Abstract
Background. Constraint-induced movement therapy (CIMT) is used to counteract learned nonuse observed following stroke in humans and has been shown to improve function. Variations of CIMT used in animal models of stroke have the potential to inform and improve our understanding of this intervention. Objective. To conduct a systematic review of studies investigating constraint in experimental stroke. The authors aimed to assess the quality and establish the efficacy of constraint on neurobehavior, cognitive function, infarct size, and stress and mortality and to determine the optimal dose or time to administration. Methods. Systematic review with meta-analysis was used. Data were analyzed using DerSimonian and Laird weighted-mean-difference random effects meta-analysis. Results. The quality scores of the 8 articles (15 studies) included were moderate (median 5/10; interquartile range, 4.8-6.0). There was a trend for animals with constraint to have worse neurobehavioral scores (−10% worse; 95% confidence interval [CI] = −20 to 0; P = .06). Infarct volumes were not significantly different between groups (−3% larger with constraint; 95% CI = −15 to 9; P = .63). Cognitive function was significantly better after constraint, although this estimate was based on only 28 animals from 2 studies. Insufficient data prevented analysis of the effect of constraint on stress and mortality. Conclusions. This meta-analysis showed no benefit of constraint on neurobehavioral scores, which is at odds with some human studies. Animal models may help us efficiently explore the biological basis of rehabilitation interventions; however, review of the data in this study raise uncertainty about its effectiveness in humans.
Introduction
Weakness, impaired sensation, loss of dexterity, and spasticity are examples of impairments of the upper limb that contribute to disability after stroke. Upper-limb dysfunction persists beyond 6 months in 38% of people after stroke, with arm dysfunction being the second most important cause of stroke-related disability. 1 As a result of upper-limb impairments, many disabled persons come to rely on their unaffected upper limb for performing daily activities. This adaptive behavior, also known as learned nonuse, often continues despite the capacity for movement in the affected limb.
Following experimental observations made on de-afferented monkeys, Taub described a phenomenon of “learned non-use” or “learned helplessness” after central nervous system injury, in which animals avoided use of the affected upper limb long after movement had returned. 2 Negative reinforcement from unsuccessful use of the affected upper limb and positive reinforcement from successful use of the unaffected upper limb during this early phase combines to result in this behavioral dysfunction. 2
Restraining the unaffected upper limb to force use of the affected upper limb in normal daily activities and during rehabilitation was pioneered by Ostendorf and Wolf. 3 This is a key feature of what is referred to in experimental studies and clinical rehabilitation as forced use, constraint, or constraint-induced movement therapy (CIMT). 4 The large EXCITE randomized controlled trial 5 showed sustained improvements in motor function and use of the affected upper limb, compared with no therapy, following 2 weeks of restraint plus 6 h/d of task-specific upper-limb training. Subsequent clinical trials have yielded mixed results.6-8
Restraint of the unaffected upper limb to force use of the affected upper limb—or more generally, constraint—has also been applied in animal models of stroke. Meta-analyses of animal studies has yielded important information not apparent from analyses of the individual studies. 9 Our aim was to conduct a systematic review and meta-analysis of animal models of stroke to determine study quality and the effect of constraint on neurobehavior, cognitive function, infarct size, stress, and mortality. Furthermore, we wished to determine the optimal dose and timing of administration of constraint.
Methods
Identification of Studies
Searches were performed in January 2010 and updated in April 2011. Electronic databases MEDLINE, BIOSIS, Web of Science, and Embase were searched for English-language publications using the following strategy to identify any poststroke training used in animal models of stroke: (stroke OR CVA OR cerebral (ischemia OR ischaemia) OR cerebrovascular accident OR MCA occlusion OR middle cerebral artery occlusion OR brain (ischemia OR ischaemia) OR (ischemic OR ischaemic) stroke OR experimental brain infarction OR focal brain (ischemia OR ischaemia) OR experimental stroke OR experimental brain (ischemia OR ischaemia)) AND (therapy OR skilled reaching OR train* OR rehabilitation OR motor task OR exercise OR learning OR physical OR motor activity OR constraint OR forced use) AND (recovery OR outcome OR rehabilitation OR compensation OR neuroplasticity OR function OR functional OR plasticity OR adaptation OR (behavior OR behaviour) OR infarct volume OR lesion size OR mortality OR efficacy OR cognition) NOT (cerebral palsy OR hyperbaric oxygen OR heatstroke OR epilepsy OR obesity OR myocardial OR cardiac OR renal OR cancer OR spinal cord injury OR human). LIMIT: Animals. Additionally, reference lists of retrieved relevant publications were manually searched to check for any omissions.
Inclusion Criteria
Interventions that restrained the unaffected limb of a stroke-affected animal to force use of the affected limb were considered for inclusion. Studies were included if they investigated constraint in animal models of focal cerebral ischemia or intracerebral hemorrhage, were controlled, and contained data on any of the outcomes of interest. Neurobehavior (score on a test examining sensorimotor function) was the primary outcome. Secondary outcomes were cognitive function (learning and/or memory) infarct size (volume or area), stress, and mortality. Animals that did not receive any form of therapy were designated as the control group.
