Abstract
Background. The relative effect of a transcutaneous peroneal nerve stimulator (tPNS) and an ankle foot orthosis (AFO) on spatiotemporal, kinematic, and kinetic parameters of hemiparetic gait has not been well described. Objective. To compare the relative neuroprosthetic effect of a tPNS with the orthotic effect of an AFO using quantitative gait analysis (QGA). Design. In all, 12 stroke survivors underwent QGA under 3 device conditions: (1) no device (ND), (2) AFO, and (3) tPNS. A series of repeated-measures analyses of variance (rmANOVAs) were performed with dorsiflexion status (presence or absence of volitional dorsiflexion) as a covariate to compare selected spatiotemporal, kinematic, and kinetic parameters for each device condition. Post hoc pairwise comparisons and/or subset analysis by dorsiflexion status were performed for significant effect. Results. Stride length was improved with both the AFO (P = .035) and the tPNS (P = .029) relative to ND. Those with absent dorsiflexion had longer stride length with the tPNS relative to ND (P = .034) and a higher walking velocity with a tPNS relative to the AFO (P = .015). There was no device effect on dorsiflexion angle at initial contact; however, a significant Device × Dorsiflexion status interaction effect favored the AFO relative to ND (P = .025) in those with dorsiflexion present. Conclusion. This study suggests that level of motor impairment may influence the relative effects of the tPNS and AFO devices in chronic hemiparetic gait; however, the small sample size limits generalizability. Future studies are necessary to determine if motor impairment level should be considered in the clinical prescription of these devices.
Introduction
The neuroprosthetic effect of a transcutaneous peroneal nerve stimulator (tPNS)1-4 and the orthotic effect of an ankle foot orthosis (AFO) 5 on various gait parameters in hemiparetic gait have been previously described. However, although the tPNS is a clinically accepted alternative device, the rehabilitation intervention considered standard of care for poststroke footdrop in the United States remains the AFO.1,2 A paradigm shift in stroke rehabilitation clinical care requires evidence-based research demonstrating, at a minimum, an equivalency of effect between the 2 devices. The few studies that have directly compared the neuroprosthetic effect of a tPNS with the orthotic effect of an AFO suggest equivalency in walking speed,6-9 activity level, 9 and functional mobility 6 and superiority of the tPNS in balance control 7 and device satisfaction. 9
The relative effect of a tPNS and an AFO on spatiotemporal, kinetic, and kinematic parameters of hemiparetic gait using quantitative gait analysis (QGA) has not been well described in the literature. Voigt and Sinkjaer 10 presented a case series of 8 hemiparetic individuals with stroke, comparing kinematic and kinetic gait parameters with and without a tPNS device but did not compare performance with an AFO. Van Swigchem et al 11 presented a single case study of within-subject AFO, transcutaneous, and implanted PNS device performance in an individual who had not tolerated a tPNS device because of skin breakdown. Kottink et al 12 randomized participants to receive either an implantable PNS device or a standard-of-care intervention (defined as either an AFO, shoe, or no device [ND]). Baseline and end-of-treatment spatiotemporal and kinematic gait parameters were compared, but neither relative device effects, using an intrasubject study design, nor kinetic parameters were studied. Second, spatiotemporal, kinematic, and kinetic analysis of an implantable PNS device performance would not necessarily be expected to yield the same results as tPNS performance because of variability of placement (stimulating electrode, heel switch, or tilt sensor) and specificity of muscle stimulation between devices. Stroke rehabilitation physicians in the United States do not have the clinical option of having their patient implanted with a PNS device because the device is not currently FDA approved; thus, the relative effect of an implantable PNS device in comparison with a standard-of-care device is less clinically relevant. Presently, there are 3 FDA-approved tPNS devices being prescribed by physicians providing clinical care to stroke rehabilitation patients in the United States, yet the relative effect of a tPNS device and AFO is not well understood for any given patient.
