Abstract
Background. Upper extremity use in daily life is a critical ingredient of continued functional recovery after cerebral stroke. However, time-evolutions of use-dependent motion quality are poorly understood due to limitations of existing measurement tools. Objective. Proof-of-concept study to determine if spectral analyses explain the variability of known temporal kinematic movement quality (ie, movement duration, number of peaks, jerk) for uncontrolled reach-to-grasp tasks. Methods. Ten individuals with chronic stroke performed unimanual goal-directed movements using both hands, with and without task object present, wearing accelerometers on each wrist. Temporal and spectral measures were extracted for each gesture. The effects of performance condition on outcome measures were determined using 2-way, within subject, hand (nonparetic vs paretic) × object (present vs absent) analysis of variance. Regression analyses determined if spectral measures explained the variability of the temporal measures. Results. There were main effects of hand on all 3 temporal measures and main effects of object on movement duration and peaks. For the paretic limb, spectral measures explain 41.2% and 51.1% of the variability in movement duration and peaks, respectively. For the nonparetic limb, spectral measures explain 40.1%, 42.5%, and 27.8% of the variability of movement duration, peaks, and jerk, respectively. Conclusions. Spectral measures explain the variability of motion efficiency and control in individuals with stroke. Signal power from 1.0 to 2.0 Hz is sensitive to changes in hand and object. Analyzing the evolution of this measure in ambient environments may provide as yet uncharted information useful for evaluating long-term recovery.
Introduction
Optimal and meaningful recovery after stroke requires spontaneous use and seamless integration of the paretic arm and hand into the natural environment.1,2 Use of the paretic upper extremity (UE) is directly tied to an overall sense of recovery after stroke; stroke survivors and their families reported that arm impairment was not only of extreme detriment to quality of life but also poorly understood, appreciated, and addressed. 3 From a clinical perspective, we know that arm use and recovery are important for behavioral and neurological changes. Imaging studies have shown that after a brain injury such as stroke, cortical areas tied to a damaged limb can be “taken over” by adjacent areas. Countering this phenomenon requires increased use of the paretic limb to promote positive, functional neuroplasticity in cortical areas representing that limb.4,5 Thus, from the perspective of the patient, and from neurobehavioral studies, skilled, voluntary use of the stroke-affected upper limb is required to optimize restitution and recovery. Because recovery after stroke depends on a number of variables including the type and dose of therapy, understanding normal patterns of recovery and use is necessary to guide monitoring of future patients. 6 Unfortunately, in spite of its well-established importance, there is a lack of understanding of the time-evolution of arm recovery after stroke.
In certain laboratory settings, kinematic data have provided insight into recovery associated with motoric changes. Rohrer et al demonstrated in some participants that poststroke recovery can be characterized by increased motion smoothness, as measured by mean squared jerk cost (the derivative of acceleration, first cited as a measure of performance by Flash and Hogan 7 ). 8 Panarese et al demonstrated recovery of UE motor function as measured by velocity, path error, and the number of peaks in the velocity profile. 9 Colombo et al demonstrated that changes in mean movement velocity and forces applied to a robotic manipulandum correlated with changes in Fugl-Meyer and Motor Status Score scales. 10 These studies indicate that kinematic measures provide some understanding of the mechanisms of recovery. More recently, researchers have sought to relate kinematics to known functional assessments.11,12 These authors used tasks from the Wolf Motor Function Test and the Fugl-Meyer Assessment to correlate quantitative measures with clinician assessments. This body of work focuses on controlled, ballistic targeted reaching tasks. How these movements might correspond to activities of daily living (ADL)–inspired uncontrolled movements is not known. Here, uncontrolled refers to movements for which the instructions provide a defined end goal, but no restriction on the strategy used to achieve it (eg, put the pencil in the cup). Uncontrolled movements are more similar to the types of movements performed in ambient settings.
We are interested in developing novel tools with the sensitivity to detect and potentially reward incremental and immediate changes in upper limb use in natural environments based on spectral analyses. Such analyses have been used to determine (a) critical changes in performance for those on medication for Parkinson’s disease, 13 (b) behavior that anticipates falls in elderly walkers, 14 (c) differentiation of lower extremity movements after stroke, 15 and (d) UE practice of skilled tasks by nondisabled individuals.16,17 From these studies, frequency ranges between 0 and 2 Hz, and up to 10 Hz, were useful in distinguishing relative levels of motion quality during task performance. Such studies have yet to be conducted for UE task performance in the chronic poststroke population. This article presents the first of a 2-part research project designed to capture meaningful information about motor patterns of people with motor deficits in real-world settings. We believe that spectral analyses of unsegmented continuous in-home data are promising tools for revealing meaningful information regarding motion quality. This is important because, in ambient environments, standard temporal kinematic measures cannot be obtained without knowing, according to a secondary measure, how to extract, or segment, a gesture. The first step is to establish the meaningfulness of these spectral measures on manually segmented gesture data, which is the focus of this article.
