Abstract
Background. By involving additional degrees of freedom, the nervous system may preserve hand trajectories when making pointing movements with or without trunk displacement. Previous studies indicate that the potential contribution of trunk movement to hand displacement for movements made within arm reach is neutralized by appropriate compensatory shoulder and elbow rotations. For beyond-the-reach movements, compensatory coordination is attenuated after the hand peak velocity, allowing trunk movement to contribute to hand displacement. Objective. To investigate if the timing and spatial coordination of arm and trunk movements during beyond-the-reach movements is preserved in stroke. Methods. Eleven healthy control subjects and 11 individuals with mild-to-moderate chronic unilateral hemiparesis participated. Arm and trunk kinematics during 60 target reaches to an ipsilaterally placed target were recorded. In 30% of randomly chosen trials, trunk movement was unexpectedly prevented (blocked-trunk trials) by an electromagnetic device, resulting in divergence of the hand trajectory from that in free-trunk trials. Hand trajectories and elbow–shoulder interjoint coordination were compared between trials. Results. In stroke participants, hand trajectory divergence occurred at a shorter movement extent and interjoint coordination patterns diverged at a relatively greater distance compared to controls. Thus, arm movements in stroke participants only partially compensated trunk displacement resulting in the trunk movement contributing to arm movement earlier and to a larger extent during reaching. Conclusion. Individuals with mild-to-moderate stroke have deficits in timing and spatial coordination of arm and trunk movements during different parts of a reaching movement. This deficit may be targeted in therapy to improve upper limb function.
Introduction
The specific coordination between trunk and arm movements when reaching to objects placed beyond arm’s length is a hallmark of normal motor control.1-5 Identification of deficits in this organization in stroke may be important for enhancing upper limb (UL) motor recovery.
When healthy individuals reach to beyond arm’s length objects, the trunk begins to move slightly before or simultaneously with the hand, but trunk displacement only begins to contribute to hand movement extent after hand peak velocity. Ghafouri and Feldman 6 found that this time matches the end of the central shift in the arm equilibrium position, when the hand virtually reaches its limit of extension (~90% arm length).7,8 The difference between the equilibrium and actual arm positions leads to force generation driving the arm to the final position. Up until this time, the trunk contribution to hand movement is minimized by compensatory arm joint rotations in the opposite direction. 1 Compensatory arm–trunk coordination is also seen at the end of reaching when the trunk continues to move after arm movement offset while the hand remains motionless. The earlier trunk recruitment at reaching onset is likely due to larger trunk inertia that has to be accelerated before the time of peak hand velocity to be smoothly integrated into the latter part of the reach. An analogy is that of a relay competition in which the second runner starts to run before the previous runner arrives in order to match their speeds prior to the hand-off of the baton. Previous studies suggest that although the trunk and arm move in parallel, the central commands responsible for the contribution of arm and trunk movements to hand movement extent are applied sequentially, that is, the contribution of the trunk to the hand movement extent begins after the arm has virtually reached its extension limit. 1
Recovery of UL movement after stroke not only involves the ability to isolate joint movement and control movement throughout the joint range 9 but also to regain interjoint and intersegment coordination, deficits that have received less emphasis in traditional rehabilitation. For within-arm length reaching that normally does not require trunk displacement, individuals with stroke often use atypical compensating strategies such as excessive shoulder abduction10,11 and/or trunk displacement to compensate for limited arm extension, and/or flex and adduct the shoulder.12-16 The amount of atypical compensatory movement varies with the motor deficit severity 13 and may also be related to agonist/antagonist muscle weakness, 17 deficits in selective movement production,10,18 abnormal movement synergies or torque compensation,19-23 and interjoint/segment timing deficits.24-27
When the trunk is abnormally recruited for within-reach tasks, temporal arm and trunk recruitment is similar to healthy individuals for reaches beyond arm length, but spatial coordination is disrupted such that the trunk segment contributes to arm movement extent earlier in the reach. 16 This disruption in coordination may be one factor underlying decreased arm use in activities of daily living (ADLs), even in clinically well-recovered patients. 28 Since many ADLs also require reaching beyond the arm workspace, similar disruptions in spatial and/or temporal coordination may also contribute to decreased recovery. Understanding of arm–trunk coordination during beyond arm length reaching combined with knowledge of coordination deficits for within-reach movements in patients with stroke may provide guidelines for the development of treatments aimed at improving UL functional recovery.
We hypothesized that unlike healthy subjects, individuals with stroke would have deficits in arm–trunk coordination when reaching beyond arm length such that trunk movement would contribute to arm movement extent earlier in the reach.
The hypothesis was tested by comparing hand trajectories and arm interjoint coordination during reaching when subjects moved the trunk (free-trunk condition) and when trunk motion was unexpectedly prevented (blocked-trunk condition) in participants with stroke compared to healthy individuals. The rationale for comparison of kinematics in the 2 types of trials is the following. According to the previous study in healthy participants, 1 when the trunk was unrestrained, forward trunk movement was initially compensated by appropriate arm joint rotations so that, during the initial reaching phase, the arm trajectory was not affected by trunk motion. Blocking trunk movement just stopped the compensatory arm–trunk coordination, and thus initial arm trajectories were the same in the 2 types of trials, only diverging at the limit of arm extension. In contrast, in poststroke subjects, arm extension limit is usually diminished, 15 and therefore compensatory arm–trunk coordination should cease earlier to allow the trunk to contribute to hand movement extent. This could lead to an earlier divergence in hand trajectories and velocity profiles between the 2 trial types. Deficits in compensatory arm–trunk coordination were also identified by comparing arm interjoint coordination between trial types. Preliminary results have appeared in abstract form. 29
Methods
Participants
Eleven participants (age = 66.1 ± 15.5 years; Table 1) with hemiparesis due to cortical and/or subcortical stroke and 11 healthy individual (age = 51.6 ± 14.5 years) participated after signing informed consent forms approved by the CRIR Ethics Committee. Sample size was determined from preliminary data and a 95% confidence level of detecting a between-group difference in endpoint divergence times. Participants had a stroke >6 months previously with unilateral hemiparesis and at least 3/7 on the Chedoke-McMaster Scale. 30 Those with stroke due to trauma or tumor, proprioceptive or vestibular deficits, UL pain or shoulder subluxation, marked apraxia, or cognitive deficits according to medical charts and clinical evaluations were excluded. Healthy subjects were excluded if they had a history of neurological disorders or physical deficits in arms or trunk.
