Abstract
Background. Recent evidence suggests that motor training may be beneficial for slowing the onset of motor impairments in Parkinson’s disease (PD). Objective. To examine the impact of targeted rehabilitation on limb motor and cranial motor function and the corresponding corticospinal and corticobulbar circuits in a rodent model of PD. Methods. Baseline performance of limb (reaching) and cranial (licking) motor function were established prior to and 6 weeks following unilateral intrastriatal 6-hydroxydopamine (6-OHDA) infusions. Animals then received 6 weeks of limb motor rehabilitation (LMR) or cranial motor rehabilitation (CMR), after which motor performance was reassessed. Intracortical microstimulation (ICMS) was used to generate motor maps of corresponding corticospinal (forelimb) and corticobulbar (tongue) movement representations within the motor cortex ipsilateral to the 6-OHDA infusion. Quantitative tyrosine hydroxylase (TH) immunohistochemistry was performed to determine levels of striatal TH depletion in 6-OHDA animals using near infrared densitometry. Results. (1) unilateral intrastriatal dopamine depletion impaired both reaching accuracy and lick force; (2) targeted LMR ameliorated impairments in reaching performance; however, CMR did not improve lick force impairments; (3) unilateral dopamine depletion significantly reduced forelimb but not tongue motor map topography; (4) LMR partially restored forelimb motor maps, whereas CMR did not alter tongue motor maps; and (5) significant correlations were observed between skilled reaching accuracy, forelimb motor map area, and TH depletion, but no relationships were revealed for cranial motor function, motor maps, or TH depletion. Conclusions: These data demonstrate dissociation between striatal dopamine depletion, limb versus cranial motor function, and targeted motor rehabilitation in a rodent model of PD.
Keywords
Introduction
Parkinson’s disease (PD) is a neurodegenerative disease characterized by bradykinesia, tremor, rigidity, and postural instability. Movement dysfunction of the upper-aerodigestive tract contributes to significant impairment in cranial motor functions, including speech and swallowing 1 that result in reductions in quality of life 2 and mortality. 3 Although PD has now come to be recognized as a multisystem neurodegenerative disease, 3 most motor symptoms are believed to be the clinical offprint of dysfunction within the basal ganglia thalamocortical (BGTC) loop. Loss of nigrostriatal dopamine results in hyperactivity of the basal ganglia, resulting in hypoactivity of motor cortical areas4,5 and causing the hypokinetic symptoms of PD. 6
Several rodent models of nigrostriatal degeneration have been developed to recreate the behavioral and neurophysiological abnormalities seen in PD. Using such a model, we have previously demonstrated that striatal dopamine depletion induces significant forelimb motor impairments that manifest neurophysiologically as a reduction in cortical forelimb movement representations and that forelimb impairments were correlated with both the level of striatal dopamine depletion and motor map loss. 7 We have also noted, however, that cranial motor impairments (licking), were not correlated with striatal dopamine depletion or associated with a loss of oral motor movement representations. 8 Thus, the relationship between striatal dopamine depletion, motor impairments, and motor cortex dysfunction appear to differ for limb motor versus cranial motor systems.
Surgical and pharmacological treatments directed at recovering BGTC function are effective in treating limb motor dysfunction, 9 and dopaminergic replacement therapy has been noted to restore decreased limb movement cortical activity in PD patients.5,10 These same treatments, however, fail to confer reliable improvements in cranial motor functions.11-14 Recent evidence suggests, however, that targeted motor training may be effective for improving the onset of both limb15-17 and cranial18-20 motor impairments in individuals with PD, which are hypothesized to be the result of a slowing or reversal of dopamine neuron degeneration.21-23 These findings suggest that motor training during the early stages of PD may restore function within the BGTC loop and ameliorate aberrant inhibition of motor cortical areas. The purpose of this investigation was to examine the effects of targeted limb versus cranial motor training on (1) limb motor and cranial motor function and (2) corticospinal and corticobulbar circuits in an animal model of early-stage PD.
Methods
This investigation was conducted in compliance with the University of Florida Animal Care and Use Committee. The experimental timeline is depicted in Figure 1. A total of 40 male Long Evans hooded rats (~100 days old, 325-425 g) completed baseline testing (test I) of limb motor and cranial motor function. Animals were then randomized to a 6-hydroxydopamine (6-OHDA; n = 20) or control (n = 20) group. Animals in the 6-OHDA group underwent unilateral intrastriatal 6-OHDA infusions to induce striatal dopamine depletion. Animals were then retested (test II) to assess the impact of unilateral dopamine depletion on limb and cranial motor function. Immediately following postlesion behavioral testing, animals were further randomized to either a limb motor rehabilitation (LMR; n = 20) or cranial motor rehabilitation (CMR) group (n = 20). Animals then underwent daily LMR or CMR for 6 weeks. Immediately following rehabilitation, animals were retested (test III) to examine the impact of targeted training on limb motor and cranial motor impairment. Intracortical microstimulation (ICMS) was used to generate high-resolution motor maps of cranial and limb movement representations within the motor cortex.
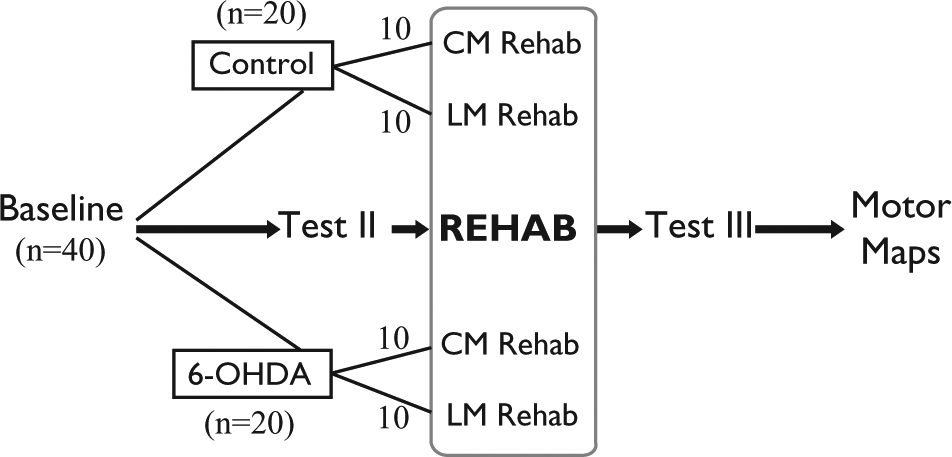
Experimental timeline.