Our primary study hypothesis was that the tPNS and AFO devices would have equivalent effects on the selected spatiotemporal gait parameters (walking velocity, stride length, cadence, and double support time). Our second hypothesis was that ankle dorsiflexion angle at initial contact (DFIC) would be improved by both the tPNS and AFO devices but that peak dorsiflexion, knee flexion, and hip flexion during swing would be greater in the tPNS device condition because of a lower extremity flexion withdrawal response 13 elicited specifically by the tPNS. Our third hypothesis was that peak ankle power at push-off would be less in the AFO device condition, relative to the tPNS device condition, because of inhibition of active ankle movement by the AFO.
Methods
Study Design
This case series evaluated 12 chronic hemiparetic stroke survivors who were participating in a randomized controlled trial (RCT), which compared the lower-limb motor relearning effect of a tPNS with usual care treatment. The 110 participants enrolled in the RCT were stratified by motor impairment level as evidenced by the presence (≥1/5 on the Medical Research Council [MRC] scale) or absence (0/5, MRC) of volitional ankle dorsiflexion while in the non–weight-bearing state, prior to being randomized to either a tPNS or usual care group. In all, 54 were randomized to the tPNS group, and 56 were randomized to usual care. The individuals then participated in a 12-week ambulation training treatment period using the assigned device in their home and community setting for up to 8 h/d. All those randomized to the tPNS group who had used an AFO prior to enrollment in the RCT were offered the opportunity to participate. This present study evaluated the 12 individuals in the tPNS group who met that criterion and agreed to participate in this additional analysis. These individuals underwent 1 QGA session near the end of the 12-week tPNS treatment period under the 3 device conditions, in the same device order (ND, AFO, and tPNS).
Participants
The protocol of the larger RCT included this optional protocol, which was available to individuals randomized to the tPNS group only and was approved by the institutional review boards of the involved academic medical centers. Each participant gave prior written consent for participation. All participants were ≥18 years of age, >3 months poststroke, and medically stable. They demonstrated unilateral hemiparesis with ankle dorsiflexion strength of no greater than 4/5 on the MRC scale. Each individual demonstrated dorsiflexion weakness during ambulation, such that gait instability or inefficient gait patterns were exhibited. Each one was required to ambulate 10 m with minimal assistance or less and scored ≥24 on the Berg Balance Scale because of safety considerations in performing the QGA. Individuals were excluded for concomitant neurological diagnoses, uncompensated hemineglect, severely impaired cognition and communication, fixed ankle contracture, peroneal nerve injury, genu recurvatum, or history of botulinum toxin injection to the affected lower extremity in the preceding 3 months. Those enrolled in this protocol were close to completing the 12-week treatment period of the RCT, were independently using the PNS device for daily ambulation, and also had in their possession a custom AFO that was being used for correction of footdrop prior to enrollment in the RCT. None of the participants required an ambulatory assist device (ie, cane) for safe participation in the QGA procedure.
Devices
The tPNS device was the Odstock Dropped-Foot Stimulator 2 (Odstock Medical Limited, Salisbury, UK), which is a single-channel device consisting of a 9-V battery-powered stimulator and skin surface electrodes. Stimulation is initiated at preswing with detection of heel rise by the 3-mm insole pressure-sensing footswitch. The participants used their own physician-prescribed AFO that was in their possession prior to entry into the study, and thus, the AFOs were not standardized. Each AFO was custom molded, hinged, and fabricated using conventional, clinically accepted techniques.