Demographic and Clinical Data for Participants With Stroke and Mean Age Values for Healthy Control Subjects.
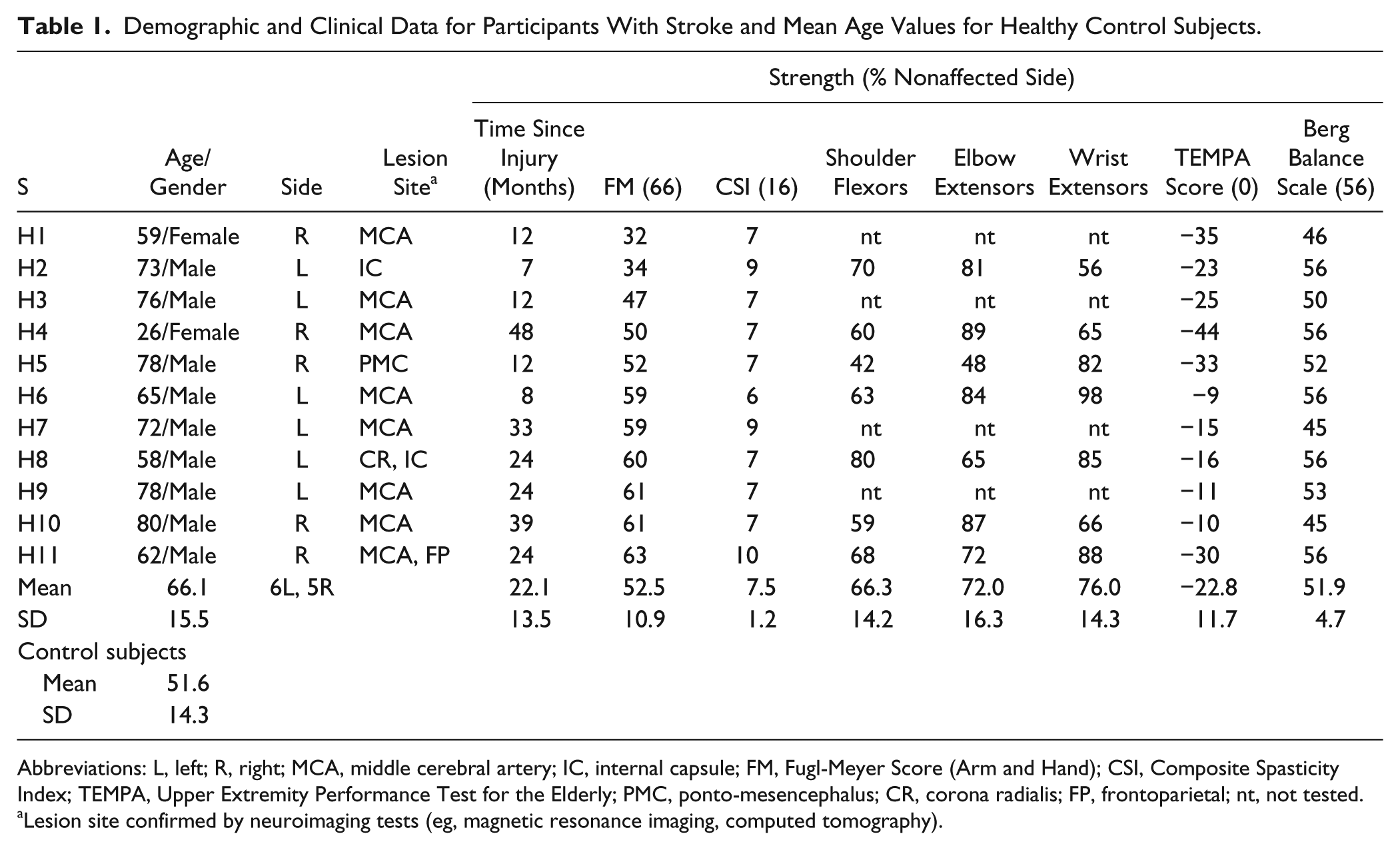
Abbreviations: L, left; R, right; MCA, middle cerebral artery; IC, internal capsule; FM, Fugl-Meyer Score (Arm and Hand); CSI, Composite Spasticity Index; TEMPA, Upper Extremity Performance Test for the Elderly; PMC, ponto-mesencephalus; CR, corona radialis; FP, frontoparietal; nt, not tested.
Lesion site confirmed by neuroimaging tests (eg, magnetic resonance imaging, computed tomography).
Clinical Evaluation
Arm and hand motor impairment and functional ability and sitting/standing balance were assessed with valid and reliable scales in stroke participants by experienced clinicians (Table 1).
Motor impairment was assessed with the following: Fugl-Meyer Scale (FM),31,32 Composite Spasticity Index (CSI),33,34 and measures of isometric force of shoulder flexors, elbow, and wrist extensors. 35 FM scale was used to determine arm and hand impairment (voluntary movement, passive range of motion, reflex function) on a 66-point scale as well as sensation, proprioception, and pain. Participants had mild (63) to moderate (32) impairment and no significant sensory deficit. CSI evaluated elbow spasticity as (a) resistance felt by the examiner to moderate-speed elbow flexor stretch, (b) biceps brachii tendon reflex excitability, and (c) wrist clonus. Spasticity scores ranged from mild 6/16 to severe 10/16.
Shoulder flexor, elbow extensor, and wrist extensor isometric force was tested with the arm in neutral using hand-held dynamometry (Nicholas, MMT, Lafayette—Model 01160) placed on the forearm, arm, and hand dorsum, respectively. Maxima of 3 exertions per muscle were expressed as percentages of contralateral arm values. Strength scores (n = 8) in the 3 muscles were ~71% of the less-impaired side (range = 42% to 98%).
Hand and arm functional ability was assessed with the Upper Extremity Performance Test for the Elderly (TEMPA), 36 evaluating 9 ADL tasks (4 unilateral, 5 bilateral). A score of “0” indicates normal function with below normal functioning ranging from −150 to 0. Scores ranged from −9 to −44 indicating mild dysfunction.
