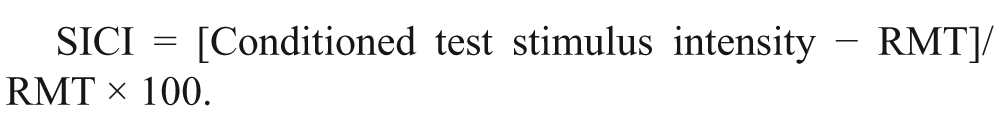Abstract
Background. With the advent of novel brain stimulation techniques aimed at improving functional outcome, understanding poststroke plasticity becomes critical for the appropriate selection of patients and optimal timing to introduce neuromodulatory interventions. Objective. To better define the temporal evolution of central and peripheral neuroplastic changes in the first 3 months after stroke and their clinical implications. Methods. Transcranial magnetic stimulation, peripheral nerve excitability, and clinical assessments were undertaken longitudinally in 31 acute stroke patients, comprising a total of 384 clinical studies. Results. During the hyperacute phase (<7 days), short-interval intracortical inhibition (SICI) was significantly reduced in lesioned (4.3% ± 1.3%) and contralesional hemispheres (3.6% ± 1.9%) compared with controls (11.4% ± 1.3%, P = .001). There were also significant alterations in accommodative properties of motor axons in the affected limb. At follow-up, SICI remained suppressed in both hemispheres in the context of significant clinical improvement. Conclusion. Simultaneous assessment of central and peripheral motor pathways has identified bilateral plastic changes that develop throughout the neural axis in acute stroke patients. It is proposed that these changes represent an adaptive response and that the persistent bihemispheric reduction in SICI may act to promote stroke recovery through cortical reorganization.
Introduction
Despite improvement in acute stroke management, many patients are left with residual motor deficits affecting function and independence. In part, this variability in outcome reflects differences in patterns of motor reorganization. 1 Specifically, unilateral lesions result in disruption of the complex interplay between inhibitory and excitatory influences responsible for the balance of excitability between hemispheres.1-3 Debate remains concerning the nature, evolution, and clinical significance of cortical excitability changes that develop in both hemispheres,4-10 particularly regarding the role of the unaffected hemisphere in terms of stroke recovery. Whereas some investigators have reported persistence in contralesional cortical hyperexcitability in association with poor functional recovery,7,10-12 others have reported positive correlations with recovery.4,13,14
In part, this discrepancy may be attributed to heterogeneity in duration from time of stroke to assessment, as well as the cross-sectional design of most studies. Further elucidating the nature of excitability changes and its potential influences on functional recovery may have therapeutic implications, especially when assessing the effects of emerging strategies that use different modalities (pharmacological and electrophysiological) to neuromodulate the balance in cortical excitability between hemispheres in an attempt to improve stroke recovery.15,16 Clearly, the optimal timing of such interventions will need to be better defined in order to avoid detrimental effects particularly if delivered during the phase, when the balance between the hemispheres is adaptive for functional recovery after stroke.
Furthermore, peripheral changes have been identified in motor axons in the affected limb of chronic stroke patients. 17 Of further relevance, ipsilateral (“unaffected”) limbs have demonstrated impairment in grip strength and dexterity using kinetic and kinematic measures in the subacute 18 and chronic phases 19 of stroke patients. These studies provide evidence for plasticity involving the peripheral motor pathways bilaterally, but again, given the cross-sectional nature of these studies, the timing and clinical significance of these changes poststroke has yet to be established. As such, the present study combined clinical assessment, with central and peripheral motor threshold-tracking techniques using paired-pulse transcranial magnetic stimulation (TMS) and nerve excitability studies, to assess the entire neural axis (bilaterally) in acute stroke patients from time of ictus, followed longitudinally over 3 months. The aim was to define the nature and temporal evolution of poststroke excitability changes in central and peripheral motor pathways, and their association with functional improvement during the acute recovery process. Furthermore, we hypothesized that cortical disinhibition, particularly over the contralesional hemisphere, will become less prominent in the later stages following an acute stroke, as access to alternative neural networks become better established. Second, we also hypothesized that nerve excitability changes may develop in peripheral motor axons immediately following an acute stroke in response to the changes occurring in the central nervous system.
Methods
Subjects
Patients diagnosed with a first-time unilateral ischemic stroke and motor deficit were recruited for the study in the acute period (within 7 days post-ictus), from a specialized acute stroke unit. Acute stroke was confirmed by diffusion-weighted magnetic resonance imaging. Patients with disorders affecting the central and/or peripheral nervous system or taking medications that could potentially affect these and thereby influence excitability parameters, or patients with contraindications to TMS, were excluded. Written informed consent was obtained from the participants and the study was approved by the local Human Research Ethics Committee.
The study cohort consisted of 31 stroke patients (15 men, 16 women; age range, 29-90 years; mean, 69.4 ± 15.0 years) recruited within the acute period (mean, 6.3 days). Subcortical lesions were evident in 52% of patients, 32% had cortical lesions, 9% had embolic lesions involving both cortical and subcortical regions, and 7% had pontine lesions. Clinical, functional, and electrophysiological assessments were performed within the same session on patients within 1 week of the acute stroke and were repeated at 3-month follow-up (range, 85-109 days; mean, 96.5 days). Thirty controls (18 men; age range, 43-92 years; mean, 60.4 ± 15.9 years) were also studied, with cortical stimulation over the left hemisphere and peripheral stimulation of the right hand. No side-to-side differences in cortical and peripheral excitability parameters in controls have been demonstrated by our laboratory and previous studies.4,5,17
Clinical Assessment
Formal clinical and functional impairment was assessed using the National Institute of Health Stroke Scale (NIHSS), the modified Rankin scale (mRS), the upper limb component of the Fugl-Meyer score (FM), and the Barthel index (BI). The measure of reflex activity was excluded from the FM assessment, to form a modified FM score with a maximum of 60, giving a more unidimensional assessment, as previous studies found that the reflex items reflected a different motor control construct than the other items. 20
Cortical Function
Measures of cortical excitability and thereby function were assessed by applying a 90-mm circular coil and assessing stroke affected and unaffected hemispheres separately with recordings measured over the contralateral abductor pollicis brevis muscle (APB). The side stimulated depended on which side of the coil (labeled A or B) is facing up. The induced current flow was from the posterior to anterior direction in both hemispheres, such that when stimulating the left motor cortex, side A of the coil was facing up ensuring a posterior-to-anterior current flow within the left motor cortex, whereas side B was facing up when stimulating the right motor cortex. The coil was adjusted over the patient’s scalp until optimal position for a motor-evoked potential (MEP) was obtained from the APB muscle. Currents were generated by 2 magnetic stimulators connected via a BiStim2002 (Magstim Co, Whitland, UK), such that conditioning and test stimuli could be independently set and delivered through one coil.