Our objective is to determine the nature of the relationship between spectral measures and known temporal measures of performance (movement duration, DUR; number of peaks, PKS; and mean squared jerk cost, MSJ) during uncontrolled goal-directed reaching and grasping movements inspired by ADLs. These dependent measures were selected because their relationship to motor ability metrics such as efficiency, motor control, smoothness, and coordination has been well established for controlled movements.8,18-22 For this proof-of-concept study, we hypothesize that spectral measures will explain the variability of known temporal measures of motion quality. To test this hypothesis, we use a 2-stage process. First, we find how both temporal and spectral measures vary under different task performance conditions. Second, we investigate the ability of the spectral measures to explain the variability of the temporal measures. We designed a study wherein participants performed a number of simulated unimanual ADLs in a laboratory environment using the paretic and nonparetic limb in separate runs. Differences in performance with the paretic and nonparetic limb are reflective of differences in motor control, spasticity, and strength. 23 To elicit motoric changes within a task, participants were also asked to perform each task with and without the task object present. Object affordance was shown by Wu and colleagues to create noticeable changes in motion quality 18 reflective of differences in planning.18,24
Our long-term aim is to use spectral analyses of inertial sensor data to develop a novel measure of UE use that can be determined without a priori knowledge of the task being performed. The advantage of such a measure, if proven to be valid and reliable, is that it can be applied to data sets obtained in unsupervised, natural environments such as the home to measure motion quality and to quantitatively measure recovery of paretic limb use.
Methods
Participant Recruitment
Recruitment and screening were performed at the University of Southern California’s Center for Health Professions according to the specifications of the University of Southern California Institutional Review Board. Our primary inclusion criterion was the ability, while seated, to grasp and lift an object of moderate weight (eg, a full bottle of water) above a table surface and hold it for 2 seconds with the paretic arm. Furthermore, we required participants to be right handed premorbidly and at least 6 months poststroke.
Experimental Procedure
The study was designed as a cross-sectional, within-subjects repeated-measures design with 2 independent variables: hand (nonparetic vs paretic) and object (with vs without object affordance). Tri-axial accelerometers manufactured by Gulf Coast Data Concepts, LLC, were secured to each of the participants’ wrists using sweat-bands; participants then performed a battery of tasks, listed in Table 1. For seated tasks, participants sat at a comfortable distance from a table on which task performance items were placed (this “comfortable distance” was defined as the minimum distance at which participants could reach their hands from their lap to the table top without being obstructed by the table’s edge). The locations of the task items were standardized using a tabletop template. For the standing tasks, markers on the floor indicated where participants should stand relative to the task performance items. Each task was performed once to obtain the natural, “unpracticed” representation of the gesture.
Gesture List With Descriptions (Sit or Stand Indicate Sitting or Standing Tasks, Respectively).

The sequence of task performance is illustrated in Figure 1. Prior to performing each task, participants were instructed to keep their hands in a resting “home” position (in the lap while seated; at the sides while standing). Using a stopwatch, the experimenter instructed participants to wait 5 seconds at rest, then to perform the gesture with the nonparetic, then paretic, then nonparetic without the object, then paretic without the object. We define the 4 performances of a single task as a task block. The 5-second wait periods were used to facilitate automatic gesture extraction from the accelerometer data. Participants were instructed to move at a comfortable pace (to more closely mimic “real-world” task performance). All sessions were recorded with a camcorder.
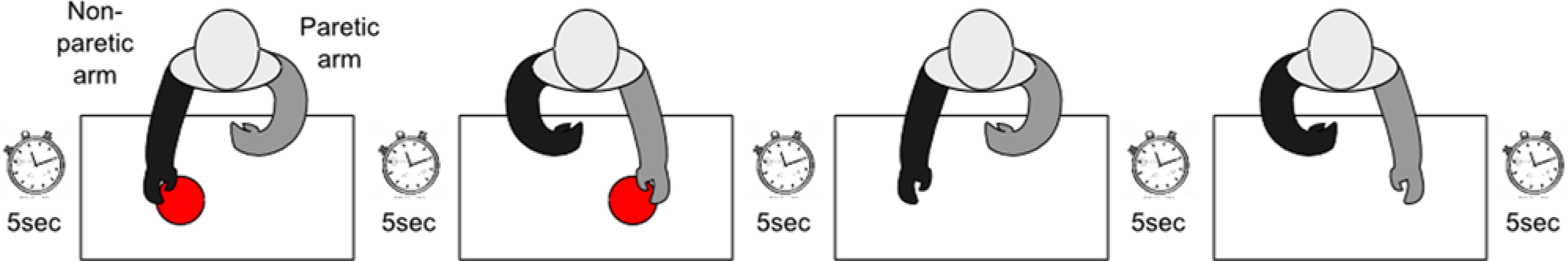
One task block. For each given task, participants performed in the following order: with/nonparetic, with/paretic, without/nonparetic, without paretic (with 5-second pauses in between each performance).
