Abstract
Introduction
Between 50% and 70% of stroke survivors have persistent upper-extremity motor deficits,1,2 which significantly decrease activity and participation.
3
Many tasks can be accomplished with only 1 hand. Thus, after unsuccessful attempts to use the impaired arm after stroke, patients begin to use compensatory strategies such as relying on the intact limb to perform various self-care activities.
4
As a result, patients avoid using the affected limb, even though some degree of function remains; a phenomenon known as
Following stroke and other forms of central nervous system injury in humans, a component of the deficit observed occurs as a result of the learned suppression of movement in addition to the brain injury itself. 7 Although such compensatory strategies can be advantageous, stroke patients who develop learned nonuse may have less, or slower, spontaneous recovery and may not benefit as much from rehabilitation as those who do not develop learned nonuse. 7 Constraint-induced movement therapy involves restraining the ipsilateral arm to encourage use of the contralateral arm, and it has been well established that it improves upper-extremity deficits in stroke patients, in part by overcoming the effects of learned nonuse. 8
There are striking similarities between rodent and human reaching movements,9,10 and rats exhibit persistent reaching impairments and aspects of learned nonuse as in human stroke. 11 Jones and colleagues11-13 trained rats to retrieve food reward pellets with the ipsilateral forelimb following cortical injury and found both worsened motor function of the paretic forelimb and decreased responsiveness to subsequent rehabilitative training, as described in humans. Overreliance on the ipsilateral forelimb, as occurs in learned nonuse, may contribute to maladaptive plasticity that disrupts recovery of the impaired forelimb.11-13 Several issues warrant further investigation, however, such as the timing and persistence of forelimb training effects. 13
Thus, the purpose of this study was to evaluate the persistence of forelimb deficits following ipsilateral forelimb training after stroke in rats. We modified the training paradigm described by Jones and colleagues11-13 in an attempt to produce chronic upper-limb deficits as occurs in stroke patients. 2 A model of chronic learned nonuse in rats would be valuable for further study of the maladaptive plasticity that occurs with learned nonuse and in optimizing treatments designed to overcome it.
Methods
Participants
A total of 32 adult (~3 months old) male Sprague Dawley rats (Charles River Laboratories, Quebec, Canada) weighing ~300 g at the time of surgery were used in this study. Rats were housed in pairs in standard PLEXIGLAS cages on a 12-hour light/dark cycle (lights on at 07:00), and behavioral training and testing occurred during the light phase. All animals were handled daily for 3 days prior to behavioral training and accessed food and water ad libitum unless otherwise stated. All experimental procedures were approved by the Memorial University Animal Care Committee, conformed to the Canadian Council on Animal Care guidelines, and were conducted by experimenters blinded to group identity.
Behavioral Training
Staircase skilled-reaching task
Animals were food restricted to ~90% of their free-feeding weight 2 weeks prior to surgery and trained to retrieve food reward pellets (45-mg food pellets; TestDiet, Richmond, IN) in the staircase-reaching task. 14 Training consisted of two 15-minute trials per day separated by 4 to 5 hours over 10 days. 15 The staircase apparatus contains 14 steps (7 on each side) with a central platform between staircases (Figure 1A). Rats retrieve food reward pellets (3 pellets per step; maximum of 21 pellets/side) on the left staircase only with the left forelimb and pellets on the right staircase with only the right forelimb. The number of pellets consumed on each side was recorded as a measure of independent forelimb reaching ability, and the average number of pellets retrieved during the last 8 training trials was taken to indicate baseline performance. The exclusion criterion consisted of rats obtaining at least 12 pellets out of a possible 21 (±standard deviation of 2 pellets) with the preferred limb over the last 8 training trials. 16 The limb that rats obtained the most pellets with during training was referred to as the “preferred limb”. On average, rats retrieved approximately 17 pellets with the preferred limb, and none were excluded at this time.