Paired-pulse threshold-tracking TMS techniques were used to assess cortical function according to a previously reported method 21 and was developed to overcome the variability in MEP amplitudes with consecutive stimuli that resulted in limitations using the constant-stimulus method. 22 While the threshold-tracking method used in this study is different from the traditional methods, this method was first developed by Fisher et al 23 in 2002, later validated by Vucic et al 21 in 2006, and is currently used and published widely in numerous pathological studies.24-29 Importantly, as suggested by Fisher et al, 23 measurement of intracortical excitability at a constant MEP response using threshold-tracking methods limits the contribution of spinal and peripheral elements to the output measurement. The threshold-tracking strategy used a target response of 0.2 mV (±20%) located in the middle of the established linear relationship between the logarithm of the MEP amplitude and the stimulus intensity. 21 As such, selecting a target that is located in the steepest portion of the stimulus–response (SR) curve, relatively large variations in the MEP amplitude translate to relatively small variations in stimulus intensity.
In the initial part of the protocol, resting motor threshold (RMT) was defined as the stimulus intensity required to consistently (average 10 trials) produce and maintain this target MEP response of 0.2 mV, with the patient seated in a comfortable chair and testing limb completely relaxed. Voluntary electromyographic (EMG) activity was continually monitored using audio and visual feedback displayed on a projected computer screen, to ensure that these did not contaminate the recordings. Furthermore, a “time window” for capturing the MEP response was set such that no prestimulus EMG activity was recorded or captured for analysis. If voluntary EMG activity occurred within the window, then the entire protocol was repeated.
Stimulus–response curves for cortical stimulation were determined by increasing the intensity of the magnetic stimulus to the following levels: 60%, 80%, 90%, 100%, 110%, 120%, 130%, 140%, and 150% RMT. Three stimuli were delivered at each intensity level and the maximum MEP amplitudes determined from the SR curve.
Cortical silent period (CSP) was induced by single-pulse TMS with intensity varied as for the SR curve and recorded while patients performed a weak voluntary contraction with visual feedback displayed on a monitor and broadly estimated by the investigators as representing 10% to 30% of maximum voluntary contraction. The CSP duration was measured from the beginning of MEP to the return of EMG activity 30 at 150% RMT stimulus intensity.
Short-interval intracortical inhibition (SICI) and intracortical facilitation (ICF) were measured according to a previously described protocol. 21 Subthreshold conditioning stimuli (70% RMT) were delivered in sequential order at the following interstimulus intervals: 1, 1.5, 2, 2.5, 3, 3.5, 4, 5, 7, 10, 15, 20, and 30 ms. In this paired-pulse paradigm (Figure 1), stimuli were delivered sequentially as a series of 3 channels: Channel 1 tracked the stimulus intensity required to produce the unconditioned test response (ie, RMT, such that the RMT was continuously monitored throughout the testing to ensure it remained stable); channel 2 monitored the subthreshold conditioning stimulus (70% RMT) so as to ensure that an MEP response was not produced and that the subject remained relaxed; and channel 3 tracked the stimulus required to produce the target MEP when conditioned by a subthreshold stimulus equal in intensity to that on channel 2. Tracking was deemed acceptable when the test stimulus produced 2 consecutive MEP responses that were within 20% of the target response (0.2 mV) or consistently oscillated about the target. The 3 channels were applied sequentially and the computer advanced to the next interstimulus interval (ISI) only when tracking met the target criteria. The interval between stimuli was 5 seconds.
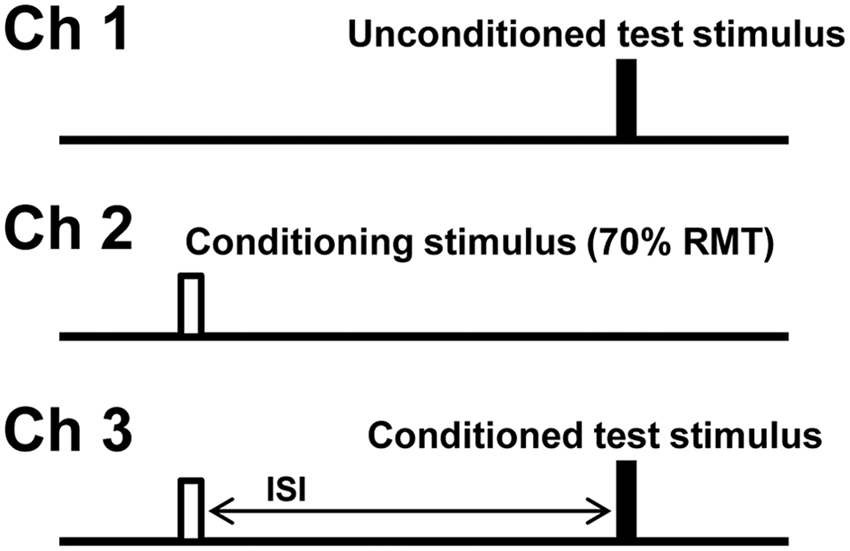
Experimental paradigm and configuration of stimulus patterns for measurement of short-interval intracortical inhibition (SICI) and intracortical facilitation (ICF). Cortical excitability was assessed by measuring changes in stimulus intensity required to generate a target motor-evoked potential (MEP) response of 0.2 mV. Channel 1, unconditioned test stimulus, measuring resting motor threshold (RMT). Channel 2, conditioning stimulus, set to subthreshold (70% RMT). Channel 3, conditioned test stimulus at different interstimulus intervals (ISIs).
