Abstract
Introduction. Limited evidence suggests that visual impairments may influence outcome after stroke. The degree of recovery from these impairments is poorly characterized. Objectives. To describe recovery and to determine whether visual impairments influence functional outcome and quality of life. Methods. We extracted demographic and outcome data from the Virtual International Stroke Trials Archive (VISTA). We examined horizontal eye movement disorders and hemianopia using the Best Gaze and Visual domains of the National Institutes of Health Stroke Scale (NIHSS) and described recovery at 30 and 90 days. Proportional odds modelling was used to examine the association between impairments at baseline, modified Rankin Scale (mRS), and European Quality of Life Score (EQ-5D) at 90 days. Results. Visual impairments were reported in 7,204/11,900 (60.5%) patients at baseline. Complete recovery occurred in 1,398/3,285 (42.6%) and 3,243/7,204 (45.0%) patients by 30 and 90 days respectively. The burden of persistent visual impairment in survivors was 1,135/4,028 (28.2%) at 30 days and 1,915/9,338 (20.5%) at 90 days. Partial gaze palsy (P < .0001; OR = 0.81; 95% CI = 0.74-0.87), forced deviation (P < .0001; OR = 0.48; 95% CI = 0.43-0.53), and complete homonymous hemianopia (P < .0001; OR = 0.67; 95% CI = 0.62-0.73) at baseline were associated with poor mRS at 90 days. Conclusions. The rate of recovery was greater in the first month after stroke, suggesting a potential time frame for interventions. The associations between visual impairments and poor mRS suggest that these impairments should be considered in multidisciplinary assessments and interventions.
Keywords
Introduction
Stroke can cause visual impairments such as visual field defects, eye movement disorders, and visuospatial neglect. The reported prevalence of visual impairments varies considerably: visual field defects present in 20% to 74% of stroke patients.1-5 A recent multicenter prospective cohort study based on referrals used simple screening to identify patients with suspected visual difficulties. Of 323 patients, 92% had confirmed visual impairments; 68.4% had eye movement disorders, and 46.1% had visual field defects. 6
Visual impairments can affect functional ability, quality of life, participation in rehabilitation, and discharge destination following stroke.7-11 Those with visual impairments after stroke are at increased risk of falling, report difficulties reading, and experience emotional disorders, including anxiety and depression.9,12,13 The presence of visual impairments can also affect visual memory, recognition, forward planning, and decision making. 14 As the incidence of stroke increases with age, a significant proportion of stroke patients may have coexisting age-related visual problems, 14 which if untreated, can further affect quality of life. 12
One barrier to greater understanding of the impact of visual impairments on rehabilitation after stroke is the uncertain prevalence and natural history of recovery4,15-17; estimates of recovery range between 7% and 85%.17-21 Opinions differ as to the degree of visual field restoration that occurs 22 and the time frame for maximal recovery, with estimates ranging from 48 hours to 30 days.4,23,24 Investigations of visual restitution training in a non–stroke-specific population have reported that some recovery may be possible even years after injury. 25
We therefore sought to explore the recovery from visual impairments during the first 3 months post-stroke, the associations between recovery and key demographic, and clinical variables and their association with functional outcome and quality of life at 3 months poststroke.
Methods
We used anonymized stroke trial data from the acute ischemic, intracerebral hemorrhage and rehabilitation sections of the Virtual International Stroke Trials Archive (VISTA). 26 To avoid reanalysis of trial treatment effects per VISTA guidelines and to avoid the confounding effect of active treatment, we sought data from nonthrombolysis trials that had an overall neutral outcome. We included data from trials in which patients were thrombolyzed as part of standard care and adjusted our analyses accordingly. We compiled an analysis data set of age, medical history, functional outcomes by modified Rankin Scale (mRS), quality of life outcomes using the European Quality of Life Score (EQ-5D), and poststroke impairment described using the National Institutes of Health Stroke Scale (NIHSS) domain scores.
The EQ-5D 27 assesses quality of life, measuring domains such as mobility, self-care, usual activities, anxiety/depression, and pain/discomfort; a visual analogue scale enables the patients to assess their own health state. The mRS is a global functional outcome scale graded from 0 to 6 (0 = no symptoms at all, 1 = no significant disability despite symptoms, 2 = slight disability, 3 = moderate disability, 4 = moderate to severe disability, 5 = severe disability, 6 = death). The NIHSS 28 measures a range of impairments, including level of consciousness, dysarthria, limb weakness, ataxia, and sensory and visual field deficits. The total NIHSS is scored from 0 to 42, with 42 indicating the worst outcome. We scored the presence of either a horizontal eye movement disorder or a visual field defect using the NIHSS domain scores (Best Gaze: 1 = partial gaze palsy [inability to move one or both eyes completely to both directions], 2 = forced deviation [conjugate deviation of the eyes to the right or left, even with reflexive movements]; Visual: 1 = partial hemianopia, 2 = complete hemianopia, and 3 = bilateral hemianopia). According to published, validated instructions, hemianopia is assessed by confrontation, using finger counting or visual threat as appropriate. 28 Horizontal eye movement disorders are scored using voluntary or reflexive eye movements.