Limb Motor Testing
Animals were trained over 14 days to reach for a single pellet with their dominant forelimb, as has previously been described by Boychuk et al. 24 Animals were food restricted throughout limb motor testing and performance videotaped for offline analysis. Percentage reaching accuracy was calculated as follows: [(Number of successful retrievals/The total number of reaches) × 100] and averaged over the final 2 testing days.
Cranial Motor Testing
A lick-force recording apparatus previously described in detail25,26 evaluated licking dynamics. Rats were water restricted and completed daily 2-minute sessions for 14 days with a lingual force requirement set at 2 g and licking behavior reinforced by delivery of 0.06 mL of water after every 12th lick (FR12). Median lick peak force (g) was averaged over the final 2 days of testing.
6-OHDA Infusions
Animals assigned to the 6-OHDA group received 4 unilateral 6-OHDA infusions into the contralateral hemisphere to their dominant limb following the protocol of Kirik et al. 27 Rats were anaesthetized with isoflurane (4% induction, 1.5% maintenance) and placed in a stereotaxic instrument (Kopf, Tujunga, CA) with the incisor bar set to skull flat. Infusion coordinates relative to Bregma were as follows: (AP +1.3 mm, ML −2.6 mm; −5.0 mm DV); (AP +0.9 mm, ML −3.0 mm; −5.0 mm DV); (AP −0.4 mm, ML −4.2 mm; −5.0 mm DV); (AP −1.3 mm, ML −4.5 mm; −5.0 mm DV). At each infusion site, a microbur was made through the skull, and a pulled glass micropipette was lowered into the striatum at each coordinate. Using a computer-assisted nanoinject microinfusion pump (World Precision Instruments, Sarasota, FL), 7 µg (free base weight) of 6-OHDA hydrochloride (Sigma, Oakville, ON) dissolved in 0.1% ascorbic acid was infused at a rate of 1 µl/min and left in place for 5 minutes to aid infusate diffusion before being slowly retracted from the brain. The scalp was then sutured.
Rehabilitation
Immediately following test II, animals commenced daily rehabilitation for 6 weeks and a total of 40 sessions. Those assigned to the LMR group performed daily single-pellet reaching rehabilitation for 15 minutes. Animals in the CMR group commenced a progressive lingual resistance training protocol developed by Connor et al. 28 This involved placing animals in the lick-force chamber and progressively increasing the force target required for obtaining a water reward over the 40-day training period. Maximum lingual force was first determined in each animal; they then completed an individualized progressive resistance protocol at 50% of their estimated maximum lingual strength for training days 1 to 10, 60% for training days 11 to 20, 70% for training days 21 to 30, and 80% for training days 31 to 40.
Intracortical Microstimulation
Within 1 week of the final testing session, ICMS was used to generate detailed maps of forelimb and tongue regions.29,30 Animals were anesthetized with ketamine hydrochloride (70 mg/kg intraperitoneally) and xylazine (5 mg/kg intraperitoneally). A craniotomy was performed over the ipsilesional motor cortex, and a digital image of the cortical surface was taken and saved in CANVAS 10.0 (ACD Systems, Vancouver, Canada) on a Macintosh Computer (Cupertino, CA). A glass microelectrode was used to make systematic penetrations across the cortex using the cortical surface image with a 300-µm superimposed grid as a guide. At each penetration site, the electrode was lowered to 1550 µm (cortical layer V). Stimulation consisted of 13 cathodal pulses (200 µs) delivered at 350 Hz from an electrically isolated stimulation circuit at a rate of 1 Hz. Sites where no movement was detected at ≤60 µA were recorded as unresponsive. Movements of the distal forelimb, proximal forelimb, and tongue were visually confirmed by an experimenter. Outcome variables included total motor map area (mm2) and minimum stimulation thresholds (µA) for forelimb and tongue.
Immunohistochemistry
Animals were perfused intracardially and decapitated for brain extraction. Brains were postfixed in 4% paraformaldehyde (24 hours) and cut into 40-µm coronal sections using a freezing microtome. Floating sections were washed with 0.01 M phosphate buffered saline (PBS) and treated for 15 minutes with 0.5%H2O2 + 10% methanol in 0.01 M PBS. Sections were preincubated with 3% normal horse serum + 0.1% Triton X-100 in 0.01 M PBS, then incubated overnight with a 1:2000 dilution of a mouse antityrosine hydroxylase (anti-TH) antibody (Chemicon, Temecula, CA). Following incubation, tissue was washed and incubated for 2 hours with a secondary antibody directed against the species in which the primary antibody was raised. The reactions were incubated with a near-infrared secondary antibody for TH densitometry, IR dye 800 (LI-COR Biosciences, Lincoln, NE). Sections were mounted on subbed slides, dehydrated in ascending alcohol concentrations, cleared in xylene, and coverslipped in Permount.
Near-Infrared Densitometry
TH-labeled sections were scanned at a resolution of 21 µm and a 5.5 sensitivity setting on a LI-COR Odyssey scanner (LI-COR Biosciences). Striatal sections were outlined as well as the value of the integrated intensity obtained from the right and left striatum of each section. TH depletion was calculated using the following formula: [(ϵ Lesioned integrated intensity/ϵ Intact integrated intensity) × 100]. 31
The following statistical analyses were performed: (1) a 2 × 2 (Condition × Time) mixed-model ANOVA on limb and cranial motor function pre-6-OHDA and post-6-OHDA lesion; (2) a 4 × 3 (Rehabilitation group × Time) mixed-model ANOVA on limb and cranial motor tests; (3) a 1-way, between-groups ANOVA on motor map area and movement thresholds across rehabilitation groups; and (4) a series of Pearson’s correlation analyses between TH depletion, motor map area, and limb and cranial motor function. Post hoc analyses consisted of a Scheffé test when significant interactions for condition or rehabilitation were observed and a paired t test when significant interactions with time were noted. We set α at .05 and SPSS version 19.0 (SPSS Inc, Chicago, IL) was used to compute all data.