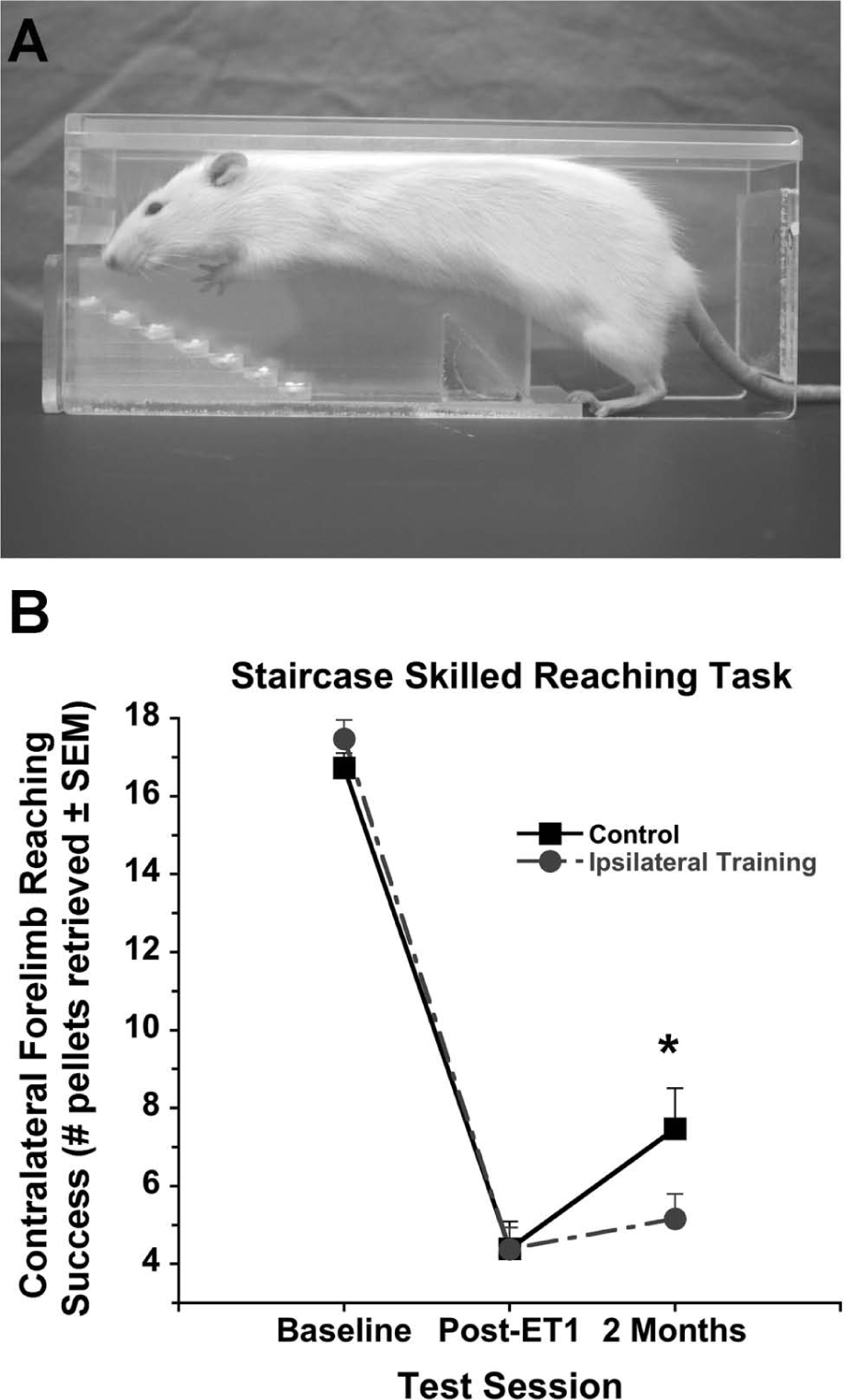
Rats retrieved food rewards pellets in the staircase test (A). Performance was similar at baseline and 5 days following focal ischemia. At 2 months, rats that received training with the ipsilateral forelimb consumed significantly fewer pellets with the contralateral forelimb compared with controls (B).
Cylinder forelimb-use asymmetry task
Rats were placed in a clear PLEXIGLAS cylinder (diameter = 20 cm; height = 35 cm; Figure 2A), and spontaneous exploration was videotaped from below. For each rat, recording took place until at least 20 forelimb contacts were made with the cylinder wall (~5 minutes). 17 The number of ipsilateral, contralateral, and bilateral forelimb contacts was recorded, and percentage contralateral forelimb use was calculated as follows15,18:
Percentage contralateral forelimb use = [(Number of contralateral contacts + ½ Bilateral contacts)/Total contacts] × 100.