Data Extraction
Titles and abstracts of references retrieved from all databases were merged then screened by independent reviewers (SS and HJ). Studies that failed to meet the previously outlined criteria were excluded. Uncertainty or differing opinions between reviewers regarding eligibility were addressed after obtaining a full list of publications. A third independent reviewer (NJS) determined eligibility in those circumstances where it was still not possible to reach a consensus. For each comparison and for each control and treatment group investigators (SS and HJ) identified and extracted data according to the standard methodology used in previous published meta-analyses of animal stroke studies.10,11 Pertinent data, including species, time of commencement of the constraint relative to stroke, time of assessment, housing conditions of animals, type of ischemia, duration of ischemia (for temporary occlusions), method of ventilation, method of anesthetic induction, dose (h/d) and length (days) of constraint, method of limb constraint, and other features of constraint (ie, additional reach training), were extracted for the purpose of exploring the effect such variables had on each outcome via stratified analyses. For each control and treatment group in each study, investigators (SS and HJ) identified and extracted the number of animals per group, mean outcome, and standard error or standard deviation. Where data were presented serially, only data from the last time point were included in the analyses. If data were only presented graphically, attempts were made to obtain data from authors; if this was not available, values were measured from the published graphs.
In the interests of increasing numbers and statistical power, animals that were exposed to a physical or cognitive intervention in addition to constraint (a cotreatment) were included where both groups were exposed to the cotreatment. The presence of a cotreatment was recorded. Multiple articles (repeat publications) of the same study were noted, and all relevant data were allocated to the original group of animals. When a single control group served multiple treatment groups, the size of the control group entered into the meta-analysis was adjusted by dividing by the number of treatment groups served.
Data from animals that died during surgery or in the immediate postsurgical period were excluded from all calculations. Data from animals that died after treatment allocation were used in the estimate of mortality. Data from animals that were removed retrospectively on the basis of small histological lesion (postintervention) were excluded for the estimate of infarct size.
