Abstract
Background. Bodily sensations are an important component of corporeal awareness. Spinal cord injury can leave affected body parts insentient and unmoving, leading to specific disturbances in the mental representation of one’s own body and the sense of self. Objective. Here, we explored how illusions induced by multisensory stimulation influence immediate sensory signals and tactile awareness in patients with spinal cord injuries. Methods. The rubber hand illusion paradigm was applied to 2 patients with chronic and complete spinal cord injury of the sixth cervical spine, with severe somatosensory impairments in 2 of 5 fingers. Results. Both patients experienced a strong illusion of ownership of the rubber hand during synchronous, but not asynchronous, stroking. They also, spontaneously reported basic tactile sensations in their previously numb fingers. Tactile awareness from seeing the rubber hand was enhanced by progressively increasing the stimulation duration. Conclusions. Multisensory illusions directly and specifically modulate the reemergence of sensory memories and enhance tactile sensation, despite (or as a result of) prior deafferentation. When sensory inputs are lost, and are later illusorily regained, the brain updates a coherent body image even several years after the body has become permanently unable to feel. This particular example of neural plasticity represents a significant opportunity to strengthen the sense of the self and the feelings of embodiment in patients with spinal cord injury.
Keywords
Introduction
The sense of one’s own body is an important aspect of one’s sense of self. The loss of online sensorimotor signals following spinal cord injuries (SCIs) results not only in the inability to move but also in dramatically reduced perceptual bodily information from the affected body parts. 1 The insentient body is often perceived by patients with SCI as devastating and disturbing. 2 The inability to feel leads to neuroplastic rearrangements of the inherent nature of bodily representations, 3 resulting in disturbances to the bodily self.1,4
Recent studies have shown that bodily illusions evoked by multisensory conflicts can be a powerful tool for changing and normalizing altered body representations. 5 Herein, we used the rubber hand illusion (RHI) paradigm 6 to manipulate tactile awareness and the sense of the body in 2 patients with SCI. In this paradigm, the synchronous stroking of an observed fake hand and one’s own hidden hand leads to an illusory feeling of touch in the fake hand. This illusion has previously been proven to change various aspects of the bodily self, including awareness, tactile acuity, proprioceptive judgment and even body temperature. We hypothesized that the RHI could increase tactile sensations and feelings of bodily ownership in patients with SCI.
Methods