Outcome Measures
QGA was performed using a Vicon system (Vicon Motion Systems Limited, Oxford, UK), a motion measurement and analysis system that tracked the trajectories of reflective markers in the field of view of multiple cameras mounted around the periphery of the laboratory. All participants wore low-heeled, laced shoes during the trials. Retroreflective markers were adhered to the skin at anatomic locations following a modified Helen Hayes marker set14,15; the second metatarsal and calcaneal markers were secured directly to the shoe, and thus, all marker positions were consistent between trials. Participants were asked to ambulate 10 m at a self-selected comfortable rate. A minimum of 20 strides (approximately 10 trials) were collected for each device condition. AMTI Biomechanics Platforms (Advanced Mechanical Technology, Inc, Watertown, Massachusetts) were embedded in the walkway of the laboratory. Illumination, motion capture data, and analog to digital conversion of transducer input were synchronized and controlled by the Vicon system, which was in turn controlled by a Pentium-based PC. Data were processed using the Vicon Plug-In-Gait biomechanical model in Vicon supplied to generate joint angles, moments, powers, and spatiotemporal parameters of gait. At completion of the single QGA session, study participation was concluded. For purposes of this analysis, specific spatiotemporal, kinematic, and kinetic gait parameters were identified a priori for evaluation (Table 1). Spatiotemporal parameters (walking speed, stride length, cadence, and double support time) were chosen based on clinical relevance for gait efficiency and symmetry. Kinematic parameters chosen were the following: (1) peak hip flexion, knee flexion, and dorsiflexion during swing to detect a tPNS-induced flexion withdrawal response; (2) DFIC as both a tPNS and AFO affect weight-bearing stability via ankle positioning at early stance; and (3) peak knee extension in stance because an AFO can affect knee extension depending on the degree of dorsiflexion incorporated into its design. Kinetic parameters chosen were the following: (1) knee extensor moment, caused by possible device effect on location of ground reaction force relative to the knee joint; (2) peak hip power, because of its contribution to walking speed and forward propulsion; and (3) peak ankle power, as a result of concern that an AFO may inhibit ankle movement and thus ankle power. All QGA sessions were supervised and performed by the same research physical therapist and gait laboratory engineer using standardized procedures.