Results
Densitometry
Three animals were excluded from the 6-OHDA group because of insufficient TH depletion (<50%). Densitometry analysis revealed that mean TH depletion for the 6-OHDA group (n = 17) was 88.27% (range = 50.6-100; standard error of the mean [SEM] = 3.39). Fluorescent immunoreactivity staining indicated that striatal 6-OHDA infusion resulted in loss of TH immunoreactivity in the striatum (Figure 2). Significant reductions in TH-integrated intensity values were revealed for 6-OHDA animals compared with controls: t(16) = −4.49; P < .001. In addition, a significant reduction in striatal TH levels for the intact versus lesioned hemisphere was revealed in 6-OHDA animals: t(16) = −7.77; P < .001. No significant differences were revealed for TH depletion levels between 6-OHDA rehabilitation groups: t(16) = −1.58; P > .05. Mean TH depletion for lesioned hemispheres in LMR versus CMR 6-OHDA groups (Figure 2B), respectively, were 16.56 (5.60) versus 6.30 (2.75).
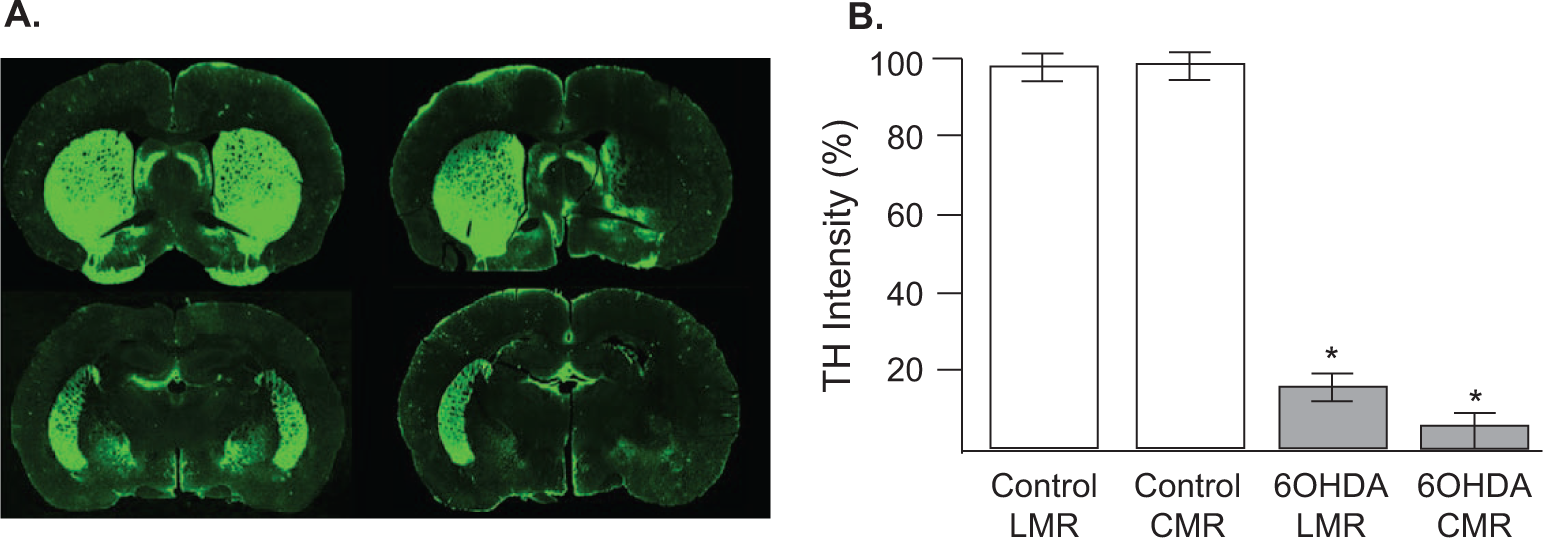
A. Representative coronal sections of a control (left) and 6-OHDA animal (right) immunohistochemically labeled for tyrosine hydroxylase (TH). B. Mean (±standard error of the mean) striatal TH depletion levels across groups: (ϵ Lesioned integrated intensity/ϵ Intact integrated intensity) × 100. Percentage TH depletion was significantly reduced in 6-OHDA animals but did not differ as a function of training condition (*P < .05).
Impact of Unilateral Dopamine Depletion on Cranial and Limb Motor Performance
A significant Time × Condition interaction was revealed for reaching accuracy between the first 2 behavioral testing points—F(1) = 20.53, P < .001—indicating significant impairment in forelimb reaching abilities following unilateral striatal dopamine deletion. Subsequent post hoc analysis revealed that 6-OHDA animals demonstrated significantly lower reaching accuracies at test II (postlesion, prerehabilitation) in comparison to both their own baseline reaching performance—t(16) = 5.03, P <.001—and to controls at test II—t(38) = −5.53, P <.001. A significant Time × Condition interaction was revealed for lick force: F(1) = 4.36; P < .04. Subsequent post hoc tests revealed a significant postlesion reduction in lick force for 6-OHDA animals: t(16) = 4.04; P < .001.