To minimize the loss of statistical power that results from the analysis of multiple outcomes, a single measure of escape latency (in seconds) to reach the hidden target from the Morris Water Maze test was chosen for the outcome of cognitive function.
Quality was assessed against the Collaborative Approach to Meta Analysis and Review of Animal Data from Experimental Stroke (CAMARADES) study quality checklist, 12 which is a well-established measurement tool incorporating important features of experiment validity.9,13
Statistical Analysis
All meta-analyses were performed using the DerSimonian and Laird random-effects model. Continuous data (neurobehavior, cognitive function, learning, and stress) are presented as weighted mean difference (WMD) and 95% confidence intervals (CIs). Dichotomous data (mortality) are presented as odds ratios (ORs) and 95% CIs. The significance level was set at P < .017 to allow for multiple comparisons. Heterogeneity for each data set was examined using the I2 statistic, 14 and the significance level was set at P < .05. Stratified meta-analyses to identify possible sources of heterogeneity were performed on aspects of study quality and design in heterogeneous data sets. Publication bias assessment was planned using the using the Egger method. 15 Results for each of the outcomes are presented in forest plot graphs, where the size and location of the point represents the number of animals in each study and the mean treatment effect, respectively, with the horizontal lines on either side of the point representing the 95% CIs.
Results
Flow of Studies Through the Review
A total of 3577 articles were returned from the electronic database search (Figure 1); 8 articles (15 studies) met the inclusion criteria and contained complete data sets (Table 1). The median quality score was 5/10 (interquartile range [IQR] = 4.8-6.0). The majority of articles (n = 7) reported blinded assessment of outcome, and half (n = 4) reported randomization (see Table 2). No article included a statement regarding sample size calculations. The small number of studies that contributed to each outcome resulted in insufficient power to statistically assess publication bias.
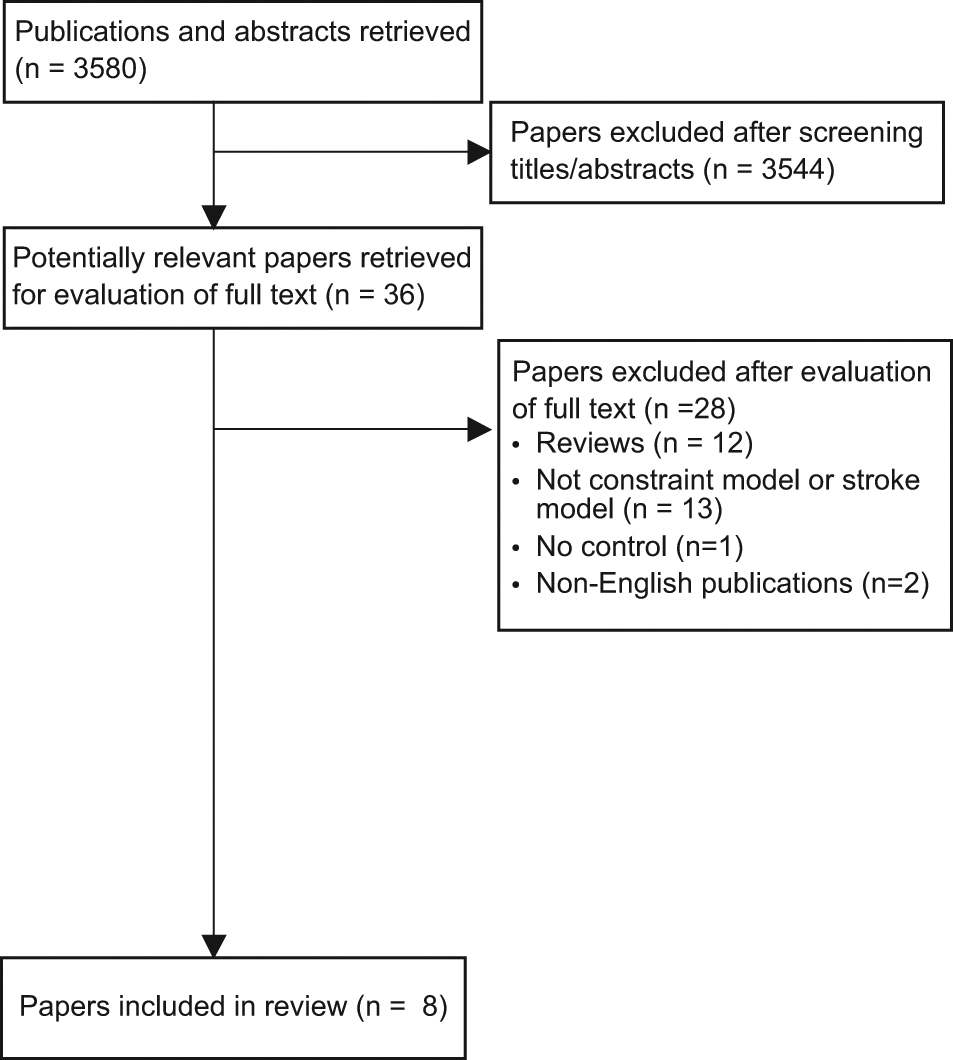
Flow of studies through the review.
Characteristics of Included Studies
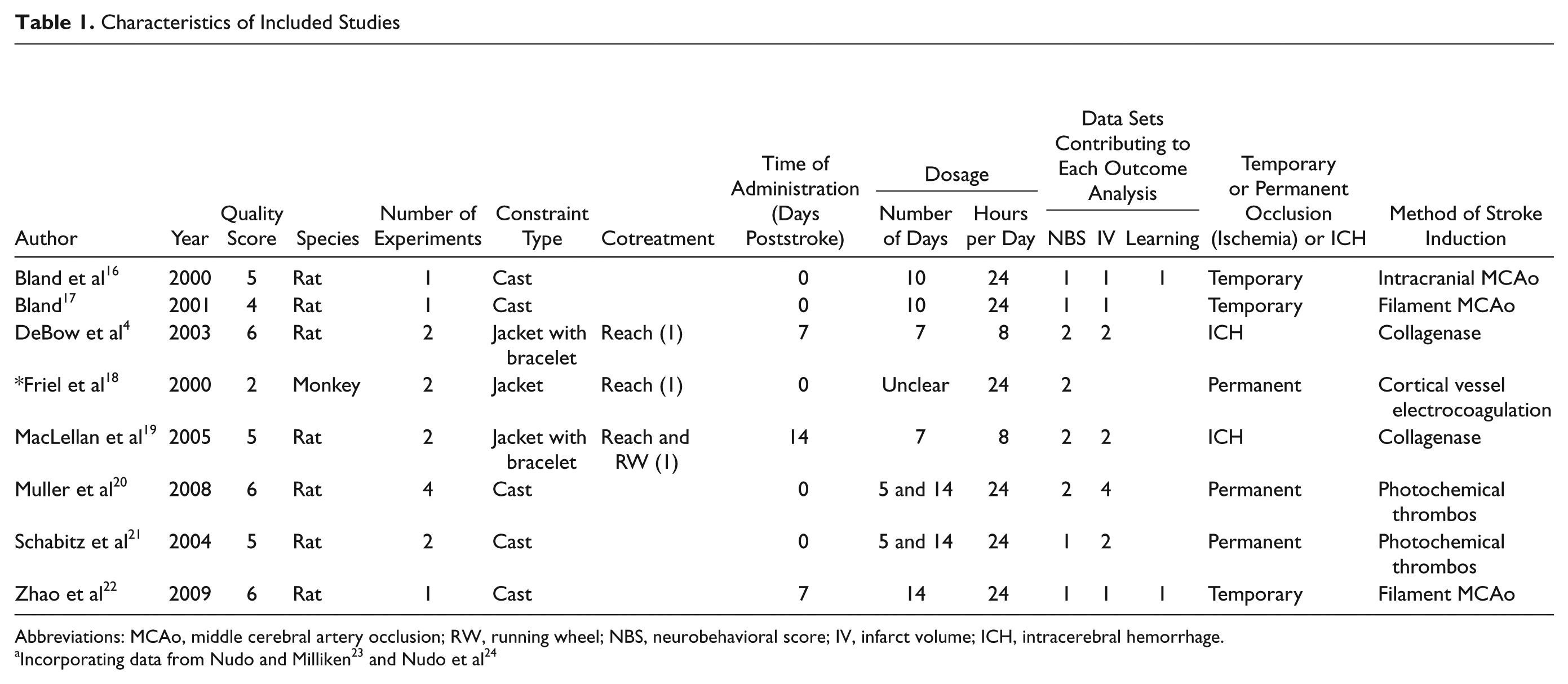
Abbreviations: MCAo, middle cerebral artery occlusion; RW, running wheel; NBS, neurobehavioral score; IV, infarct volume; ICH, intracerebral hemorrhage.