Mean Spatiotemporal, Kinematic, and Kinetic Parameters by Device Group (SD).
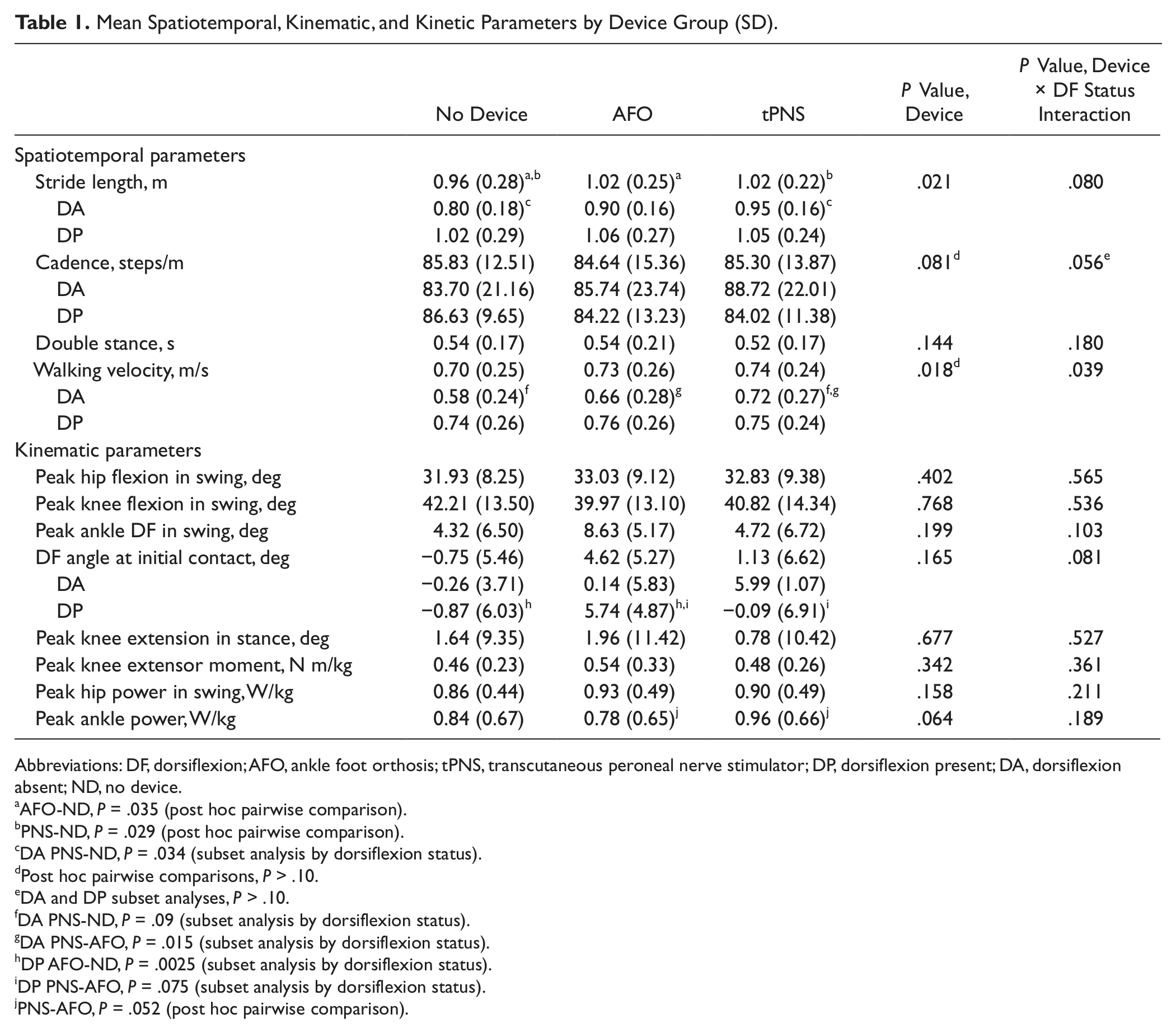
Abbreviations: DF, dorsiflexion; AFO, ankle foot orthosis; tPNS, transcutaneous peroneal nerve stimulator; DP, dorsiflexion present; DA, dorsiflexion absent; ND, no device.
AFO-ND, P = .035 (post hoc pairwise comparison).
PNS-ND, P = .029 (post hoc pairwise comparison).
DA PNS-ND, P = .034 (subset analysis by dorsiflexion status).
Post hoc pairwise comparisons, P > .10.
DA and DP subset analyses, P > .10.
DA PNS-ND, P = .09 (subset analysis by dorsiflexion status).
DA PNS-AFO, P = .015 (subset analysis by dorsiflexion status).
DP AFO-ND, P = .0025 (subset analysis by dorsiflexion status).
DP PNS-AFO, P = .075 (subset analysis by dorsiflexion status).
PNS-AFO, P = .052 (post hoc pairwise comparison).
Statistical Analysis
Demographic data, including age, sex, interval poststroke, stroke etiology, and involved hemisphere, were evaluated for mean, standard deviation, and/or frequency. Repeated-measures analysis of variance (rmANOVA) testing was performed to compare the means of each parameter by device condition (ND, AFO, and PNS) using motor impairment level as evidenced by dorsiflexion status (presence or absence of active dorsiflexion) as a covariate. Post hoc pairwise comparisons were performed if a device effect or Device × Dorsiflexion interaction effect was found. Because of the exploratory nature of this study, post hoc analyses using the least-significant differences method were performed if rmANOVA yielded a P value ≤.10 for device effect or Device × Dorsiflexion interaction effect. A P value ≤.05 was defined as a statistically significant difference for all post hoc analyses.
Results
Demographic data are presented in Table 2. Spatiotemporal, kinematic, and kinetic data for the 3 device conditions, post hoc pairwise comparisons, and subset analyses based on dorsiflexion status are presented in Table 1.
Participant Characteristics, Including Means (Standard Deviation).