Impact of Targeted Rehabilitation on Cranial and Limb Motor Performance
A significant Time × Rehabilitation × Condition interaction was observed for reaching accuracy (Figure 3A): F(1) = 4.03; P < .05. Post hoc analyses revealed significantly lower skilled reaching accuracy for both 6-OHDA rehabilitation groups versus both control rehabilitation groups at test II. This included a significant difference in 6-OHDA LMR versus control LMR (P = .009), 6-OHDA LMR versus control CMR (P = .008), 6-OHDA CMR versus control LMR (P = .002), and 6-OHDA CMR versus control CMR (P = .002). At test III, the 6-OHDA CMR group demonstrated significantly reduced reaching accuracy compared with both control groups (LMR, P = .01; CMR, P < .005). Following 6 weeks of rehabilitation in forelimb reaching, LMR 6-OHDA animals demonstrated significantly higher reaching accuracy than the 6-OHDA animals in the CMR group (P = .01) and no significant differences compared with either of the 2 control groups (P > .05). A post hoc paired t test revealed a significant reduction in skilled reaching accuracy for the 6-OHDA LMR group—t(8) = 3.06, P < .02—and 6-OHDA CMR group—t(8) = 3.92, P < .01—from baseline testing to test II. Between test II (postlesion, prerehabilitation) and test III (postlesion, postrehabilitation), a significant increase was noted in skilled reaching accuracy for 6-OHDA animals engaging in daily LMR—t(8) = 3.06, P < .03—whereas a significant decrease was noted for 6-OHDA animals in the CMR group—t(8) = 2.96, P < .02.
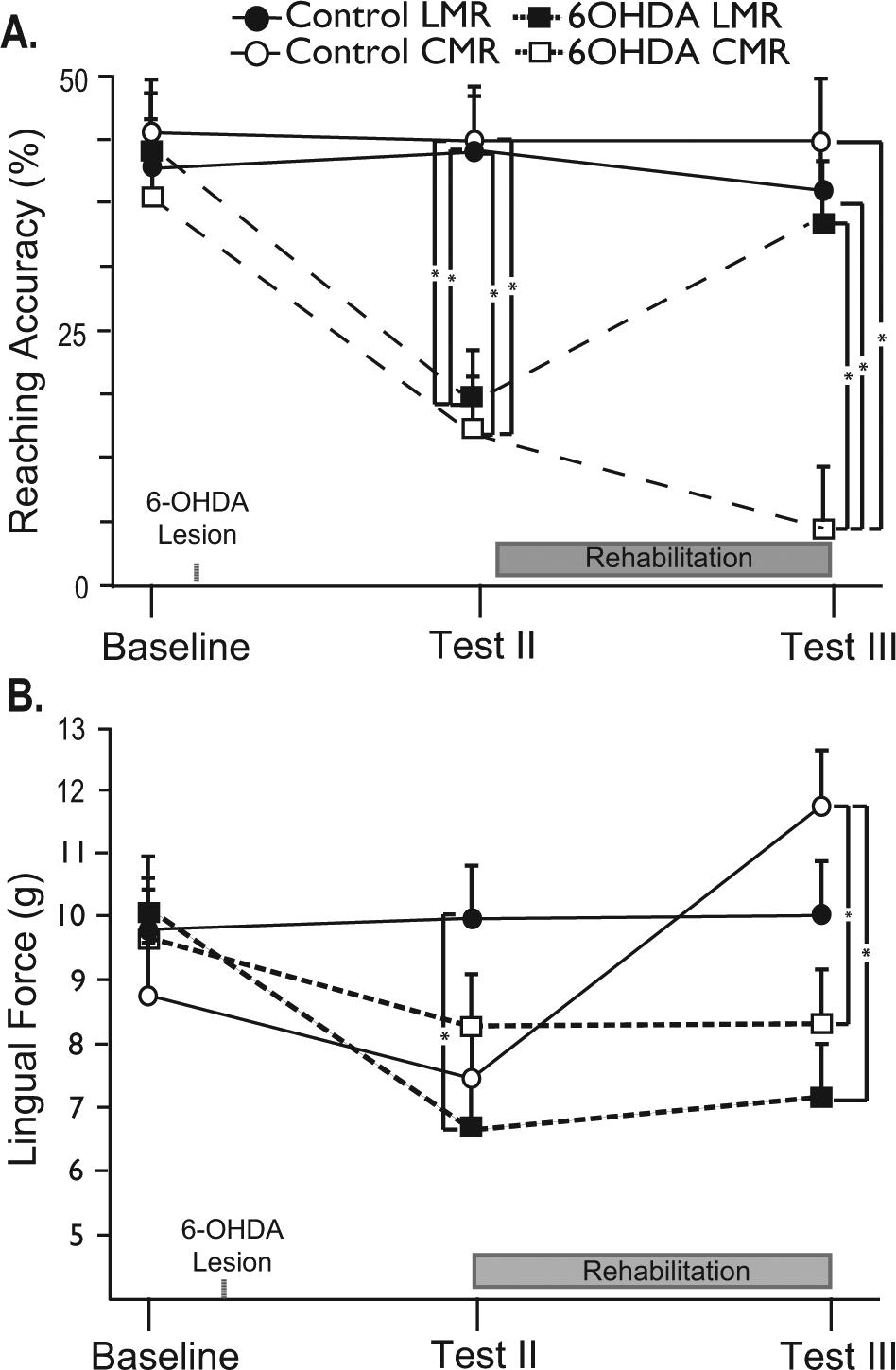
Impact of targeted training on limb and cranial motor function: A. 6-OHDA animals showed significantly reduced reaching accuracies at test II (postlesion, prerehabilitation) compared with control animals. However, 6 weeks of LMR ameliorated forelimb reaching deficits in 6-OHDA animals. B. 6-OHDA animals showed significantly reduced lingual force at test II compared with control animals; 6 weeks of CMR did not improve lingual force in 6-OHDA animals but did significantly increase lingual force in control animals.