Short-interval intracortical inhibition was measured as the increase in test stimulus intensity required to evoke the target MEP, and calculated as follows21,23:
Facilitation was measured as the decrease in the conditioned test stimulus intensity required to evoke the target MEP. SICI was determined over the ISIs of 1 to 7 ms, whereas ICF was determined over the intervals of 10 to 30 ms.
All parameters except CSP were recorded with target muscle at rest, which was closely observed for the presence of voluntary activity as discussed above.
Peripheral Nerve Function
Peripheral nerve excitability studies were performed on affected (AL) as well as unaffected (UL) upper limbs of stroke patients, using a previously described protocol.31,32 Motor axons of the median nerve were electrically stimulated at the wrist and the compound muscle action potential (CMAP) obtained from APB was recorded. Test current pulses of 1-ms duration were applied to produce a threshold tracking target that was 40% of the maximum CMAP.
From the SR curves obtained, strength–duration time constant (τSD) was calculated using Weiss formula.33,34 Subsequently, threshold electrotonus (TE) was recorded using prolonged 100-ms subthreshold depolarizing (TEd) and hyperpolarizing (TEh) pulses set at 40% of the control threshold current.31,35 Recovery cycle parameters following a supramaximal conditioning stimulus was next recorded and included relative refractory period (RRP), superexcitability, and late subexcitability. 31 The final part of the protocol measured the current threshold (I/V) relationship by applying a 1-ms test stimulus after onset of subthreshold 200-ms duration polarizing currents altered stepwise from +50% (depolarizing) to −100% (hyperpolarizing) of control threshold in 10% steps. 31
Recordings of CMAP and MEP were amplified and filtered (3Hz to 3kHz) using a GRASS ICP511 AC amplifier (Grass-Telefactor, Astro-Med Inc, West Warwick, Rhode Island) and sampled at 10 kHz with a 12-bit data acquisition card (National instruments PCI-MIO-16E-4). Data acquisition and stimulation delivery were controlled by QTRAC software (Institute of Neurology, Queen Square, London, UK).
Statistical Analysis
Group data are expressed as mean ± standard error of the mean and compared using either Student t test or Mann–Whitney U test depending on normality of data distribution and applied when analyzing patient against control data. Likewise, side-to-side paired data for each considered variable obtained at baseline were compared using either paired t test or Wilcoxon test. A repeated-measures analysis of variance with time as the within-group factor, and study side as the between-group factor, and follow-up pairwise comparisons, were performed to test the evolution of neurophysiological data. The Bonferroni correction was applied. Clinical data between the study time points were compared using the Wilcoxon test. Correlation between excitability indices and clinical scores were analyzed by Spearman rank test. A P value of <.05 was regarded as statistically significant. It is noted that in our control data for SICI for conditioning stimulus 70%, 21 no age-related changes were evident between younger and older subjects. Peripheral nerve excitability measurements were compensated for age and temperature before statistical analysis, using relations obtained in controls.31,36-37
Results
Acute Stroke Baseline
Clinical assessment
