Abstract
Background. Since the brain is intact, persons with a spinal cord injury (SCI) might benefit from a brain–computer interface (BCI) to improve mobility by making use of functional near-infrared spectroscopy (fNIRS). Objective. We aimed to use fNIRS to detect contralateral primary motor cortex activity during attempted foot movements in participants with complete SCI. Methods. A 6-channel fNIRS, including 2 reference channels, measured relative concentration changes of oxy- (HbO) and deoxy-hemoglobin (HbR) in the contralateral motor cortex for the right foot. Seven subjects, studied within 18 months after injury, performed 12 trials of attempted right foot and real hand movements. Results. T tests revealed significant HbO and HbR responses of the left motor cortex for attempted foot movements, but not for right hand movements. A 2-way repeated-measures analysis of variance revealed a larger decrease in HbR for attempted foot movements compared to hand movements. Individual results show major interindividual differences in (number of) channels activated and the sensitive chromophore (HbR or HbO). Conclusions. On group level, activity in the motor cortex of the foot can be measured with fNIRS in patients with complete SCI during attempted foot movements and might in principle be used in future BCI studies and applications.
Keywords
Introduction
The incidence of spinal cord injury (SCI) over the past decades has not changed substantially, but the percentage of complete SCI has slightly decreased as a result of new and improved acute treatment modalities. 1 However, the dramatic increase in life expectancies for SCI patients since World War II emphasizes the need to optimize their quality of life. 2 SCI patients report a decrease in hand and arm function (quadriplegic) and a decrease in mobility as one of their major problems. 3 Therefore, developing alternatives for these patients in terms of independence in mobility would be a great advantage. For this reason, and since the brain is still completely intact, SCI patients might in principle benefit from a brain–computer interface (BCI) to improve the mobility. 4 Several successful attempts are made using invasive BCIs to allow quadriplegic SCI patients to control computer cursors or assistive devices.5-8 However, these invasive methods are still under debate and also major successes have been made in the field of BCI using noninvasive neuro-imaging techniques.
As noninvasive neuroimaging techniques in BCI, functional magnetic resonance imaging (fMRI), positron emission tomography, and magnetoencephalography are regularly used to demonstrate the ability of the brain to control and adapt to an external device or computer. 9 Since there are various practical limitations using the aforementioned techniques, a lot of research on BCIs is performed using electroencephalography (EEG) as the neuroimaging technique applicable in daily life. Various EEG studies successfully demonstrated the possibility to control a EEG-based BCI by using intentional movements, which are closely related to that what is controlled.10-12 Functional near-infrared spectroscopy (fNIRS), a noninvasive neuroimaging technique that measures cortical hemodynamic changes, could be of additional value in the field of BCI. Although fNIRS has its limitations, in contrast to EEG, fNIRS is not susceptible to electrical noise from surrounding equipment, muscle contractions, or head movements. 13 Other advantages are the portability, accessibility, low cost, and the ease of using the system. Therefore, fNIRS will play a role in future studies on cortical control of gait. The present study still used isolated foot movements, which are commonly used in fMRI studies as a substitute for locomotion.14-16 Nagaoka et al 17 demonstrated a proof of principle of an fNIRS-based BCI in healthy subjects who controlled an upper extremity muscle with functional electrostimulation. Other neuroimaging techniques have also shown potential for controlling a BCI by SCI patients18,19 and several invasive BCI even demonstrated real working BCIs in SCI patients.5,6,8 In addition to these invasive BCI accomplishments, some BCI studies in SCI patients used a noninvasive neuroimaging technique. Kauhanen et al 20 demonstrated results of a BCI experiment that enabled SCI patients to move a circle on a monitor by imagined left and right hand movements. In addition, Pfurtscheller et al 21 demonstrated a quadriplegic patient that controlled the upper extremity by functional electro-stimulation using imagined hand movements. Hence, although some BCI studies have been performed, non-invasive BCI research in SCI patients is still sparse.