Quality of Included Studies a
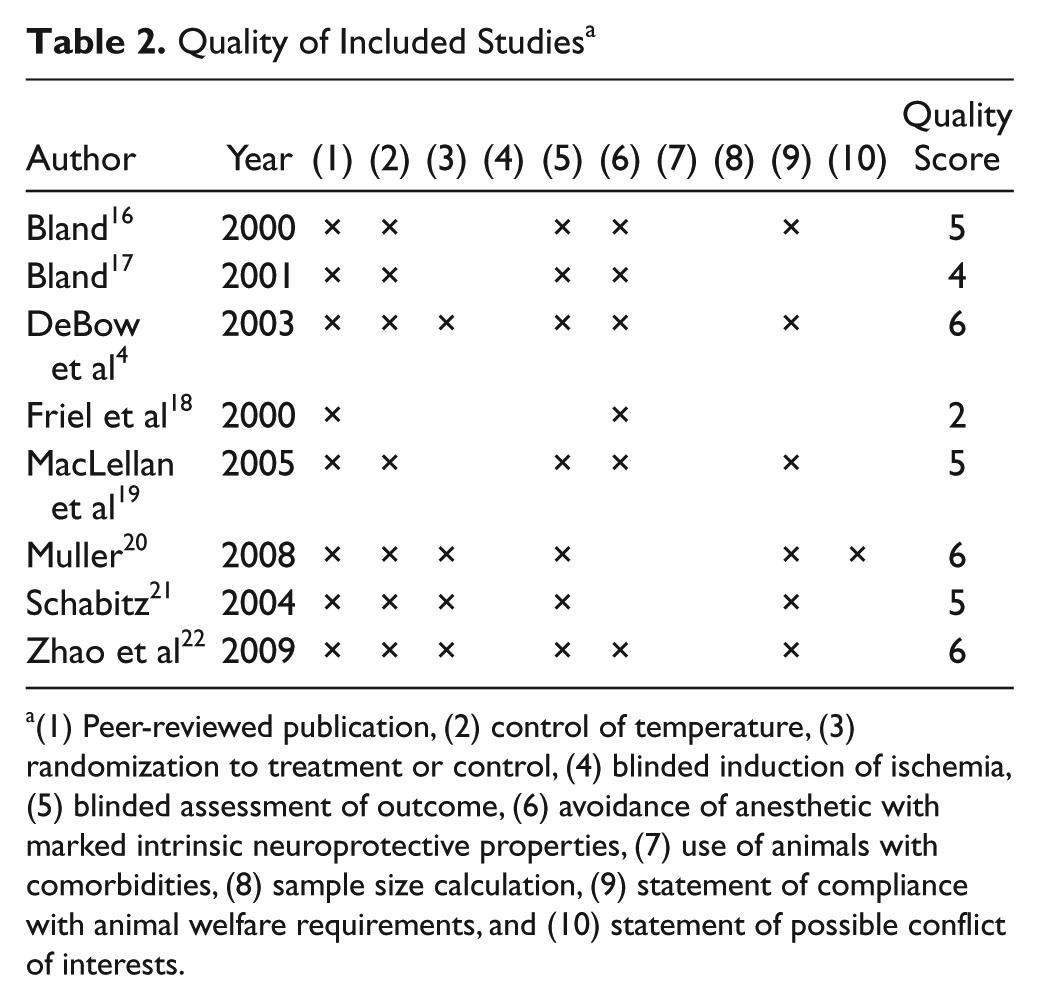
(1) Peer-reviewed publication, (2) control of temperature, (3) randomization to treatment or control, (4) blinded induction of ischemia, (5) blinded assessment of outcome, (6) avoidance of anesthetic with marked intrinsic neuroprotective properties, (7) use of animals with comorbidities, (8) sample size calculation, (9) statement of compliance with animal welfare requirements, and (10) statement of possible conflict of interests.
Similarity Between Studies
Studies varied across the 8 articles4, 16-22 (see Table 1). All but one 16 used rats; 2 studied models of intracerebral hemorrhage.4,17 Methods of constraint of the unaffected limb included jackets, jackets with bracelets, and plaster casts, of which plaster casts were the most commonly used (9/15). Time to commencement was either immediately (n = 10), 7 days (n = 3), or 14 days (n = 2) poststroke. Constraint dose ranged from 5 to 35 days (median = 10) and in all but 4 studies4,17 was applied for 24 h/d. The majority of studies investigated constraint alone (simply retraining the limb). Two investigated the effect of constraint in combination with running and reach training. 17 One article 16 incorporated results from 2 previous publications18,19 in their analyses to explore the efficacy of providing reach training in conjunction with constraint. Housing conditions of animals, method of stroke induction, and the neurobehavioral tests used varied between studies.
Effect of Constraint on Neurobehavior
Of the 15 studies from the 8 articles, 12 had complete data for neurobehavior (n = 173 animals). Neurobehavioral testing was performed only after cessation of the constraint in all studies. The most frequently used neurobehavioral tests were the cylinder (n = 5), staircase (n = 4), and ladder tests (n = 4). The median time point of assessment of neurobehavior used in the analysis was 42 days poststroke (IQR = 38-56). The median time point from cessation of constraint to assessment was 28 days (IQR = 28-42).
