Abstract
Background
During post-stroke motor rehabilitation, patients often receive feedback from therapists or via rehabilitation technologies. Research suggests that feedback may benefit motor performance, skill acquisition, and action selection. However, there is no consensus on how extrinsic feedback should be implemented during stroke rehabilitation to best leverage specific neurobehavioral mechanisms to optimize recovery.
Objective
To identify the existing evidence and research gaps regarding the effects of extrinsic feedback on upper extremity motor function in stroke survivors, and to map the evidence onto neurobehavioral concepts of motor performance, motor learning, and action selection.
Methods
The MEDLINE, PsychInfo, EMBASE, and CINHAL databases were searched for relevant articles. A sequential screening process and data extraction were performed by 2 independent reviewers, and the results were reported according to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses for Scoping Reviews guidelines.
Results
A total of 29 studies were identified that met the criteria for inclusion. Beneficial effects of feedback were reported for clinical outcomes of rehabilitation interventions as well as motor performance, motor learning, and action selection post-stroke. Three studies showed that the addition of rewarding elements to positive performance feedback benefited learning or recovery.
Conclusions
Extrinsic feedback has the potential to improve outcomes of stroke rehabilitation through effects on motor performance, motor learning, or action selection. To understand how these specific neurobehavioral processes contribute to recovery, clinical trials should include more granular behavioral measures. Rewarding feedback may be particularly beneficial, but more research is needed regarding the specific implementation of feedback.
Introduction
Sensory feedback about the execution and results of our actions plays an essential role in the control of movement. Interventions that provide extrinsic feedback can improve the performance and learning of motor skills in healthy people, and could potentially facilitate relearning of skilled movements following stroke.1,2 Virtual reality, robotics, and motion tracking technologies are commonly used to provide extrinsic feedback during rehabilitation.3,4 There is a growing interest in using “gamification” to improve outcomes of stroke rehabilitation, in part by promoting engagement and motivation through feedback. 5 However, there is no consensus regarding how extrinsic feedback should be implemented during stroke rehabilitation, nor what specific effects of feedback should be targeted. These questions may be usefully informed by neuroscientific and behavioral research which has characterized the powerful ways in which feedback shapes motor behavior and neural plasticity.
Work in healthy populations has shown that motor behavior and neuroplasticity are shaped by multiple, distinct mechanisms that are sensitive to specific types and parameters of feedback. It has long been recognized that extrinsic feedback about the size and direction of movement errors can guide learners to improve their performance through error correction. 6 Recent evidence has demonstrated that motor performance and learning are also influenced by rewarding feedback and punishment in response to task successes and failures.7,8 Feedback conveying information about movement errors, rewards, and punishment each drive distinct forms of learning and neuroplasticity.9,10 and are sensitive to specific parameters of feedback including sensory modality,11,12 timing,13,14 frequency,15,16 valence, 17 and motivational incentives.18-20 The specific characteristics of extrinsic feedback delivered during neurorehabilitation may therefore substantially affect its clinical benefits.
The effects of extrinsic feedback may differ for motor performance, motor learning, and action selection, with each component process contributing in distinct ways to stroke rehabilitation. Effects on
Feedback can also influence the voluntary selection of particular movements or movement patterns among alternatives, which we refer to as
Here, we perform a scoping review with the objective of identifying the existing evidence and knowledge gaps regarding the effects of extrinsic feedback on upper extremity motor function in stroke survivors. In order to identify evidence specific to the effects of feedback, we consider studies that included multiple conditions in which stroke survivors performed similar motor tasks but which differed in the feedback that was provided. We map the effects of feedback onto distinct neurobehavioral concepts of motor performance, motor learning, and action selection in order to gain a more specific understanding of how extrinsic feedback may benefit stroke rehabilitation. We further characterize the use of extrinsic feedback according to sensory modality, timing, frequency, valence, and motivational incentives, as these factors have been shown to influence the effects of feedback on motor behavior. Previous reviews covering the effects of extrinsic feedback on upper limb motor function post-stroke are more than 10 years old.26,27
Methods
A protocol for this review was constructed according to the guidelines of the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) extension for scoping reviews and registered through publication on the Open Science Framework (https://doi.org/10.17605/OSF.IO/C625E).
Eligibility Criteria
We included peer-reviewed journal articles written in English that meet the following criteria: (1) The participants included adult stroke survivors (greater than 18 years of age). (2) The participants performed a task or therapy involving the more affected upper extremity, which included the provision of extrinsic feedback contingent on task success or motor performance relative to a specific goal. Interventions that distorted visual or proprioceptive feedback in order to modify the magnitude of movement errors were not considered. We direct the reader to Israely and Carmeli 28 and Liu et al 29 for recent reviews specifically covering the effects of such error augmentation and error reduction paradigms in upper limb stroke rehabilitation. (3) At least 2 experimental groups or conditions were tested, in which the provision of feedback was different but participants otherwise performed equivalent interventions. (4) Clinical or behavioral outcomes were measured regarding motor function of the more affected upper extremity.
Information Sources and Search Strategy
A health sciences librarian developed the search strategy and performed the literature searches in MEDLINE (Ovid), Embase (Ovid), PsycInfo (Ovid), and CINAHL (EBSCO) on December 21, 2022. No date limit or language limit was applied. The MEDLINE strategy was developed with input from the project team and adapted for use in the other databases. The complete search strategies are available in an institutional data repository: https://doi.org/10.5683/SP3/ADFAGD.
Selection of Sources of Evidence
The search results were exported to Endnote software for deduplication, and exported to Rayyan software for screening. Articles were selected for inclusion through a sequential screening process in which 2 reviewers independently examined first the titles and abstracts, then the full texts of each record. All conflicts between reviewers were resolved through discussion.