The 2 male patients (SCIP1 and SCIP2) who participated in our study both had a traumatic, complete cervical lesion at the sixth cervical vertebra of the spinal cord and no brain injury. Neurological examination indicated that both patients presented with paralysis of the legs and trunk, as well as a variable degree of sensory loss and partial paralysis of the upper limbs. They were also unable to feel 2 of their 5 fingers. Demographic and clinical data are presented in Figure 1A. This study was approved by the local ethics committee in accordance with the Declaration of Helsinki. The experimental paradigm is shown in Figure 1B.
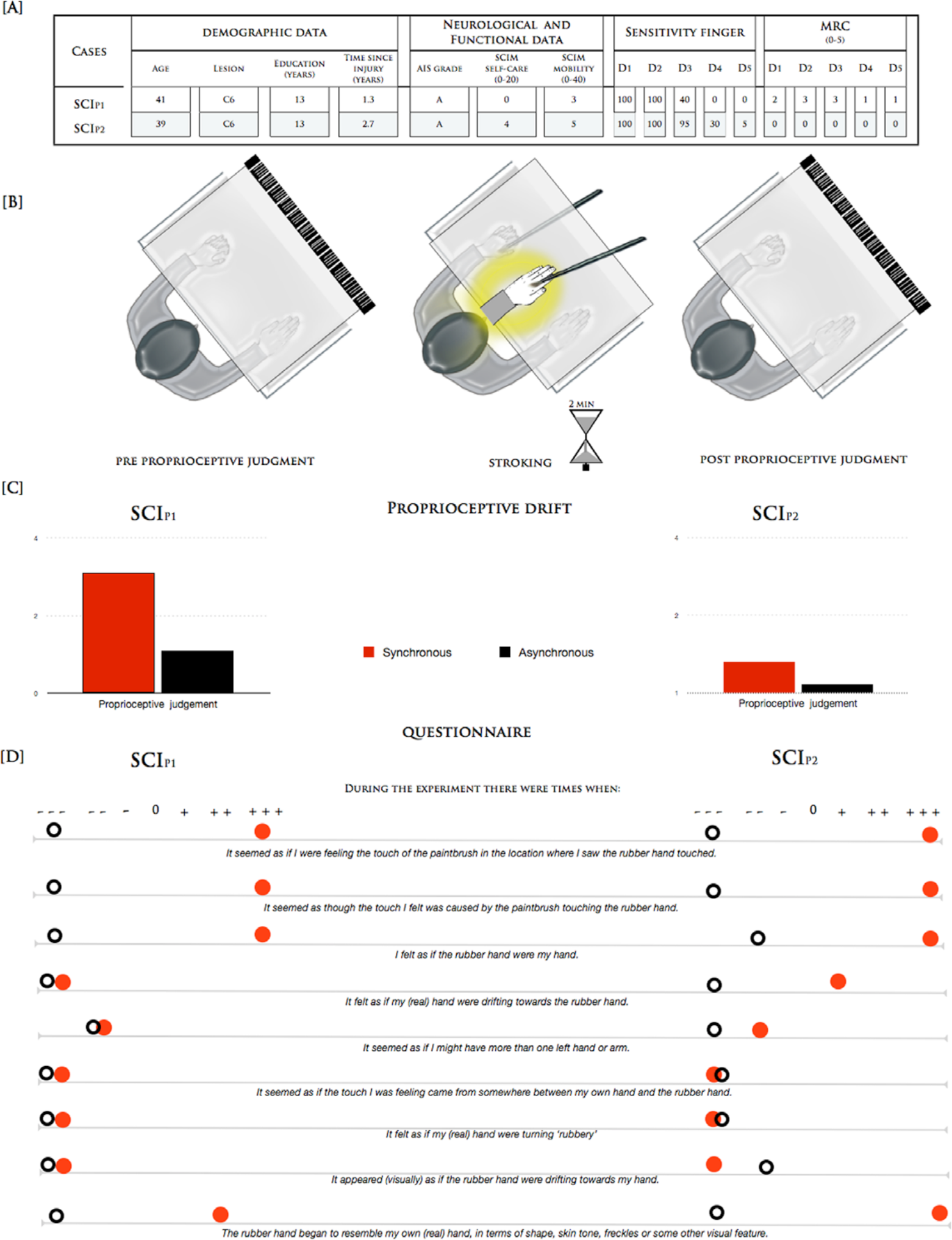
(A) Clinical and demographic characteristics of participants. The neurological levels of lesion and injury, as determined by the American Impairment Scale (AIS), are indicated. The third version of the Spinal Cord Independence Measure (SCIM III; subscales: Self-care and Management and Mobility) was used to quantify functional ability in each patient. The sensitivity of the finger indicated the tactile sensation assigned by the patients to each finger compared with complete tactile sensation on the face. The muscle strength for each finger was determined by the Medical Research Council (MRC) scale (5 represents normal strength). (B) Schematic representations of the experimental procedure. Patients were seated in front of a table and were asked to observe a rubber hand. They were instructed not to move either their hands or head during the test phase. The researcher placed the participants’ arms in a standard anatomical position inside a wooden box. A life-sized and realistic-looking rubber left hand was placed inside the box in front of the participants and aligned with their midline with a distance of 13 cm to the real hand. In the synchrony and asynchrony conditions, 2 identical paint brushes were used to stroke the rubber hand and the participants’ hidden hand at an approximate rate of one brush stroke/s. Immediately after, a wooden board with a ruler was inserted into the box covering the participants’ real hands and the rubber hand. They were told to verbally indicate as precisely as possible under which number on the ruler the middle point of their left index finger was located. The participants’ judgment regarding the occluded left index finger position in the absence of stimulation was requested 5 times before the onset of stimulation as a baseline measure. 4 (C) Perceived finger position in synchrony/asynchrony conditions (red and black bars) relative to the individual pretest baseline for the 2 patients (SCIP1 and SCIP2). (D) Subjective ratings of illusion during the stimulation as assessed by an Italian version of the 9-item questionnaire. 6
Results
During synchronous stroking in the classical RHI setup, both patients spontaneously reported vivid tactile sensations in their previously numb fingers (Figure 1B). They were incredibly surprised about this phenomenon given that they had experienced no such feelings since their injury. The typical RHI measurements indicated that they both experienced strong illusory ownership (maximal scores on the illusion-relevant questions of the RHI questionnaire 6 ) and mislocalization (proprioceptive drift, difference between proprioceptive judgment after the synchronous vs asynchronous condition: SCIP1 2.0 cm and SCIP2 0.5 cm) of the sense of touch toward the rubber hand (Figure 1C and D).