A possible problem for BCI applications in SCI patients might be the reorganization of the motor representations on the cortex in this patient group. Several fMRI studies in chronic SCI patients showed preserved motor representations long after the injury.22-24 Nevertheless, some motor cortical reorganization has been demonstrated in some studies with paraplegic SCI. 25 This is convincingly shown by the study of Lotze et al, 26 who demonstrated a medial shift of almost 20 mm in the location of the intact cortical area involved in the control of the elbow flexor in complete thoracic SCI patients. In addition, reorganization is also noticed in the motor cortical regions formerly responsible for movements of body parts below the level of injury. Nevertheless, Müller-Putz et al 18 demonstrated typical motor cortex activity during “attempted” foot movements in an SCI patient 4 months after injury. In contrast, SCI patients, measured longer periods after date of injury, showed atypical broad areas of activation or a lack of motor cortex activity. Since the study of Lotze et al 26 also revealed a more pronounced reorganization with increasing time after SCI injury (ranging from approximately 4 months to 34 years), this factor might account for the inconsistent findings of preserved motor control. Therefore, the present fNIRS study focused on a group of SCI patients measured within a relative short period after the SCI (ie, 1-16 months). Starting early after the SCI with the use of a BCI might also be relevant for the patients in their future use of BCI, since preservation of motor representations might occur by the continued stimulation of the motor areas. It was hypothesized that it would be possible to detect hemodynamic responses in the medial part of the motor cortex for attempted foot movements in paraplegic SCI patients early after the SCI. In addition to attempted foot movements, hemodynamic responses on the medial part of the motor cortex were studied during real hand movements. These hand movements were added since hemodynamic changes in fNIRS studies show that basically the foot and hand motor areas can be discriminated but still there is considerable overlap.27,28 Therefore, the task of hand movements was included to check for the selectivity of the activations. We hypothesized that attempted foot movements would reveal larger hemodynamic changes in the medial part of the motor cortex compared to the real hand movements.
Methods
Subjects
Seven patients with complete SCI participated in the study (mean age = 33.8 years, SD = 14.5). All the patients had a trauma-induced SCI at thoracic (n = 6) or cervical (n = 1) level. The duration of the lesion was on average 7.3 months (SD = 5.8) and ranged from 1 to 16 months. Individual characteristics are presented in Table 1. All subjects voluntary participated in the study and signed an informed consent.
Subject Characteristics and Significant Channels.

Abbreviations: ASIA, American Spinal Injury Association; Th, thoracic; C, cervical; HbO, oxy-hemoglobin; HbR, deoxy-hemoglobin; ch, channel.
These patients showed extremely low signal-to-noise ratios at channel 4.
This patient suffered from neuropathic below-level spinal cord injury pain. The pain was also regularly present during the experiment.
The channel numbers in the last four columns with an asterisk indicate significance (P < .01), those with a dagger symbol indicate a trend (.01 < P < .05), and the numbers between brackets indicate a significant response with an inverse amplitude (eg, a decrease in HbO or an increase in HbR).
Instrumentation
Oxy-hemoglobin (HbO) and deoxy-hemoglobin (HbR) concentration changes were measured using a pulsed continuous-wave NIRS instrument, the OXYMON (Artinis Medical Systems, Zetten, The Netherlands). Data acquisition was performed with 2 different wavelengths of near-infrared light (760 and 860 nm) and sampled at a frequency of 10 Hz. Two receivers and 4 light-emitting optodes were used to create a 6-channel setup of 2 reference channels and 4 channels for cortical activity detection (see Figure 1). Reference channels were used to correct for superficial hemodynamic fluctuations unrelated to cortical activity.29,30 For the 2 reference channels, a distance of 10 mm was used; for 2 of the long-distance channels, an interoptode distance of 30 mm was used and for the other 2 channels, the interoptode distance was 35 mm (see Figure 1). The distance between the 2 receivers was 18 mm. We determined the Cz position using the international 10-20 system and positioned the optodes just left to this position on the motor cortex of the right foot (Figure 1); that is, only the contralateral motor cortex was measured. Foam of 10 mm thickness with holes for the optodes was used to ensure stable positioning of the equipment.
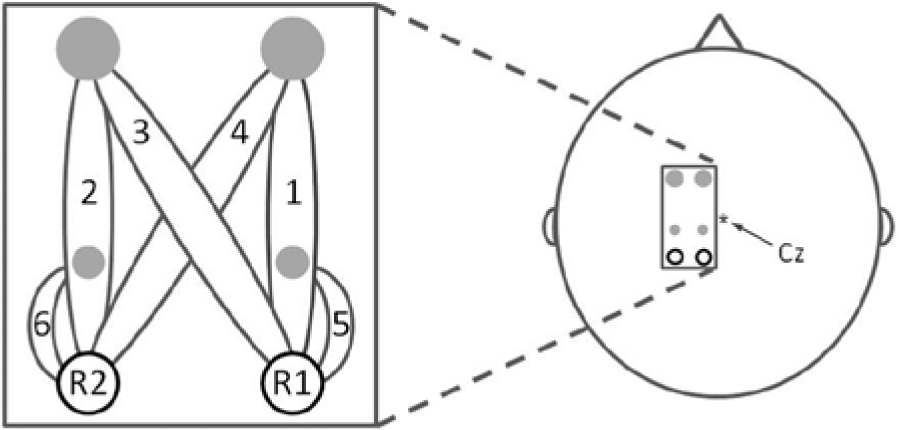
Optodes and channel configuration.
