Abstract
Spasticity may be associated with a variety of neurological conditions that produce cerebral or spinal upper motor neuron damage. The lost inhibition of α or γ motor neurons results in a velocity-dependent hypertonia that may present clinically as stiffness, spasms, pain, decreased mobility, and increased difficulty with activities of daily living.
Stretching exercises, physical/occupational therapy, and oral antispasticity agents have long been the mainstay of treatment for spasticity. Patients with severe, functionally limiting diffuse spasticity may benefit from intrathecal baclofen (ITB) therapy. This FDA-approved therapy involves delivery of a small dose of the poorly lipophilic GABA-B agonist into the subarachnoid space. This treatment involves a surgical procedure and carries well-known risks, including surgical complications, infection, catheter malfunction, pump stall, baclofen withdrawal, and baclofen overdose. 1 Thus, careful patient selection and testing are warranted.
Favorable outcomes with ITB are well documented for patients with spinal cord injury, traumatic brain injury, stroke, and multiple sclerosis.2,3 In patients with severe spasticity refractory to oral symptomatic medications and who undergo a successful test injection, ITB therapy can decrease pain associated with spasms and stiffness, prevent contractures, complement other rehabilitation strategies, and facilitate hygiene and other care.
Spasticity and painful spasms are commonly encountered in patients with motor neuron diseases (MNDs) such as amyotrophic lateral sclerosis (ALS) and primary lateral sclerosis (PLS).4,5 Marquardt and Seifert 6 discussed 2 ALS patients with good response to ITB. Milano et al 7 reported improvement in quality of life related to ITB treatment in a patient with PLS. Sadiq and Wang 3 included 3 patients with MND in their report of safe ITB therapy in 36 ambulatory patients. McClelland et al 8 described pain relief associated with ITB therapy in 8 ALS patients (a subset of our cohort).
ITB therapy may not be strongly considered in this population of patients because of concerns for increased weakness and loss of ambulation associated with the treatment. 3 The limited life expectancy of these patients may also be a consideration. The purpose of this article is to report on our experience with a cohort of patients with upper motor neuron predominant MND (U-MND) referred for discussion of ITB therapy.
Methods
Participants
The data used for this analysis were extracted from a clinical spasticity registry that was approved by the Cleveland Clinic Institutional Review Board. A total of 44 patients with a diagnosis of ALS, PLS, or MND were referred to our spasticity clinic. All patients had severe spasticity refractory to oral medications. All patients were previously evaluated by a neurologist specialized in the treatment of motor neuron disorders (EPP).
Patients were considered candidates for ITB therapy if they had severe diffuse spasticity in the lower extremities with adverse consequences (eg, discomfort and interference with limb and trunk posture, ease of care, or the ability to perform daily activities) and had failed oral antispasticity medications, either as a result of lack of efficacy at high doses or as a result of dose-limiting side effects. Potential ITB candidates were evaluated by a physiatrist (FB) and were educated about ITB therapy. During the evaluation process, specific individualized goals were agreed on with the patient, based on the observed negative effects of spasticity and grounded in realistic expectations about what could be achieved with spasticity relief. Patients interested in pursuing a test injection received a 25- to 75-µg bolus of baclofen via lumbar puncture. The dose of the bolus was determined based on the severity of the spasticity, the severity of underlying weakness, the patient’s ambulation status, and the perceived risk of side effects (eg, compromised respiratory status). Those who experienced reduced stiffness, spasms, and pain and/or improved lower-extremity function were further educated about the risks and benefits of ITB therapy. These outcomes were based both on clinical standardized measures (described below) and on the patient’s qualitative feedback on the perceived benefits from the injection.
Patients were allocated to an ITB group or a non-ITB group based on their ultimate decision to proceed with ITB therapy or to continue conservative management. A virtual surgery date was calculated for the non-ITB group based on the average time to surgery for the ITB group (approximately 4 weeks). Two follow-up end points were defined: early (less than 6 months) and late (more than 6 months) follow-up. Only those patients with sufficient data available at baseline and late follow-up were included in the analysis. Most patients who did not get a baclofen pump did not have an early follow-up visit.