Clinical scores of patients ranged from mild to severe, with inability to achieve independence in 35%. The mean for FM score was 46.3 (range, 0-60), NIHSS was 5 (range, 0-19), BI was 65.8 (range, 0-100), and mRS was 2.9 (range, 0-5), translating to moderate levels of motor, global neurological and functional impairment.
Cortical function
Affected hemisphere motor cortices were inexcitable in 19% of patients (absent target MEP response at maximum stimulator output), all of whom exhibited moderate–severe functional impairment (mean FM = 28, NIHSS = 9, BI = 36.7, mRS = 4.2). In the remainder of patients, RMT was neither significantly different in controls (59.8% ± 1.8%) when compared with affected (59.4% ± 3.4%, P = .89) and unaffected hemispheres (58.1% ± 1.9%, P = .5), nor between hemispheres (P = .32).
Assessment of intracortical excitability established that averaged SICI (over 1-7 ms ISIs) was significantly reduced in both affected (4.3% ± 1.3%, P = .001) and unaffected (3.6% ± 1.9%, P = .001) hemispheres compared with controls (11.4% ± 1.3%; Figure 2), without side-to-side differences (P = .86). Averaged ICF was not different in affected (1.1% ± 1.2%, P = .19) and unaffected (1.2% ± 1.0%, P = .13) hemispheres when compared with controls (−1.4% ± 1.3%), and between hemispheres (P = .72).
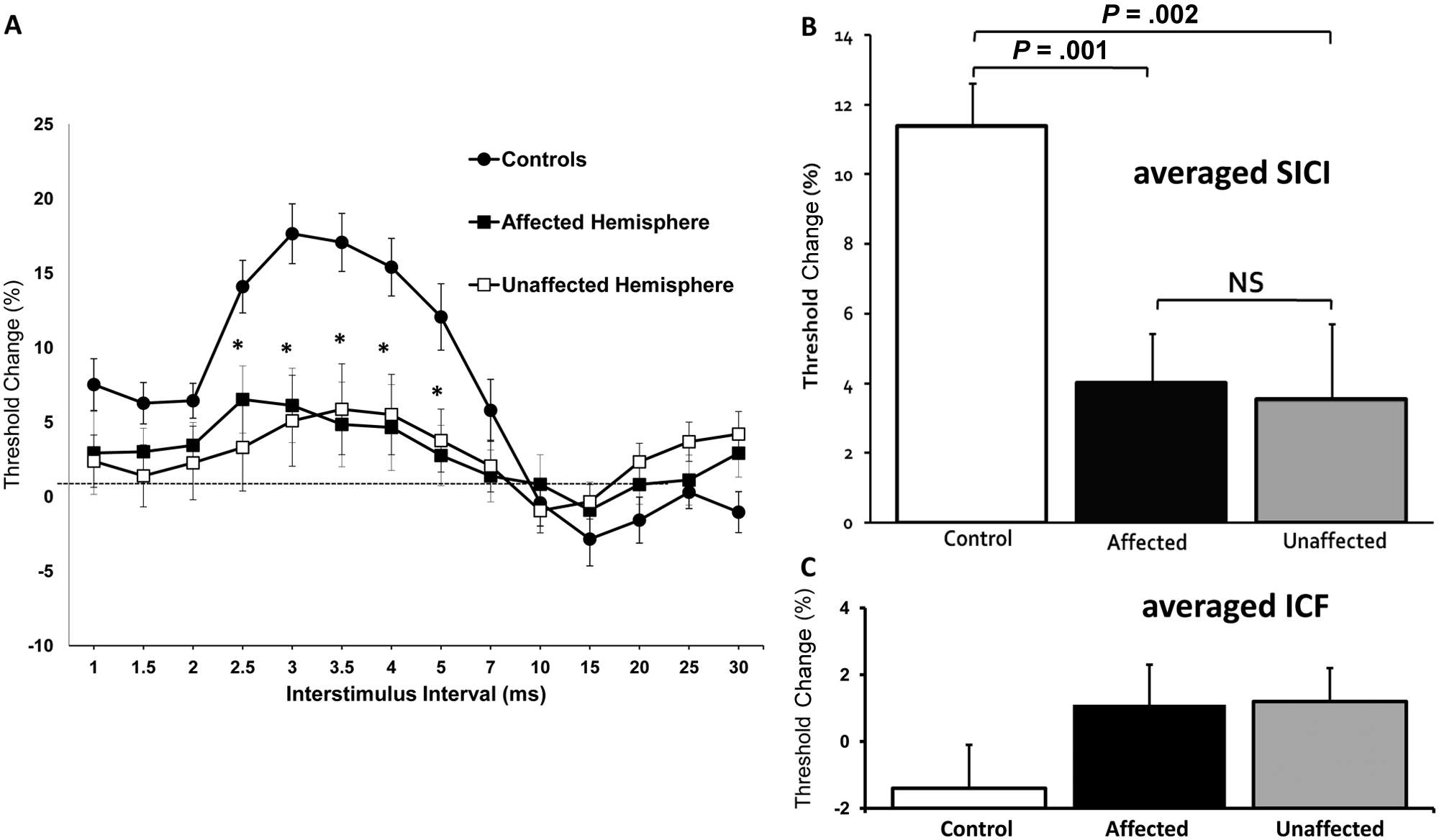
Baseline paired-pulse subthreshold conditioning transcranial magnetic stimulation demonstrating (A) reduction in short-interval intracortical inhibition (SICI) in affected and unaffected hemispheres; (B) significant reductions in averaged SICI (between interstimulus intervals of 1 to 7 ms) in affected and unaffected hemispheres compared with controls; (C) no significant differences in averaged intracortical facilitation (ICF) between affected, unaffected hemispheres, and controls. *P < .01; NS, not significant; RMT, resting motor threshold. Threshold change is the difference in test stimulus intensity required to evoke the target motor-evoked potential (MEP) calculated as follows: [Conditioned test stimulus intensity − RMT]/RMT × 100.
Assessment of other parameters of cortical function established no significant differences in CSP duration (controls = 215.8 ± 5.2 ms; AH = 221.5 ± 10.6 ms, P = .59; UH = 218.7 ± 6.1 ms, P = .23) and maximum MEP amplitude (expressed as percentage of CMAP amplitude) (controls = 24.6% ± 3.5%; AH = 38% ± 8.1%, P = .98; UH = 44.2% ± 6%, P = .72) between hemispheres in stroke patients and controls, as well as between hemispheres (CSP, P = .79; MEP amplitude, P = .39)
Peripheral nerve function
There was a significantly greater change to hyperpolarizing currents observed in affected (AL) [TEh(90-100 ms): −132.9% ± 4.5%] compared with unaffected limbs (UL) (−121.6% ± 3.6%, P = .03) and with controls (−112% ± 3.2%, P = .003; Figure 3A). The difference between unaffected limbs and controls, however, was not statistically significant (P = .13).
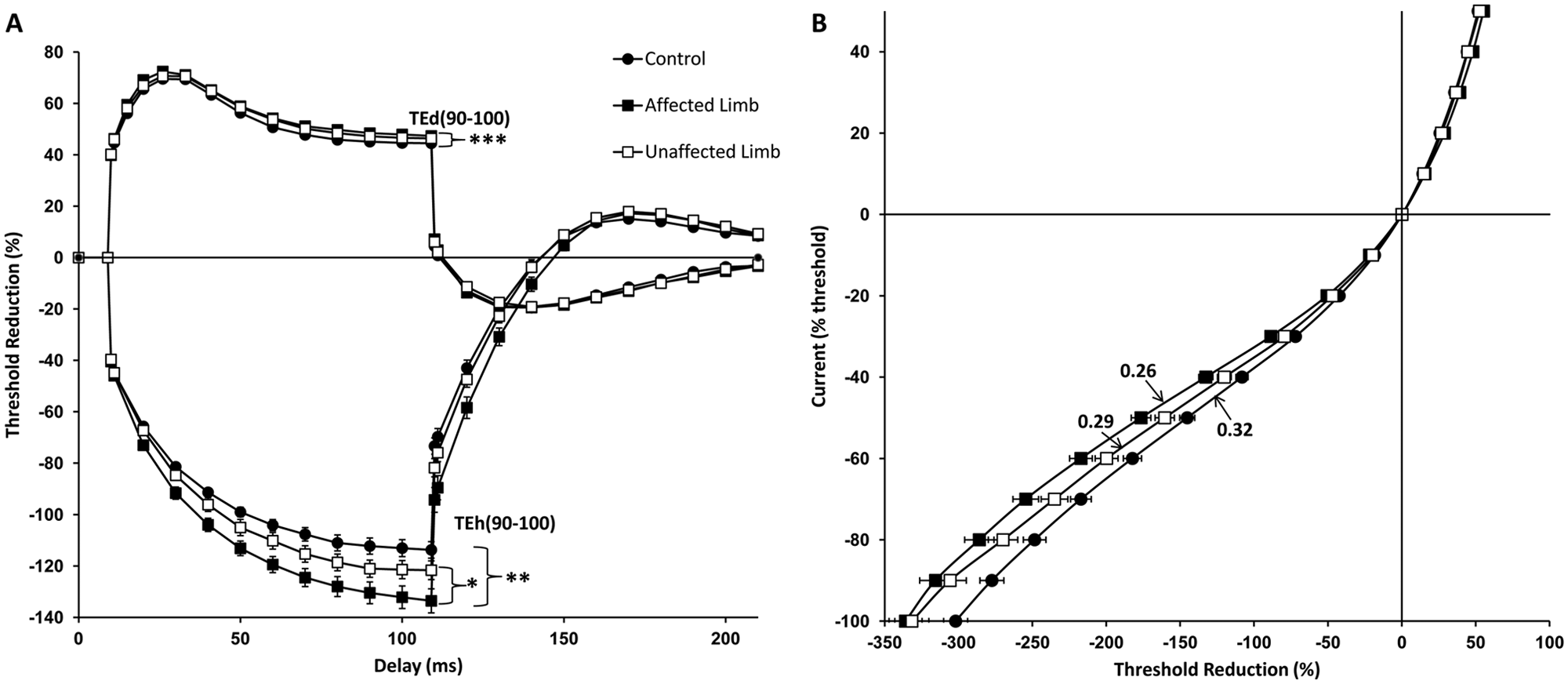
(A) Threshold electrotonus demonstrating significant reductions in accommodation to hyperpolarizing currents [TEh(90-100)] in affected limbs compared with both unaffected limbs and controls. Affected limbs also showed greater change to depolarizing currents [TEd(90-100)] compared with controls. (B) Current–threshold relationship demonstrating significantly reduced hyperpolarizing slope in affected limbs compared with unaffected limbs and controls. *P < .05, **P < .01, ***P < .001. TEh(90-100), threshold change produced by prolonged subthreshold hyperpolarizing currents measured at 90 to 100 ms delay; TEd(90-100), threshold change produced by prolonged subthreshold depolarizing currents measured at 90 to 100 ms delay.