Outcome Measures
Temporal Measures. Accelerometer data were sampled at 40 Hz and then filtered using a second-order Butterworth bandpass filter with cutoff frequencies at 0.1 and 10 Hz. The low-frequency cutoff was chosen to eliminate contributions of sensor drift, gravity, and pose orientation. 25 The high-frequency cutoff was chosen because few observable harmonics of volitional movement exist beyond 10 Hz. 16 We performed a sinusoidal “shake” of all sensors, on camera, before and after affixing them to the participant to facilitate synchronization of sensor data (see the appendix for detailed information regarding gesture extraction from accelerometer data).
For each gesture, the resultant acceleration, ar, was computed using the following formula:
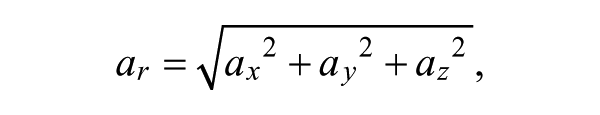
where ax, ay, and az are the x-, y-, and z-direction accelerations (respectively) in m/s2. Once individual gestures were extracted from accelerometer data, movement duration was obtained by subtracting the movement onset from the movement offset. The number of peaks was found using an in-built command in Matlab (Version 2011a, The MathWorks Inc, Boston, MA) that counts the number of changes in slope sign within a vector of filtered data. 21 The mean-squared jerk cost was computed using the following formula:
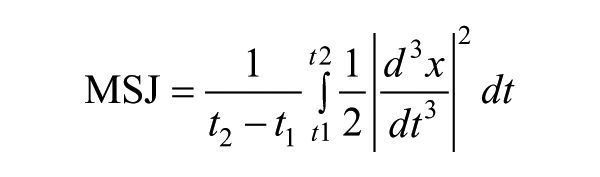
from Hogan and Sternad. 26 Note that there are a number of jerk metrics used in the literature. This metric was selected so that smoothness would be independent of movement duration. 22
Spectral Measures
To evaluate spectral measures, we first determined which frequency bands were sensitive to the types of differences observed in poststroke movements. Because there are no existing studies of spectral analyses of UE movement after stroke, we searched around harmonic frequencies cited as important loci for changes in performance, as determined in the Hotraphinyo and Hansson works.16,17 In both studies, the authors assessed skilled movements (those of surgeons and industrial workers, respectively). We use the study outcomes of signal power as guidelines—in particular, both found spectral components of intentional wrist motion below 8 and 5 Hz, respectively, with unintentional motion (eg, tremor) above these values. Using this entire range as a region of interest for our spectral analyses, we first computed the power spectral density (PSD) of ar using the Welch method. Using the aforementioned frequency ranges as a guide, we computed the relative contributions of power up to frequency fi, to the total power in the 0.1 to 10 Hz range (recall that the signal was bandpass filtered at 0.1 and 10 Hz). This was computed using
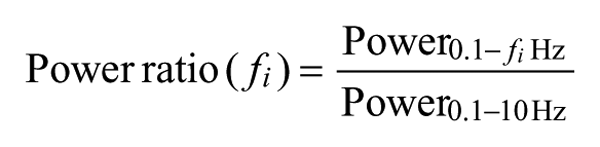
This was done for all fi ∈ [0.1, 10]. This analysis revealed frequencies at which the harmonics of the resultant acceleration signals changed significantly in magnitude, between 1 and 2 Hz (this was confirmed by analyses of pilot participant data). Thus, we chose to evaluate separately the power ratios for 0.1 to 1 Hz (the lowest frequency range of interest), 1 to 2 Hz (the higher frequency range of interest), and 2 to 10 Hz (the remainder of the available signal). We denote these as PRa, PRb, and PRc.
Data/Statistical Analyses
Dependent Variable Descriptives
We explored the descriptive nature of the dependent measures using the Shapiro–Wilk test for normality. The temporal (DUR, PKS, MSJ) and spectral measures (PRb and PRc) displayed positive skew, and PRa displayed negative skew. To normalize these data, we performed natural log transforms of each measure (and an inverse natural log transform for PRa).
Effects of Conditions on Kinematic and Spectral Measures
To determine the main effects of hand and object on the log transformed temporal and spectral dependent measures, we performed a 2-way repeated-measures analysis of variance (ANOVA) on each measure. For each ANOVA, a Bonferonni correction was applied to adjust the significance level for repeated tests (because there are 2 levels of each factor, this correction required P < .025 for significance).
Relationship Between Temporal and Spectral Measures
To determine the extent to which the spectral measures explain the variability of the kinematic measures derived from the same gesture, we performed a stepwise multiple linear regression analysis (using the 3 spectral measures as explanatory variables and the temporal measures as response variables). Due to the exploratory nature of this study, there are few existing standards for a good coefficient of determination (R2) value. We conservatively used R2 = 0.36, typical for biological data analyses, as a cutoff for the ability of our explanatory variables to explain the response variable. 27 All statistical analyses were conducted using PASW Statistics (Release 18.0, 2009).