Sitting and standing balance was assessed with Berg Balance Scale (BBS),37,38 for performance on 14 balance tasks (maximum score = 56 points). BBS evaluates difficulties stabilizing the body after reduction of base of support or when center-of-mass perturbations occur. Participants had very good (45 points) to excellent balance (56 points).
Pointing Task
Participants sat in a cut-out section of a transparent Plexiglas table allowing forward trunk bending (Figure 1A). Targets (red lights) were placed within custom-made openings beneath the table. Initially, the hand was in the sagittal body midline, 30 cm from the sternum. The reaching target was placed in the ipsilateral arm workspace, 45° to the sagittal plane, at 1.3 times arm length measured from the acromion to the index. Since precision reaching was not required, the control arm was not matched for dominance. Controls performed the task with their nondominant arm and participants with stroke used their affected (hemiparetic) arm.
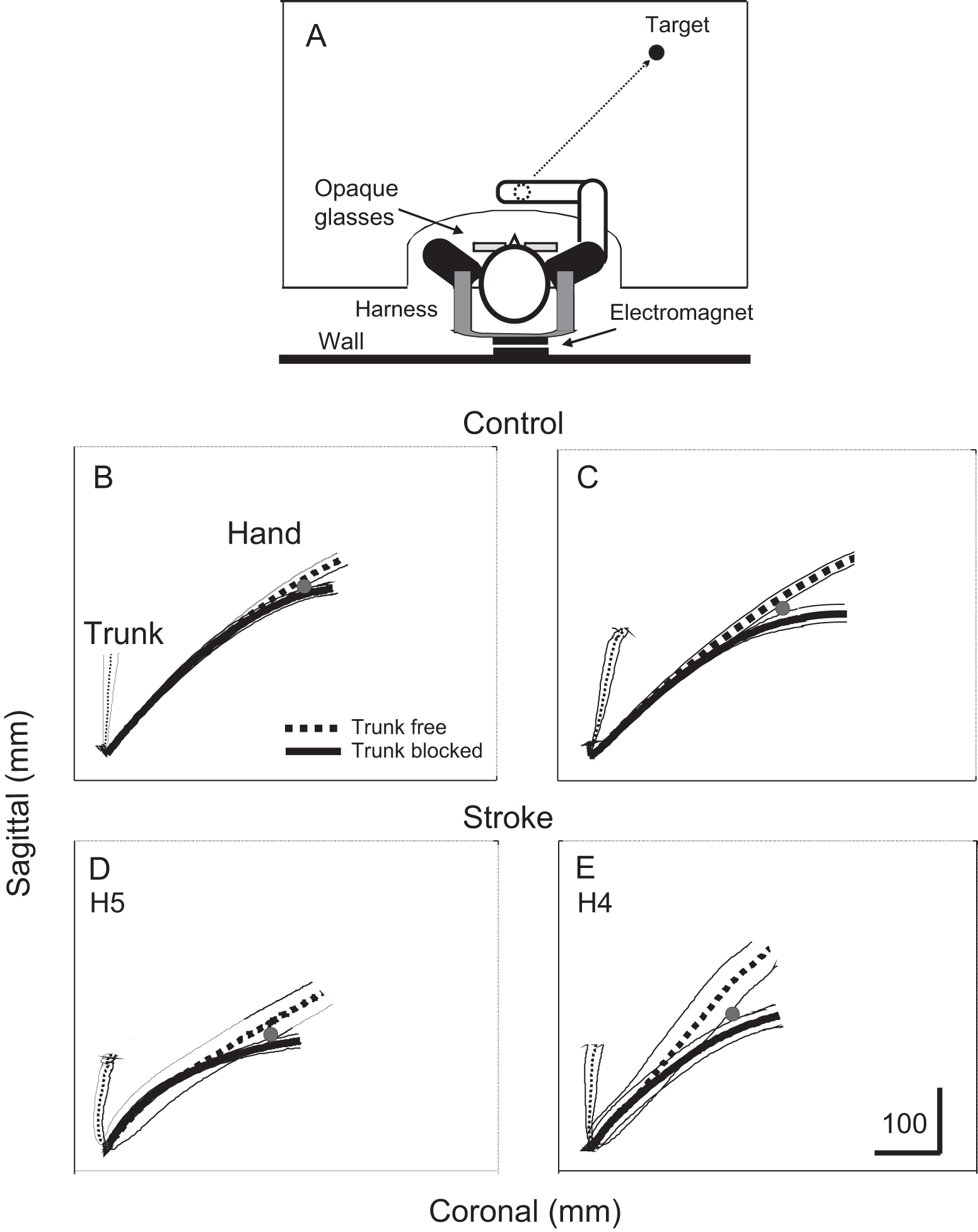
(A) Experimental set-up. The subject sat in a cut-out section of a plexiglass table and wore goggles obstructing vision during movement. Initial and final targets were lights placed beneath the table. Initial target was located 30 cm in front of the sternum. A metal plate attached to the back and an electromagnet attached to the wall minimized trunk movement in 30% of randomly selected trials. (B-E) Mean hand (thick lines) and trunk (thin lines) trajectories for reaches to the target for 2 control (B, C) and 2 subjects with stroke (D, E). Trajectories are shown for free-trunk (dashed lines) and trunk-blocked (solid lines) movements. Divergence between trajectories in 2 conditions indicated by gray dots. Despite differences in the trunk motion between conditions, hand trajectories for blocked-trunk trials initially coincided with those for free-trunk movements. The amount of coincidence was less in stroke subjects.
At a “ready” cue (light), subjects lifted their hand 2 to 3 cm above the initial position (Figure 1A, white dot), and at an auditory “go” signal, they moved the hand to the target (Figure 1A, black dot), which required trunk displacement. To minimize relative head-on-trunk displacement, subjects leaned the head and trunk forward as a single unit. Lateral trunk inclinations were permitted in the limits determined by the table cut-out (~10° per direction).