To better quantify the subjective alteration in tactile perception experienced during the RHI, stroking was applied for 2 minutes during 3 different conditions: (a) tactile-only stroking of the real hand, (b) synchronous stroking of both the real and the rubber hands, and (c) asynchronous stroking of the real and rubber hands (Figure 2A). The patients were instructed to look at the rubber hand in all of the conditions. These 3 conditions allowed us to dissociate effects related to the pure tactile stimulation from those related to the bodily self. After each 30-second period, the patients were asked to rate how strongly they felt the tactile sensation during the stroking. This intensity was rated on a continuous scale for each individual finger. The scores ranged from 0 (absent) to 100 (normal, complete tactile sensation, as would be felt on the face).
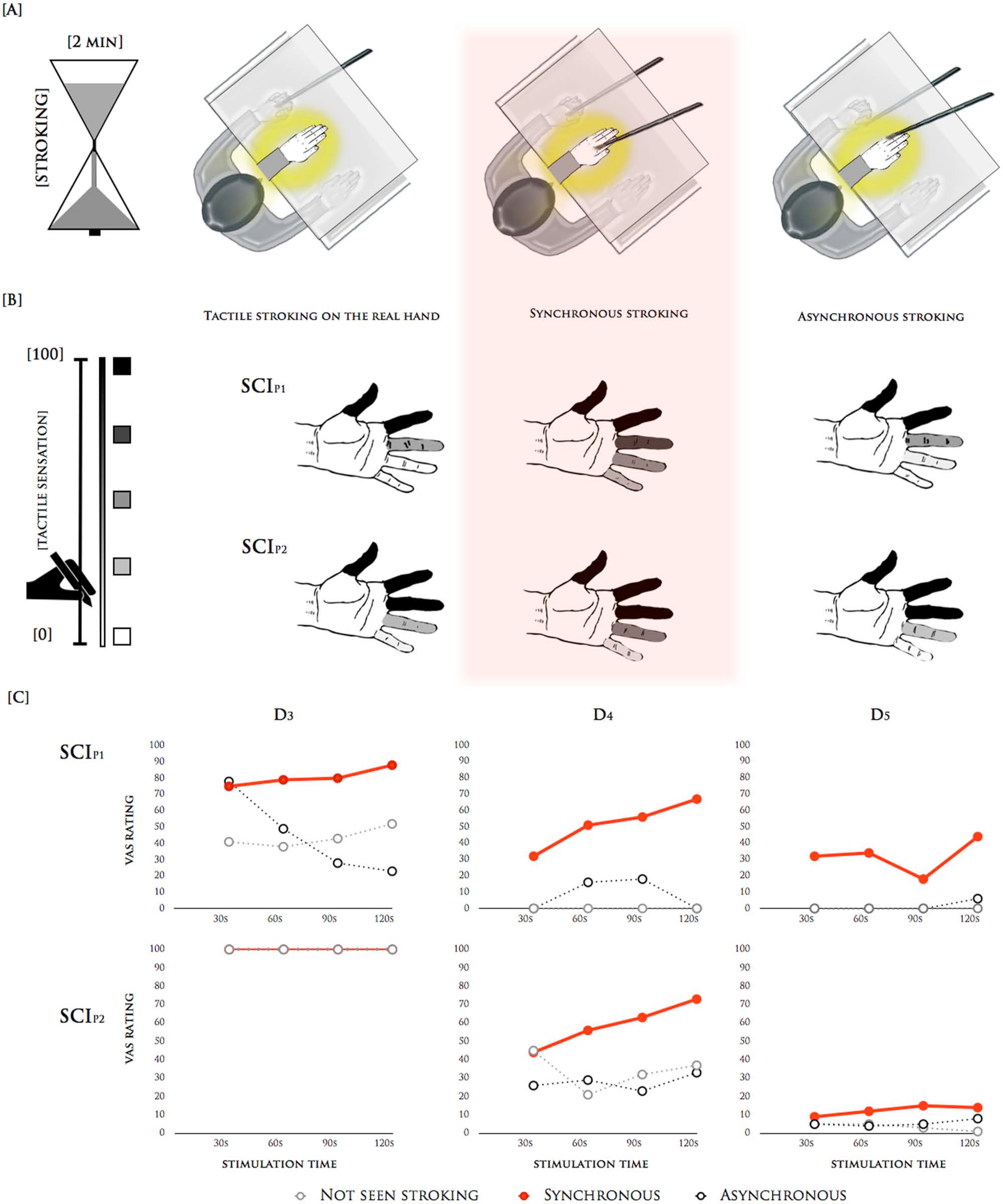
(A) The 3 different experimental conditions during rubber hand illusion (RHI): tactile-only stroking, synchronous stroking, and asynchronous stroking. In the tactile-only condition, the researcher brushed the real hands of the patients, the rubber hand was not stroked, and the patients simply observed the rubber hand. In the synchrony and asynchrony conditions, the brush strokes were applied to the dorsal surface of all fingers for 2 minutes at an approximate rate of 1 brush stroke/s. At the end of each 30-second period, the patients were asked to rate on a continuous scale for each finger how strongly they felt the tactile sensation during the stroking. (B) The subjective rating of tactile sensation on the stimulated hand in SCIP1 and SCIP2. The mean values of the strength of the tactile sensation on each stimulated finger compared with the face are represented as a grayscale from white (no sensation) to black (complete sensation, as on the face). (C) Subjective report of strength of touch sensation after 30, 60, 90, and 120 seconds for fingers with reduced sensitivity (middle finger, D3; ring finger, D4; and little finger, D5) in SCIP1 and SCIP2.