The findings in TE were accompanied by significant reductions in hyperpolarizing I/V gradient in affected (0.26 ± 0.01) when compared with unaffected limbs (0.29 ± 0.14, P = .005) and controls (0.32 ± 0.11, P = .001; Figure 3B). The difference between unaffected limbs and controls did not reach statistical significance (P = .088). The greater threshold change to hyperpolarizing currents observed in TE accompanied by the lower gradient in hyperpolarizing I/V slope demonstrated in affected limbs, suggests impaired accommodation to hyperpolarizing currents reflecting reduction in inward-rectifying (I H) conductance of motor axons. 31 There were no differences in depolarizing threshold electrotonus between affected and unaffected limbs of patients [TEd(90-100 ms): AL 47.6% ± 0.7%, UL 46.5% ± 1.1%, P = .49], although affected limbs showed larger threshold changes compared with controls (44.3% ± 0.6%, P < .001) whereas there was a trend for this difference between unaffected limbs and controls (P = .06; Figure 3A).
In terms of other peripheral functional parameters, CMAP amplitudes were no different between stroke patients and controls (AL = 5.7 ± 0.6 mV, P = .30; UL = 5.4 ± 0.5 mV, P = .12; controls = 6.9 ± 0.6 mV;) or when comparing the limbs (P = .82). In addition, τSD, which reflects nodal persistence Na+ conductance 38 was also comparable between the 3 groups (AL = 0.48 ± 0.02 ms, P = .09; UL = 0.45 ± 0.02 ms, P = .40; controls = 0.43 ± 0.02 ms; paired, P = .29).
There were no differences in recovery cycle measures between limbs of patients compared with controls. Specifically, the relative refractory period was similar between groups (AL = 3.3 ± 1.0 ms, P = .24; UL = 3.1 ± 1.0 ms, P = .051; controls = 3.1 ± 1.0 ms; paired, P = .30), as was superexcitability (AL = −22.0% ± 1.0%, P = .27; UL = −21.6% ± 1.1%, P = .24; controls = −23.1% ± 1.0%; paired, P = .79) and late subexcitability (AL = 14.3% ± 1.0%, P = .71; UL = 13.6% ± 0.7%, P = .48; controls = 14.5% ± 0.6%; paired, P = .67). The absence of differences in τSD and recovery cycle parameters suggests comparable resting membrane potentials in affected and unaffected limbs of stroke patients. 34