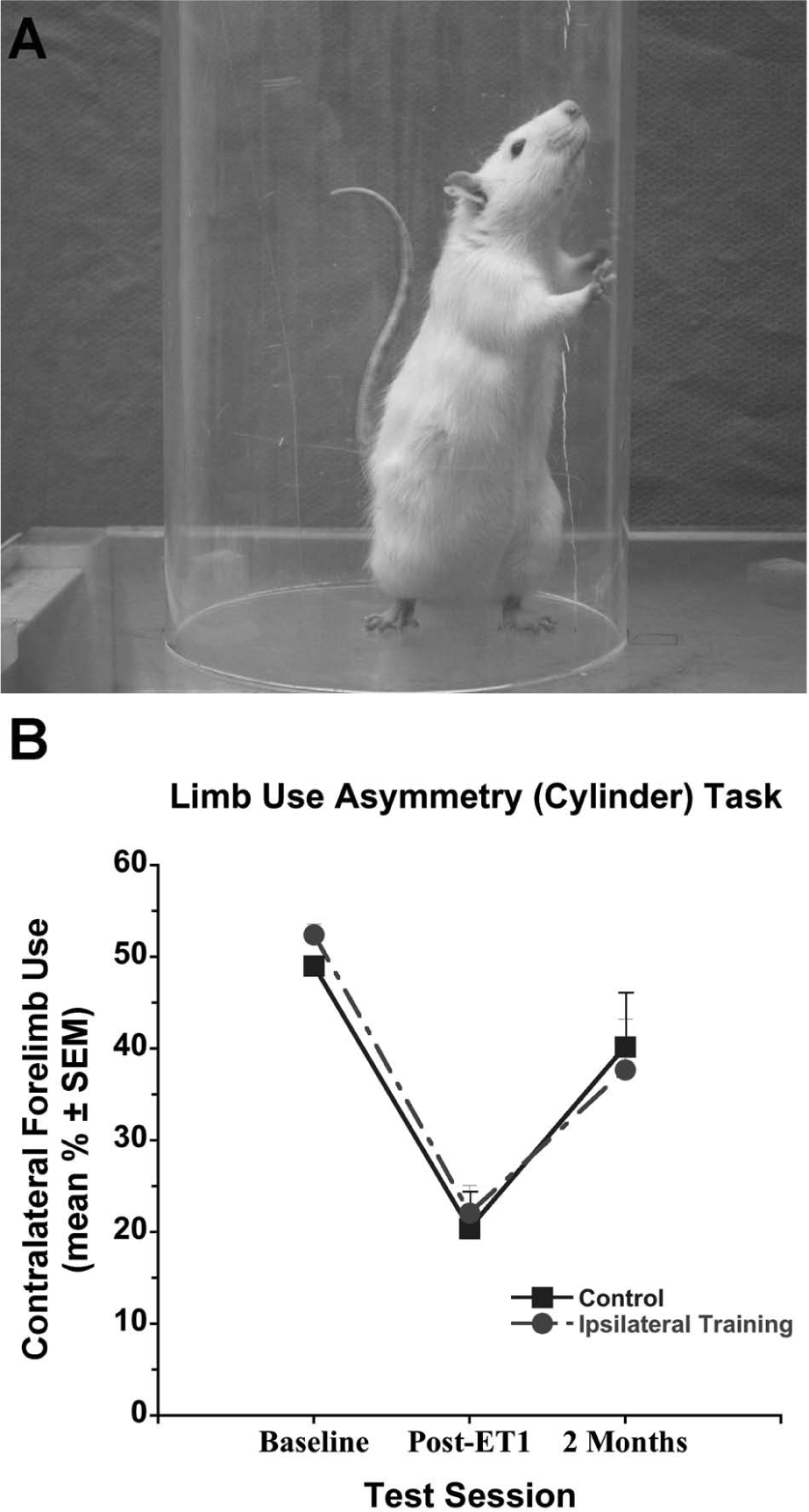
Spontaneous forelimb use was assessed in the cylinder task (A). Both groups used the contralateral forelimb to explore the cylinder walls similarly at all time points (B). Abbreviations: SEM, standard error of the mean; ET, endothelin-1.