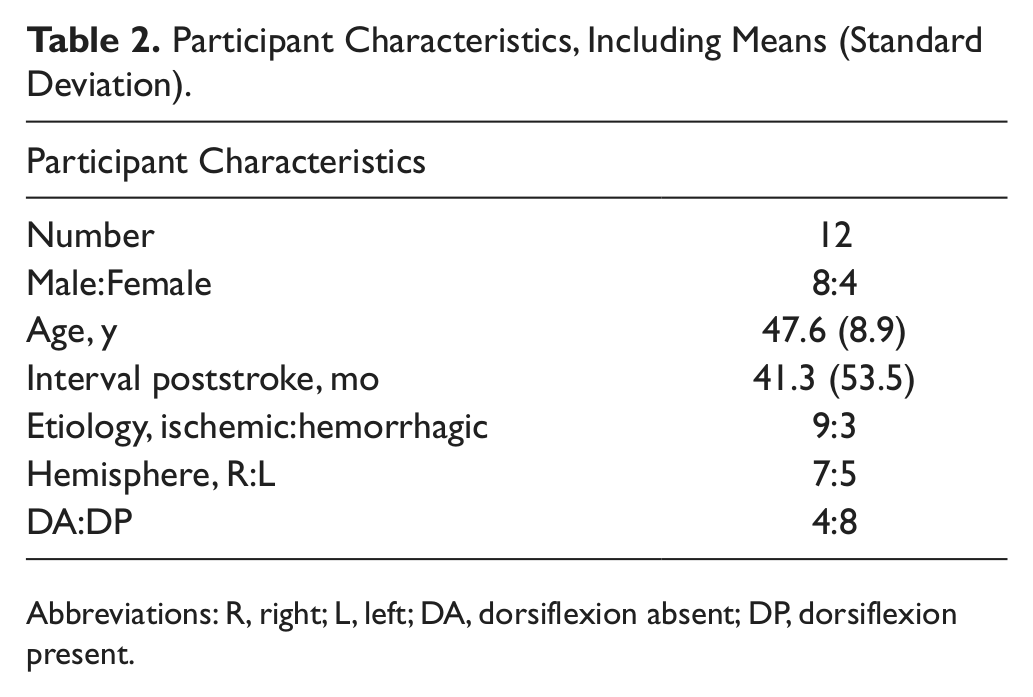
Abbreviations: R, right; L, left; DA, dorsiflexion absent; DP, dorsiflexion present.
Spatiotemporal Parameters
There was a significant difference in stride length—F(2, 18) = 4.812, P = .021—between device conditions; post hoc pairwise comparisons found that stride length was significantly improved with both the AFO (1.02 ± 0.25 m, P = .035) and the tPNS (1.02 ± 0.22 m, P = .029) relative to ND (0.96 ± 0.28 m). An interaction between device and dorsiflexion status on stride length was found, which approached significance—F(2, 18) = 2.910, P = .08—and post hoc analysis showed a significant device effect in favor of PNS (0.95 ± 0.16 m) relative to ND (0.80 ± 0.16 m, P = .034) in those with absent dorsiflexion (DA). There was a significant difference in walking velocity between device conditions—F(2, 18) = 5.092, P = .018; however, post hoc pairwise comparisons failed to detect the differences between conditions. A significant interaction between device and dorsiflexion status on walking velocity was found—F(2, 18) = 3.917, P = .039—and post hoc analysis showed a significant effect in favor of the tPNS (0.72 ± 0.27 m/s) relative to the AFO (0.66 ± 0.28 m/s, P = .015) in those with DA. The difference in cadence between device conditions—F(2, 18) = 2.901, P = .081—and device dorsiflexion interaction effect—F(2, 18) = 3.389, P = .056—both approached significance; however, post hoc pairwise comparisons and subset analysis based on dorsiflexion status were nonsignificant (P > .05). There was no device effect or interaction between device and dorsiflexion status on double support time.
Kinematic Parameters
There was no device effect or interaction between device and dorsiflexion status on peak hip flexion in swing, peak knee flexion in swing, peak dorsiflexion in swing, or peak knee extension in stance. There was no device effect on DFIC; however, there was an interaction between device and dorsiflexion status that approached significance—F(2, 16) = 2.946, P = .081. Post hoc analysis found a significant device effect on DFIC in favor of the AFO (5.74° ± 4.87°) as compared with ND (−0.87° ± 6.03°, P = .025), and statistical significance was approached in favor of the AFO over the PNS in those with dorsiflexion present (DP; P = .075).
Kinetic Parameters
There was no device effect or interaction between device and dorsiflexion status on peak knee extensor moment in stance or peak hip power. The difference in ankle power at push-off between device conditions approached significance—F(2, 18) = 3.217, P = .064—and post hoc pairwise comparisons found a difference that approached significance and favored the tPNS (0.96 ± 0.66 W/kg) over the AFO (0.78 ± 0.65 W/kg, P = .052). There was no interaction between device and dorsiflexion status on peak ankle power at push-off.