Longitudinal 3-Month Stroke Follow-Up
Clinical assessment
At 3-month follow-up, there were significant improvements in all clinical scores compared with baseline assessments (Figure 4A-D) reflecting functional independence in all patients at follow-up.
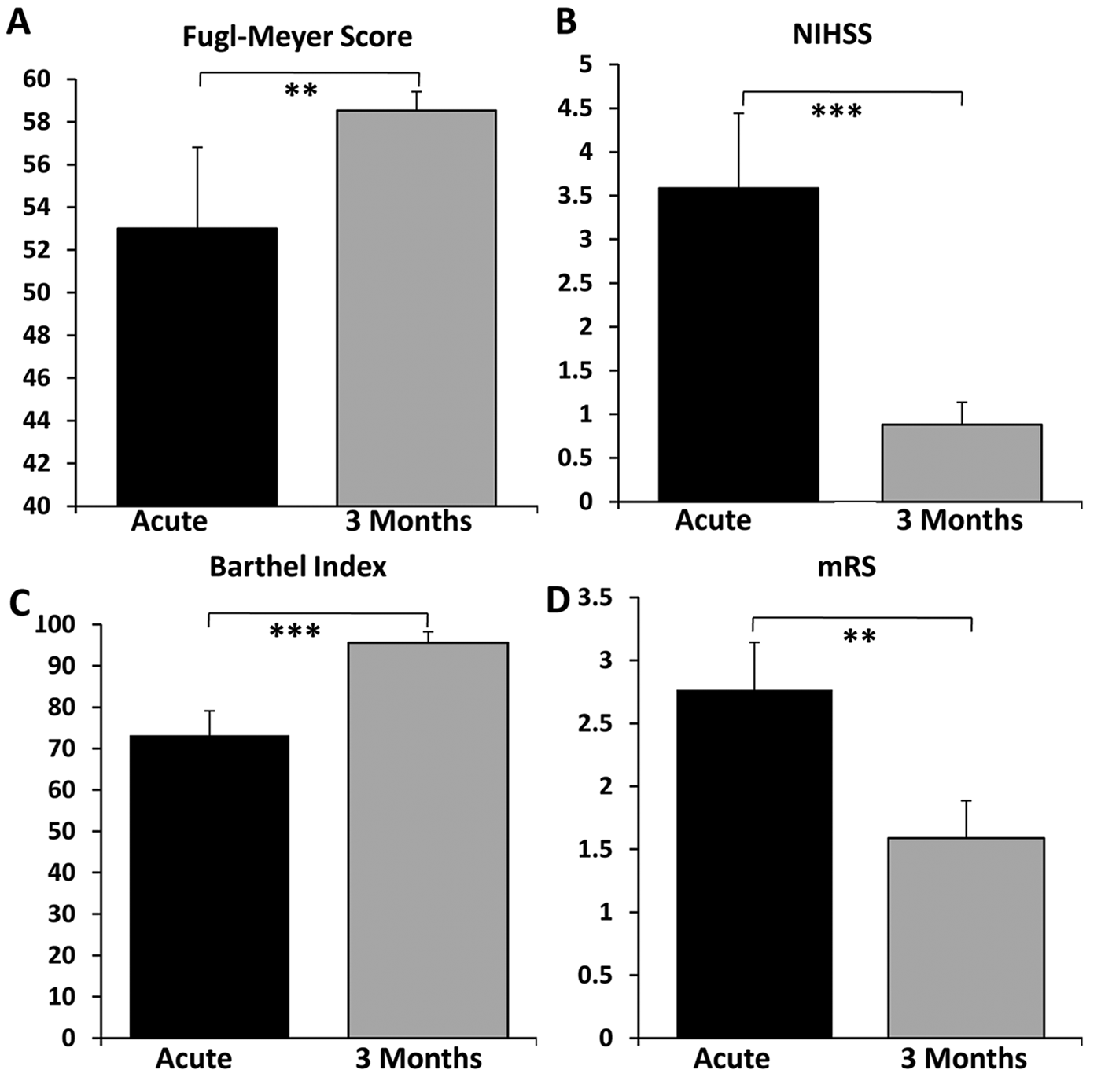
Significant improvements in all clinical scores (A-D) at 3-month follow-up compared with baseline. **P < .01, ***P < .001.
Cortical function
Averaged SICI remained suppressed in both hemispheres compared with controls at 3-months (AH, 4% ± 2.3%; UH, 5% ± 2.3%; P = .006) but were no different compared with baseline recordings and to each other (analysis of variance: F 1,24 = 0.04; P = .84; Figure 5). Of the cohort of stroke patients with inexcitable affected hemisphere cortices at baseline, all had normalization of cortical excitability (RMT = 60.9% ± 8%), associated with improvement in clinical scores from moderate–severe levels of disability to functional independence (mean FM = 54.25; NIHSS = 2.25; BI = 86.25; mRS = 2.5).
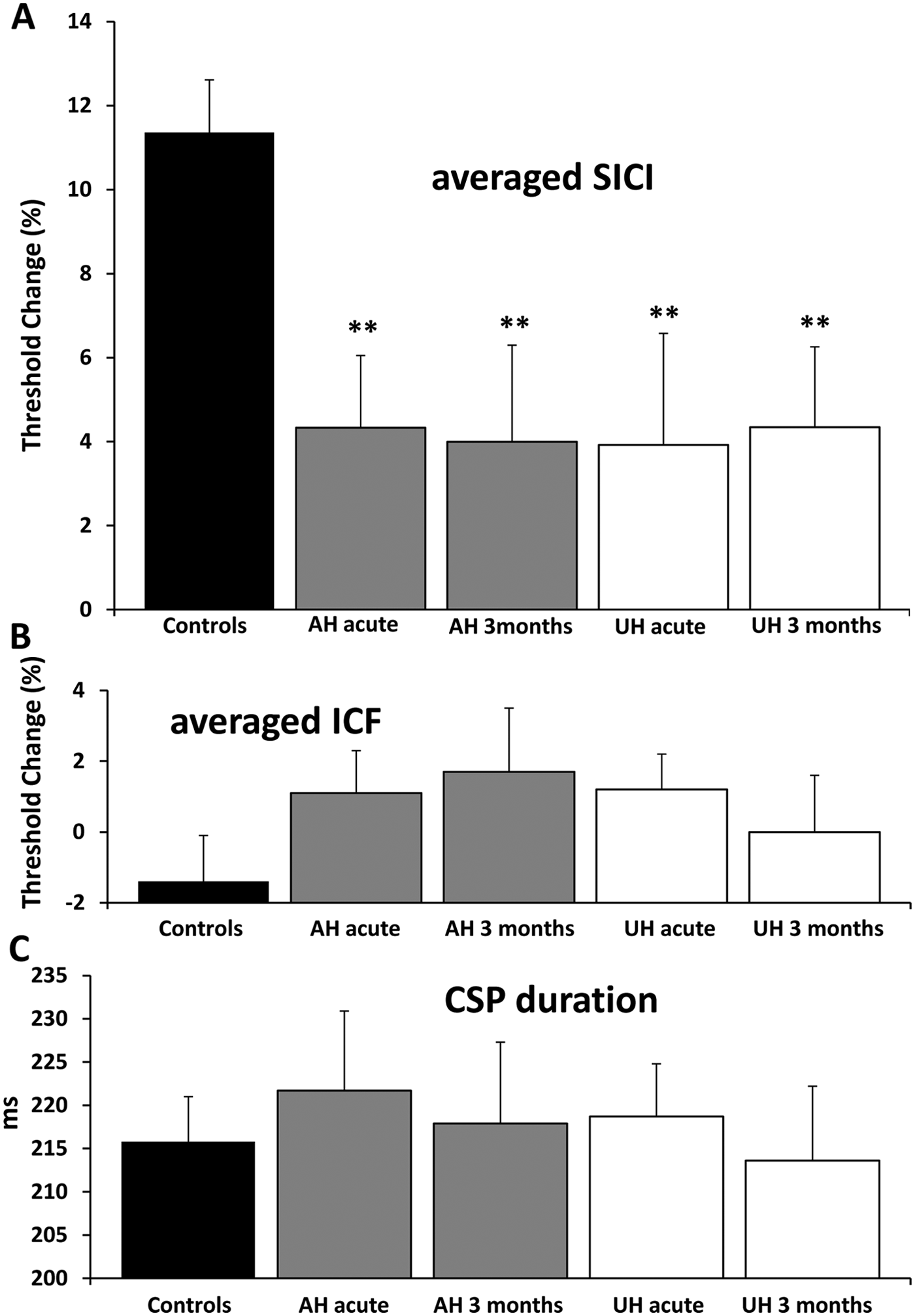
(A) Persistently suppressed averaged short-interval intracortical inhibition (SICI) in affected (AH) and unaffected (UH) hemispheres at 3 months. (B, C) No significant differences in averaged intracortical facilitation (ICF) and cortical silent period (CSP) at 3 months compared with baseline and controls. **P < .01. RMT, resting motor threshold. Threshold change is the difference in test stimulus intensity required to evoke the target motor-evoked potential (MEP) calculated as follows: [Conditioned test stimulus intensity − RMT]/RMT × 100.