Focal Ischemia
Rats were anesthetized with isoflurane (4% induction, 2% maintenance in 30% O2 and 70% N2O) and placed in a stereotaxic frame. A local anesthetic (Marcaine, Sanofi; Markham, ON, Canada) was injected under the scalp ~5 minutes before a midline incision was made. Using aseptic technique, a small burr hole (~1 mm diameter) was made at each of the 3 injection sites in the hemisphere opposite to the rat’s preferred forelimb as determined in staircase training (ie, baseline performance 17 ). The vasoconstrictive peptide endothelin-1 (ET-1, 400 pmol/µL in H2O; CalBiochem, La Jolla, CA) was injected into 2 regions of the forelimb motor cortex (2 µL per site) and dorsolateral striatum (1 µL) at the following coordinates relative to bregma.19,20 The dorsoventral (DV) coordinates are from the surface of the skull:
Cortex 1: AP (anterior-posterior): 0.0 mm; ML (medial-lateral): ±2.5 mm; DV: −2.6 mm
Cortex 2: AP: +2.3 mm; ML: ±2.5 mm; DV: −2.6 mm
Striatum: AP: +0.7 mm; ML: ±3.8 mm; DV: −7.3 mm
The forelimb region of the motor cortex and dorsolateral striatum were targeted because ET-1-induced injury to these areas results in sensorimotor impairments (ie, in skilled reaching, balance, and limb use), and this model can therefore be used to study recovery of function. 19 At each site, ET-1 was infused at a rate of 1 µL over 2 minutes, and the needle was left in place for an additional 3 minutes to avoid back flow. The wound was sutured and a topical anesthetic (1.0% Xylocaine gel) and antibiotic ointment were applied. Body temperature was maintained near normothermia (~37°C) using a rectal temperature probe and electric heating blanket (Harvard Apparatus, Holliston, MA).
Postoperative Behavioral Testing
Performance was assessed in the staircase task (4 test trials over 2 days) and cylinder task (1 test session) on days 5 and 6 postischemia. Rats with minor impairments in the staircase task (ie, >65% of baseline performance; n = 2) were excluded from the study. Our experience suggests that this occurs because of ineffective ET-1 lesions. To ensure similar degrees of reaching deficits between groups prior to intervention and to avoid separating cage mates, pairs of rats were pseudorandomized into treatment condition as described below. 17
Treatment Condition
Beginning 7 days after ischemia, half of the rats (ipsilateral training, n = 15) were placed in the tray-reaching apparatus (length = 20 cm; width = 15 cm; height = 15 cm) and trained to reach for food reward pellets with their ipsilateral forelimb for 2 h/d over 20 days (Figure 3B). Briefly, rats reached through a 1-cm wide opening to retrieve food pellets from a tray (~5 cm from the floor) placed in front of the opening. A clear PLEXIGLAS wall was placed near the opening on 1 side to allow use of only the ipsilateral forelimb for reaching. 17 The 2-hour duration was chosen from pilot work that showed that rats were satiated (displayed little or no reaching attempts) after 1.5 to 2 hours of reaching with the ipsilateral forelimb. During the training session, reaching attempts decline over the 2 hours, but trays were refilled with reward pellets every 10 minutes to encourage rats to continue reaching. All rats had free access to food and water, and rats in the control group (control; n = 15) remained in their standard cages and were given an equivalent quantity of food reward pellets each day (~12 g/rat). Following this period, all rats remained in their home cages for an additional 4 weeks. See Figure 3A for timeline of experimental procedures.
