additionally, there appears to be an advantage of our robot in functional scales. Gains were significant in the ARAT after only 12 hours of our robot therapy, while in the Pneu-WREX and T-WREX studies, significant gains were not achieved in the Rancho Functional Test for the Hemiplegic/Paretic Upper Extremity. Similarly, recovery rate in the Motor Assessment Scale during GENTLE/s training was not larger than during the multiple baseline period. This advantage in functional scales might have been due to use of the HandSOME device. No hand assistance was provided in GENTLE/s, and grasp practice in the T-WREX and Pneu-WREX was performed virtually using a grip sensor.
Limitations
The limited therapy hours could mask potential gains that would have been made with additional therapy. The robotic training cannot be easily customized to a patient’s specific goals. For example, if a patient expresses the goal of playing tennis, this can be easily integrated into conventional therapy, but cannot be practiced in the robot. In addition, the use of many novel components makes it difficult to pinpoint the exact cause of the benefits of robotic therapy. The use of virtual reality and games could have provided additional motivational benefit over conventional therapy; however, a recent review reported that the benefits of virtual reality during stroke therapy were inconclusive. 41
The small sample size raises the possibility of randomization bias. However, several points argue against this. Firstly, subjects were randomized between starting with robot or conventional therapy. These two groups of subjects were not different in age, time poststroke, and all the baseline clinical measures. Secondly, the crossover design protects against randomization bias to some degree because all subjects are exposed to both interventions, and the main comparisons are within subject. It is possible that the most responsive subjects were randomized to receive robot therapy first, and that neither intervention was effective when delivered second because gains had already saturated. This would be randomization bias in favor of the robot group. However, this is unlikely because order effects were not significant in the ARAT and FM, indicating that treatment gains were independent of whether the treatment was given in the first or second therapy block. The significant order effect in the B&B was because of greater gains from conventional therapy when received after robotic therapy. Therefore, this also argues against randomization bias.
Future Directions
Our data show that robotic therapy provides a stimulus for recovery that is different from conventional therapy. However, because of the small sample size, these results need to be confirmed in a larger scale study using a parallel study design. Considering the ARAT, the effect size (mean difference/standard deviation) of the treatment difference was d = 1.2, considered a large effect according to Cohen’s definition. 42 Power analysis for a parallel design study indicates that 16 subjects are needed in each arm of the study to detect this effect size with 95% power. One important future area of work are robots that can implement the training modes used in this study, and that also have low inertia to allow realistic simulation of “free” movements. Alternatively, robots could be used as a precursor to reduce impairments, prior to application of conventional therapy or “free” movement practice. With additional advancements, extended periods of unsupervised robotic therapy would be possible, reducing cost and providing patients more therapy hours.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Funding for this work provided by the US Army Medical Research and Materiel Command (W81XWH-05-1-0160) and the Department of Veterans Affairs (B4719R).