The NIHSS Extinction/Inattention sub-item, which describes neglect of 1 side, does not score visual neglect separately, so we reported combined visual, tactile, auditory, spatial, or personal inattention. We described the prevalence and types of visual impairment at baseline as a proportion of the entire population.
Recovery from visual impairments was defined as a score of 0 on both the Visual and Best Gaze domains of the NIHSS at 30 or 90 days; partial recovery was assessed by comparing the shift from complete hemianopia to partial hemianopia or forced deviation to partial gaze palsy by 30 and 90 days. We used proportional odds modeling to examine the factors that were associated with complete recovery from poststroke visual impairments, adjusting for patient age, medical history, the vision-unrelated NIHSS domain scores (total baseline NIHSS score = Best Gaze + Visual Scores), recombinant tissue plasminogen activator (rt-PA) administration, and type of stroke.
We compared the distribution of mRS and EQ-5D scores at 90 days in those patients with and without visual impairments. We formally compared these distributions using proportional odds modeling, adjusting for patient age, rt-PA administration, vision-unrelated NIHSS domain scores, type of stroke, and medical history.
Results
Preexisting Visual Impairments
Our analysis data set contained anonymized data on 11,900 patients. Of these, 693 patients (5.8%) had a preexisting visual problem, the most common of which were glaucoma (n = 247, 35.6%), cataract (n = 215, 31.0%), macular degeneration (n = 50, 7.2%), blindness (n = 42, 6.1%), and retinopathy (n = 20, 2.9%).
The Natural History of Recovery From Visual Impairments
Baseline
At baseline, 7,204/11,900 (60.5%) patients presented with some form of visual impairment (Figure 1), classified as an eye movement disorder or hemianopia. Complete homonymous hemianopia was most common, affecting 4,158/11,900 patients (35.0%), closely followed by partial gaze palsy (n = 3,691/11,900; 31.0%).
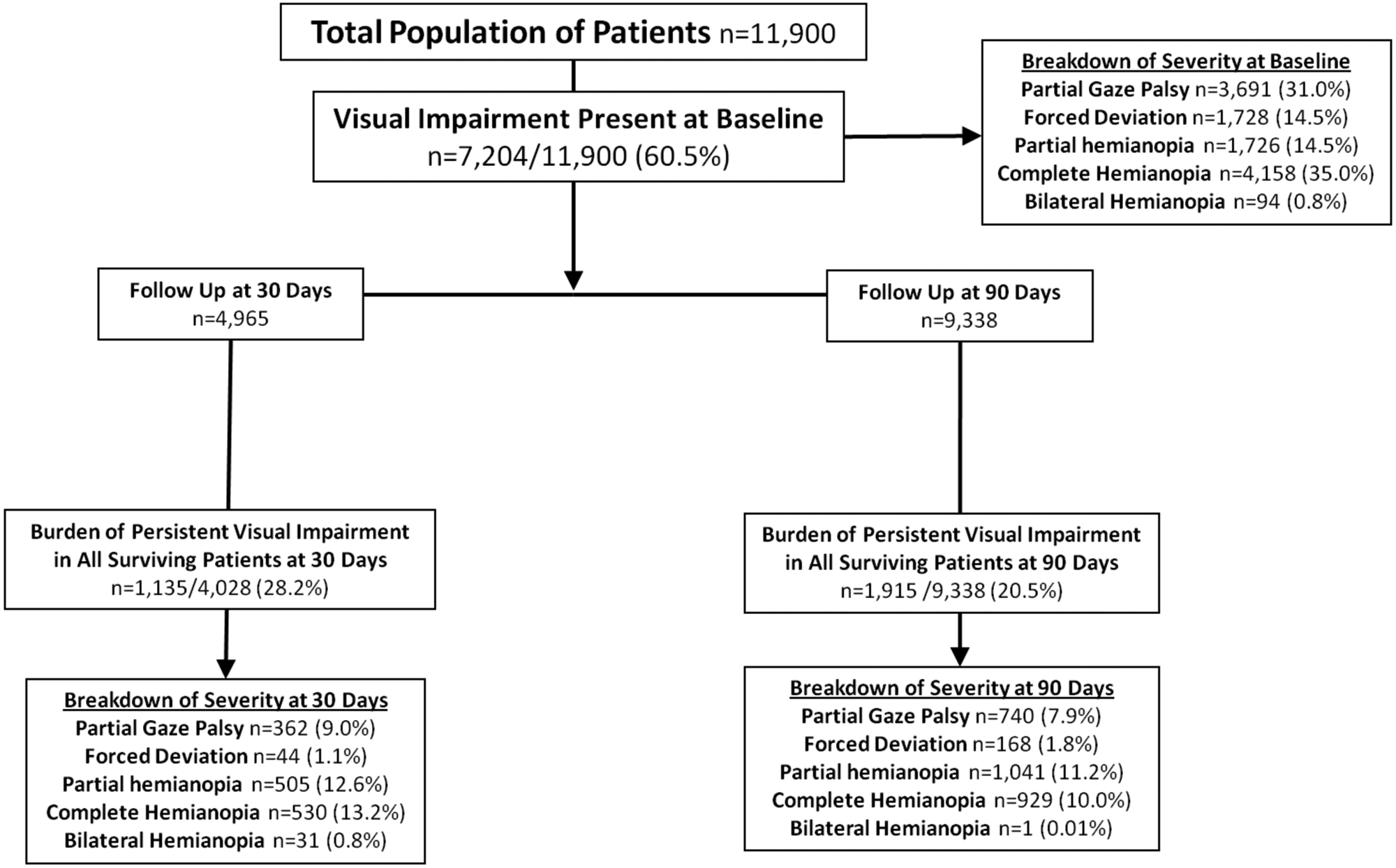
The burden of persistent Visual Impairment was 28.2% at 30 days and 20.5% at 90 days.