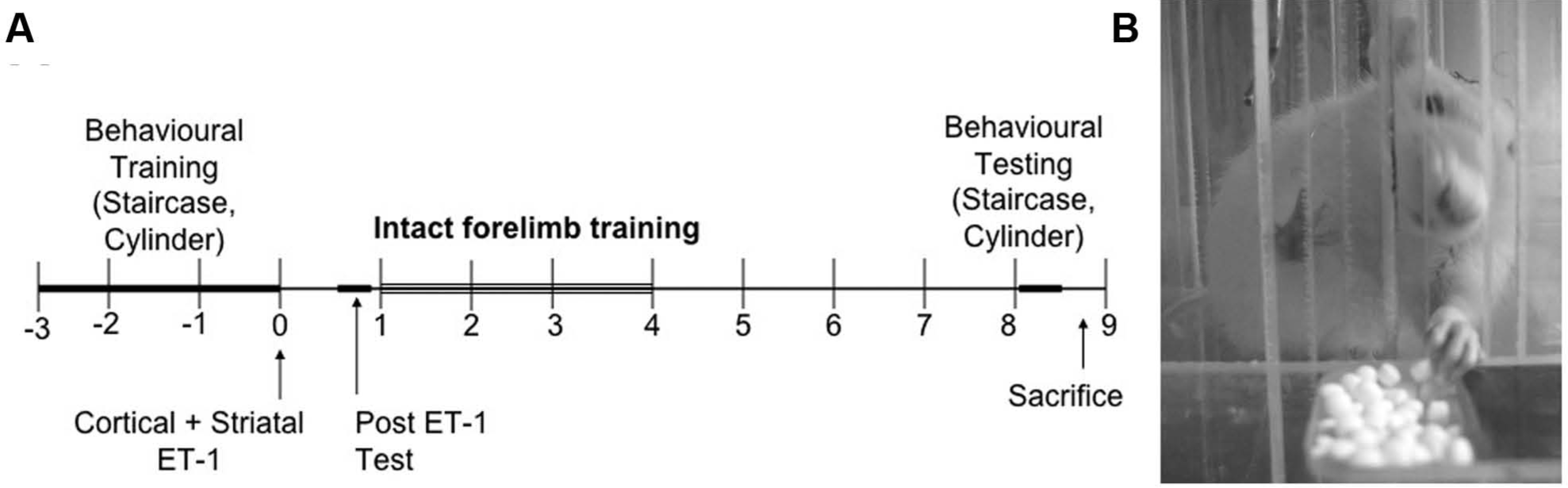
Timeline of experimental procedures in weeks relative to endothelin-1 (ET-1) focal ischemia (A). Half of the rats were trained to reach for food reward pellets in the tray task with the ipsilateral forelimb 2 h/d over 20 days (B).
Functional Outcome
Functional outcome was assessed in the staircase and cylinder tasks 2 months following ischemia. Testing was delayed for approximately 1 month after the end of ipsilateral forelimb training to determine whether effects of ipsilateral forelimb training were long-lasting. Earlier testing was not done in order to prevent possible rehabilitative effects of testing on the contralateral forelimb. All rats were tested over 2 days (2 trials/d as described above) in the staircase task and once in the cylinder task. Food reward pellets were available on both sides of the staircase during testing.
Volume of Brain Injury
Following the last behavioral assessment (~2 months postischemia), rats were anesthetized with 4% isoflurane and transcardially perfused with ice-cold 0.9% heparinized saline and 4% paraformaldehyde. Brains were immediately extracted from the skull, postfixed in 4% paraformaldehyde for 90 minutes, and then placed in 20% sucrose in phosphate-buffered saline until saturated. Brains were frozen and 40-µm thick coronal sections were cut using a cryostat. Sections were slide mounted and stained with cresyl violet. Brain injury was assessed from at least 8 sections per brain