Data Collected
Demographic and medical characteristics included diagnosis, duration of neurological symptoms, age, gender, ambulation status, total number of medications used for spasticity, and dose of baclofen and tizanidine (the 2 most commonly used antispasticity agents).
The following data were collected during baseline assessment and at routine follow-up visits:
pain Numerical Rating Scale (NRS) from 0 to 10, where 10 indicates maximal pain 9 ;
manual muscle testing (MMT) on the standard 0 to 5 scale, where 5 is full strength 10 ; hip flexor strength scores for each side were averaged;
Modified Ashworth Scale (MAS) assessing resistance to passive movement on a 0 to 4 scale, where higher scores reflect more severe spasticity; scores for individual muscle groups (hip adductors, knee extensors, knee flexors, and ankle plantar flexors) were averaged to produce an overall lower extremity spasticity index 11 ;
self-reported Spasm Frequency Scale to estimate spasm frequency, from 0 (no spasm) to 4 (>10 spasms per hour) 12 ; and
timed 25-Foot Walk Test to measure maximum gait speed (in cm/s) over a short distance, for ambulatory patients; the patients were instructed to walk as fast as possible (but safely) from a standing start. 13
Statistical Analysis
Descriptive statistics were generated at each end point as appropriate (frequency, mean, and standard deviation). Between-group and within-group differences were tested using nonparametric tests (Mann Whitney
Results
Of the 44 patients with U-MND in our registry, 23 patients underwent implantation of an ITB infusion systxm. In all, 21 patients did not receive a pump either because they declined the screening injection (n = 14) or because they decided not to proceed with pump placement (n = 7). The reasons for declining a test injection were not systematically documented. The reasons for declining pump surgery included fear of complications, insufficient benefit perceived during the test injection, and patients “not liking the way the medication makes them feel.”
Sufficient data were available at baseline and late follow-up for only 35 of the 44 patients. Three patients who received baclofen pumps did not have follow-up data; 3 patients who declined the test injection had baseline data but no follow-up data; and 3 patients in the non-ITB group had no baseline or follow-up data. These 9 patients were excluded from the data analysis. There were no statistically significant differences between excluded and included patients for age, sex, or disease duration. Table 1 summarizes the baseline characteristics of the 35 patients (20 ITB, 15 non-ITB). Average baseline pain scores were significantly lower in the non-ITB group. The non-ITB group had a lower average MAS score, faster gait speed, and a lower proportion of patients using a walker than the ITB group. Although these differences were not statistically significant, they suggest that patients who opted for ITB therapy were somewhat more severely affected by spasticity. There was no difference in the average hip flexor strength, spasm frequency, or requirement for oral spasticity agents.
Patient Characteristics and Baseline Clinical Measures.