Experimental Setup
Subjects were sitting in their own wheelchair at a table in front of a screen that presented instructions before and during the experiment. First, a check was performed with 2 trials of attempted foot movements to ensure a hemodynamic response was measured. If no response was seen the channels were relocated and once again checked for a hemodynamic response. The relocation procedure was repeated until a visually detectable average hemodynamic response over 2 trials was seen with a maximum of 3 replacements (1 cm more cranially, 1 cm more caudally, and back to the original position). Subsequently, 12 trials of real right hand tapping (“HAND”) and 12 trials of attempted right foot tapping movements (“FOOT”) were randomly performed. Each trial lasted 25 seconds and was alternated with a baseline period ranging between 20 and 30 seconds. After the first 12 trials, a rest period of approximately 5 minutes was provided. Before each part of the experiment (hemodynamic check and experiment parts I and II), 1 minute of rest data were recorded for reference channel factor calculations, which were performed afterward. During this 1-minute rest period, subjects were instructed to focus on a cross on the screen and not to move or attempt to move.
Before each task period the instruction “FOOT” or “HAND” was presented for 2 seconds on the screen to indicate which task had to be performed. Subsequently, a bar started to move upward and downward with a frequency of 0.8 Hz. Participants were instructed to perform right hand tapping at the same rate as the moving bar in case of the HAND condition and to attempt plantar and dorsal flexion of the right ankle at the same rate as the moving bar in case of the FOOT condition. Baseline periods started by the instruction “REST” for 2 seconds and subsequently a cross was presented for the remaining baseline period. During the baseline periods participants were requested to focus on the cross and not to move.
Data Analysis and Statistics
Changes in optical densities were converted into changes in HbO and HbR using the modified Beer–Lambert law and the age-dependent pathlength factor (DPF = 4.99 + 0.067 × AGE0.814), as described by Duncan et al, 31 Figure 2 shows an overview of all the following data analysis steps. First, HbO and HbR concentrations were filtered with a second-order high-pass Butterworth filter with a cutoff frequency of 0.01 Hz to remove the drift in the signals and a second-order low-pass Butterworth filter with a cutoff frequency of 1.25 Hz was applied to remove high-frequency noise. To correct for superficial fluctuations, multiplication factors were calculated for each channel by correlating the one minute rest data of each channel with the corresponding reference (short distance) channel. Using these multiplication factors, the concentration changes of the long-distance channels were corrected by subtracting the reference channel data multiplied with the factor.29,30 Finally, a second-order low-pass Butterworth filter with a cutoff frequency of 0.5 Hz low-pass filter was used to remove remaining heart rate and high-frequency fluctuations. All filters used in the data analysis were noncausal. Data of all trials were averaged time locked to the start of the task for each channel and each condition. For HbO and HbR separately, the mean hemodynamic response was calculated by subtracting the mean hemodynamic response of the 10 seconds before the start of the task instruction from the mean hemodynamic response of the whole task period. These calculations were separately performed for each trial and each channel.