spaced 320 µm apart. An average of 8 to 10 sections were used per brain, and this encompassed sections at both the rostral and caudal extent of the lesion that had negligible damage. Volume of tissue lost was determined using Scion Image J (downloaded from the public domain: National Institutes of Health, USA, http://rsb.info.nih.gov/ij/) according to the following formulae 21 :
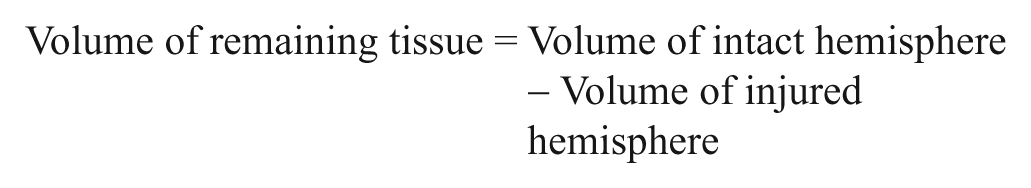
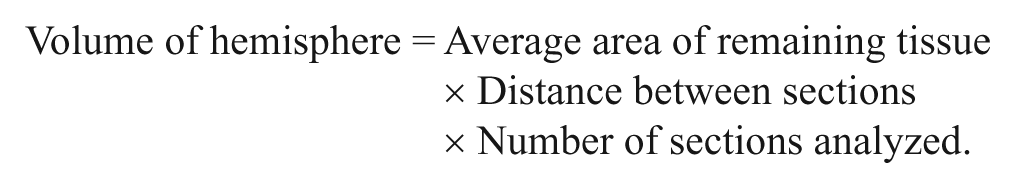
Statistical Analysis
Data were analyzed using the Statistical Package for Social Sciences (IBM SPSS Statistics; v 19.0.0; Armonk, NY) and are presented as mean ± standard error of the mean. Behavioral data (staircase and cylinder tasks) were analyzed using a repeated-measures ANOVA. In the case of a significant interaction, data were further analyzed with independent-samples
Results
Volume of Tissue Lost
Focal ischemia resulted in injury to the forelimb motor cortex and dorsolateral striatum. Representative photomicrographs of injury are presented in Figure 4A. The volume of tissue lost did not differ between the ipsilateral training and control groups (63.60 ± 13.2 mm3 and 68.5 ± 13.4 mm3, respectively;
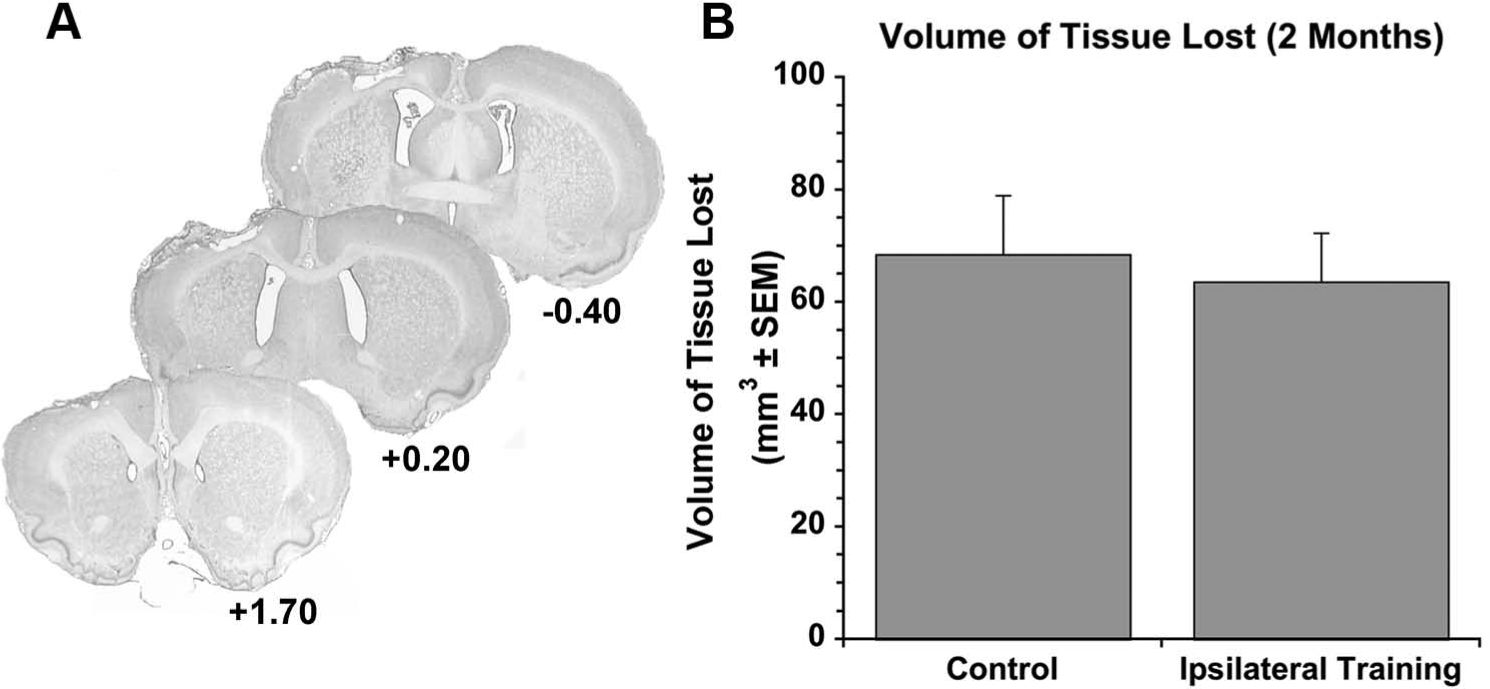
Endothelin-1 was injected into the forelimb motor cortex and dorsolateral striatum to create focal ischemia (A). The number below each photomicrograph corresponds to the bregma level. The volume of tissue lost at 2 months (~66 mm3) did not differ between groups (B).