Participants performed 60 reaches without vision to the remembered target to avoid voluntary corrections. Subjects wore liquid crystal glasses (Translucent Technologies, Plato S2 Spectacles) that obstructed vision at the “go” signal. An electromagnet was attached to the wall behind the subject. In each trial, the electromagnet was initially locked to a ferromagnetic plate on the back of a harness worn by the subject. In 70% of random trials (n = 42 trials), the electromagnet was unlocked 100 ms after the “go” signal so that trunk movement was not obstructed (free-trunk condition). In the other 30% of trials (n = 18 trials), the electromagnet remained locked and trunk movement was unexpectedly blocked (blocked-trunk condition). Blocked-trunk movement was characterized by the subject straining forward against the harness. In these trials, the hand could not reach the target because of the limits of shoulder and elbow excursion. This protocol permitted the comparison of arm movement coordination with and without trunk movement when the same motor task was planned. The high number of blocked-trunk trials was necessary to assess response variability. Before recording, subjects practiced 3 to 5 free-trunk movements with vision for task familiarization. Subjects could touch the table only after the hand returned to the initial position and the glasses became transparent. Participants were instructed to perform the same type of trunk-assisted reaching in each condition and not to correct movements when trunk arrests occurred. Indeed, corrective movements in blocked-trunk trials were unlikely since arm movement was restricted by shoulder and elbow range limitations. Subjects were also instructed to move at their comfortable speed, which was monitored online for consistency. Atypical slow or fast trials were repeated.
Data Collection and Analysis
Arm and trunk kinematics were recorded by a 3D motion analysis system (Optotrak 3010, Northern Digital, Waterloo) for 4 seconds at 120 Hz. Six infrared light-emitting markers were placed on bony landmarks of the index (hand), ulna (wrist), epicondyle (elbow), right and left acromions (shoulders), and mid-sternum (trunk).
Variables were computed for hand and trunk movements made in free- and blocked-trunk conditions. Primary outcome variables were the points of divergence of (a) hand trajectories and (b) elbow/shoulder angle/angle coordination patterns between conditions. Secondary outcomes included other reaching characteristics: peak and time-to-peak hand and trunk tangential velocities, hand and trunk trajectory lengths, and trunk, elbow, and shoulder displacements.
Movements were aligned on their onsets and averaged. Movement beginning and end were defined as the times that hand and trunk tangential velocities, computed from x, y, and z positions of index and sternum markers, respectively, exceeded and remained above and then fell and remained below 5% of their maximal values. Movement time was computed between movement beginning and end. Hand and trunk marker displacements were used to calculate 3D trajectory lengths of hand and trunk movement respectively.
Spatiotemporal divergence points between free- and blocked-trunk hand trajectories were determined. Mean and standard deviations of hand trajectories for each condition (blocked, free) were aligned on onsets and plotted together. To determine the divergence point, t tests (P < .05) were done on each successive pair of points in the spatial domain of each mean trajectory. The null hypothesis was rejected if the difference between pairs exceeded a 10-mm threshold. The divergence point was characterized by (a) the absolute trajectory length between movement onset to the first point of significant spatial difference between trajectories, maintained for at least 5 successive points and (b) the corresponding time. Divergence point determination was made in both directions, that is, from trajectory beginning to end and then confirmed by reapplying the algorithm in the reverse direction. Spatial divergence points were also expressed as a percentage of the total trajectory length for each individual. For temporal divergence point estimation, since movement time differed between subjects and was consistently longer for stroke participants, divergence latency was normalized by expressing it as a percentage of the time to peak velocity for each subject. In each case, smaller numbers indicate that the traces diverged at a shorter distance or earlier.
Interjoint coordination between shoulder horizontal adduction/abduction and elbow flexion/extension and between shoulder flexion/extension and elbow flexion/extension were analyzed as the main components of reaching. Elbow and shoulder angles were computed from the dot product of vectors defined by the coordinates of adjacent markers. Elbow angle was defined by vectors formed between wrist and elbow markers and those between the elbow and the ipsilateral acromion. Shoulder horizontal abduction/adduction was measured as the angle between vectors formed by elbow and ipsilateral shoulder markers and the 2 acromion markers, projected horizontally. Shoulder flexion was the angle between elbow and ipsilateral shoulder markers and the straight line through the trunk. In addition, trunk rotation around the vertical axis was computed as the angle between the 2 shoulder markers and the frontal plane. Joint movements were calculated between movement beginning and end, defined above. The divergence point was also expressed as the angle at which the shoulder and elbow joint displacements diverged in a similar way as defined above for trajectory divergence. Divergence points were normalized by expressing them as a percentage of the maximal joint excursion for the free-trunk condition for each individual.
Statistical Analysis
Because of the relatively small sample size, nonparametric statistics were used for between-group comparisons of kinematic data (Mann–Whitney U tests) and for between-condition comparisons in each group (free-trunk, blocked-trunk; Wilcoxon signed ranks tests) using SPSS V.15. Spearman correlations were done between clinical status scores and movement parameters. Minimal significance levels of P < .05 were used.