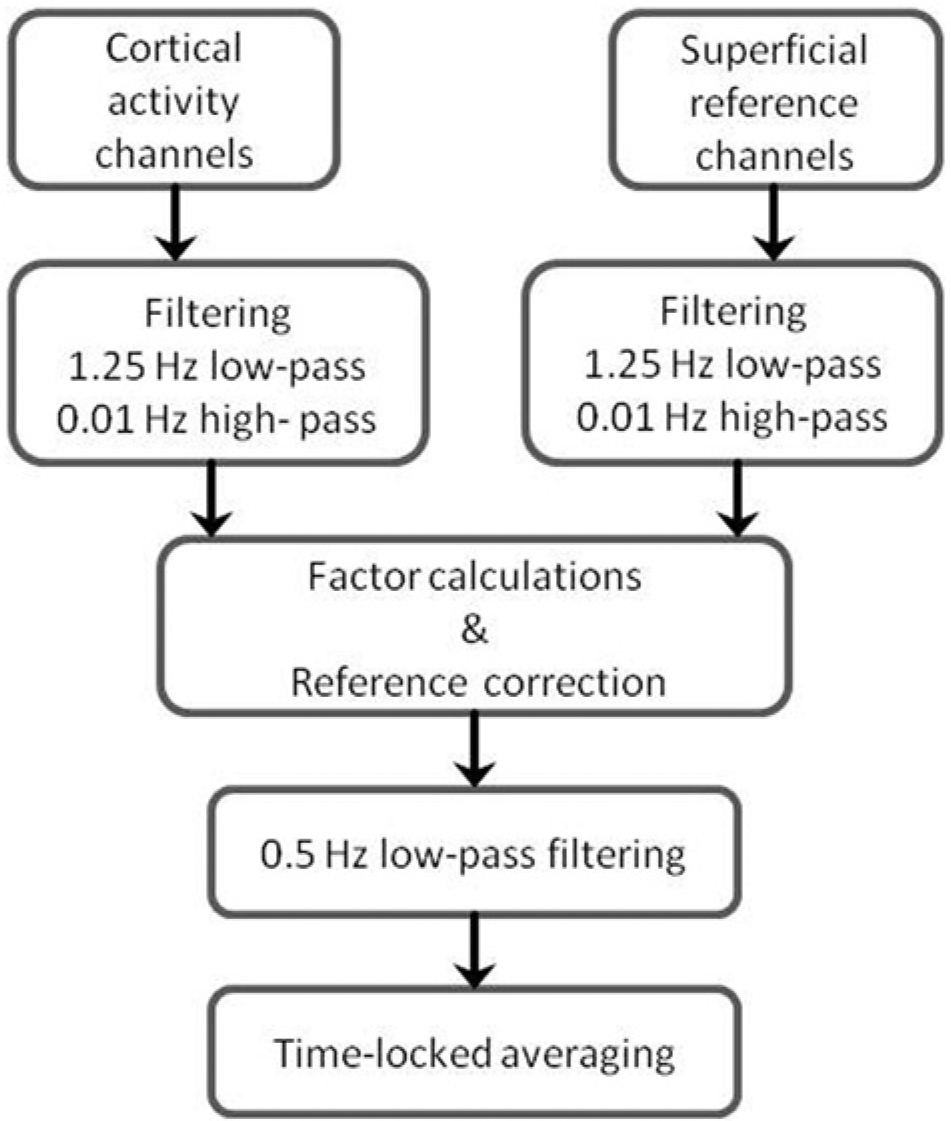
Flowchart of data analysis.
Using the mean hemodynamic responses (as described above) for each subject as outcome parameter, t tests on group level were used to test for significant responses on the separate conditions and channels. Two-way repeated measures analyses of variance (ANOVAs) were used to test differences between execution of hand movements and attempted foot movements (“conditions” factor) and between the long-distance channels (“channels” factor) for the HbO and HbR mean hemodynamic responses on group level. Because of low signal-to-noise ratios for channel 4 in 2 of the 7 patients, the ANOVAs were performed using channel 1 to 3. For the ANOVAs, the individual data were normalized to the highest increase or decrease over all trials of all conditions and all long-distance channels, since large interindividual differences in response amplitudes are usually present in fNIRS data. Finally, on group level, the hemodynamic responses were correlated with the time after SCI for the 2 conditions and 4 channels separately. The significance level for the aforementioned statistics was set at P < .05.
For each individual, t tests were used to test for significant responses for each condition and each channel. In addition, the difference between the 2 conditions at each channel was tested for significance with a t test. To compensate for multiple testing, we considered results with P < .01 as significant and P < .05 as trends.
Results
For the attempted foot movements condition, the most centrally located channel (channel 1) revealed a significant HbO increase of 0.30 µM (SD = 0.32; P = .048) and a significant HbR decrease of 0.28 µM (SD = 0.29; P = .045) for the complete SCI patient group. In addition, channel 3 revealed a significant HbO increase of 0.43 µM (SD = 0.33; P = .01) and a significant HbR decrease of 0.32 µM (SD = 0.27; P = .02) for attempted foot movements. Figure 3 illustrates the hemodynamic changes during attempted foot movements in channel 3 across all subjects. A typical hemodynamic response of an increase in HbO reaching a plateau during the task period and a decrease in HbR was seen. Subsequently, HbR and HbO returned to baseline after the task period. In contrast to the attempted foot movements, real hand movements revealed no significant hemodynamic responses in all channels (P values >.2, except for HbO channel 1, P = 0.13; HbO channel 3, P = .14; and HbR channel 4, P = .19). Figure 4 gives an overview of the group results for the mean hemodynamic responses of HbO and HbR at the 4 channels positioned on the motor cortex of the foot during real hand and attempted foot movements. The hemodynamic responses for attempted foot movements were not significantly correlated with the time after the SCI. Correlation coefficient values across the channels ranged from −0.38 to 0.26 (P values >.4). For hand movements, the correlations coefficients ranged from −0.04 to 0.64, but here too no significance was found. Noteworthy, the mean hemodynamic responses (HbO of channels 1 and 2 and HbR of channel 3) for hand movements tended to increase with increasing time after SCI (r values [P values]: 0.58 [.17], 0.60 [.15], and 0.64 [.12], respectively, all other P values were higher than .2).