Staircase Skilled-Reaching Task
Two rats did not exhibit contralateral forelimb skilled-reaching deficits after ischemia and were therefore excluded from the study. Groups performed similarly at baseline (overall mean = 17.1 ± 0.43 pellets per session;
Forelimb Use Asymmetry (Cylinder) Task
Two additional rats (1 from each group) displayed very little cylinder exploration at 2 months (fewer than 5 independent wall touches) and were therefore excluded from the repeated-measures analysis. The repeated-measures analysis revealed a significant effect of time (
Discussion
Training rats to retrieve food reward pellets with the ipsilateral forelimb decreased reaching success with the contralateral forelimb 2 months following stroke. This effect cannot be explained by differences in the volume of tissue lost. These results support growing evidence that ipsilateral forelimb training impedes learning a novel skilled-reaching task and impedes recovery of skilled reaching after stroke-like injury.11 -13 Here, we show long-term deficits in skilled-reaching performance and propose that ipsilateral forelimb training results in maladaptive brain plasticity and in learning an overreliance on the ipsilateral forelimb for reaching activities. Thus, we suggest the paradigm described here be used to model learned nonuse in rats.
The present findings build on existing literature in several ways. First, this ipsilateral forelimb training paradigm (2 h/d over 20 days) was more intense and prolonged than other paradigms used by Jones and colleagues. Rats retrieved approximately 12 g of food reward pellets per day, which translates to at least 275 successful reaches. We hypothesized that more intense, prolonged ipsilateral forelimb training would result in significant and long-term impairments of the contralateral forelimb and more accurately model hand deficits and learned nonuse found in stroke patients. Indeed, human stroke survivors generally have poor recovery of hand motor function after motor cortex injury 22 and may learn to rely on the less-affected hand to accomplish activities of daily living (learned nonuse). Furthermore, evidence suggests that the intensity of reaching rehabilitation critically determines rehabilitation efficacy. A recent study showed that intense, enriched rehabilitation was associated with greater functional recovery and brain-derived neurotrophic factor after stroke in rats compared with less-intense or no rehabilitation. 15 Clinical data also suggest that more intense rehabilitation provides the greatest benefit for stroke survivors.23-26
In other paradigms, rats received 10 minutes of ipsilateral forelimb training (or 60 trials; whichever came first) over 10 to 15 days. They then received rehabilitative training of their contralateral limb in a reaching task for an additional 10 days. Studies suggest worsened performance of the contralateral forelimb.11-13 However, by the end of rehabilitative training, significant reaching deficits are not always detected (eg, Allred and Jones 13 ), and it is not known whether skilled deficits exist beyond this time period. Together, these results suggest that the intensity of training is important and may contribute to long-term deficits such as ours, which were detected 1 month following the end of the training period. It is possible that intense training resulted in even larger deficits immediately after the training period. The goal of the current study, however, was to detect long-term (ie, 2 months poststroke) effects of the training period, and we therefore did not assess animals at earlier time points in order to prevent possible rehabilitative effects of testing on the contralateral forelimb. Further rehabilitative training with the contralateral forelimb following our ipsilateral forelimb training protocol would likely improve reaching success of the contralateral forelimb.