Data on extinction/inattention were available for 10,437 patients at baseline. Visual, tactile, auditory, spatial, or personal inattention was present in 2,887/10,437 (27.7%) patients, and 2,240/10,437 (21.5%) had profound hemi-inattention of more than 1 modality. All patients with visual impairments at baseline also experienced motor impairment.
Visual impairment at 30 days
Data on NIHSS domain scores at 30 days poststroke were recorded in 4,965 patients. Of these patients, 742 had died between admission and 30 days. Visual impairments had resolved in 1,398/3,285 (42.6%) patients by 30 days. However, the burden of persistent visual impairments in surviving patients at 30 days was 1,135/4,028 (28.2%, Figure 1). At 30 days, complete homonymous hemianopia affected 530/4,028 (13.2%) patients, whereas partial homonymous hemianopia affected 505/4,028 (12.6%) patients.
Of those who presented with forced deviation at baseline, 380/515 (73.8%) had recovered by 30 days, and forced deviation had evolved to partial gaze palsy in 110/515 (21.4%) patients at 30 days (Figures 2a-2c).
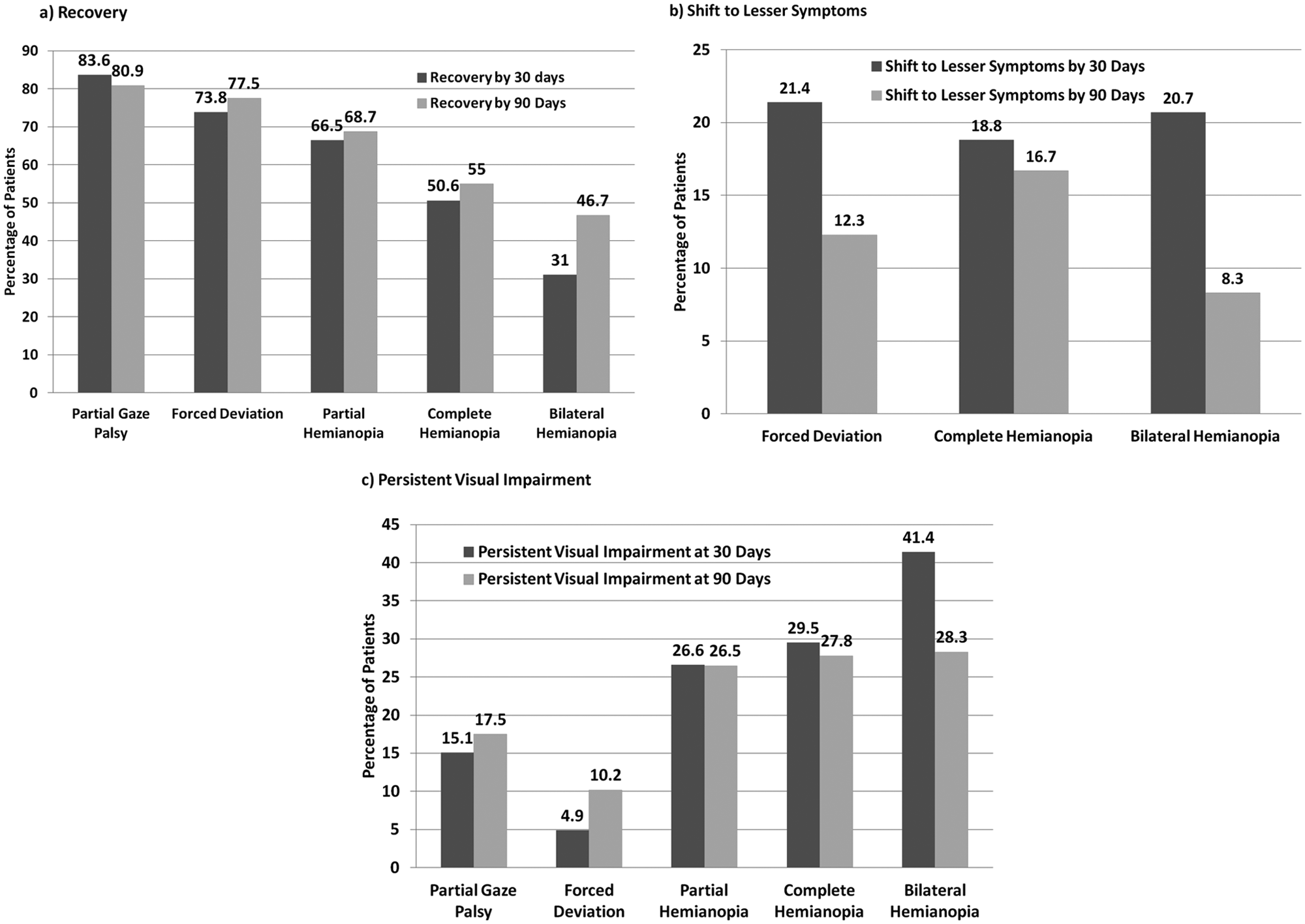
Recovery from horizontal eye movement disorders and hemianopias were greatest within the first 30 days after stroke, and continued at a much slower rate up to 90 days.