A significant interaction was revealed for Time × Rehabilitation × Condition for lick force: F(1) = 7.04, P < .01. Post hoc analyses indicated several significant differences between groups. At test II, average licking force for 6-OHDA CMR animals was significantly lower than for control LMR animals (P = .002), whereas at test III, both 6-OHDA rehabilitation groups demonstrated significantly lower lingual forces than the control CMR group (6-OHDA CMR, P = .000; 6-OHDA LMR, P = .003). Finally, a paired t test revealed significant reductions in licking force from baseline to test II for the 6-OHDA LMR animals—t(8) = 2.47, P = .04—and 6-OHDA CMR animals—t(7) = 3.60, P = .009—whereas a significant increase in lingual force was noted between test II and test III for control CMR animals—t(10) = −4.51, P = .01—demonstrating a training effect for the controls. The mean (±SEM) lick forces for control animals during the rehabilitation phase at weeks 1, 3, and 5 were as follows: 7.5 g (1.2), 9.6 g (1.5), and 11.8 g (1.6), respectively. The mean (±SEM) lick forces for 6-OHDA animals were as follows: 8.2 g (1.5), 8.1 g (1.6), and 8.2 g (1.7) at weeks 1, 3, and 5 of the rehabilitation period.
No significant between- or within-group differences were revealed for licking rhythm—F(2) = 1.31, P > .05—or licking frequency—F(2) = 1.01, P > .05.
Cortical Movement Representations
No significant differences in the amount of anesthesia as a function of body weight and duration of surgery were noted for ketamine—F(3) = 1.28, P = .30—or xylazine—F(3) = 0.67, P = .67—across the 4 rehabilitation groups for the ICMS procedure. Motor map area was significantly reduced for forelimb—F(1) = 34.21, P = .000—but not tongue—F(1) = 0.24, P > .05—movement representations in 6-OHDA animals compared with controls. Although differences in motor map size were noted across disease states, no significant differences were observed in motor map excitability for forelimb—F(1) = 0.60, P > .05—or tongue—F(1) = 0.06, P > .05—movement thresholds. Mean forelimb motor thresholds were 29.17 (0.78) and 30.36 (1.52) µA for control and 6-OHDA animals. Mean tongue motor thresholds were 38.64 (5.37) and 37.06 (2.18) µA for control and 6-OHDA groups. Representative motor maps across disease and rehabilitation states are shown in Figure 4.
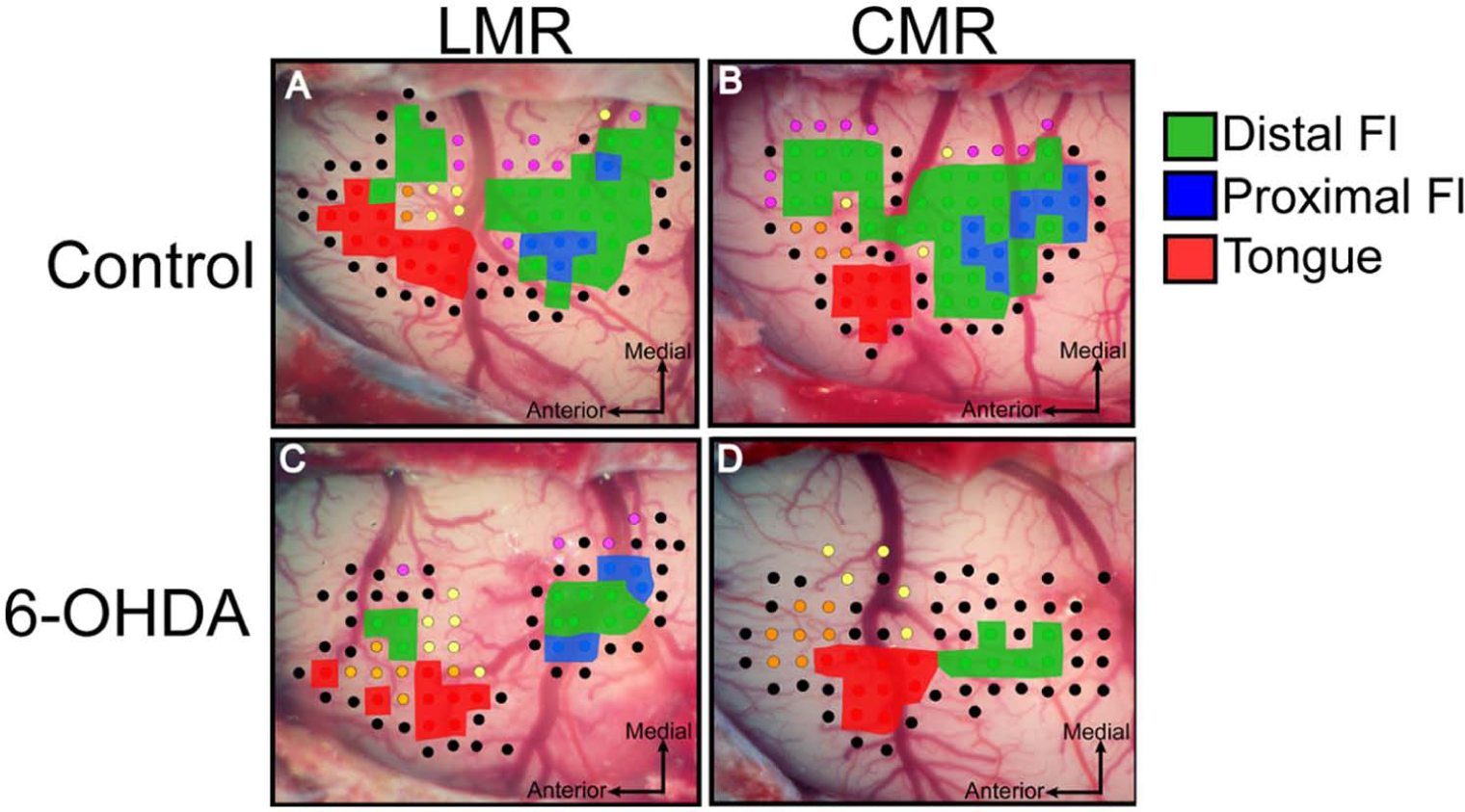
Representative motor maps for control and 6-OHDA animals across rehabilitation groups: A. control LMR. B. control CMR. C. 6-OHDA LMR. D. 6-OHDA CMR. The distal forelimb (wrist/digit) is shown in green, proximal forelimb (elbow shoulder) in blue, and tongue in red. Border sites included no response (black), jaw (orange), whisker (purple), and neck (yellow).