Results
Examples of mean (±SD) hand and trunk trajectories in free- and blocked-trunk conditions in 2 representative control and stroke subjects are shown in Figure 1B to E. Figure 2 shows examples of mean arm and trunk tangential velocity traces and corresponding arm and trunk displacements in 1 control and 2 stroke subjects. In all subjects, the trunk began to move before or nearly simultaneously with the hand (hand-trunk delay <50 ms for both) and h
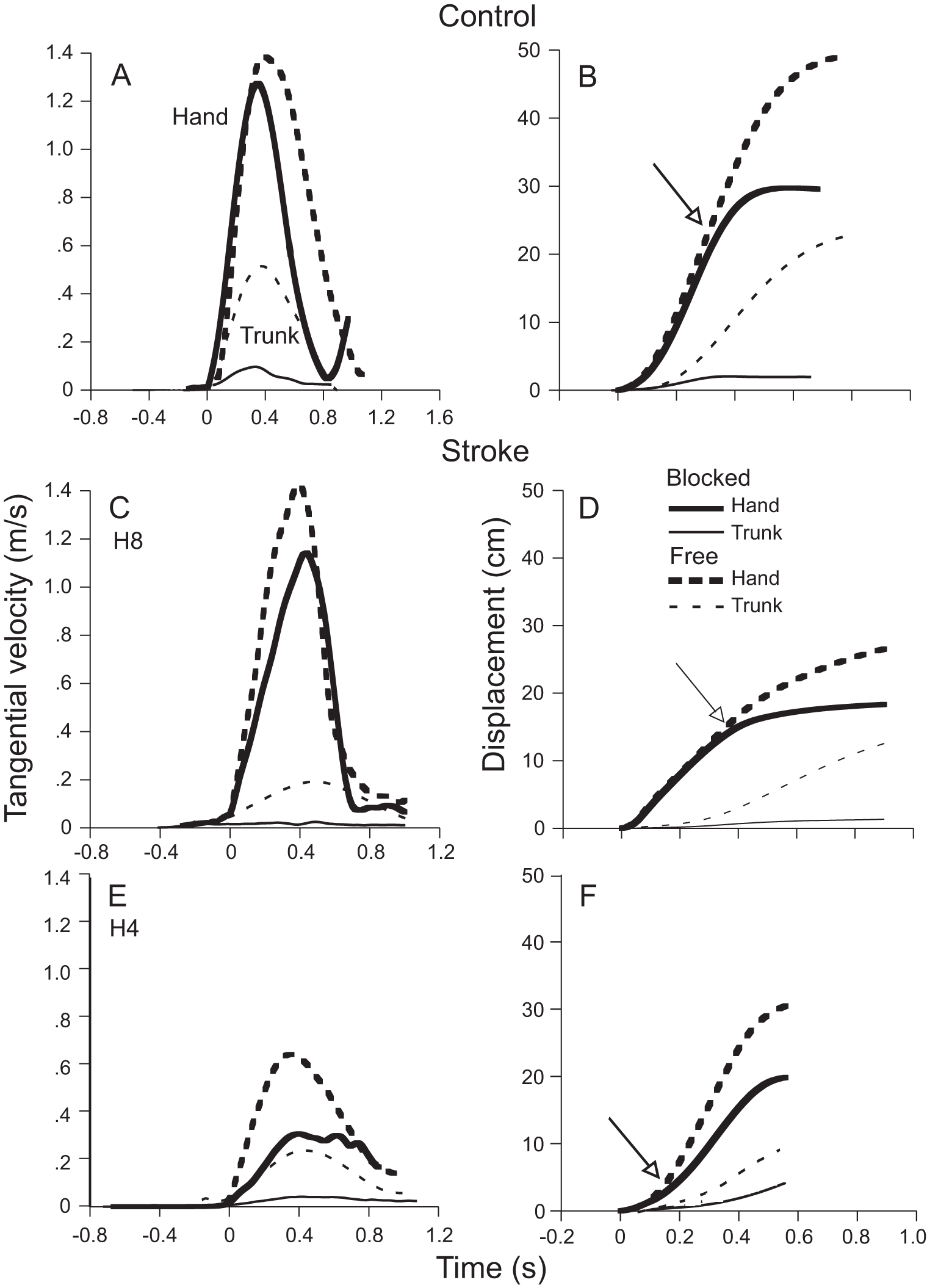
Mean tangential velocity profiles of the hand (thick lines) and trunk (thin lines) for the free-trunk (dashed lines) and blocked-trunk (solid lines) conditions in 1 control (A) and 2 individuals with stroke (C, E). Corresponding displacements of hand and trunk segments shown in B, D, and F. The points of divergence in the hand trajectory between the 2 conditions is indicated by arrows in each panel.
Comparison of Data From Healthy Control Subjects and Individuals With Stroke Making Reaching Movements When the Trunk Was Free to Move (Free) or Restrained (Blocked).
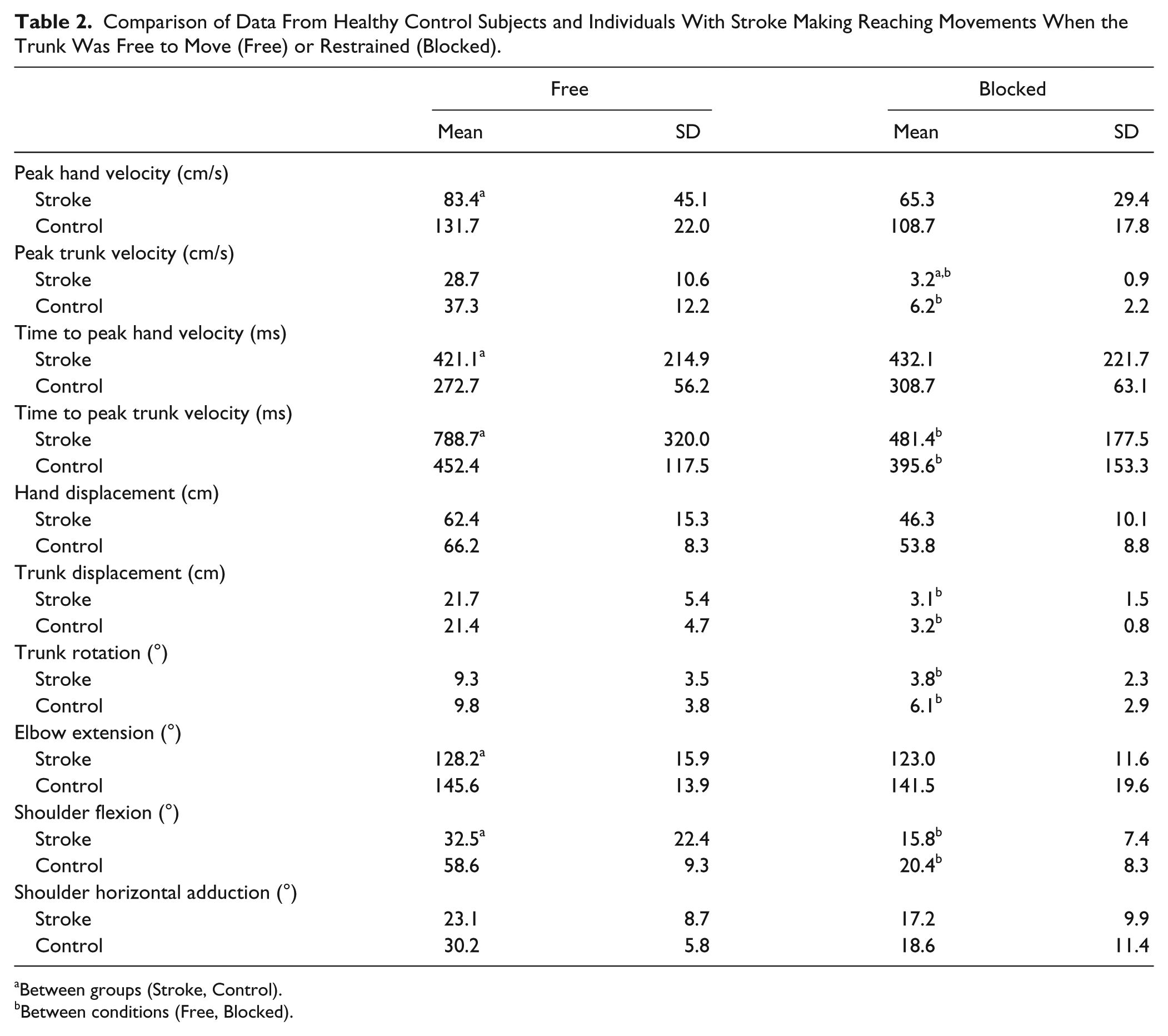
Between groups (Stroke, Control).