Discussion
A primary finding of this analysis is a significant device and dorsiflexion interaction effect on stride length and walking velocity that favored tPNS in those with DA, with an overall equivalent device effect on spatiotemporal parameters when dorsiflexion status was not considered. In this study, 8 of the 12 participants had partial preservation of dorsiflexion, and 4 participants had DA on clinical examination at study enrollment. Participants were presumed to have a higher level of motor impairment if dorsiflexion was absent and a lower level of motor impairment if dorsiflexion was present. For all participants, the significant increase in stride length noted with both the tPNS and AFO, relative to ND, is consistent with both clinical expectation and a prior temporal spatial analysis of the orthotic effect of an AFO. 5 The lack of a significant difference in walking velocities between device conditions, however, is not consistent with previous studies that have demonstrated increases in hemiparetic walking speed, which ranged between 14% and 29%1-3,16 for a tPNS and 32% for an AFO. 5 The lack of a robust effect of either device on walking speed for whole-group analysis may be a result of limitations of the QGA methods. Using QGA, mean walking speeds are determined based on multiple walking trials recorded over a relatively short distance of 10 m while encumbered with multiple retroreflective 3-dimensional markers adhered to key bony prominences from the pelvis to the ankles; therefore, maximal comfortable gait speeds, for any given device condition, may not have been achieved. A more accurate measurement of walking velocity may have been obtained if the 10-m walking velocity had been measured separately and not in the context of the QGA procedure. Regardless of methods of measurement, these findings are generally consistent, however, with the few studies that have attempted to directly compare the effect of a tPNS versus an AFO on walking speeds and found equivalency of effect.6-9 The lack of difference in double support time suggests that neither device significantly affects dynamic balance. The lack of difference in cadence between device conditions, which is not consistent with prior gait analyses by Esquenazi et al 5 and Ring et al, 7 who reported improvement in cadence with an AFO and tPNS device, respectively, may similarly be a result of the relatively short distances ambulated.
The interaction between device and dorsiflexion status is an intriguing finding in this present study, which has not been previously reported. Stride length and walking velocity were significantly enhanced by the tPNS, relative to ND and the AFO, respectively, in the subgroup of participants with DA. A possible explanation for this finding is that the tPNS, as compared with an AFO, provides dorsiflexion assist during swing while not compromising stance phase proprioceptive input (joint position sense, sensation), which may be more important to enhance walking performance in those with a higher level of motor impairment (those with DA). Participants with a lower level of motor impairment (those with DP) may have better proximal motor control and strength for compensation and may thus be less reliant on proprioceptive input for gait. Because spatiotemporal parameters are influenced by walking velocity, future studies should focus specifically on this subgroup of more severely impaired individuals with the goal of measuring device effect while maintaining a constant walking velocity.