There was no significant difference in neurobehavior between groups; however, there was a strong trend toward worse performance in the constraint group (constraint group −10% worse than control; 95% CI = −21 to 0; P = .06). Heterogeneity was moderate (52%; Figure 2A). Stratified analysis revealed that study quality (P = .03), method of stroke induction (P = .01), time to commencement of constraint (P = .05), and time of assessment (P = .03) account for a significant proportion of between-study heterogeneity (Figure 2B).
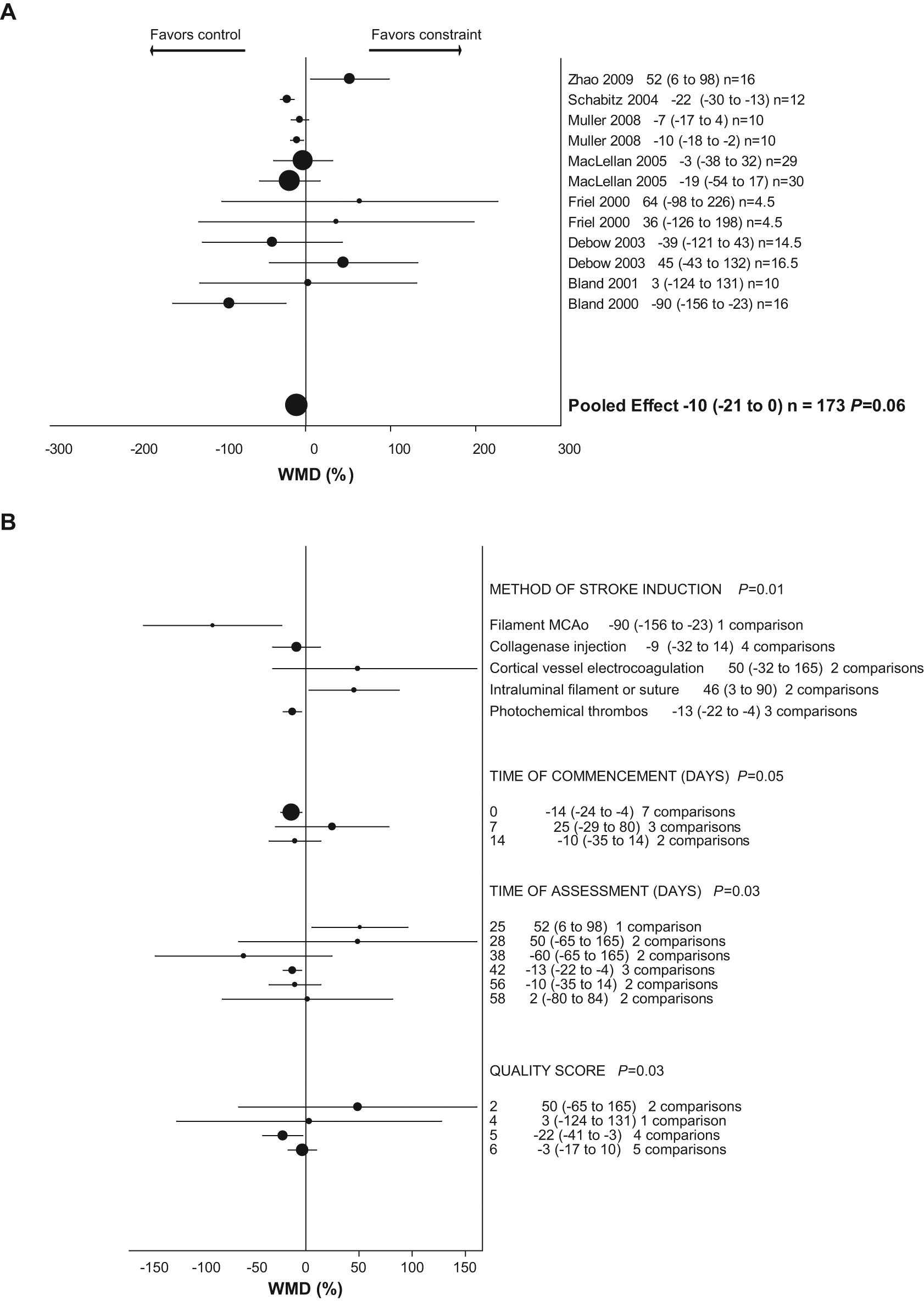
Weighted mean difference (WMD) as percentage (95% confidence interval) for effect of constraint on (A) neurobehavioral score and (B) neurobehavioral score, stratified analysis.
Effect of Constraint on Infarct Size
Of the 15 studies from the 8 articles, 13 had complete data for infarct size (n = 194 animals). All data presented lesion size as infarct volume (mm3). Time points of assessment ranged from 7 to 42 days poststroke. There was no significant difference in infarct size between groups (constraint group −3% larger than control; 95% CI = −15 to 9; P = .63). Heterogeneity was low (8%; Figure 3).