Results
Participant Demographics
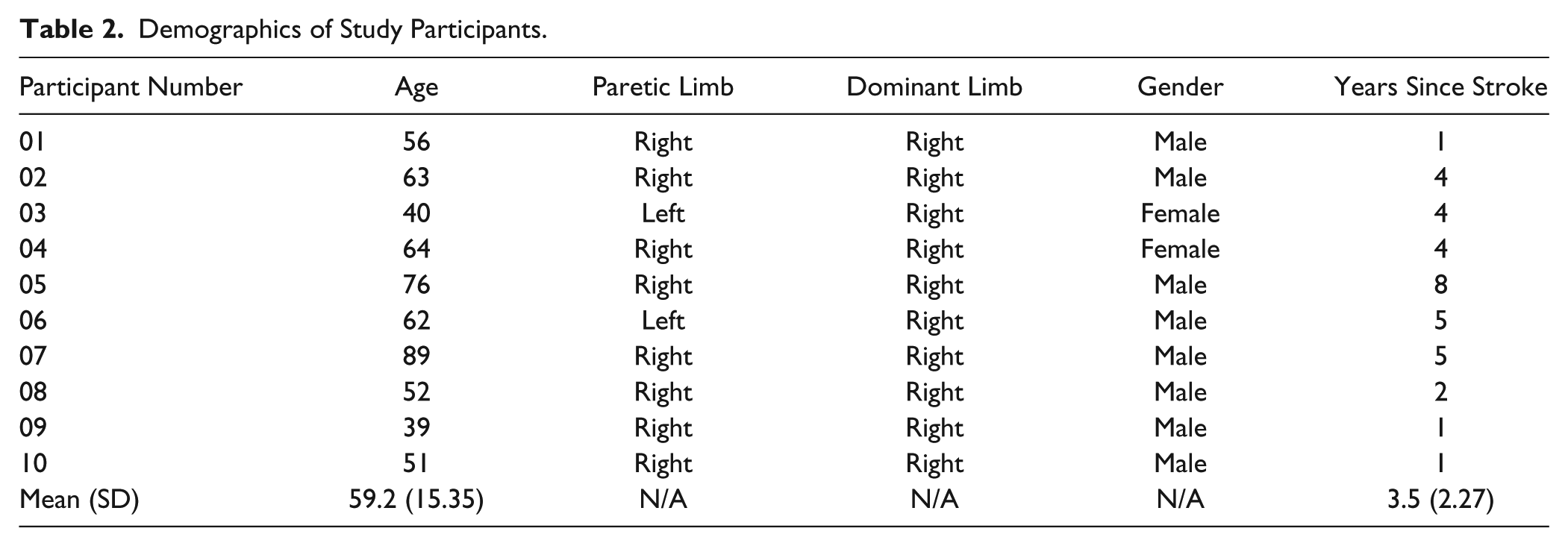
We recruited 10 hemiparetic individuals ranging from 1 to 8 years in stroke duration. All were right-handed premorbidity; 8 of the 10 were more affected on the right side (for demographics, see Table 2). All were living and functioning relatively independently in a residence with a family member or caregiver. The inclusion criteria required that each enrolled participant have a moderate degree of hand and arm function to be able to perform the ADL tasks. Therefore, in general all 10 participants were only mildly to moderately impaired in arm and hand function.
Demographics of Study Participants.
Temporal Measures
Not surprisingly, DURs with the paretic hand were longer than DURs with the nonparetic hand, F(1, 79) = 55.11, P < .001; see Figure 2A). DURs with the object were greater than DURs without the object, F(1, 79) = 28.05, P < .001. Not surprisingly, PKS with the paretic hand were greater than with the nonparetic hand, F(1, 79) = 28.20, P < .001; see Figure 2B). PKS with the object were greater than without the object, F(1, 79) = 41.65, P < .001). MSJ of the paretic hand was greater than that of the nonparetic hand, F(1, 79) = 27.09, P < .001; see Figure 2C). There were no differences in MSJ with or without the object (P = .10). There were no interactions between hand and object for any of the 3 temporal measures.