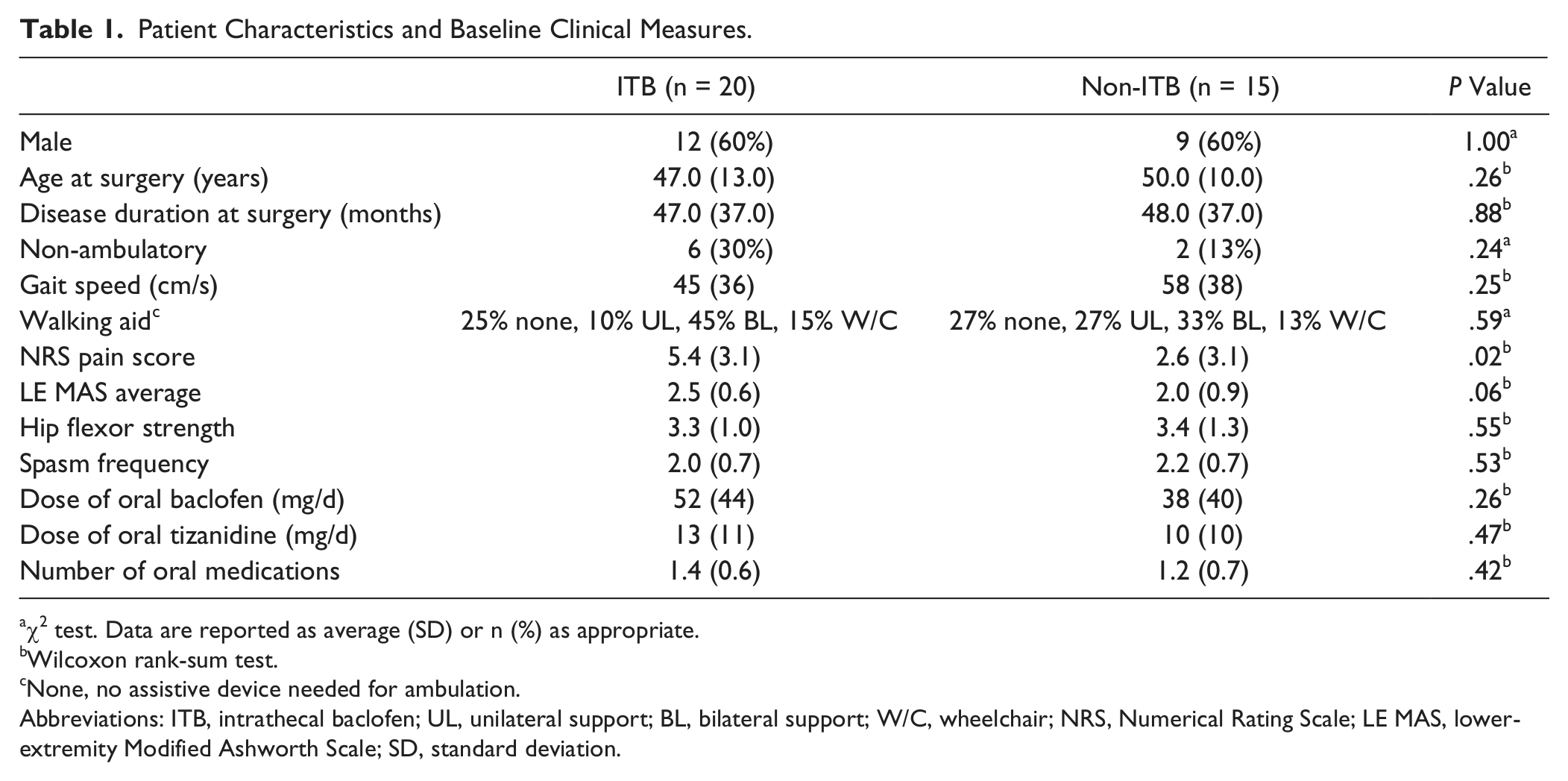
χ2 test. Data are reported as average (SD) or n (%) as appropriate.
Wilcoxon rank-sum test.
None, no assistive device needed for ambulation.
Abbreviations: ITB, intrathecal baclofen; UL, unilateral support; BL, bilateral support; W/C, wheelchair; NRS, Numerical Rating Scale; LE MAS, lower-extremity Modified Ashworth Scale; SD, standard deviation.
ITB Procedure and Postoperative Care
All patients electing for ITB therapy were implanted with a programmable infusion system via a standard surgical technique. 14 The pump was filled and started at a low rate in the operating room. Our clinical team made adjustments as necessary during the acute hospital stay. Subsequent rate adjustments or refills were performed either in our clinic or by a home care agency. The average (standard deviation) daily rate of ITB was 99.8 (71.4) µg/d at early follow-up and 233.9 (263.2) µg/d at the late follow-up. One patient developed a catheter malfunction but recovered without sequelae after surgical revision. No other complications requiring surgery were noted.
Comparison of Outcomes Between Groups at Late Follow-up (Table 2)
The late follow-up time period within the ITB group averaged 304 ± 99 days, and in the control group 297 ± 77 days. There was a statistically significant difference in change in MAS score (
Change Between Baseline and Late Follow-up.