All other parameters, including ICF, RMT, CSP duration, and MEP amplitude remained unchanged in both hemispheres compared to baseline.
Peripheral function
Changes in nerve excitability in limbs of stroke patients persisted at 3 months. In particular, indices that were significantly different compared with controls at baseline showed little change at follow-up.
Clinical Correlations
At baseline and 3 months, correlations were examined between cortical and peripheral measures and clinical assessment. There were no relationships between SICI in both hemispheres and clinical scores; nor were there correlations between peripheral excitability parameters and clinical measures. However, RMT and CMAP of the affected sides correlated with all functional scores (FM, NIHSS, BI, and mRS; Table 1). In other words, better clinical scores were associated with lower RMT and higher CMAP amplitude. Correlations were sought between central and peripheral measures to explore the relationship between changes in upper and lower motor neuron pathways. This demonstrated a negative correlation between RMT and CMAP (r = −0.51, P = .036). Together, these suggest that corticomotoneuronal dysfunction following an ischemic lesion is associated with disruption to impulse conduction downstream and consequently greater functional impairment.
Correlations Between Functional Scores, Central Studies, and Peripheral Studies at Baseline a
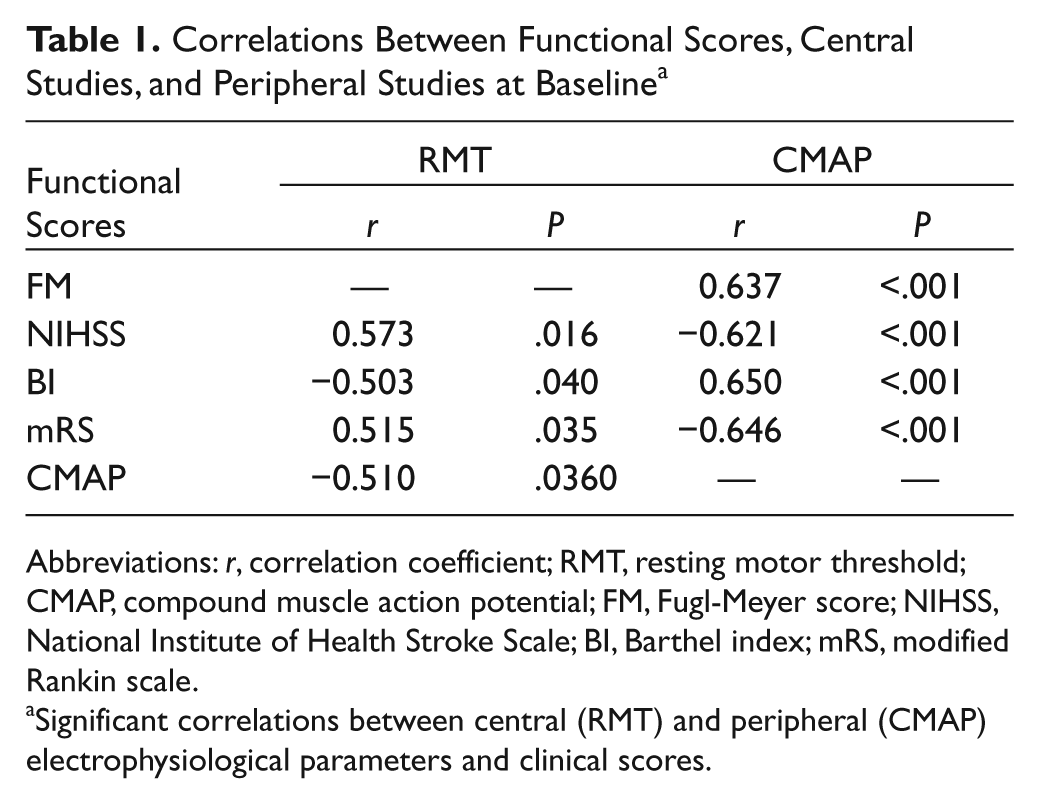
Abbreviations: r, correlation coefficient; RMT, resting motor threshold; CMAP, compound muscle action potential; FM, Fugl-Meyer score; NIHSS, National Institute of Health Stroke Scale; BI, Barthel index; mRS, modified Rankin scale.
Significant correlations between central (RMT) and peripheral (CMAP) electrophysiological parameters and clinical scores.
Discussion
Using a novel combination of threshold-tracking techniques, combined with clinical assessment to simultaneously investigate central and peripheral neuronal excitability in acute stroke patients, the present study has identified evolution of neuroplastic changes that develop at central and peripheral levels shortly after stroke onset. Persistent reductions in SICI in both affected and unaffected hemispheres, combined with persistent peripheral nerve excitability abnormalities, followed by significant improvements in functional measures over 3 months, suggest an adaptive process promoting recovery after stroke.
Although similar results were observed by previous investigators that used a cross-sectional design, our longitudinal study was able to critically determine the evolution of these changes. In addition, the present study has identified significant functional improvements in a cohort of acute stroke patients who initially exhibited inexcitable motor cortices, suggesting that the absence of ipsilesional responses in the hyperacute period does not always correlate with poor functional outcome.39,40 Furthermore, it has been proposed more recently that the presence of an MEP response is more useful for predicting outcome when combined with clinical assessment and novel imaging modalities such as diffusion tensor magnetic resonance imaging to better define the structural integrity of the corticomotor pathways.41,42 In contrast to previous studies by Swayne et al, 10 the acute disinhibition observed over the contralesional cortex in our study was not demonstrated in their cohort and may be related to the difference in sample size with a larger population size used in our present study.