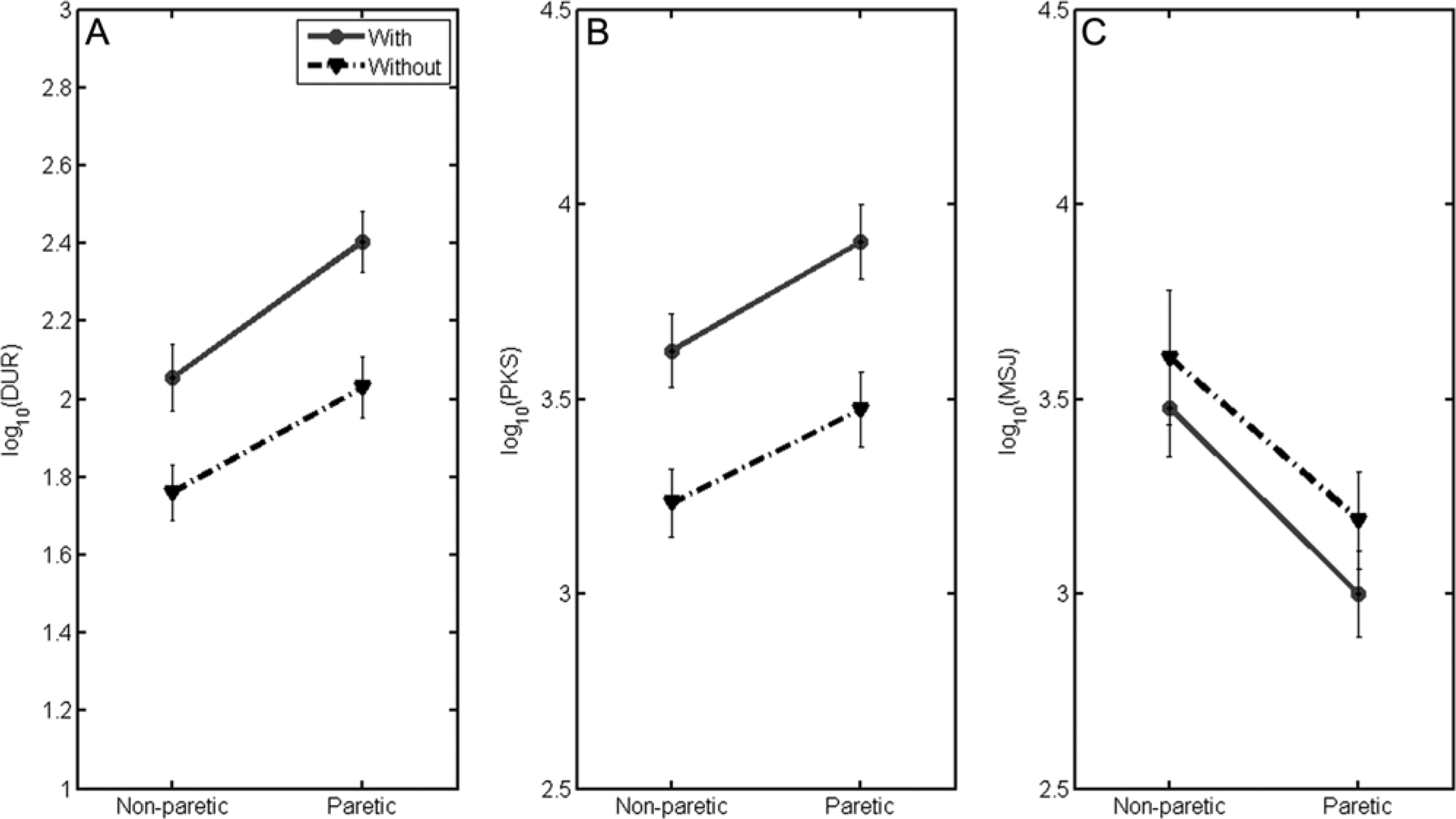
Plots indicating interactions between object and hand conditions on the dependent measures (A) movement duration, (B) number of peaks, and (C) mean squared jerk cost.
Spectral Measures
There was no difference in the PRa of the nonparetic and paretic hand (P = .283). PRa with the object was greater than PRa without the object, F(1, 79) = 8.24, P < .01. There was a significant interaction between object and hand, F(1, 79) = 4.64, P < .05.
PRb of the paretic hand was greater than PRb of the nonparetic hand, F(1, 79) = 4.58, P < .05). PRb with the object was greater than PRb without the object, F(1, 79) = 12.25, P < .01. Finally, there a was significant interaction between object and hand, F(1, 79) = 4.37, P < .05.
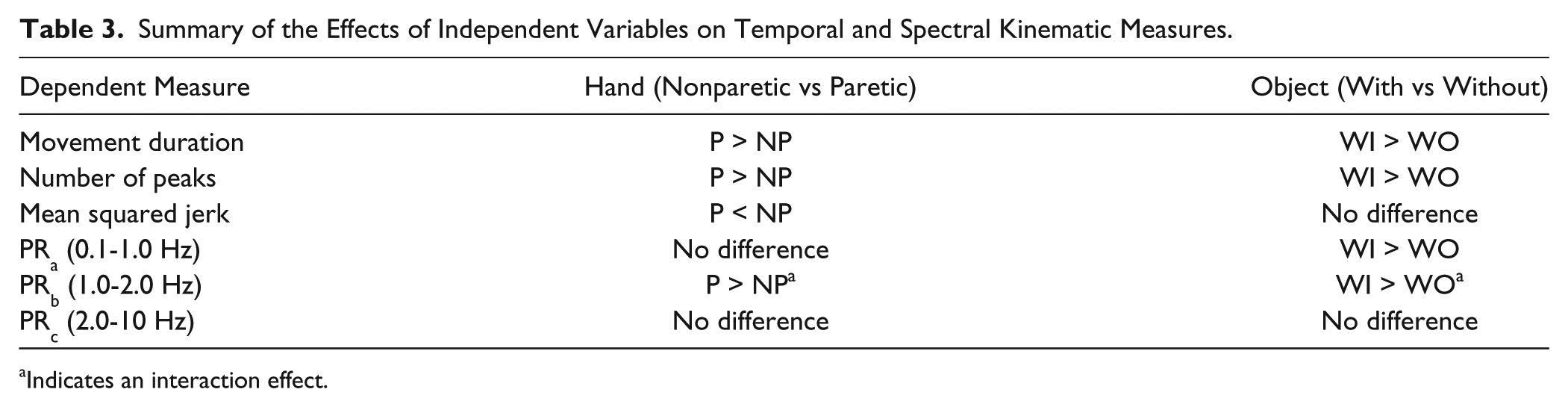
There was no difference in PRc of the nonparetic and paretic hand (P = .11). There were no differences with and without the object (P = .47) and no interaction (P = .12). Results of all ANOVAs are shown in Table 3.