Of those who presented with complete homonymous hemianopia at baseline, 752/1,486 (50.6%) had recovered by 30 days, 439/1,486 (29.5%) had persistent complete homonymous hemianopia, and 279/1,486 (18.8%) had shifted to lesser symptoms by 30 days (Figures 2a-2c).
Visual impairment at 90 days
By 90 days, 2,118/11,900 (18.0%) patients had died, and 212/11,900 (1.8%) patients were lost to follow-up. Visual impairments had resolved in 3,243/7,204 (45.0%) patients, but the total burden of persistent visual impairments at 90 days in all stroke survivors was 1,915/9,338 (20.5%, Figure 1).
Of those patients who presented with partial gaze palsy at baseline, 2,154/2,664 (80.9%) had recovered and 465/ 2,664 (17.5%) had persistent partial gaze palsy at 90 days. Of 2,848 who presented with complete homonymous hemianopia at baseline, 1,567 (55.0%) had recovered, 477 (16.7%) exhibited lesser symptoms (partial homonymous hemianopia), and 793 (27.8%) had persistent complete homonymous hemianopia at 90 days (Figures 2a-2c). Recovery from visual impairments improved between baseline and 90 days (Figure 3).
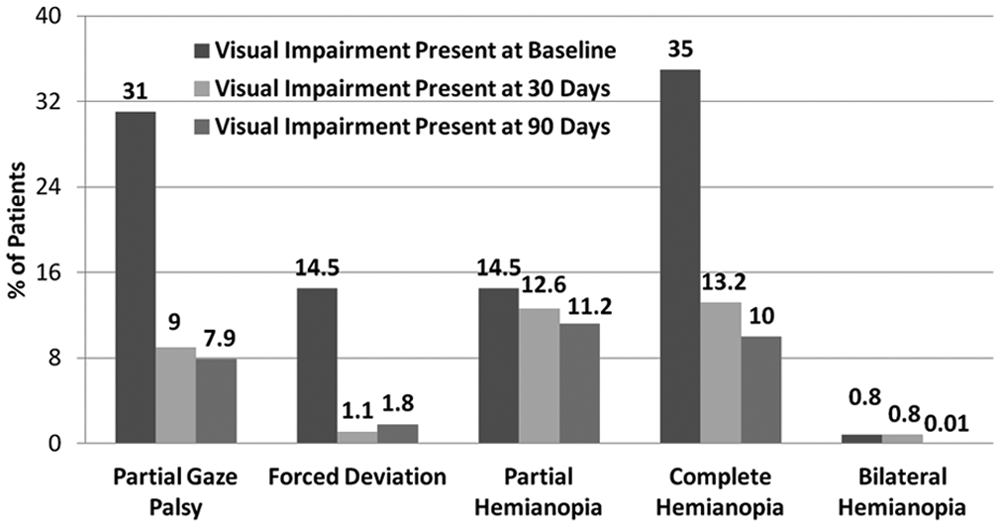
Complete homonymous hemianopia was the most common visual impairment at baseline and persisted in the highest proportion of patients at 30 days. Partial Gaze Palsy, though present in 31% at baseline, was only present in 9% at 30 days and 7.9% at 90 days.
Of 3,243 who recovered from visual impairments at 90 days, 304 (10.6%) patients had visual, tactile, auditory, spatial, or personal inattention, whereas 53 (1.9%) had profound hemi-inattention to more than 1 modality; our data did not allow us to separate visual from tactile, personal, spatial, or auditory inattention.
Recovery between 30 and 90 days
In our analysis data set 1,567/9,338 (16.8%) patients presented with complete homonymous hemianopia at baseline and had recovered by 90 days, compared with 752/4,028 (18.7%) who had recovered by 30 days. Furthermore, 2,154/9,338 (23.1%) patients presented with baseline partial gaze palsy and had subsequently recovered at 90 days, compared with 1,046/4,028 (26.0%) who had recovered by 30 days.
Association between recovery and clinical and demographic variables
Treatment with thrombolytics improved recovery from visual impairments at 90 days (P < .0001; odds ratio [OR] = 2.6; 95% confidence interval [CI] = 2.2-3.0). In surviving patients, older age (P < .0001; OR = 0.99; 95% CI = 0.98-0.99), a history of prior stroke (P ≤ .0001; OR = 0.70; 95% CI = 0.60-0.81), or diabetes (P = .002; OR = 0.78; 95% CI = 0.67-0.92) were associated with poorer recovery from visual impairments.