Cortical Plasticity
Stroke alters the balance between cortical excitatory and inhibitory circuits, and the excitability changes occurring bihemispherically may shift over time in association with stroke recovery. 43 The present study has identified reductions in intracortical inhibition (SICI) in both the ipsilesional and contralesional hemispheres in the immediate period (<1 week) following acute stroke. These changes persisted at 3 months, in the context of significant improvements in all clinical scores, suggesting that persistent disinhibition during the first 3 months of acute stroke is associated with functional recovery.
The reduction in SICI, a reflection of downregulation in GABAergic circuits via GABAA receptors, 43 enables the unmasking of functionally latent networks, permitting cortical reorganization critical in stroke recovery.1,12,43 Specifically, the inhibitory neurotransmitter GABA (γ-aminobutyric acid) is critical for cortical plasticity as blockades of GABAergic signaling are associated with changes in cellular excitability, including long-term potentiation that mediates learning and memory. 14 Focal and ischemic cortical lesions in rat models have demonstrated downregulations in GABAA-receptor function and associated decreases in GABAergic intracortical inhibition in both lesioned and contralesional motor cortices.44,45 Accompanying this, rat models have also demonstrated axonal sprouting, dendritic growth, and pruning as well as synaptogenesis and enhanced synaptic efficiency in both hemispheres.46-49 Of further relevance, bihemispheric reorganization of cortical networks may result in enhanced interhemispheric connectivity, 2 thereby contributing to a recovery of motor function. Moreover, the role of GABAA-mediated processes in stroke recovery is further supported by the findings that reducing GABAA-mediated inhibition using an antagonist resulted in improvements in stroke outcome in mice, 50 as well as worsening of stroke deficits in patients who were administered midazolam, which increases GABAergic inhibition. 51
Although decreased cortical inhibition in the affected hemisphere appears consistent in both the acute and chronic periods following functional improvement,12,52,53 the evolution of excitability changes in the unaffected hemisphere as a function of recovery remains complex. The balance between excitability changes is controlled by mutual interhemispheric inhibition, and although contralesional hyperexcitability occurs acutely after stroke, the concern remains whether persistent hyperexcitability may result in potentially detrimental inhibitory effects on the ipsilesional hemisphere.3,12 Previous reports have demonstrated that chronic stroke patients with poor recovery have increased interhemispheric inhibition from the contralesional cortex and that improved clinical scores were obtained following training and associated with reductions in this transcallosal inhibition. 54 One study, however, demonstrated that the contralesional hyperexcitability was not associated with an increase in interhemispheric inhibition from the contralesional to ipsilesional motor cortex, 55 but the study was performed on stroke patients within 6 weeks of ictus and the interhemispheric influences may change over time. Other cross-sectional studies have reported discrepancies regarding the prognostic influence of persistent contralesional cortical disinhibition on ultimate recovery, with some advocating a return to normal inhibition in association with recovery7,10-12 and others demonstrating the converse.4,13 Some investigators have found that patients with good recovery of hand function had reductions in inhibition in the contralesional hemisphere whereas those with poor recovery did not. 4 Furthermore, the contralesional hyperexcitability occurred regardless of alterations in interhemispheric inhibition from the ipsilesional cortex, suggesting that this may represent changes in neurotransmission associated with stroke recovery, as opposed to a release phenomenon. 55
The role of the unaffected hemisphere in stroke recovery may be multifactorial. Specifically, it may act as a relevant anatomic substrate for functional reorganization by recruitment of latent ipsilateral corticospinal tracts to the paretic limb to aid in motor recovery. 11 Such a hypothesis is supported by evidence for ipsilateral MEPs evoked in stroke patients using TMS. 56 The transcallosal transfer of information from the intact to lesioned hemisphere may change from inhibitory to facilitatory to aid adaptive plastic changes and to facilitate perceived difficulty in movement of the paretic limb. 57
Plasticity of Peripheral Motor Axons
The present study demonstrated significant changes in nerve excitability parameters in the affected limb of patients in the immediate period after ictus. The impaired accommodation to hyperpolarizing currents, as reflected by alterations in TEh(90-100 ms) and hyperpolarizing I/V slope, were suggestive of reductions in I H conductances. However, there were also significant differences in accommodation to depolarizing currents [TEd(90-100 ms)] observed in the affected limb compared with controls suggesting an additional biophysical alteration in motor axons (reduction in slow K+ conductance) following stroke.
The subclinical involvement of the unaffected limb may be explained by downstream changes in response to contralesional hemispheric disinhibition and involvement of ipsilesional uncrossed fibers. The exact mechanisms for the observed biophysical changes may not be as easily elucidated as those that occur in response to peripheral nerve insult, in particular those observed in peripheral nerve ischemia, 58 but may provide a window of insight into events occurring in the central nervous system.
Potential limitations to the present study include the nonrandomized delivery of ISIs during the paired-pulse paradigm by nonblinded investigators, which may have resulted in a systematic effect in the MEP response. Although the CSP duration was measured from the onset of the facilitated MEP to return of EMG activity as per convention,21,30,59,60 this may differ from other investigators that report CSP duration as measured from the onset of stimulus.
Conclusion
We demonstrated bihemispheric reductions in SICI from time of ictus to 3 months after stroke associated with functional recovery, suggesting that persistent bilateral disinhibition during this period may be associated with a process promoting ongoing utilization of additional networks to aid recovery. A critical question to address is when the balance in cortical excitability transforms from being adaptive to become maladaptive. Furthermore, in keeping with the hypothesis that the role of the contralesional hemisphere may become less critically important during stroke recovery, longitudinal studies extending beyond 3 months will be required to establish when the excitability changes begin to normalize. This becomes more critical with the advent of novel neuromodulating approaches aimed at reducing the excitability of the intact hemisphere while increasing that of the affected hemisphere, such as noninvasive brain stimulation with repetitive TMS, peripheral nerve stimulation, and transcranial direct current stimulation15,61-64 for neuromodulation to facilitate stroke recovery.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: WH is receiving the postgraduate scholarship from the National Health & Medical Research Council of Australia (NHMRC). AVK was supported by an NHMRC Career Development Award (Grant No. 568680).