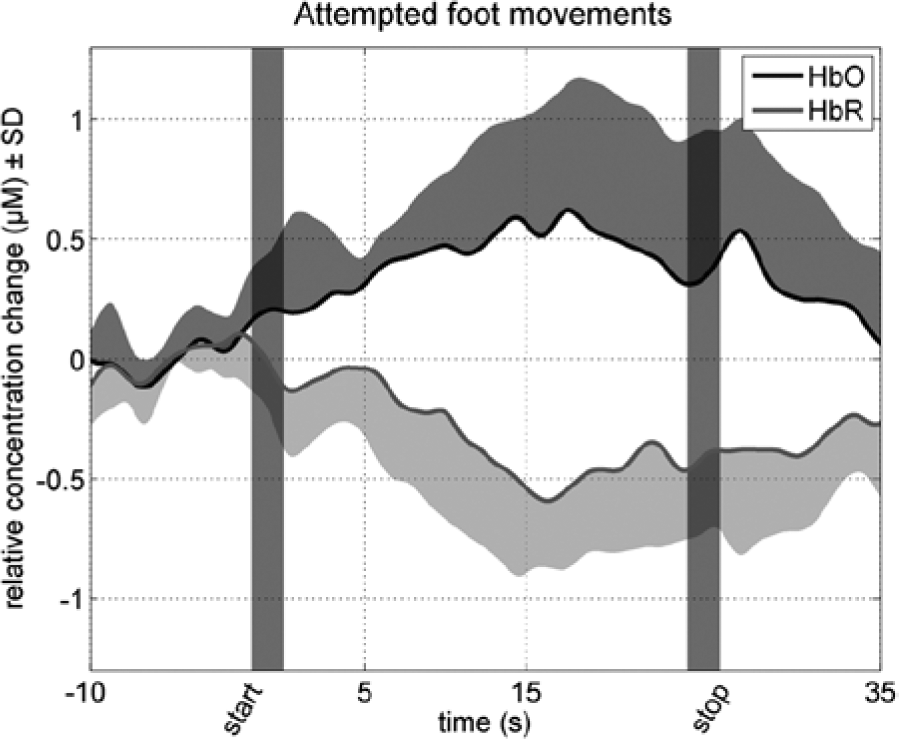
Hemodynamic changes during attempted foot movements.
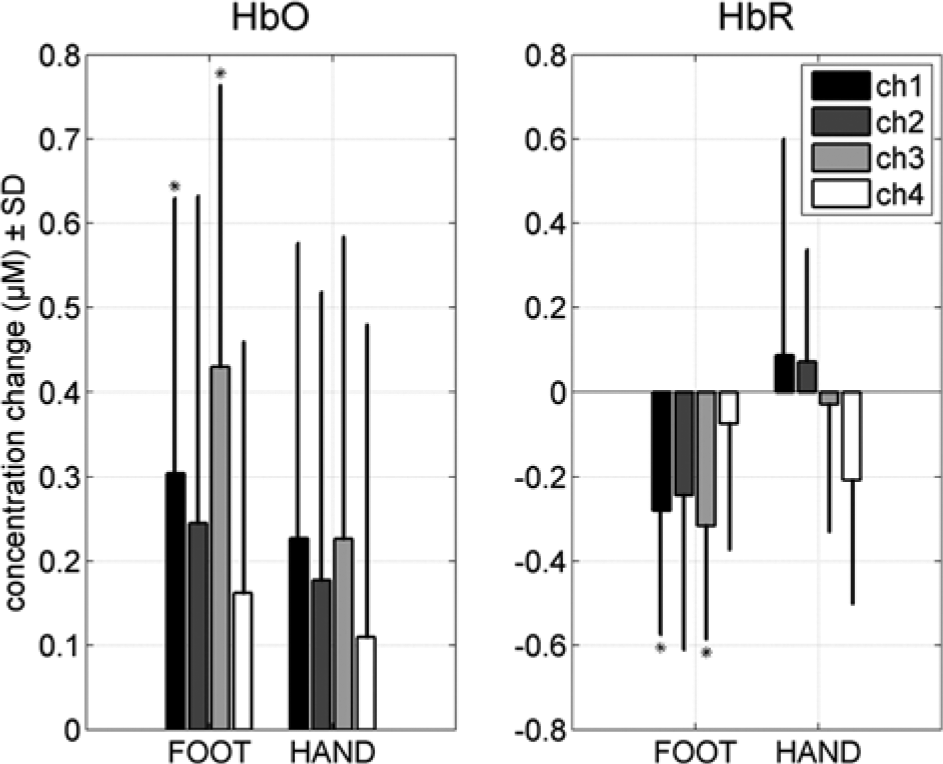
Group hemodynamic responses.