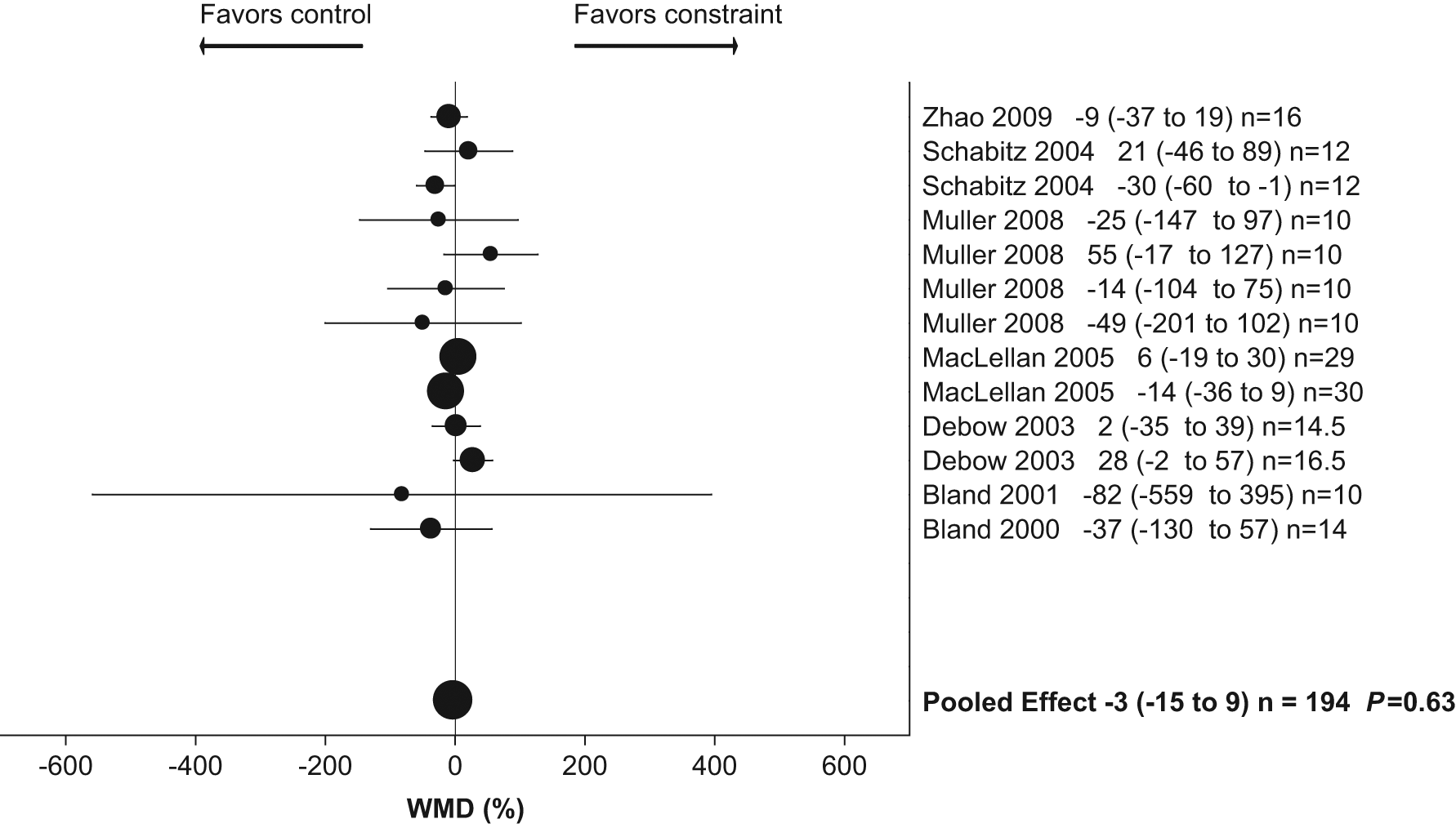
Weighted mean difference (WMD) as percentage (95% confidence interval) for effect of constraint on infarct volume.
Effect of Constraint on Cognitive Function
Of the 15 studies, 2 had complete data for cognitive function, of which both presented latency results from the Morris Water Maze Test (n = 28 animals).20,21 Animals with constraint poststroke had significantly better cognitive function (ie, shorter latency measures; constraint 50% better than control; 95% CI = 10-91; P = .02; see Figure 4). These data were homogeneous (I2 = 0%; P = .73).