Between conditions (Free, Blocked).
Sagittal hand displacement extent (ie, in the main direction of trunk movement) did not differ between groups (Table 2). Peak tangential hand velocities ranged from 102 to 159 cm/s for controls and from 45 to 166cm/s for stroke (Table 2). Overall, stroke participants had lower hand but not trunk peak velocities (U = 21, z = −2.192, P < .03) compared to controls. Times to peak hand (U = 21, z = −2.197, P < .03) and trunk (U = 20, z = −2.628, P < .005) velocities were also longer in stroke compared to controls (Table 2). However, although arm movements were slower in stroke participants, velocity relationships between arm and trunk movements were similar between groups. Thus, in controls, the arm moved approximately 3.5 times faster than the trunk compared to about 3.0 times faster for the stroke group. For joint ranges, controls used more elbow extension (U = 15, z = −2.989, P < .002) and shoulder flexion (U = 22, z = −2.532, P < .01) than stroke subjects (Table 2) but a comparable amount of shoulder horizontal adduction.
Trajectory Divergence Points
In controls, hand trajectories (Figure 1A and B) and velocity profiles (Figure 2A and B) of blocked-trunk trials initially matched those from free-trunk trials and then began to diverge. The spatial divergence between arm paths occurred at a significantly shorter distance in stroke (31.4 ± 9.9 cm from movement onset) compared to controls (44.3 ± 10.8 cm from movement onset; U = 17.0, z = −2.496, P < .02; Figure 1B-E). This corresponded to a mean divergence at 51.7 ± 18.9% of the total hand path distance in stroke compared to 67.4 ± 15.5% for controls (U = 17.0, z = −2.496, P < .02; see Figure 4A).
Movements were slower in stroke participants compared to controls, and raw divergence point latencies did not differ between groups (stroke: 624.7 ± 257.3 ms; controls: 442 ± 105 ms). When divergence times were expressed relative to peak hand velocity times, trajectory divergence occurred at approximately the same time in both groups (stroke: 166.7 ± 91.5%; controls: 163.1 ± 29.9% of time to peak velocity).
Angle–Angle Divergence Points
To determine how compensation was accomplished, we analyzed the coordination between arm joint pairs contributing to the hand movement. In controls, shoulder flexion/elbow extension interjoint coordination patterns differed in the 2 types of trials (Figure 3A and B), as indicated by a spatial divergence point between these patterns (26.9 ± 20.1° or 34.2 ± 21.8% of the total free-trunk elbow excursion). The spatial divergence in the shoulder flexion/elbow extension coordination patterns occurred later in stroke (32.7 ± 16.0° or 64.1 ± 28.4% of total elbow excursion; U = 24.5, z = −2.366, P < .02; Figure 4) but not those between shoulder horizontal adduction/elbow extension.
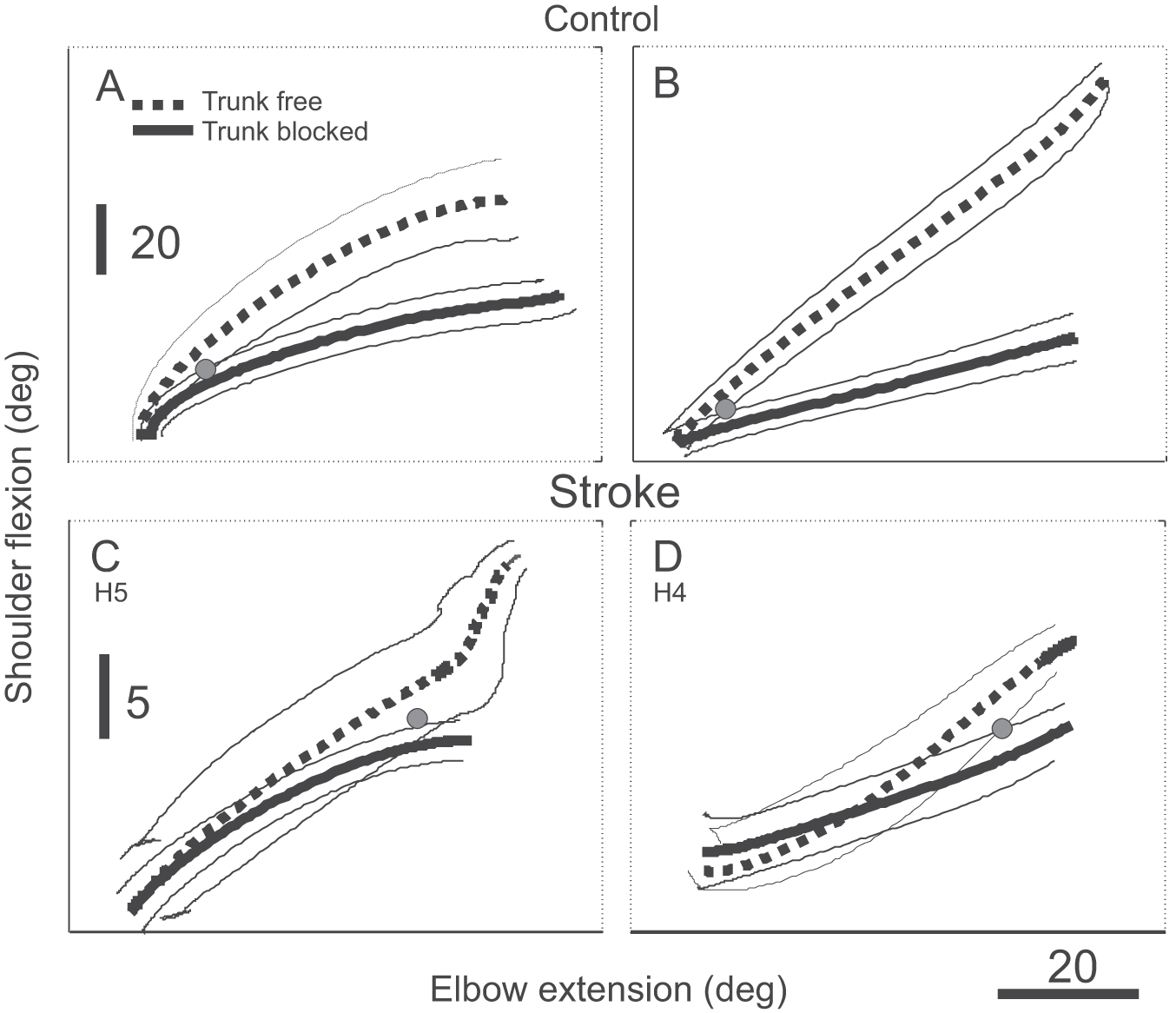
Typical traces of (mean) interjoint coordination patterns between the elbow (flexion/extension) and the shoulder (flexion/extension) for free-trunk (dashed lines) and blocked-trunk (solid lines) conditions for 2 control subjects (A, B) and 2 participants with stroke (C,D). The point of divergence between patterns is indicated by gray dots. The divergence points occurred after a bigger change in the elbow angle in the stroke subjects. Note the difference in the vertical scales in the graphs for control and stroke subjects.