The ANOVA to compare the mean HbO responses between the different conditions and channels revealed no interaction effect, F(2, 6) = 0.77, P = .48. In addition, no main effect for condition, F(1, 6) = 0.57, P = .48, and no main effect for channel was found, F(2, 6) = .66, P = .54). For HbR, on the other hand, the ANOVA revealed a significant condition effect, F(1, 6) = 11.09, P < .02, but no significant interaction effect, F(2, 6) = 0.08, P = .93, and no main channel effect, F(2, 6) = 0.83, P = .46. Larger HbR decreases were found for attempted foot movements (−0.28 µM, SD = 0.30) compared with real hand movements (0.04 µM, SD = 0.36).
Table 1 shows the channels revealing a significant hemodynamic response amplitude for each individual. Five of the 7 SCI patients revealed a trend (P < .05) or significant (P < .01) responses for foot movements in 1 or more of the 4 channels, in HbO, HbR, or both. Remarkably, channel 3, the channel with the largest interoptode distance (35 mm) and thereby measuring the largest cortical area, showed significance or trends during foot movements in each of these 5 subjects. One patient (S2), the only patient who suffered from neuropathic below-level SCI pain (also during the experiment), showed no significant responses at all. Subjects S1 and S5 revealed a trend of a larger response for the attempted foot movements compared to the real hand movements of 1.2 µM (SD = 1.4) in HbO at channel 1 (P = .01) and of −1.0 µM (SD = 1.1) in HbR at channel 2 (P = .02). To provide more insight into the differences between subjects, the time courses of the hemodynamic responses on channel 3 for 3 typical subjects in the foot and hand conditions are shown in Figure 5. In the left panels of Figure 5, the time courses of the hemodynamic responses are shown for S1, the subject who revealed a significant difference between real hand and attempted foot movements. In addition to S1, the hemodynamic responses over time for hand and foot movements in subject S2 (center panels) and S3 (right panels) are shown. Subject S2 did not have a significant average hemodynamic response in both conditions. Finally, subject S3 showed similar hemodynamic responses for hand and attempted foot movements.
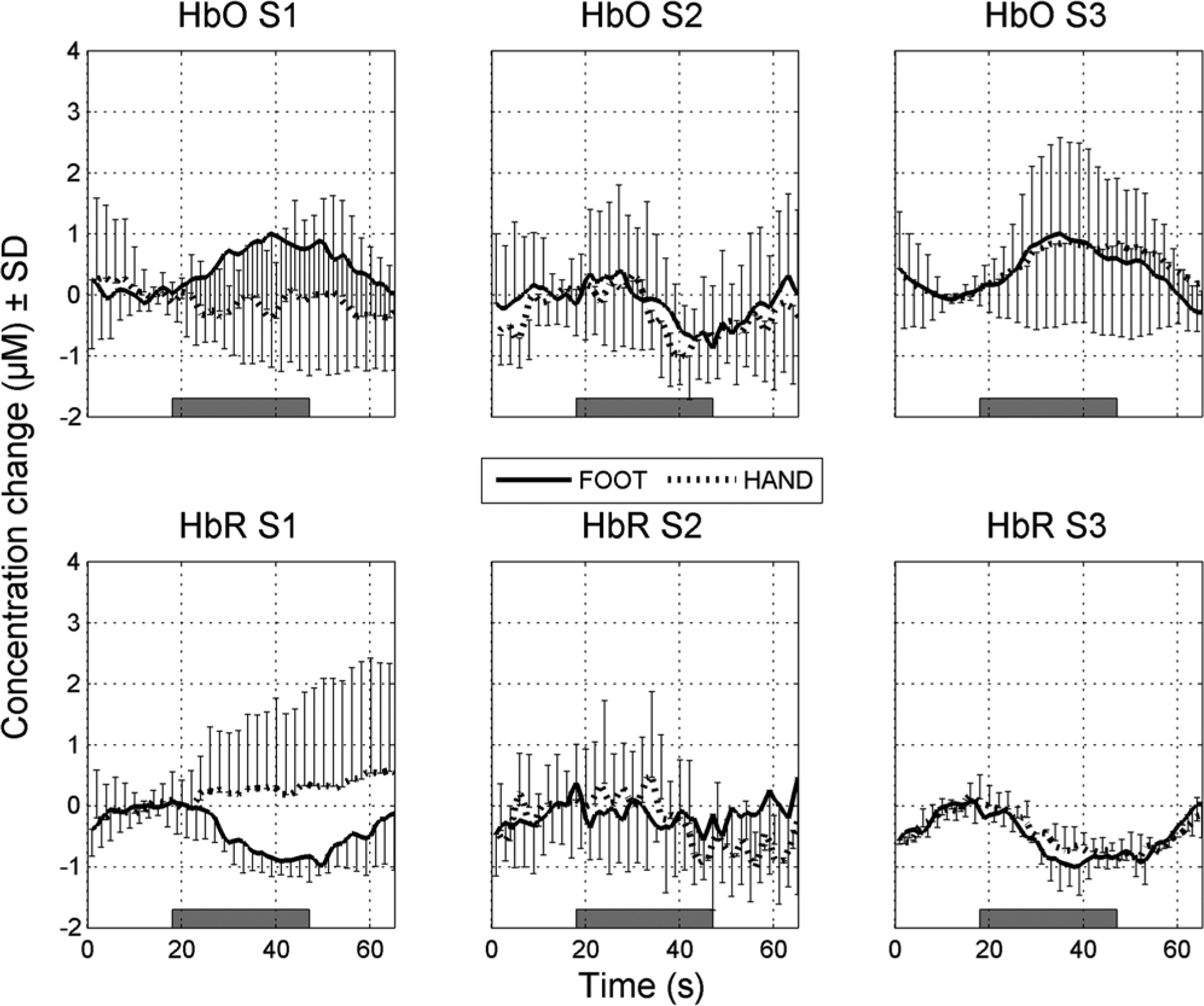
Typical hemodynamic responses.
Discussion