Summary of the Effects of Independent Variables on Temporal and Spectral Kinematic Measures.
Indicates an interaction effect.
Explaining Temporal Variables
Having noted the effect of hand on the kinematic measures, we performed separate regression analyses for the nonparetic and paretic limbs. The spectral measures showed moderate-to-good ability to explain DUR and PKS (according to the threshold set at R2 = 0.36), but only explained MSJ variability in the nonparetic limb. For the paretic limb, PRa, PRb, and PRc together explained 41.2% of the variability of DUR (F = 35.5, P < .001), 51.1% of the variability of PKS (F = 54.39, P < .001), but were unable to explain MSJ (F = 91.82, P = .061). The relative contributions of PRa, PRb, and PRc are shown in Figure 3B, where the R2 values are shown, with the relative contributions of each spectral band depicted by different shades. For the nonparetic limb, PRa, PRb, and PRc explained 40.1% of the variability of DUR (F = 35.75, P < .001), 42.5% of the variability of PKS (F = 38.41, P < .001), and 27.8% of the variability of MSJ (F = 20.10, P < .001; see Figure 3A).
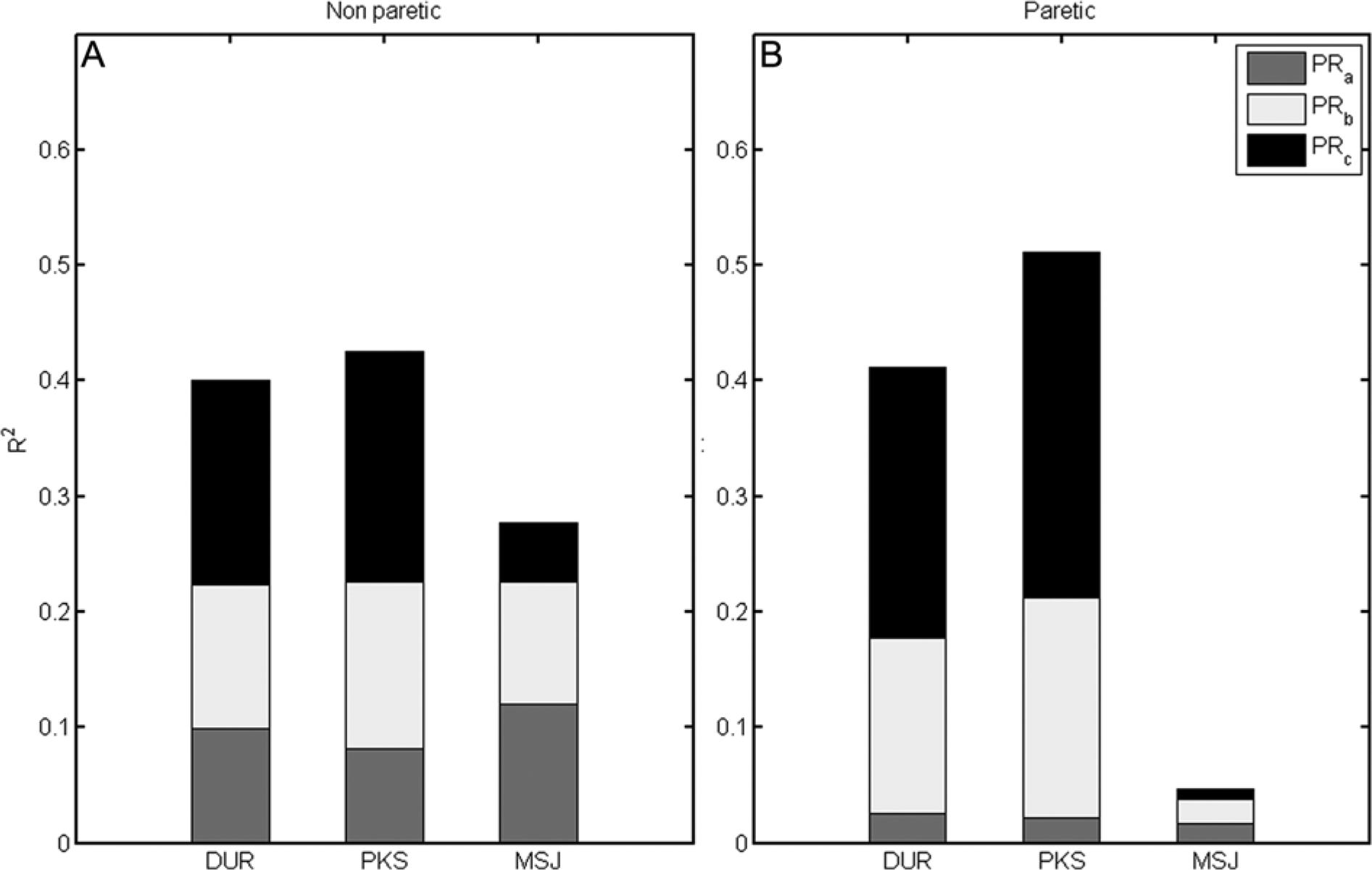
The relative contributions of the spectral measures PRa, PRb, and PRc to DUR, PKS, and MSJ. For each dependent measure, the value of R2 is plotted; the relative weight of each spectral measure is represented by the amount of corresponding grayscale shade within a given bar. The 2 plots represent the analyses of the nonparetic hand and the paretic hand.