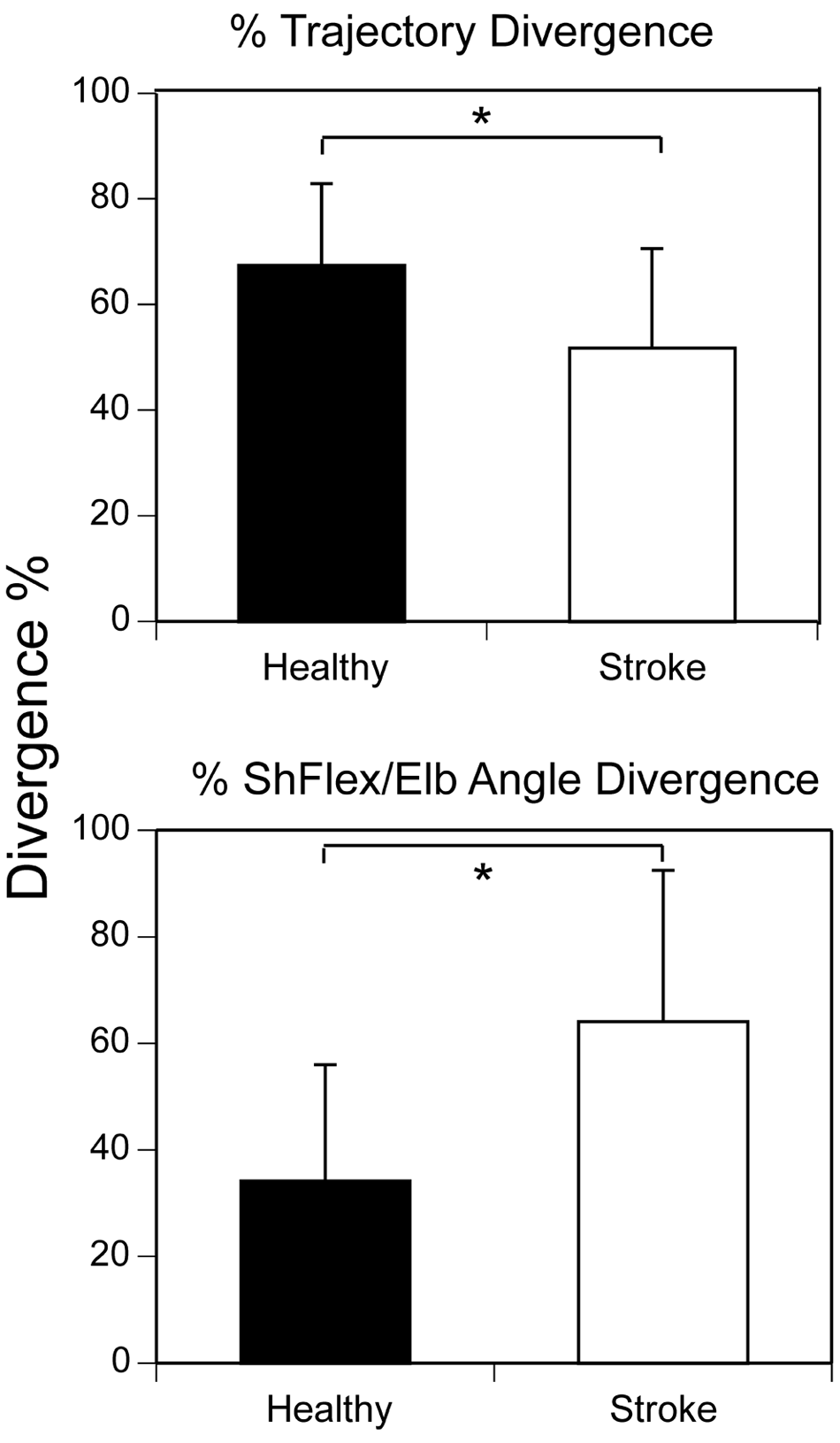
Trajectory divergence (top) and interjoint angle divergence (bottom) for control (black bars) and stroke subjects (white bars) expressed as a percentage of the total trajectory or angular length in the free condition; *P < .02.
To determine the relationship between changes in coordination of reaching and stroke severity, we correlated trajectory and joint angle divergence values with clinical tests of impairment (FM, muscle force, CSI, BBS). In participants with more severe clinical impairment, hand trajectories diverged earlier (BBS: 0.55, P < .04; FM: 0.52, P < .05) and in those with greater spasticity, interjoint coordination patterns diverged later (CSI: 0.58, P < .04).