In a group of patients with complete SCI, studied within a relatively short time after the date of injury (1-16 months), attempted foot movements revealed a typical hemodynamic cortical group response of a HbO increase and HbR decrease. In contrast, as expected and hypothesized, the real hand movements condition revealed no significant group responses on the motor cortex of the foot. Moreover, the tested differences in activity on the motor cortex of the foot revealed significant larger HbR decreases for the attempted foot movements compared with the control condition of real hand movements. On individual level, 5 of the 7 SCI patients revealed significant hemodynamic changes during attempted foot movements. Since only 7 patients were included in the present study, the results on group level should be carefully interpreted. The low number of subjects was a result of the scarcity of patients that met our specific inclusion criteria. Nevertheless, the group statistics revealed a significant hemodynamic response during attempted foot movements.
The main purpose of the present study was to determine whether activity in motor cortical areas, formerly in control of below-lesion movements, could still be observed in complete SCI patients using fNIRS. Although several studies assumed that in the acute phases of SCI the motor cortex network is still functioning, 18 other studies have indicated that patients early after SCI (a few years) demonstrated a distorted force control pattern in motor cortex activation. 19 Furthermore, Jurkiewicz et al 23 revealed a shift in cortical activations during attempted foot movements to the parietal cortices. However, the present study indicated cortical activation of the medial part of the primary motor cortex during attempted foot movements in complete SCI patients measured within 16 months after the SCI. These results are generally in line with those obtained in fMRI studies. One of these studies demonstrated the capability of a group of quadriplegic SCI patients to evoke activity in a motor cortical region that prior to the injury controlled the upper extremity. 22 Similarly, preservation of the representations on the motor cortices for the lower extremity has been demonstrated in multiple fMRI studies.23,32-35 However, fMRI is not practical for BCI purposes and therefore other studies have used EEG.18,19 These studies also showed preserved cortical control and even preserved force control for attempted movements in SCI patients.18,19 A major disadvantage of EEG remains the distortion of the EEG signals by muscle contractions originating from muscle closely located to the EEG cap (eg, eye and neck muscles). Therefore, it has been proposed to combine EEG with fNIRS and eventually create a hybrid BCI that uses multiple neuroimaging techniques.36,37 Since the present study demonstrated the capability of fNIRS to detect hemodynamic changes in the medial part of the motor cortex during attempts to move the lower extremity in a complete SCI group early after the date of injury, the authors encourage the application of fNIRS for BCI.