In the ND condition, ankle DFIC for the group as a whole and the relative kinematic findings for the DA and DP groups are notable. The goal of both a tPNS and AFO device is to correct for dorsiflexion weakness during the swing phase of gait and maintain the ankle at neutral (90°) at initial contact, at a minimum, so as to stabilize the ankle and facilitate gait progression throughout the weight-bearing (stance) phase of the gait cycle. Thus, ankle DFIC may be the single most important kinematic parameter to examine for effect. In this study, the mean ankle dorsiflexion angle at initial contact of −0.75 ± 5.46 is greater than might be anticipated in a ND condition for the enrolled participants. This ND finding may be a result of the fact that individuals with varying degrees of dorsiflexion weakness (dorsiflexion strength range ≤4/5 on MRC) were enrolled. In clinical practice, those with mild dorsiflexion weakness may have sufficient strength to achieve dorsiflexion at initial contact positioning close to neutral as measured by QGA, yet still clinically manifest footdrop during the swing phase of gait. In terms of DA versus DP performance, the 2 ankle kinematic parameters that might be expected to most dramatically differentiate those with DA from those with DP are peak ankle dorsiflexion in swing and DFIC in the ND condition. However, the lack of an interaction between device and dorsiflexion status for peak ankle dorsiflexion in swing and review of the raw data for both kinematic parameters suggest that these data points were not significantly affected by participant designation as either DA or DP. At entry to the larger RCT, the presence or absence of dorsiflexion was determined with the participant seated in a non–weight-bearing fashion and with the lower leg extended. Clinically, this is the most common position from which a clinician measures dorsiflexion strength and range of motion. It is likely that many of the participants who exhibited 0/5 dorsiflexion strength when measured statically manifested some degree of dynamic dorsiflexion during gait associated with both tone and proximal knee and hip flexion. Thus, dorsiflexion strength and active ankle range of motion were likely different depending on whether the parameters were measured while not bearing weight in the context of a clinical examination or dynamically during gait as measured by QGA. The larger RCT found that the mean baseline lower-extremity Fugl-Meyer scores (a standardized measure of motor impairment) for the DP group were significantly higher than those for the DA group as a whole. Thus, designation of the participants as either DP or DA based on clinical exam reasonably designated them into higher- and lower-level impairment groups.
A secondary finding is no device effect on the kinematic parameters when dorsiflexion status was not considered, yet a significant device and dorsiflexion interaction effect on ankle DFIC, which favored an AFO in those with DP. A significant improvement in ankle DFIC in the subgroup of participants with DP, and not the DA group, is likely a result of better ankle positioning for weight bearing associated with not simply the rigidity of a plastic AFO but also greater proximal lower-extremity strength in this less-impaired subgroup. The inability of the tPNS to achieve DFIC of a similar magnitude, irrespective of dorsiflexion status, may theoretically be explained by inadequate positioning of the stimulating electrode over the common peroneal nerve, suboptimal stimulus parameters (frequency, amplitude, and pulse width), or lack of precision of the timing of stimulus initiation as a result of footswitch placement. However, tPNS device setup is unlikely to be contributory because stimulation parameter settings and electrode placement were optimized by the research therapist prior to the commencement of each QGA session. The lack of significant DFIC enhancement in the tPNS device condition may also be a result of muscle fatigue of the anterior tibialis associated with device testing order (ND, AFO, tPNS). Muscle fatigue may be an even greater limiting factor in the DP group because of the effect of repetitive volitional muscle contraction. Alternatively, the lack of effect of tPNS on DFIC may reflect limitations in the sensitivity of the QGA software and procedure itself. The DFIC angle is recorded at the moment when the vertical ground reaction force passes a 10 Newton threshold, whereas the tPNS stimulation turns off at the moment weight bearing is sensed by the heel switch. A subtle mismatch of these signals, despite optimal device setup, could result in the QGA underreporting the actual maximal DFIC in all participants.
A third finding in this study is the lack of evidence of a tPNS-induced flexion withdrawal response in the kinematic data. A lower-extremity flexion withdrawal response is a spinal reflex consisting of a widespread contraction of flexor muscles and simultaneous relaxation of extensor muscles. Prior clinical studies have suggested that clearance of a hemiparetic lower limb during the swing phase of gait may be enhanced by a flexion withdrawal reflex elicited by peroneal nerve stimulation,13,17-19 though no previous study has attempted to measure this response to tPNS using QGA. Interestingly, our results are in accord with a recent study by Kottink et al, 12 who found no evidence that an implantable PNS device triggered hip and knee flexion, and at odds with a single case report by Van Swigchem, 20 who did report an implantable PNS device–induced lower-extremity flexion withdrawal response. In the construct of this present study, a tPNS flexion withdrawal reflex would be evident by an increase in paretic peak hip flexion, peak knee flexion, and peak ankle dorsiflexion angle during swing, relative to the ND and AFO trials. Our data suggest that the neuroprosthetic application of tPNS does not elicit a lower-extremity flexion withdrawal response that is either measurable by QGA or of clinical significance. The magnitude of peak hip, knee, and ankle flexion during swing in the ND condition suggests that these individuals as a whole, although they are the appropriate clinical population for the PNS device, may have lesser lower-extremity motor impairment deficits than the broader hemiparetic stroke population and thus a lesser margin for enhancement of these parameters. Other possible explanations include inadequate tPNS-induced dorsiflexion torque; loss of range of motion at the paretic ankle, knee, and hip; or paretic extremity extensor spasticity. An important consideration is that stimulation of the peroneal nerve at the level of the fibular head activates both the anterior tibialis and peronei musculature, meaning that the PNS device corrects for footdrop by eliciting not simply ankle dorsiflexion during swing but also ankle eversion. DFIC and peak ankle dorsiflexion in swing, measured in a sagittal plane, were chosen a priori as kinematic parameters, which should theoretically be improved with a PNS, but our study methods may have been insufficient to demonstrate the full neuroprosthetic effect associated with ankle eversion measured in the coronal plane. The lack of significant difference between peak dorsiflexion in swing tPNS and ND conditions, in the setting of optimal device functioning, suggests the importance of quantifying PNS effect on ankle eversion as well as dorsiflexion to better characterize the neuroprosthetic effect of the device.