Discussion
This is the first study to investigate spectral characteristics of UE ADL task performance in individuals after stroke. The main findings are that (a) standard kinematic measures, DUR and PKS, were sensitive to hemiparesis and object affordance, while MSJ was only sensitive to hemiparesis; (b) of the 3 spectral measures, PRb was sensitive to differences in both hemiparesis and object affordance; and (c) the spectral measures can be used to explain the variability of DUR and PKS. To frame the utility of these outcomes in creating performance measures outside of the lab, we discuss these findings by offering answers to 3 related research questions: What do the temporal measures tell us; what do the spectral measures tell us; and can we use the spectral measures to explain temporal measures.
What Do the Temporal Kinematic Measures Tell Us?
Our results indicated longer DURs for the paretic limb than for the nonparetic limb, matching expectations based on observed differences in limb motion after stroke, 18 and on within person comparisons of various ballistic reaching movements. 24 Our finding that DURs with the object were greater than those without was unexpected. In previous work, performance with the object present elicited shorter movement durations than with the object on a ballistic reach-to-grasp task. 18 The assumption is that the proprioceptive feedback (provided by the physical presence of the object) enhances task performance, allowing people to scale spatial relationships without imagining target location and thus to perform more efficient movements. We suspect that the more complex, multistep nature of our tasks may be the cause of the discrepancy. In fact, a study by Wisneski and Johnson used tasks modeled on ADLs to reveal that healthy individuals showed no differences in DUR between object present and absent for drinking and feeding tasks. 24 Thus, it is reasonable to assume that specific task parameters (object size, object orientation) affect kinematic performance differently in simple ballistic reach-to-grasp tasks and ADL-modeled tasks. Furthermore, in our study, more difficult tasks (eg, pencil in a cup, which required fine dexterity) took longer for many participants (eg, those who had trouble grasping the pencil with the paretic limb). However, without the object, participants could more quickly pantomime the task without the real-world constraints of grasping and lifting the pencil.
Consistent with existing literature, we found that the paretic limb had more PKS than the nonparetic limb, indicating less motion smoothness. 18 Our finding that PKS were greater with the object (than without) was once again surprising in light of the results of Wu et al, who found that movements were smoother when the object was present and suggested that either object presence (and visual information) resulted in less reliance on external feedback, or that reaching to an object is more familiar than pantomiming resulting in a more centrally preprogrammed movement. However, for our tasks, again the complexity of the movements may have washed out some of these effects. In the Wisneski study, there were no statistically significant differences in number of peaks between the object present and absent conditions for drinking and eating tasks. Furthermore, the instructions in our study (ie, to move at a comfortable, self-selected pace) differ from those of other studies that stress moving quickly and accurately. Without the time constraints, participants may have chosen to optimize control (as opposed to speed) as a reaching strategy.
We also found that the MSJ was greater for the nonparetic limb than the paretic limb. On first inspection, this may be counterintuitive. In 1985, Flash and Hogan described jerk as a smoothness measure and it has been used as such by other researchers.7,26,28-31 MSJ is thought to capture smoothness because movement reversals result in larger values of all derivations, including jerk. However, it has also been pointed out that variation of this measure depends on more than smoothness. In a robotic rehabilitation training paradigm, Rohrer et al showed that during recovery, some participants showed an increase in the jerk metric as they improved functional ability, while others did not (inpatients showed decreases in jerk, while outpatients showed increases). 8 The authors mention that for poststroke movements, jerk minimization may not be the primary criterion governing refinements in movement patterns. Thus, jerk cost may be capturing different performance measures based on an individual’s level of recovery. Our participants were demographically similar to the outpatients of the Rohrer study. Thus, higher jerk may not be contradictory to lower smoothness observed in the paretic limbs of our participants.
What Do the Spectral Measures Tell Us?