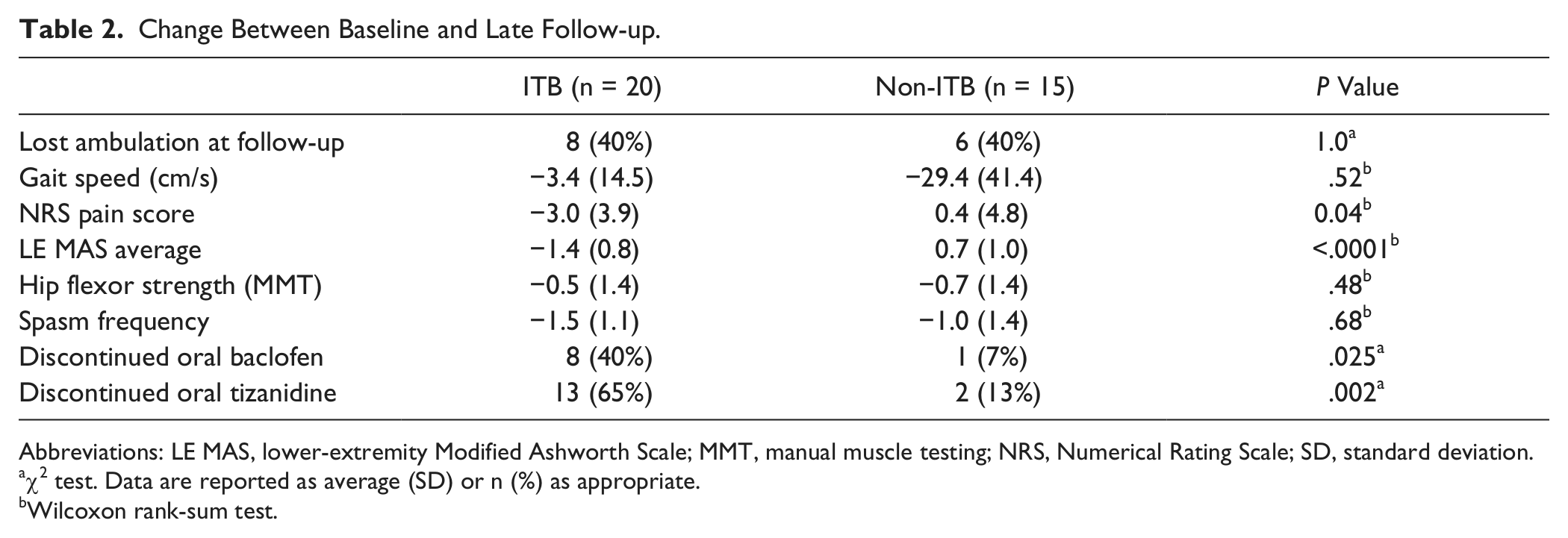
Abbreviations: LE MAS, lower-extremity Modified Ashworth Scale; MMT, manual muscle testing; NRS, Numerical Rating Scale; SD, standard deviation.
χ2 test. Data are reported as average (SD) or n (%) as appropriate.
Wilcoxon rank-sum test.
Outcomes Within the ITB Group (Figure 1)
Early follow-up data for the ITB patients were collected at 41 ± 23 days postop. Four patients (20%) became nonambulatory, and 2 previously nonambulatory patients (10%) regained the ability to walk at least 25 feet. There was a statistically significant improvement in MAS, Spasm Frequency Scale, and pain scores at early and late follow-up. The use of oral medications for spasticity was significantly reduced at early and late follow-up. Hip flexor strength was significantly improved at early follow-up only. No ITB patient elected to stop therapy by the time of late follow-up. Within the non-ITB group, there was a significant increase in MAS scores between baseline and late follow-up.