A significant difference in forelimb motor map area was revealed across groups (Figure 5A): F(3) = 15.66; P < .001. Subsequent post hoc analyses revealed 5 significant between-group differences for the forelimb motor map area. The 6-OHDA CMR forelimb map area was significantly smaller than that of control CMR (P = .000), control LMR (P = .000), and 6-OHDA LMR groups (P = .04)—that is, every other group. The 6-OHDA LMR forelimb map area was significantly smaller than that of control CMR (P = .002) and control LMR (P = .003) groups. A significant difference in forelimb movement distribution (percentage proximal vs distal representation of total forelimb map area) was noted across disease states—t(35) = 3.41, P < .003—with 6-OHDA animals demonstrating significantly larger proportions of forelimb motor maps occupied by distal forelimb sites. No rehabilitation effects were observed (P > .05), although 6-OHDA animals undergoing CMR were noted to lose proximal forelimb movement representation, whereas those undergoing daily LMR were observed to retain on average 15% proximal forelimb representation of their total forelimb map area.
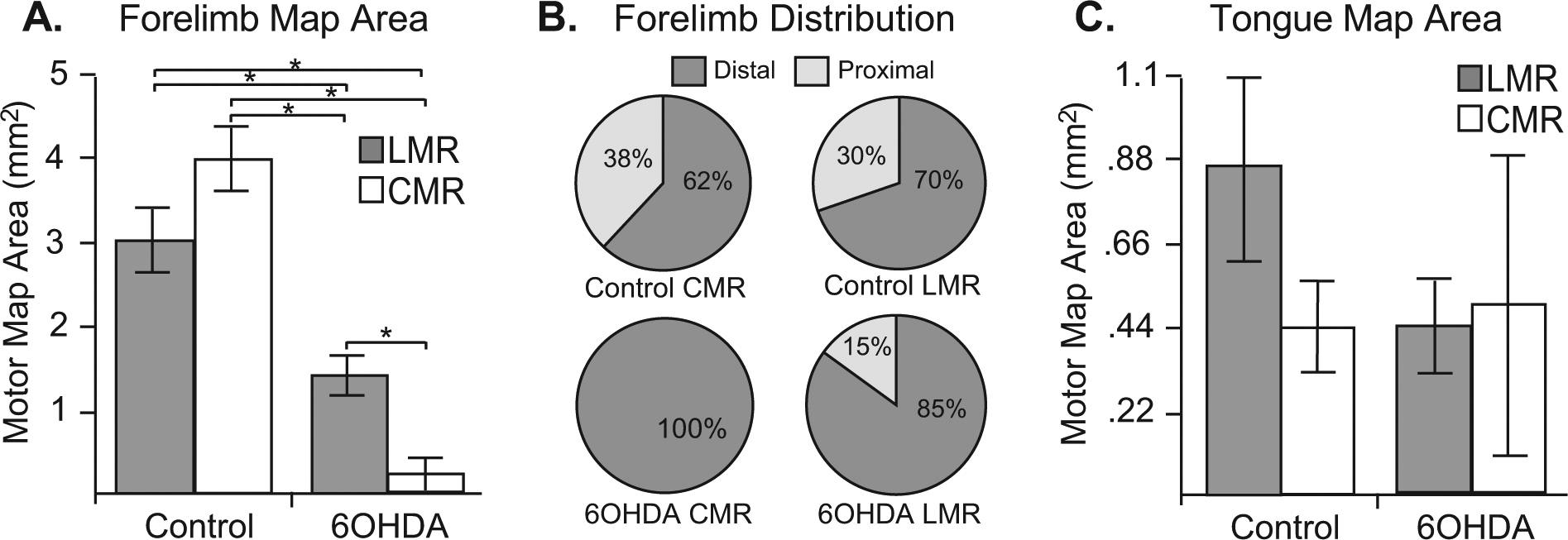
Mean (±standard error of the mean) forelimb and tongue motor map area. A. Both 6-OHDA rehabilitation groups demonstrated significantly smaller forelimb motor maps compared with both control groups. Furthermore, 6-OHDA animals in the LMR condition demonstrated significantly larger forelimb motor maps than 6-OHDA animals not engaging in LMR. B. Forelimb movement distributions across experimental conditions. A significant reduction in percentage proximal forelimb representation was noted for 6-OHDA animals (P = .003). No rehabilitation effect was observed (P > .05); however, 6-OHDA animals undergoing CMR were noted to lose proximal forelimb movement representation, whereas those undergoing daily LMR retained on average 14.44% proximal forelimb representation of their total forelimb map area. C. No significant differences were noted for tongue motor map area between any experimental conditions.
No significant differences were noted for tongue motor map area by rehabilitation group (Figure 5C): F(3) = 0.84, P > .05. No significant between-group differences were observed for rehabilitation group and motor map thresholds for either the forelimb—F(3) = 0.33, P > .05—or tongue—F(3) = 0.14, P > .05.
Relationship Between Behavior, Maps, and TH Depletion
Both reaching accuracy (r = −0.65, P = .002; Figure 6A) and forelimb motor map area (r = −0.59, P = .01; Figure 6B) were negatively correlated with TH depletion. A positive correlation was also revealed for reaching accuracy and forelimb motor map area (r = 0.81, P = .000; Figure 6C). No correlations were observed between TH depletion levels, cranial motor function, or motor maps (P > .05; Figures 6D-6F).