Functional Outcome
The presence of visual impairments was associated with poor functional outcome by mRS (Figure 4a). More patients with visual impairments at baseline had died by 90 days (25.6% vs 7.1%). Complete homonymous hemianopia (P < .0001; OR = 0.67; 95% CI = 0.67-0.73), partial gaze palsy (P < .0001; OR = 0.81; 95% CI = 0.74-0.87), and forced deviation (P < .0001; OR = 0.48; 95% CI = 0.43-0.53) at baseline were associated with poor mRS scores after adjusting for age, type of stroke, rt-PA administration, vision-unrelated NIHSS domain scores, and medical history (Table 1). Visual impairments at 90 days were significantly associated with poor mRS scores at 90 days; preexisting visual impairments did not significantly influence mRS scores at 90 days.
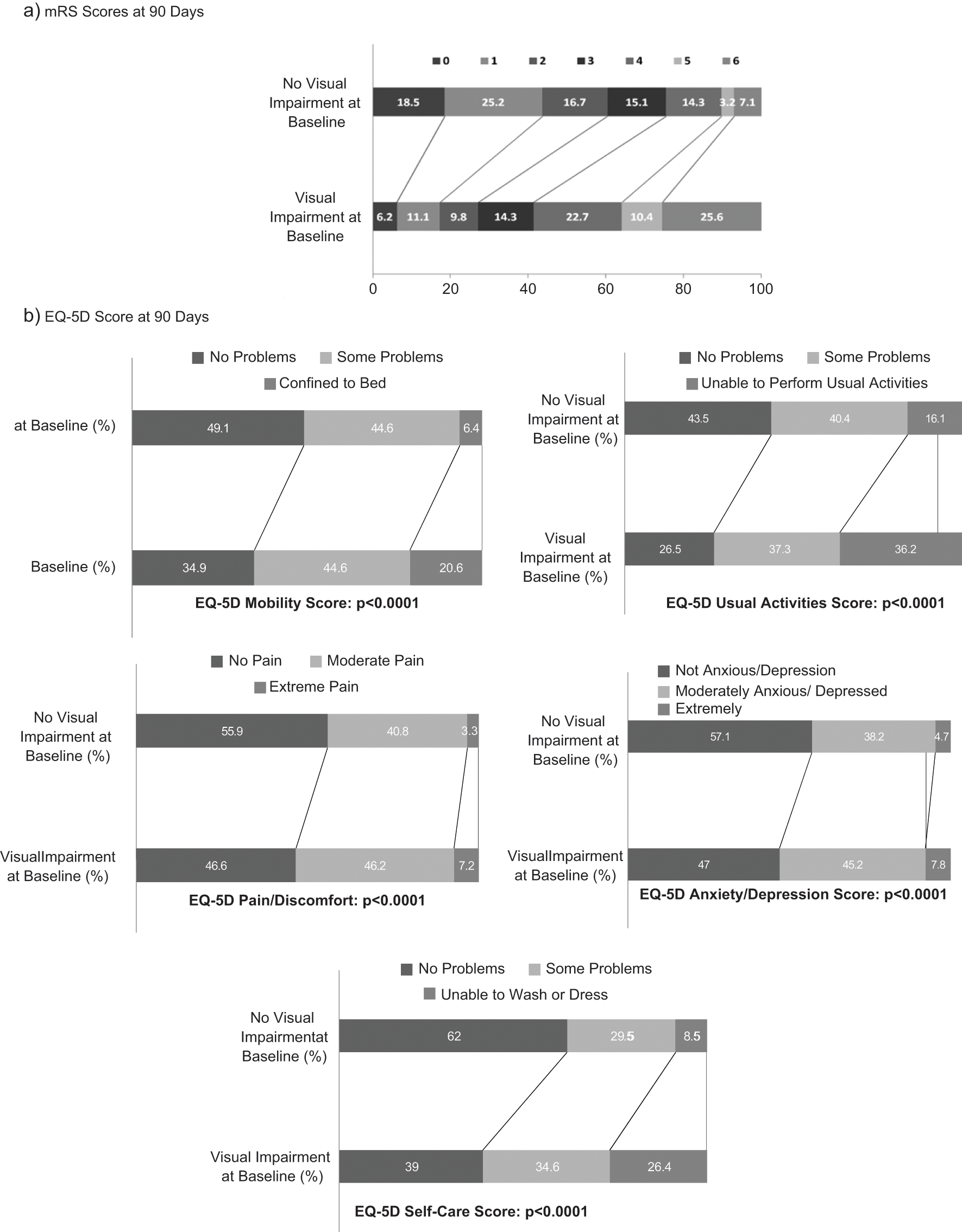
Functional outcome was better in patients who did not present with visual impairments at baseline compared to those who presented with visual impairments.