The timing of training also appears to be an important determinant of maladaptive plasticity and development of learned nonuse. There is evidence for a critical window after stroke where the brain is most sensitive to the effects of rehabilitation in the early postinjury period.16,27 It follows, then, that the brain may also be most sensitive to the maladaptive effects of training the less-affected limb soon after stroke. Jones and colleagues trained the ipsilateral limb starting 6 days after brain injury,11-13 and the training began 7 days after ischemia in the current study. The lasting detrimental effects on contralateral skilled-reaching performance suggest that experience during the first week after injury critically affects functional outcome. Erickson and colleagues 28 demonstrated decreased reaching attempts during the acute (first few days) postsurgical phase but not after the first week, suggesting that learned nonuse develops early after stroke. Monkeys and humans learn to suppress movement of the contralateral limb (despite the fact that it is not paralyzed) if they are allowed to use the ipsilateral limb for compensatory movements.6,7,29 Thus, experimental and clinical studies together suggest that early poststroke experience has lasting effects on brain reorganization and functional recovery.
Training rats to reach for reward pellets with the ipsilateral forelimb in the tray-reaching task resulted in contralateral forelimb skilled-reaching deficits in the staircase-reaching task 2 months poststroke. This generalization of reaching deficits across skilled-reaching tasks has not previously been shown. However, we did not detect decreased spontaneous use of the contralateral forelimb in the cylinder task at 2 months poststroke. There are several possible explanations for this finding. First, we delayed assessing spontaneous limb use until 1 month after the end of ipsilateral forelimb training, during which time quadripedal rats used the contralateral forelimb for weight bearing and postural support during cage exploration and for other activities such as grooming, manipulating food, or exploring the home cage, which were not assessed. There may have been a transient effect on spontaneous limb use that recovered by the 2-month test session.
There are several limitations to this work. First, although we demonstrated decreased recovery in skilled-reaching tasks as a result of ipsilateral limb training, we did not measure the intensity or duration of limb use to isolate whether decreased success is a result of motor dysfunction (eg, impairments in grasping), decreased motivation, or fewer reaching attempts or whether longer test session duration (eg, 30 vs 15 minutes) would result in increased contralateral forelimb success. A detailed qualitative analysis of performance in the staircase task 30 could be done to clarify this issue. However, Jones and colleagues presented reaching success per attempt and showed that impairments did not depend on the number of reaches. Additionally, ipsilateral forelimb-reaching success at 2 months (~90% of baseline in each group) indicates that rats are highly motivated to reach for reward pellets. An additional limitation is that we did not evaluate mechanisms of maladaptive plasticity and learned nonuse in this model. Other studies discussed above have used smaller injury to the forelimb areas of the motor cortex. The somatosensory cortex of the contralesional hemisphere and transcallosal projections appear to be components of the neural circuitry involved in the maladaptive effects of training the less-affected forelimb in those studies. 11 Reduced FosB/ΔFosB neuronal density in the peri-infarct cortex has also been found. 13 In the current study, we produced larger injury to the forelimb motor cortex and the dorsolateral striatum. We chose this stroke model because it results in significant and long-term reaching deficits as well as some spontaneous skilled-reaching recovery (performance increases by 15%-25% of baseline performance). 15 These were important components of the model because we sought to determine whether ipsilateral limb training would block this recovery, and the presence of lasting deficits permits testing therapeutic interventions. The neural mechanisms associated with worsened behavioral recovery may differ among models. For example, the contralesional cortex may play a greater role in recovery than the peri-infarct cortex in rats with this large lesion. 31 Further research is needed to explore these possibilities.
The training paradigm described here may be used to further explore the neural mechanisms of maladaptive plasticity and learned nonuse, such as the role of pericortical structures and distal areas such as the cerebellum and contralesional hemisphere in recovery, and changes to metabolism or blood flow. It can also be used to evaluate potential pharmacological or rehabilitative treatments to overcome learned nonuse as well as noninvasive brain stimulation. The long-term deficits demonstrated here make it an ideal model to evaluate features of constraint-induced movement therapy in chronic stroke, such as the optimal duration or intensity of training, complexity of training tasks, and the effects of bimanual versus unimanual training on recovery.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by a grant (to D. Corbett) from the Canadian Institutes for Health Research (CIHR). C. L. MacLellan was supported by a CIHR postdoctoral fellowship. S. Butt and A. Botsford were supported by undergraduate research awards from Memorial University of Newfoundland and Labrador.