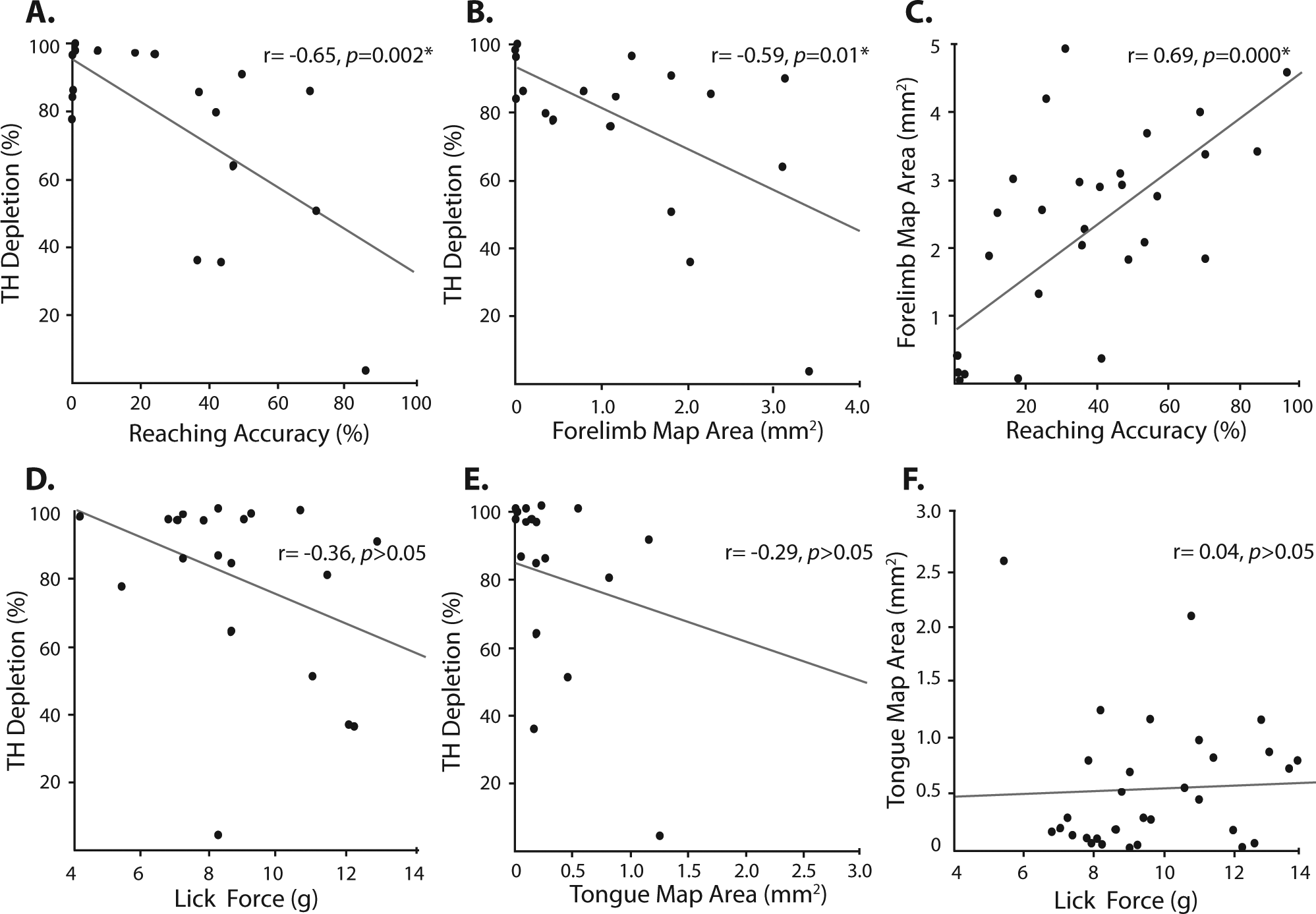
Scatterplots depicting correlations between TH depletion, motor map area, and motor performance. Significant correlations were noted between skilled reaching accuracy and TH depletion (A), TH depletion and forelimb motor map area (B), and forelimb motor map area and reaching accuracy (C). No significant correlations were revealed for lick force and TH depletion (D), TH depletion and tongue motor map area, or (E) tongue motor map area and lick force.
Discussion
Understanding the differential effect of targeted motor training on cranial versus limb motor systems in PD is a critical step toward developing comprehensive and effective treatments. The present study utilized an animal model of early-stage PD to directly compare the effects of targeted training on limb motor versus cranial motor function and the corresponding corticospinal and corticobulbar systems. Here we report that (1) unilateral intrastriatal dopamine depletion impaired both limb and cranial motor function; (2) targeted LMR ameliorated limb but not cranial motor impairments; (3) unilateral dopamine depletion significantly reduced forelimb but not tongue movement representations; (4) LMR partially restored forelimb motor map area; and (5) significant relationships were observed between skilled reaching accuracy, forelimb motor map area, and dopamine depletion, whereas no relationships were observed for cranial motor function, dopamine depletion, and corticobulbar motor map area.
Rehabilitation Improves Only Limb Motor Function in 6-OHDA Animals
We found that 6 weeks of daily targeted LMR ameliorated severe limb motor impairments, whereas targeted CMR did not overcome the moderate impairments in lingual strength in 6-OHDA animals. Although progressive lingual resistance training had no impact on tongue force in dopamine-depleted animals, this intervention was observed to enhance lingual strength in healthy animals, as has been previously documented by others,28,32,33 providing evidence that the specific CMR program utilized was of sufficient intensity to drive increases in lingual force.
Differences in the behavioral demands associated with the LMR and CMR training paradigms may have contributed to the differential responses to rehabilitation. The reaching task is a complex motor task that can be accomplished using a variety of compensatory strategies after brain injury, including changes in distal and proximal limb movement patterns and postural adjustments. 34 The ballistic, rhythmic nature of the licking task provides less opportunity for compensation in force production. Furthermore, animals in the LMR group received skilled reach training, and animals in the CMR group received lingual strength training. Therefore, different types of targeted training were being applied across the different behavioral modalities and may have affected the efficacy of the 2 rehabilitation interventions. Indeed, LMR did not cause further improvements in skilled reaching accuracy in control animals, whereas CMR did induce further increases in lick force in control animals beyond their baseline function. This suggests that the rehabilitation interventions may have been introduced at different phases of preinjury training in the LMR and CMR conditions. The LMR animals appeared to be at behavioral asymptote for reaching accuracy, whereas the CMR animals were still making gains in lingual force.