Association Between VIs and mRS at 90 Days
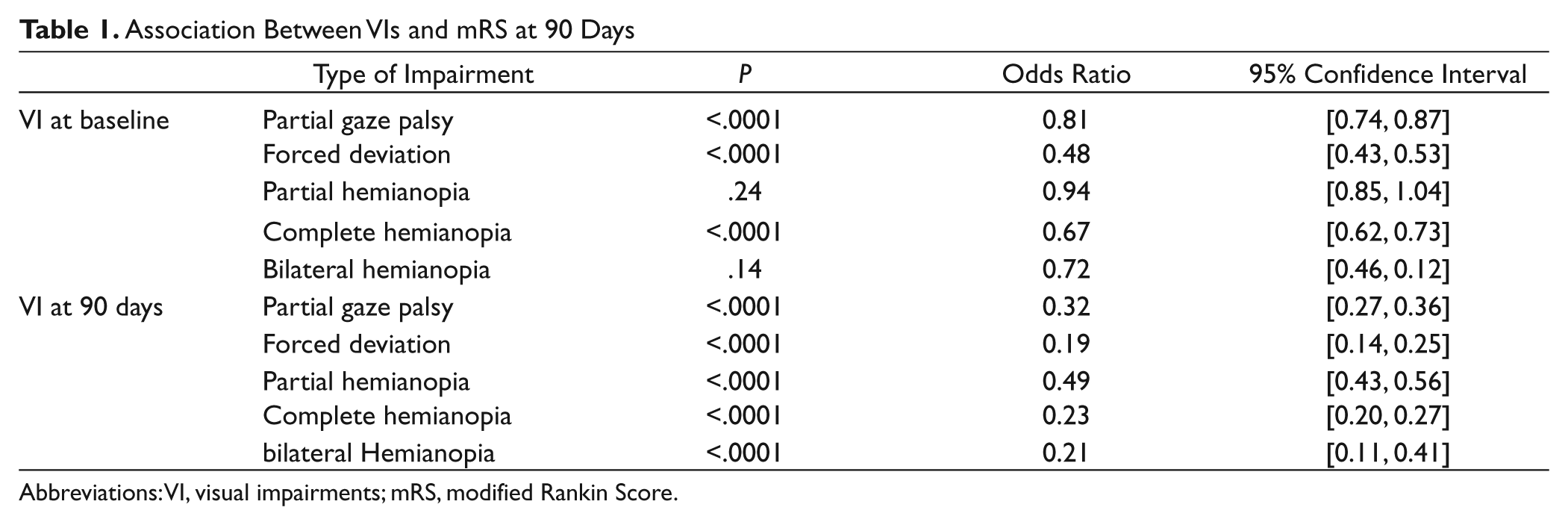
Abbreviations: VI, visual impairments; mRS, modified Rankin Score.
Quality of Life
A subset of 3,859 surviving patients had completed the EQ-5D assessment at 90 days. In this subgroup, 2,059 (53.4%) had baseline visual impairments, and the burden of persistent impairment at 90 days was 554/3,859 (14.4%). Of those patients who completed EQ-5D, 2,994 (77.9%) completed the questionnaire themselves, whereas 852 (22.2%) required assistance from a proxy. The presence of visual impairments at baseline was associated with poorer quality of life by EQ-5D, after adjusting for age, rt-PA administration, vision-unrelated NIHSS domains, and medical history variables (Figure 4b).
Specifically, forced deviation at baseline was associated with poorer scores on all domains of EQ-5D except anxiety/depression (Table 2). Complete hemianopia was associated with poor self-care and usual activities. The presence of any visual impairment at 90 days was associated with a poor outcome on almost all domains of the EQ-5D at 90 days (Table 2).
Association Between VI and EQ-5D at 90 Days
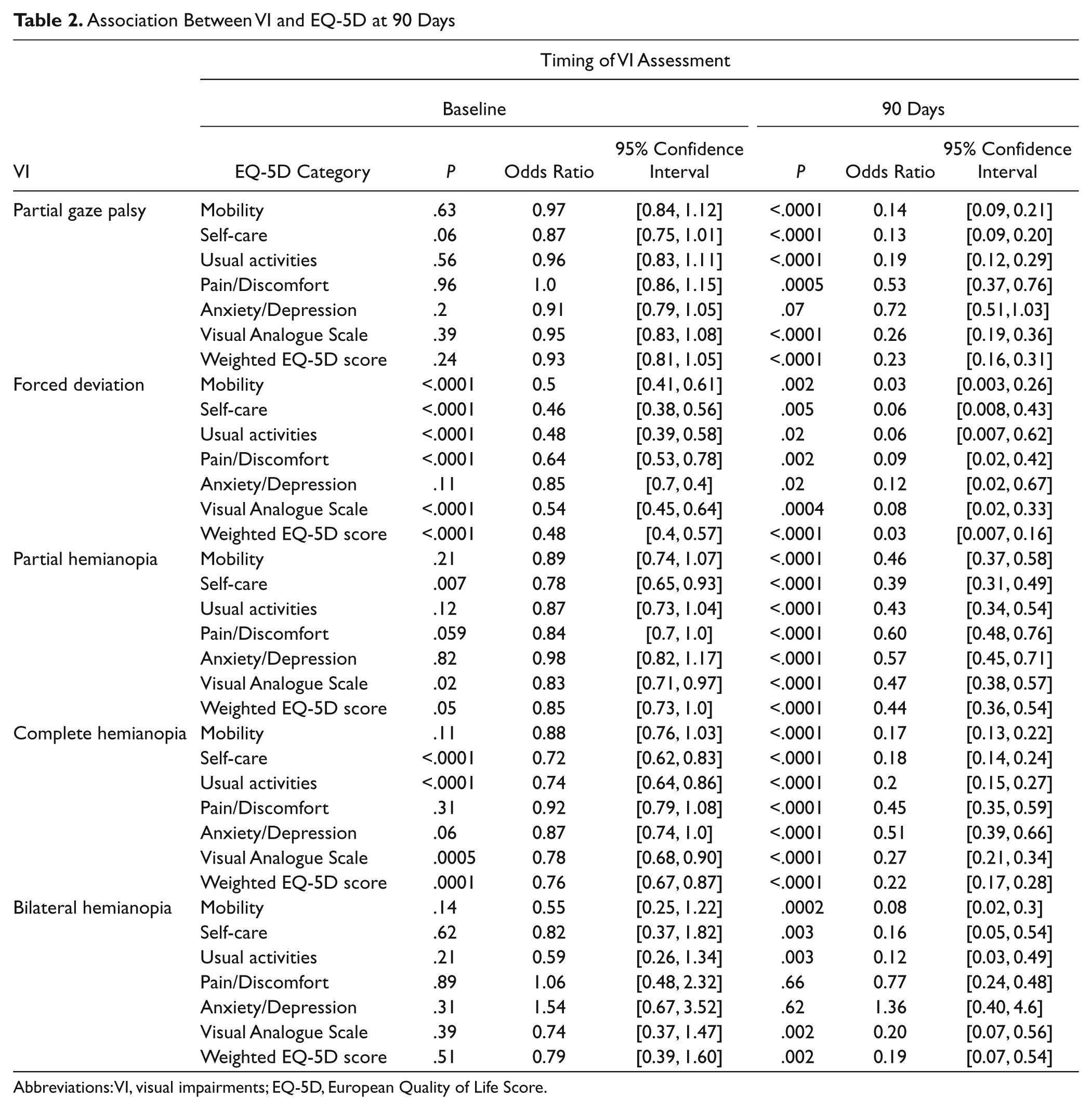
Abbreviations: VI, visual impairments; EQ-5D, European Quality of Life Score.
Discussion
In this retrospective analysis of a very large data set amassed from several large clinical stroke trials, we found that visual impairments persisted in 28.2% of patients at 30 days and 20.5% at 90 days after stroke. Recovery from visual impairments appeared to be greatest within 30 days of stroke. Horizontal eye movement disorders and complete homonymous hemianopia at baseline were significantly associated with poor mRS scores at 90 days, and treatment with thrombolytics was associated with improved recovery from visual impairments.
Previous estimates of recovery from visual impairments range between 7% and 85%.17-21 Prior studies, however, included patients with different demographic and neurological characteristics, such as trauma, stroke, tumor, and acquired brain injury23,29 or involved much smaller populations.4,5 Our study included patients with stroke in all vascular territories using a much larger sample size. We described the evolution of post-stroke eye movement disorders and hemianopia using a very large, robust data set, adjusting for numerous confounding factors. This study is unique in the size of the data set and the types of visual impairments investigated.
Gray et al 4 reported that at 28 days post-stroke, 14/81 (17%) patients with complete homonymous hemianopia at baseline had recovered from this impairment. Similarly, we found that 18.7% of patients in our sample presented with complete homonymous hemianopia at baseline and had subsequently recovered by 30 days. Zhang et al 23 2006 reported that 101/254 (38.4%) patients demonstrated spontaneous recovery from visual field defects at 1 month follow-up; however, they included patients with homonymous hemianopia resulting from trauma, tumor, or other etiology in addition to stroke, which may account for this difference.