Having established that hand and object introduced changes in performance as measured by temporal variables, we investigated how the spectral measures were affected. Only PRb was sensitive to both changes. Though the task is different, these results are consistent with spectral analyses of wrist motions of healthy subjects for a variety of ADLs, 32 of trained surgeons,16,25 and of repetitive occupational task performance, 17 which all indicate the importance of frequencies under 5 Hz in explaining motion quality. Thus, the power from 1.0 to 2.0 Hz is useful for distinguishing between changes in motor control and motor planning, as represented by temporal kinematic measures. To evaluate differences in ability of less controlled movements, we suggest measuring power in this frequency range. As for PRa, performance with the object (representative of preplanning) resulted in larger values than without the object, with an object by hand interaction. PRc was not sensitive to changes in either factor. This may imply that the tremor and unsteady motion captured between 2 and 10 Hz do not significantly vary in response to our exposures.
Can We Use the Spectral Measures to Explain the Variability of Temporal Measures?
The 3 spectral measures explain the variability of DUR and PKS for both the paretic and nonparetic limbs. Of further interest is the finding that, for the paretic limb, PRc accounts for over 57% and 58% of the explanatory ability of the spectral measures with respect to the response variables DUR and PKS, respectively. For the nonparetic limb, PRc explains less than 44% and 47% for DUR and PKS. This relative contribution of the higher frequency band may be indicative of the differences between paretic and nonparetic limb performance. In individuals poststroke, over time, the reduced contribution of PRc to the kinematic measures may be indicative of the restoration of premorbid function. This interpretation can be further investigated using longitudinally collected in-home accelerometry data.
What Next?
We have established that spectral analyses of accelerometer data are novel tools for capturing limb quality that should be further explored in order to provide new information about motion quality after stroke. Our investigation of spectral characteristics for a small population of mild-to-moderately impaired poststroke adults revealed the importance of frequencies between 1 and 2 Hz to distinguish between levels of motion quality. With data obtained continuously in a home setting, the relative contribution of these frequencies to signals can reveal progression in motor quality over time. Our focus on kinematic data is an effort to develop quantitative measures of performance and use in ambient environments. Such tools may provide insight into a number of open and compelling needs in the field of poststroke recovery. As Krakauer points out, hemiparesis is a term that incorporates effects of weakness, motor control deficits, and spasticity. 23 Understanding how these components evolve over time is necessary to understand what works optimally in helping individuals recover from stroke. This point is expanded upon by Levin and colleagues, who describe the need for tools to quantify elemental motor patterns necessary for elucidating the difference between recovery and compensation after stroke, 33 typically not distinguishable by assessments at the activity level of the International Classification of Functioning, Disability and Health model. 34
Looking forward to the development of tools for monitoring ambient activity, it will be necessary to begin evaluating less controlled, but more ecologically valid ADL-like movements in various settings to begin to understand how traditional measures of mechanisms such as efficiency and performance vary in real-world tasks. We are actively investigating the ability of spectral analyses to measure activity in ambient environments. We will make use of in-home data taken from study participants to establish the validity of spectral measures in quantifying recovery, with the ultimate goal of longitudinally evaluating how these measures change with functional recovery. One method of doing this is based on a technique demonstrated by Uswatte and colleagues, where the authors used 2-second epochs of accelerometer data to capture the amount of limb use in individuals after stroke. 35 In the same way, we can capture individual epochs of data and then apply spectral analyses to go a step further than capturing the amount of limb use, to describe the quality of that limb use as it relates to mechanisms of motor ability.
Study Limitations
The study by Wisneski et al showed that orientation and placement of items for task performance can result in significantly different reaching strategies and kinematic performances. 24 Though we standardized object locations, we did not constrain the reaching strategies of participants. Reach strategies across participants may have varied; we did not measure or account for these changes, though this may be possible using annotated video data. Also warranting further investigation is the selection of the relevant frequency bands. In our study, we use static bands across all gestures. It is likely that there is some overlap in the frequency bands we have chosen. Further studies should investigate how frequency bands of interest may vary with various classes of tasks (eg, using the limb for balance or support vs reach-to-grasp gestures). Furthermore, we did not obtain clinical descriptors on study participants. This is being incorporated into ongoing and future studies. Finally, the sample for the study was limited in size. Ongoing work will determine the generalizability of these outcomes and their meaningfulness for the greater, poststroke population.
Conclusion
In summary, we established that spectral measures of accelerometry data are sensitive to performance differences with the nonparetic and paretic limbs and with the object present and absent, for ADL-inspired tasks. This finding warrants further investigation of the ability of these measures to capture meaningful performance information from continuous motion data, potentially obtained in ambient settings. These spectral kinematic measures, when combined with clinical domain knowledge, will have the potential to provide greater insight into mechanisms of UE stroke recovery.
Footnotes
Appendix
Acknowledgements
The authors would like to acknowledge two Doctor of Physical Therapy students, Ryan Frendeway and Joseph Robinson, for their help with data extraction.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was partially supported by NIH Grant Numbers 1T32HD064578-01A1 and U01NS056256-02S1. The work was also supported by a USC Graduate School PhD Fellowship for Christina Chen.