Discussion
Basic Findings
We investigated how arm and trunk movements during reaching toward targets placed beyond arm length were coordinated in individuals with poststroke hemiparesis. We compared arm trajectories, velocity profiles, and interjoint coordination patterns during reaching when the trunk was suddenly blocked to those of free movements to determine spatial and temporal points at which hand trajectories and angular profiles in the 2 trial types diverged. The divergence point is critical for the analysis of arm–trunk coordination during reaching beyond arm reach: it indicates the point at which trunk motion begins to contribute to arm movement extent. 1
We confirmed previous findings1,23 that healthy subjects could neutralize the initial influence of trunk displacement on the hand trajectory by compensatory arm–joint rotations and that the trunk contributed to hand displacement mainly toward the end of the reach. In contrast, in stroke, the trunk contributed after a shorter hand displacement, shown by earlier hand trajectory divergence in the 2 types of trials, thus confirming the hypothesis tested in this study. The earlier divergence was associated with disrupted compensatory arm joint rotations as indicated by a later divergence in shoulder–elbow interjoint coordination patterns.
Arm–Trunk Coordination in Controls
In the previous study in healthy individuals, hand trajectory and velocity profiles in blocked-trunk trials initially matched those from free-trunk trials until the time of hand peak velocity. 1 In contrast, arm interjoint (shoulder–elbow) coordination differed in the 2 types of trials, suggesting that the influence of the trunk on the hand movement trajectory in free-trunk trials was initially compensated by adaptive changes in shoulder–elbow interjoint coordination, which occurred at a latency consistent with a rapid reflex mechanism (~50 ms). In the present study, we characterized compensatory interjoint coordination as a decrease in shoulder flexion and horizontal adduction (Table 2) in the blocked-trunk compared to the free-trunk condition (Figure 1B-E).
The compensatory arm–joint behavior during beyond arm length reaching has been explained within the context of the equilibrium-point theory of motor control. The theory suggests that reaching movement results from a feed-forward shift in the referent hand position (R).39,40 Thereby, arm muscle activation emerges depending on the distance (and its rate of change) between the actual and the referent hand positions. Shifts in referent hand position terminate at about the time of peak hand velocity. 6 In our study, this event is presumed to be associated with the moment at which the referent hand position reaches a critical distance (Rc), about 90% of the fully extended arm length. At this point, the early arm–joint compensatory changes begin to be attenuated, allowing the trunk to contribute to hand movement extent. For controls and some stroke participants, initial trunk displacement did not contribute to hand displacement until the limit of virtual arm extension was reached.
Rossi et al 1 attributed the early compensation to either vestibular and/or proprioceptive afferent signals linked to trunk motion.41,42 Additional studies in deafferented 43 and vestibulo-deficient patients44,45 favor the vestibulo-spinal mediation of this response.
Arm–Trunk Coordination in Stroke
In stroke subjects, trajectory paths spatially diverged earlier and interjoint coordination patterns diverged later than in controls. For example, in Figure 2F, hand displacements in free-trunk and blocked-trunk conditions did not coincide indicating incomplete or no compensation. The decreased trajectory coincidence was likely due to an impaired ability to rapidly adapt the interjoint coordination pattern to neutralize the trunk influence on hand movement extent. This resulted in the trunk contributing to the hand trajectory from movement onset as previously shown for within-arm length reaching. 16
Since stroke participants had no proprioceptive or vestibular deficits, differences in adaptive arm–joint movement may be attributed to other mechanisms. One possibility is a deficit in trunk control often reported in patients with stroke. 46 However, it is unlikely that chronic stroke subjects in our study had trunk control deficits since the range and timing of trunk recruitment did not differ from controls (Table 2) and they had relatively high Berg Balance Scores (Table 1).
A second possibility is that the system reacted too slowly to allow arm joints to appropriately compensate trunk displacement. This is supported by lower peak velocities and longer times to achieve peak arm and trunk velocities in stroke (Table 2). Deficits in adaptive interjoint coordination may also be related to decreased information processing speed, 47 prolonged reaction times for movement sequences with the hemiparetic arm, 48 and/or spasticity and muscle weakness.17,49 These cognitive and motor deficits may impair the ability of participants with stroke to produce appropriately timing of movement components.
Another possibility is that trunk movement contributed to the reach earlier because of a lower threshold at which the contribution was initiated. This implies that there was a decrease in the critical referent hand position, Rc, at which trunk motion began to contribute to hand movement extent in the stroke group. Previous studies have shown that when individuals with stroke reached to within-reach targets, arm interjoint coordination was disrupted,26,27 excessive (unnecessary) trunk recruitment occurred,13,15 and trunk movement contributed to arm displacement from the reach beginning. 16 Compensatory trunk displacement during reaching has been attributed to limitations in active elbow/shoulder motion range in individuals with stroke, 13 likely due to deficits in joint agonist/antagonist muscle activation,49,50 weakness,17,51,52 and difficulty controlling interactive torques.22,23 This is supported by findings of correlations between clinical impairment level and earlier divergence of interjoint coordination patterns.
Our results suggest that part of the reason individuals with poststroke hemiparesis use excessive trunk movement during reaching may be the inability of the damaged nervous system to critically coordinate arm–trunk movements in early reach. Central nervous system damage disrupts the elegant coordination necessary to produce smooth goal-directed arm movement. The damaged system may partly overcome range of motion limitations as well as in arm–trunk coordination by lowering the threshold, Rc, when the trunk begins to contribute to hand displacement, thus making use of additional body DFs to achieve the reaching goal.
Limitations
Since the study involved a small number of patients with relatively good UL recovery, generalization of results to a larger stroke population should be done with caution. It is also possible that responses recorded in the subjects with stroke might be affected by the side and type of stroke as well as dominance, which were not considered here.
Conclusions and Clinical Implications
Participants with stroke had mild-to-moderate clinical impairment, yet they still showed marked deficits in coordinated movement production. These findings cannot be extended to individuals with more severe clinical syndromes though it is likely that they would show even greater disruptions in coordination. Present findings may inform clinical practice as to what type of task components should be practiced in moderate to well-recovered patients to improve arm motor recovery for higher-order complex movement tasks. Specifically, clinicians may encourage patients to practice UL tasks involving different DFs in different combinations in order to improve interjoint and intersegment coordination. Future research may identify effectiveness of interventions targeting interjoint and intersegment coordination on UL functional recovery.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the Natural Science and Engineering Council of Canada (NSERC) and the Mach Gaensslen Foundation. MFL holds a Tier 1 Canada Research Chair in Motor Recovery and Rehabilitation.