There is no consensus as to when the maximal recovery from visual impairments occur. Visual restitution training may enhance recovery even years after initial injury; however, these studies have involved patient populations that were not restricted to stroke.25,30 Zhang et al 23 observed that the likelihood of recovery from hemianopia decreased with time. We observed that the majority of recovery from visual impairments occurred within the first 30 days post-stroke, consistent with previous observations.4,23,31 This may inform the timing of interventions for poststroke visual impairments.
A key strength of our analysis is the robustness of our data source: the VISTA repository includes government and commercially sponsored trials that involved rigorous monitoring and source data verification to meet worldwide regulatory standards. Randomized controlled trials seldom use specifically and prospectively designed neuro-ophthalmic assessment tools for visual impairments; rather, we inferred the presence of these impairments from those NIHSS sub-items that include elements of visual assessment. Detailed assessment of visual impairment is often infeasible in an acute stroke setting, particularly in the first 24 hours poststroke. Furthermore, this specialized assessment would likely only be possible in a very small subset of patients—for example, in those able to sit unsupported, who are not cognitively impaired, and who can participate in automated perimetry. The NIHSS contains 2 domains that assess hemianopia and eye movement disorders at the bedside in an efficient manner, albeit lacking sensitivity to the minutiae of diagnosis and recovery. The structure and components assessed within the NIHSS were developed in collaboration with stroke experts, and content validity has been demonstrated. 32 In addition, the NIHSS is the recommended neurological scale for stroke trials, 33 is widely used, and training materials and certification are available to ensure consistency of scoring on each of these domains.34,35 These practices should reduce the potential for variability in raters’ assessments.
We acknowledge that our results may underestimate the full degree of clinically meaningful recovery because we used a broad method for assessment of visual impairment. We reported the shift from complete to partial hemianopia; smaller changes may also be meaningful to the stroke survivor. Nevertheless, because neuro-ophthalmic assessments are rarely feasible in an acute stroke setting and because the NIHSS has been rigorously developed and tested, we consider the NIHSS Best Gaze and Visual domains to be valid and useful surrogates to describe the evolution of horizontal eye movement disorders and hemianopia by 3 months post-stroke.