Finally, a trend toward significance in ankle power at push-off between a tPNS and AFO in the group as a whole is notable because of a clinical concern that inhibition of ankle movement by an AFO may enhance weight-bearing stability in hemiparesis but at the cost of loss of ankle power necessary for optimal walking speeds 21 and forward gait progression. Prior studies of healthy adults have found that an AFO decreases ankle power during both treadmill ambulation 22 and stair locomotion. 23 In our study, all participants utilized a hinged AFO, which would theoretically diminish ankle power to a lesser degree 23 than a solid AFO. A possible explanation for greater ankle power at push-off of the tPNS relative to the AFO is that repetitive dorsiflexion contraction with the tPNS during ambulation may facilitate reciprocal strengthening or functioning of the gastroc-soleus complex.
Limitations of this study are primarily related to study methods. QGA is ideally performed barefoot with the reflective markers applied directly to the skin overlying prominent anatomic bony landmarks. Neither an AFO nor a tPNS device can be used in the absence of the supporting structure of a shoe; thus, for all trials (ND, AFO, and tPNS), QGA was performed with the participant wearing a shoe and the calcaneal and second metatarsal markers adhered directly to the shoe. In approximately half of the participants, the lateral malleolus was obscured by the AFO, which necessitated applying that reflective marker directly on the lateral portion of the AFO over the malleolus. Prior to each of the 3 gait analysis sessions, static trials, which included measurement of ankle joint width, were performed, so that the gait analysis software should theoretically have corrected for any between-device discrepancies in marker placement. Second, all participants were evaluated in the same device order (ND, AFO, tPNS) because of a concern that ambulation with the tPNS device may enhance gait performance for subsequent trials as a result of a carryover effect. The trade-off of this device sequence was that the tPNS gait performance may have been more affected by fatigue as compared with the ND and AFO trials. Of note, any between-subject nonuniformity of the AFOs evaluated in the study, although not ideal, should not have affected results, given the within-subject statistical analysis of the 3 device conditions. Finally, in this pilot study, post hoc pairwise comparisons were performed with least-significant differences in an attempt to avoid a type II error in data analysis, and interpretation of the subset analysis was limited because of small group numbers. As a result, any findings regarding relative effects of an AFO and tPNS should be confirmed in a larger RCT.
In conclusion, this study suggests equivalency of the effect of a tPNS or an AFO on spatiotemporal, kinematic, and kinetic parameters of chronic hemiparetic gait. The level of motor impairment, however, may influence the relative effects of the tPNS and AFO devices. Future studies are indicated to determine if level of motor impairment status should be considered in the clinical prescription of dorsiflex-assist devices.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the National Institute of Child Health and Human Development; National Institutes of Health, Grants No 1R01 HD044816-01A2 (PI: Chae) and K23HD060689 (PI: Sheffler); and the National Center for Research Resource Grant UL1TR000439. No commercial party having a direct financial interest in the results of the research supporting this article has or will confer a benefit on the authors or on any organization with which the authors are associated.