During the tactile-only condition, SCIP1 reported complete sensation in the thumb and index finger (mean ± standard error of the mean = 100 ± 0), some sensation in the middle finger (41 ± 5) and no sensation in the ring and little fingers (0 ± 0). SCIP2 exhibited intact sensation in the thumb and the index and middle fingers (100 ± 0), and some sensation in the ring (33 ± 5) and little (6 ± 1) fingers. Importantly, during the synchronous condition, both patients experienced a marked increase in the strength of the tactile sensation in all of the fingers with reduced sensitivity (middle finger, 80 ± 3, ring finger, 52 ± 7; and little finger, 32 ± 5 in SCIP1; ring finger, 59 ± 6 and little finger, 13 ± 1 in SCIP2). Furthermore, the data revealed growing tactile sensations in correspondence with the increasing duration of the stimulation. Indeed, tactile sensation increased by up to 75% in a previously numb finger after 2 minutes of stimulation (ring finger in SCIP1). Conversely, in the asynchronous condition, the sensation was similar to the tactile-only condition, but was clearly reduced compared with the synchronous condition (middle finger, 44 ± 13, ring finger, 9 ± 5; and little finger, 2 ± 2 in SCIP1; ring finger, 28 ± 2 and little finger, 2 ± 0 in SCIP2). All of the results are depicted in Figure 2B and C.
Discussion
Three main conclusions, with important implications for rehabilitative approaches, can be drawn from the present findings: (a) subjective tactile sensation can reemerge during a simple multisensory stimulation paradigm, despite a long period of massive deafferentation; (b) the strengthening of the sensation becomes more evident with the increasing duration of the stimulation; and (c) functional tactile perception permits the recreation of a coherent mental representation of one’s own body.
Viewing a body part improves tactile discrimination, detection and acuity. 7 Direct evidence reveals that this visual enhancement of touch effect obeys the inverse efficiency principle. From this perspective, the effect of multisensory integration is stronger in healthy participants when task difficulty increases, and in brain-damaged patients with weakened sensory channels. 8 Furthermore, since the visual enhancement of touch in the RHI is specifically related to the sense of body ownership, tactile acuity should be enhanced following synchronous as opposed to asynchronous stroking. 9 Interestingly, however, the strong reaction, and the more objective measure of both patients when seeing the rubber hand being stroked synchronously, suggest that seeing the touch on the rubber hand increased tactile awareness, while seeing their real hands being directly touched by others each day did not. Even if still speculative, patients with chronic SCI could rely more strongly on a visual body image to compensate for the complete absence of proprioceptive input. 10 The feeling of ownership of the rubber hand could mask feelings of ownership for their real hands, which is in line with previous SCI findings. 11
The illusory ownership and embodiment of the fake hand could thus induce a change in the weighting and reliability of multisensory inputs, resulting in an extraordinary effect in which previously numb fingers could be felt. This phenomenon has important consequences for SCI therapy. For example, it suggests that it is possible to reestablish tactile memories that may improve the limited capacity of sensory processing after SCI. Moreover, new treatments that manipulate the bodily self via “visual” reembodiment may be especially useful to patients with SCI, because their reduced or impaired sensorimotor abilities have disrupted their body image. 4 Perceptual effects during a full body illusion 11 paradigm may extend the potentiality of this approach to alternative forms of embodiment and could be a viable intervention for detrimental effects following SCI, such as neuropathic pain. 12
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article:
This work was supported by the International Foundation for Research in Paraplegia [grant number P133], the Swiss National Science Foundation [grant number PBELP3-133268] and the Italian Ministry of Health [grant RC12.G].