Our data did not allow us to separate visual from tactile or auditory inattention, and only patients with either horizontal eye movement disorders or hemianopia were detected by the NIHSS, omitting other visual impairments such as vertical eye movement disorders and scotomata excluding hemianopia. Additionally, hemianopia was tested using confrontation, which may lack sensitivity to small areas of visual field loss. Nevertheless, Johnson and Baloh36 examined the accuracy of visual field assessment using confrontation compared with automated perimetry. Good specificity and high positive and negative predictive values indicated that confrontation accurately identified the presence of visual field defects.
Furthermore, patients who are typically enrolled in randomized controlled trials of acute stroke interventions are not completely representative of the general stroke population. These patients typically present at hospital within the first 6 hours of stroke onset and, to be eligible for inclusion in acute intervention trials, are usually conscious at baseline, demonstrate limb weakness and possible higher-cortical dysfunction, and typically score greater than 5 points on the NIHSS. These criteria therefore potentially include a greater number of patients with visual impairment than might be anticipated within the general stroke population; patients with milder symptoms may be excluded.
The true rate of visual impairments in unselected stroke patients may differ; however, we expect the rate of recovery from stroke-related visual impairments in similarly impaired patients to be comparable. Further work to elucidate the prevalence of visual impairments could include prospective data collection from patients who underwent specialized assessment for visual impairments in the acute stroke setting.
Visual impairments may also further exacerbate poor motor recovery by restricting participation, thus diminishing the benefits of rehabilitation. A relationship between the presence of eye movement disorders and poor mobility at 90 days is to be expected because there is a known association between large infarction and motor deficits.37 This association could therefore be explained by the location and size of the stroke. As we did not have access to imaging data describing the location of stroke and lesion volume, we were unable to adjust our analysis for these variables. Nevertheless, we were able to adjust for other factors that influence recovery from visual impairments such as age, medical history, vision-unrelated NIHSS domain scores, the type of stroke, and thrombolysis. Our results were consistent with those of Pambakian and Kennard 24 who reported a negative correlation between the degree of visual field recovery, age, and a history of diabetes.
We found that 20.5% of patients in our data set continued to experience a visual impairment at 3 months. This is arguably a major stroke-related impairment that may influence outcome and is thus worthy of detailed investigation in both stroke research and clinical settings. Whether the noted functional impairment can be attributed to visual impairments per se or to another deficit that is statistically associated with visual impairments, such as motor impairment, cannot be addressed in our data. Additional studies may better establish the mechanistic link between visual impairments and functional disability.
Footnotes
Acknowledgements
VISTA Steering Committees: VISTA-Acute: K. R. Lees (Chair), A. Alexandrov, P. M. Bath, E. Bluhmki, L. Claesson, S. M. Davis, G. Donnan, H. C. Diener, M. Fisher, B. Gregson, J. Grotta, W. Hacke, M. G. Hennerici, M. Hommel, M. Kaste, P. Lyden, J. Marler, K. Muir, R. Sacco, A. Shuaib, P. Teal, N. G. Wahlgren, S. Warach, and C. Weimar. VISTA-Rehab: M. Brady (Chair), M. Ali, A. Ashburn, D. Barer, J. Bernhardt, A. Bowen, E. Brodie, S. Corr, A. Drummond, J. Edmans, C. English, J. Gladman, T. Hoffmann, L. Kalra, S. Kuys, P. Langhorne, K. R. Lees, N. Lincoln, P. Logan, L. Jongbloed, G. Mead, A. Pollock, V. Pomeroy, H. Rodgers, C. Sackley, L. Shaw, D. J. Stott, K. S. Sunnerhagen, S. Tyson, P. van Vliet, M. Walker, and W. Whiteley. VISTA-Imaging: S. Warach (Chair), G. Albers, S. Davis, G. Donnan, M. Fisher, T. Furlan, J. Grotta, W. Hacke, C. Kidwell, W. Koroshetz, K. R. Lees, M. Lev, D. Liebeskind, G. Sorensen, V. Thijs, G. Thomalla, J. Wardlaw, and M. Wintermark. VISTA-ICH: D. F. Hanley (Chair), B. Gregson, S. Davis K. R. Lees, P. Lyden, K. Muir, T. Steiner, and S. Mayer. VISTA-Plus: N. G. Wahlgren (Chair), C. Weimar, C. Molina, H. Numminen, K. R. Lees, and G. Tsivgoulis. VISTA-Prevention: H.-C. Diener (Chair), K. R. Lees and B. Ovbiagele.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: MA, MB, AP, and the Nursing, Midwifery and Allied Health Professions Research Unit are funded by the Chief Scientist Office (CSO) Scottish Government’s Health Directorate, Scotland. CH has been awarded a junior research training fellowship from the Stroke Association. This work was funded as part of a CSO postdoctoral fellowship award. The views expressed here are those of the authors and not necessarily those of the CSO.
VISTA is a not-for-profit collaboration of researchers from academia and commercial organizations. The VISTA Steering Committee members have each contributed to the organization of contributing trials, and where these have involved industry support, they have acknowledged that within the original publications.