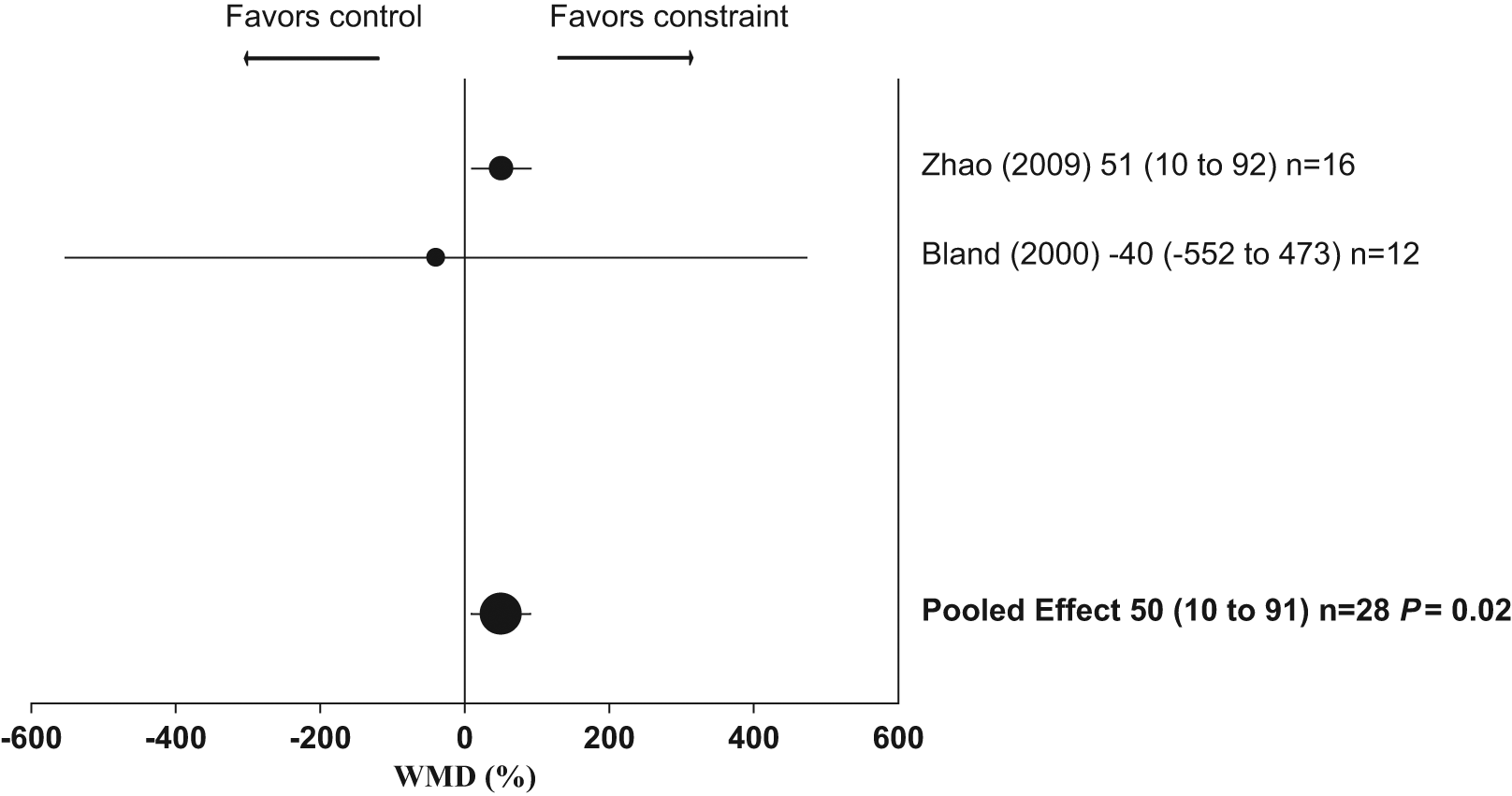
Weighted mean difference (WMD) as percentage (95% confidence interval) for effect of constraint on cognitive function.
Effect of Constraint on Stress and Mortality
Only 1 article presented data for stress. 20 Constraint in this study incorporated a plaster cast applied immediately postsurgery, with no additional exercise or task training. The cast remained on for 24 hours a day over 10 days. There was no significant difference in plasma corticosterone levels between control and constraint animals (n = 6 per group) during the period of constraint (tested at days 1, 4, 10). No article made clear statements about mortality during the intervention period.
Discussion
This is the first systematic review and meta-analysis of studies of constraint in animal models of stroke. Despite positive trials in humans showing that constraint combined with training can improve arm and hand use, 5 our results failed to mirror these findings. There were no statistically significant differences between groups for neurobehavior or infarct size and a trend toward worse neurobehavior in animals receiving constraint. The 95% CIs for this estimate excluded any scientifically significant benefit of constraint on neurobehavioral scores.
Cognitive function (spatial learning) was significantly better in animals treated with constraint. However, the small number of animals and large CIs suggest a high degree of uncertainty regarding the true effect (10%-91%). This result should be interpreted with caution. However, it is supported by evidence from human studies of structural changes within the hippocampus of stroke survivors following CIMT. 22 This highlights an area of constraint research that requires further investigation.
There were multiple sources of heterogeneity in this meta-analysis (study quality, method of stroke induction, time to commencement of constraint, time of assessment, and outcome measures). For example, across the 12 studies presenting data for neurobehavior, 11 different tests were used, making direct comparisons between studies more difficult. This heterogeneity raises the possibility that certain subgroups or types of intervention may be effective.
We were unable to determine whether there was a beneficial time to start constraint, or an optimal dose of constraint in animal models of stroke, limited again by the small number of studies and relatively small pool of animals. It is interesting to note that regarding time to commencement, the exploratory analysis suggested that there may be a relatively modest benefit of commencement at 7 days, compared with either 0 or 14 days (Figure 2B). There were only 3 comparisons (47 animals) commencing at this time, so this is hypothesis generating. Additional sources of heterogeneity included time of assessment and study quality. As would be anticipated based on previous animal studies, 23 an early time of assessment (25 days poststroke) appeared to result in greater benefit. Interpretation of the equivalent time point of assessment between humans and animal models is a vexed issue. Certainly stroke-affected rodents appear to make more rapid recovery than humans. However, this may be in part because many of the tests used assess motor function predominantly, whereas middle cerebral artery territory stroke in a rat predominantly affects the sensory cortex. 24 Whether current tests of recovery in animals are sensitive enough to detect mild but still significant deficits in behavior is open to question.