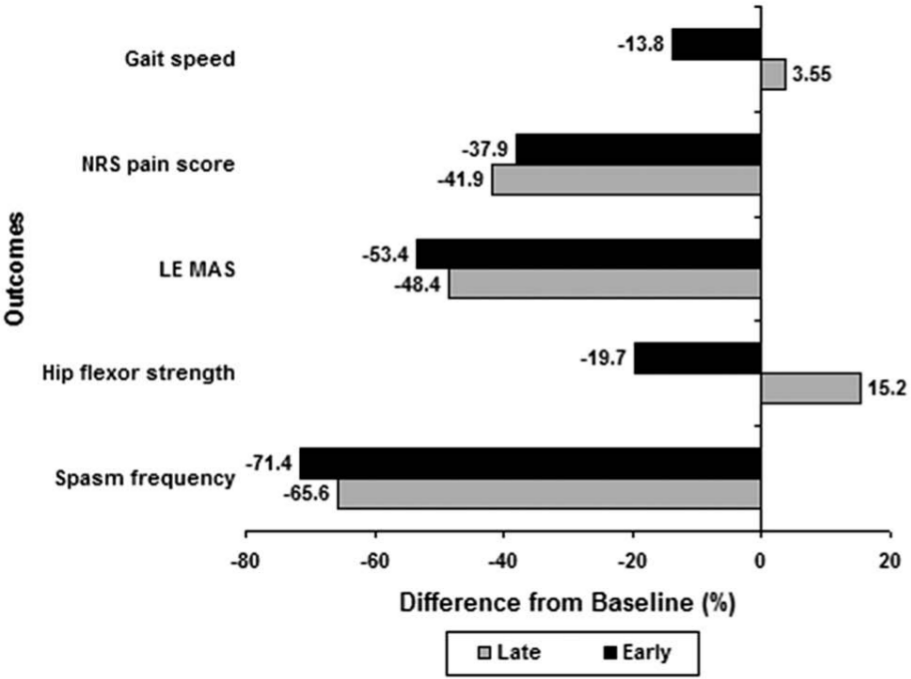
ITB effectiveness at early and late follow-up.
Discussion
Although our sample size is relatively small, this is the largest cohort of ITB-treated MND patients reported to our knowledge. Our results indicate that ITB therapy can effectively reduce spasticity and related pain and spasms in U-MND patients with severe spasticity refractory to conservative management, confirming findings published by McClelland et al 8 in a subset of 8 ALS patients from the same cohort. As expected, the reduction in symptoms was accompanied by a decrease in the use of oral antispasticity agents, which generally result in lower occurrence of central side effects such as drowsiness and dizziness (although this was not formally assessed in our study).
The improvement in spasticity and related symptoms in the ITB group was not associated with increased lower-extremity weakness or loss of functional mobility. Average hip flexor strength even improved slightly within the ITB group at the early follow-up time, suggesting that severe spasticity was partially inhibiting motor output. There was no significant difference between the ITB and non-ITB groups with regard to loss of ambulation or change in hip flexor strength at late follow-up. The gait speed of the ITB patients who remained ambulatory at late follow-up did not change significantly from baseline. There was a substantially larger decline in gait speed within the non-ITB group compared with the ITB group, which did not reach statistical significance and should be interpreted with caution since the rate of progression of the disease in each group is not known.
All the patients who appeared to lose ambulation during the follow-up period required more than 30 seconds to walk 25 feet at baseline. This suggests that these patients relied more on their spasticity to walk. It is difficult to determine if the loss of ambulation occurred earlier in these patients because of ITB therapy or if it would have occurred anyway as a result of disease progression. The substantial decline in ambulatory status in the non-ITB group seems to support the latter. The risk of functional loss and the fact that ITB does not treat the disease process had been discussed before the surgery.
None of the ITB patients (including those who lost ambulation) requested discontinuation of therapy. This suggests that the benefits of ITB on symptom relief are meaningful, even when mobility cannot be maintained, and that the therapy is well tolerated. Unfortunately, our study design did not include measures to assess the impact of pain and spasm reduction on overall quality of life. Likewise, no comparison of independence in activities of daily living before and after pump placement was available.