Unilateral Dopamine Depletion Reduced Forelimb but Not Tongue Motor Map Area
Whereas unilateral dopamine depletion significantly reduced forelimb motor map area, the same manipulation did not significantly alter tongue map area. This is consistent with prior work showing abnormalities in motor map topography several weeks after striatal dopamine depletion7,35,36 but suggests that the degradation in motor maps persist over several months. Exactly why forelimb movement representations were more sensitive to striatal dopamine depletion than the tongue is unknown. Both the tongue and forelimb areas of the striatum lie in the dorsolateral aspect of the caudate-putamen, 37 and all animals included in this study showed a loss of TH staining within this area. In cases where depletion was less than 100%, the residual staining was typically localized to the ventromedial aspect of the striatum. The results suggest, however, that the integrity of limb motor versus cranial motor neural circuits may be differentially reliant on striatal dopamine, with the cranial motor system and circuits less reliant on dopaminergic transmission as has been suggested by others.8,12,13,38-43
The observed loss of forelimb movement representations in 6-OHDA animals may be the result of a degradation of synaptic connectivity within the motor cortex. Motor map topography is highly sensitive to changes in synapse number and strength within the motor cortex. Manipulations that reduce synapse number 44 or decrease synaptic strength 45 also reduce motor map size. The observed loss of movement representations in 6-OHDA animals may therefore result from a loss of synapse number and/or strength within the motor cortex. Indeed, significant reductions in neurofilament protein, synaptophysin, and synapse number within the ipsilesional frontal cortex of animals receiving 6-OHDA injections into the SNc has been documented. 46 In support of this hypothesis, forelimb movements could be readily elicited in all 6-OHDA animals via stimulation of the internal capsule. This stimulation protocol elicits movement by directly stimulating corticospinal axons and is not dependent on transsynaptic activation of cortical pyramidal neurons within the motor cortex.
A unilateral model of dopamine depletion was utilized in this study to serve as a model for mild to moderate PD. Although it is possible to administer bilateral lesions of 6-OHDA, the lesions are typically associated with profound morbidity and a high degree of rodent mortality. One important consideration when interpreting our results is the fact that dopamine depletion was unilateral, and there is considerable bilateral control of cranial motor movements. Therefore, it is possible that the cranial motor system displayed less severe deficits than the limb motor system as a result of compensation by the contralesional hemisphere. However, Nuckolls et al 47 have studied the effects of unilateral versus bilateral dopamine depletion on tongue force and tongue motility during a licking task. They report that although both lesion groups demonstrated significant and similar reductions in lick force, only the unilateral group demonstrated tongue motility deficits. The authors suggest that the etiology of this later finding resulted from the presence of a functional imbalance in neural pathways affecting motility of the midline structure.
LMR Preserves Forelimb Map Area in 6-OHDA Animals
Although no rehabilitation effects were noted between CMR and tongue motor maps, 6-OHDA LMR–trained animals showed a significant increase in motor map area in comparison to those undergoing daily CMR. Although animals in the LMR group demonstrated lower forelimb motor map areas than both control groups, their mean forelimb motor map size was 4 times higher than 6-OHDA animals in the CMR group. Because motor map integrity is dependent on synapse number within the motor cortex,44,45 the partial restoration of forelimb motor maps may be a result of an experience-dependent increase in synapse number and/or strength. This has been observed in studies of forelimb motor rehabilitation after cortical ischemia, where skilled forelimb training both increased synapse number and improved reaching accuracy.47,48
Cortical map plasticity is also experience-specific, in that forelimb motor skill training induces map reorganization, 29 but neither aerobic 49 nor strength training 30 affect motor map topography. Rather, strength training has been noted to reduce movement thresholds in forelimb 30 and tongue 33 movements; however, this was not noted in the current study following progressive lingual strength training, likely a result of the fact that CMR trained 6-OHDA animals did not see any improvements in lingual force.
The mechanism by which LMR restores motor maps is unknown but may be related to an experience-dependent restoration of BGTC circuitry. Brown et al 50 demonstrated that stimulation of the subthalamic nucleus in 6-OHDA animals both increased motor map area and ameliorated forelimb movement impairments. In the present study, we also noted that LMR animals showed a small increase in TH staining within the striatum (83.44% depletion in LMR animals vs 93.70% depletion in CMR animals). Although this was not a statistically significant difference, we have previously reported a bimodal effect of striatal dopamine depletion on forelimb motor map integrity, whereby animals with less than 90% depletion showed partial motor maps, and animals with greater than 90% depletion showed a complete loss of forelimb motor maps. 7 This is consistent with the finding that intense limb motor activity can partially restore dopamine within the nigrostriatal system.21-23
Conclusion
These data demonstrate a triple dissociation between limb motor and cranial motor function and corticospinal and corticobulbar neural circuits in a rodent model of PD. First, unilateral intrastriatal dopamine depletion was seen to differentially impair limb motor versus cranial motor function that was reflected neurophysiologically as a loss in movement representations. Second, targeted motor rehabilitation partially restored limb function and the corresponding cortical movement representations but not cranial motor function or maps. Third, limb motor function and movement representations were related to the degree of dopamine depletion, whereas cranial motor function and movement representations were not. Together, these data demonstrate that cranial and limb motor systems are differentially affected by nigrostriatal dopamine depletion and are consistent with the clinical findings that treatments directed at restoring function of the BGTC loop are largely ineffective for ameliorating oral motor impairments in PD patients. Further work to identify the neural substrates for cranial motor deficits in PD is needed to develop comprehensive treatment strategies for the complex array of motor impairments associated with the disease.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by a National Institute of Deafness and other Communication Disorders (NIDCD) Training Grant 1F32DC010569-01A1.