Only 1 study included in our analysis investigated the effect of constraint on stress and found no significant difference between plasma levels of corticosterone in control and treatment groups. 20 With only 12 animals in this study, it is likely that there was insufficient power to rule out a scientifically important effect of constraint on stress levels, and to some extent, this is an unavoidable potentially confounding effect of all such small animal studies. Future studies should attempt to measure stress effects longitudinally and, if possible, try to modify constraint paradigms to minimize stress in animal models.
The quality of included studies was moderate. Similar to findings of previous meta-analyses of stroke models, 25 there was a trend for greater benefit of the intervention (constraint) in studies of lower quality (Figure 2B). Unlike previously published meta-analyses of animal models of stroke,10,11 a considerable number of authors reported blinded assessment of outcome. However, only half the studies randomly allocated animals to groups. None reported the presence or absence of mortality after allocation to groups. Controlling potential sources of bias is now increasingly recognized as an essential prerequisite for the successful translation of animal studies to the clinic, as reflected in multiple recent guidelines.9,26 The studies analyzed herein were conducted prior to publication of these guidelines. It is hoped that researchers will incorporate these guidelines during design of animal models of stroke in the future.
Overall, the results of this meta-analysis of animal studies contradict some human studies showing beneficial effects of constraint—for example, the EXCITE trial. 5 A recent update of the Cochrane meta-analysis of human studies of constraint (8 trials, 276 participants), however, found no significant effect of CIMT on disability 6 but a preservation of a modest benefit for arm motor function. Although human trials of constraint may show mixed findings what is at issue here is whether animal studies can add to our understanding.
There are several problems modeling constraint in animals, raising the issue of how accurate such models can be. First, the majority of studies are performed on quadruped rodents, in which the primary function of the forelimb is for locomotion, unlike the situation for humans. Humans can choose to only use their unaffected upper limb; however, rodents need to use their affected limb to fully explore their environments.
Second, constraint in particular may be associated with higher degrees of stress in animals because unlike humans, animals presumably do not have any conception that they may derive benefit. Instead of using constraint in these typically quadruped animals, which are dependent on their forelimbs for locomotion to explore and get to food and drink, a more appropriate model to enhance limb movement in animal models might be induced movement therapy. This approach relies on tailoring of the environment with novel and complex stimuli (ie, environmental enrichment) to induce exploratory behavior and consequently limb movement. This approach would reduce the stress and other potential negative consequences of animal restraint (eg, slowing movement and possibly reducing food access from overhead pellet trays).
Third, unlike constraint research in humans, which uses very specific and standardized protocols, 27 constraint in many studies is highly varied. Standardized features of poststroke behavior (learned nonuse) and motor impairment as inclusion criteria are not used in animal models of constraint. Additionally, neurobehavioral tests used with animals in these models do not reflect the upper-motor function tests that are almost universally used in human studies of constraint. 28 Having comparable models of impairments and measurement tools increases the likelihood of successful translation of any discoveries made in the laboratory to the clinical setting. 29 Considering these differences, time and resources may be better spent more clearly establishing the optimal model of constraint, because this remains uncertain, 30 and only after this should animal models be used to determine potential biological mechanisms.
Several important biological questions are best answered with animal studies. For example, how does constraint of the affected limb alter the brain at an anatomic and molecular level, and how are such alterations related to the behavioral changes seen? Furthermore, given not only the importance of learning to the rehabilitation process but the role learning plays in the acquisition of learned nonuse, it is essential that we identify the structural, physiological, and psychological drivers of the learning and memory associated with this restorative intervention. 29 However, it will not be possible to answer these questions until models are developed that faithfully replicate benefits of constraint seen in human trials. Perhaps the more pressing need is for larger scale clinical trials to confirm the findings of the EXCITE trial. This review has highlighted the importance of strong collaboration between basic scientists and clinical researchers, in particular to ensure the relevance of experimental studies and appropriately informed design of clinical trials.
Results from this review suggest that constraint as tested in animal models of stroke (ie, beginning acutely, restraining the affected limb for 24 h/d and with minimal cotherapy) is ineffective and may even do harm. Such an approach requires review, particularly considering the parallels with the most similar human trial design, where high-intensity constraint that commenced 5 to 14 days poststroke was associated with lower gains in motor function than traditional therapy or low-intensity constraint. 31 There are many clinically relevant research questions that are most rapidly and easily assessed using an animal model. Close cooperation between clinical and laboratory researchers is essential to facilitate the development of such studies. Our key recommendations would be rigorous attention to aspects of experimental design (prespecified primary and secondary outcomes, sample size calculations, randomization, and blinding being particularly important), modeling insofar as possible the approach shown to be effective in humans, and choosing functional outcome measures appropriate to the neurological deficit produced by the stroke model, with good discriminatory power at time points of at least 30 days. Only then can we begin to understand the biological basis for brain recovery and develop better therapies for patients.
Footnotes
Acknowledgements
We would like to thank Lucy Murtha for her assistance with the formulation of publication figures. Heidi Janssen is supported by a Post Graduate Public Health Scholarship from the National Stroke Foundation of Australia and the Emlyn and Jennie Thomas Postgraduate Medical Research Scholarship (top up).
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: NS was supported by a training fellowship from NHMRC (455632) and a stroke research grant from the Hunter Medical Research Institute, funded by the Greater Building Society. JB is supported by an ARC Future Fellowship. We report no conflicts of interest.