We acknowledge further methodological limitations in this retrospective study of our experience with ITB therapy in patients with MND. The sample size is small and limited to carefully selected patients. Follow-up visits were not perfectly synchronized, resulting in varying treatment durations. Patient allocation between the treatment and control groups was not random but rather the result of a clinical decision. As a consequence, the groups were not perfectly matched in baseline pain rating score or degree of lower-extremity spasticity. Our study did not include rate of disease progression or disease severity measures at baseline or at follow-up visits.
It should also be noted that our patients presented with an upper–motor neuron phenotype of MND (U-MND). Upper–motor neuron predominant ALS is associated with a younger age at onset, longer disease duration, and very late impairment of respiratory muscles compared with classic ALS.5,15 Patients included in our study had an average disease duration of 47 months at the time of surgery. Although there are no systematic studies on the course of and prognosis for upper–motor neuron predominant ALS, Soraru et al 15 reported long-term survival with a relatively slow progression to a tetrapyramidal syndrome (mean of 7.7 years) in a group of 10 patients.
Considering surgical treatment for symptom management in MND raises ethical issues. In the absence of disease-modifying therapies for MND, patient care is focused on managing symptoms, preventing complications, optimizing patient quality of life, and reducing caregiver burden. 16 Because of the rate of disease progression, the decision to evaluate a patient with U-MND for ITB therapy is sometimes made with a sense of relative urgency because the progression of bulbar symptoms and general weakness can result in a higher surgical risk. However, the potential benefits (comfort, ease of care, prevention of contractures, and possibly greater ease of functioning) must be carefully discussed and weighed against the risks and costs of the therapy, and realistic goals must be clearly defined. Our study attempts to contribute to this discussion by providing data on efficacy and safety after ITB therapy in comparison with conservative management. In our cohort, patients with higher pain scores at baseline were more likely to choose ITB therapy, suggesting that the risk-benefit ratio appeared more favorable in the presence of more severe pain associated with spasticity. Despite a worsening of symptoms and walking performance over the course of the study, none of the patients in the non-ITB group requested to start ITB therapy. This may reflect the fact that the risk-benefit ratio of this therapy remained too high in their judgment, particularly because bulbar symptoms and respiratory compromise with disease progression increase the surgical risk.
When considering long-term patient management, the dose of ITB can be easily adjusted as the disease progresses. Having intrathecal therapy already on board can help maintain patient comfort at the end of life when oral dosing of medications is no longer practical. The fact that no patient in the ITB group asked to discontinue ITB therapy suggests that the benefits of ITB therapy continue to be felt despite disease progression.
Prospective studies that include measures of quality of life, functional performance, and caregiver perceptions could provide further insights into the use of ITB as a symptomatic treatment for patients with MND. However, the ability to conduct such studies is limited by the small target population, variability in the rate of progression, and disparate goals among patients with MND (ie, preservation of function vs relief from pain). Although it is not common practice, continuous infusion of baclofen via a temporary intrathecal catheter connected to an external pump may yield more information than a single bolus dose when preservation of function in the context of significant underlying weakness is a concern.17,18
Conclusion
ITB therapy should be considered as a treatment option for MND patients with primarily upper–motor neuron symptoms and severe spasticity refractory to oral medications, who respond positively to an intrathecal test injection. Although individual responses may vary, ITB therapy does not seem to adversely affect ambulation.
Footnotes
Declaration of Conflicting Interests
The authors declared the following conflicts of interest with respect to the research, authorship, and/or publication of this article: Drs. Bethoux and Boulis receive consulting fees from Medtronic.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Authors’ Note
This work was not supported by external funding. Patient consent was obtained. Ethics approval was provided by the Cleveland Clinic Institutional Review Board. FB receives consulting fees from Medtronic, Allergan, IMPAX Laboratories, and Merz Pharmaceuticals. NB receives consulting fees from Ceregene and Medtronic and is supported by grants from NeuralStem and Genzyme/Sanofi. AM receives consulting fees from Intelect Medical/Boston Scientific and Monteris. SM, MAW, MH, LM, DS, MS, and EPP have nothing to disclose.