A second major result of the present study is that the hemodynamic changes elicited by attempted foot movements were usually larger than the hemodynamic changes over the same region (ie, medial part of the primary motor cortex) as evoked by real hand movements. This indicates the location selectivity and especially the use of fNIRS in this respect is interesting for future BCI applications. One can imagine that the control of a robot or a wheelchair by imagined/attempted foot movements should not be functioning while making hand movements (such as waving at a passerby). It should be noted that a significant larger HbR decrease was found for the attempted foot movements compared to the real hand movements but there was no equivalent change in HbO responses (despite the use of a reference channel to correct the HbO concentration changes). This lack of specificity for HbO is in line with previous findings of our own group 27 and of others.28,38 Hence, it is proposed that the HbR concentration should be taken into account as a suitable parameter to use in BCI application.
Since BCI applications are normally quite specific for each individual, it is important to point out that there were considerable differences in results for our subjects. Significant hemodynamic responses were found in 6 of the 7 SCI patients. The difference between foot and hand was also tested and revealed 2 SCI patients showing a clear and significant difference in favor of the attempted foot movements. It has to be mentioned, however, that only the mean hemodynamic response was taken into account whereas changes over time, or interactions between different channels, may add information that could be used in a BCI. The variation between the subjects might have originated from the differences in SCI between subjects or it may be due to the use of fNIRS as neuroimaging technique. Interindividual differences are regularly reported in fNIRS studies, for example, because of differences in locations of vessels and scalp–cortex distances.39,40 Furthermore, although we most likely measured the medial part of the primary motor cortex, the exact anatomical position measured with fNIRS is hard to define. Therefore, between subjects differences in the cortical area that was measured cannot be excluded. On the other hand, an EEG study by Müller-Putz et al. 18 found also quite divergent activation patterns in SCI patients during hand tapping movements. In addition, Halder et al 19 demonstrated interindividual differences in a group of SCI patients during foot tapping. The time since injury might have played a role in those studies; however, in the present study no clear correlation was seen between time since injury and the cortical activity. Moreover, the 2 patients who were measured 15 months after date of injury differed in activation patterns. One of the patients revealed no single significant response while the other patient revealed significant responses in the most channels of all patients. Another possibility that might explain the difference between subjects might be the task execution itself. Instead of attempted movements the subjects might have performed motor imagery, a phenomenon that has been previously demonstrated in patients with severe motor disabilities. 41 In a group of paraplegic SCI patients, Hotz-Boendermaker et al 24 demonstrated less primary motor cortex activity and more parietal cortex activity for imagined movements compared to attempted movements. However, the SCI patients in the present study were measured within a relatively short period (ie, within 1.5 years) after SCI. Therefore, we expect that primarily attempted movements were performed, but in some patients motor imagery might have played a role. Fortunately, in BCI one can use smart classifiers based on a training session on the specific user and adapted to the specific task performed over time.4,9,42 As a result, interindividual differences in suitable parameters will not necessarily result in poor BCI performance and a shift in task execution from motor imagery to attempted movements might be elicited. Moreover, it has been demonstrated that training over longer time periods results in distinct improvements in BCI performance. 21 In addition, in a group of tetraplegics, Kauhanen et al 20 demonstrated that BCI training with feedback can result in improvements even in a quite short period. Nevertheless, there is still a lot of research necessary to eventually use practical BCI applications for fNIRS.
In conclusion, patients with complete SCI tested within 16 months after date of injury, are capable of producing fNIRS responses in the medial primary motor cortices on group level (not all subjects revealed individual significance). These hemodynamic changes detected with fNIRS can be used in future BCI applications to control a robotic device or a wheelchair. A major additional finding is the significant difference in HbR between real hand movements and attempted foot movements on the medial part of the motor cortex, suggesting that the method is selective and that HbR is the most sensitive parameter for motor cortex activity.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The BrainGain Smart Mix Programme of the Netherlands Ministry of Economic Affairs and the Netherlands Ministry of Education, Culture and Science (SSM06011).