Data Charting Process
Data extraction was performed through a standardized data charting form using Covidence software. Data extraction for each article was performed independently by 2 reviewers, and all discrepancies in the results were resolved through discussion.
Data Items
The following items were extracted for each selected study: (1) Study design and sample size; (2) participant characteristics: age, sex, stroke type, duration, lesion location, and cognitive and motor impairment; (3) study intervention: motor task or therapy performed, number of movements performed or time on task; (4) Feedback characteristics: movement variables conveyed by feedback, timing (concurrent to movement, terminal, or summary), valence (feedback stimuli delivered for good versus bad performance), sensory modality, frequency, elements of additional motivational salience (eg, monetary reward, complex audiovisual stimuli, and social comparison); and (5) study results (mean values or differences on upper limb motor outcomes by condition).
Synthesis of Results
Extracted data items were summarized in table format, along with a descriptive overview of the results. The tables and summary were organized to separately present findings related to clinical outcomes, motor performance (immediate changes in motor function in response to feedback), motor learning (changes in motor performance measured in the absence of feedback following repeated practice of a specific task), and action selection (increased use of the affected upper extremity or decreased use of compensatory movement patterns).
Results
The searches returned 4139 records. A total of 1799 duplicates were removed. A total of 2213 records were eliminated during initial screening of titles and abstracts. A total of 127 full texts were reviewed, 97 studies were found not to meet the inclusion criteria, leaving 30 included reports. Two publications were grouped as a single study for reporting as they described analyses of different outcomes from the same dataset.30,31 Figure 1 shows the PRISMA flowchart of the search and screening process.
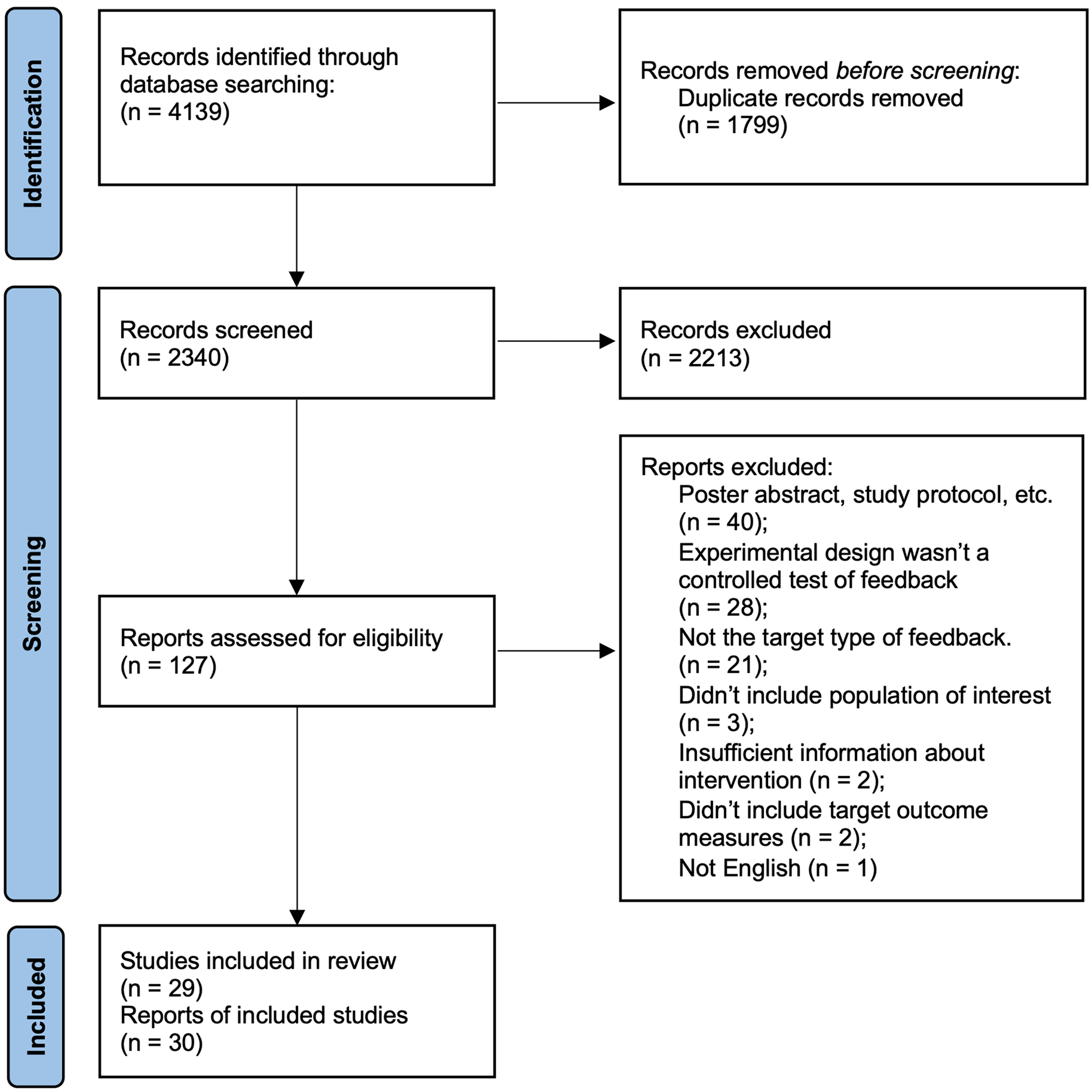
Screening and review flowchart.
Study Characteristics
We identified 15 studies with between-group designs comparing the effects of different feedback conditions. Thirteen of these studies were randomized controlled trials (RCTs), including 5 pilot studies.30-43 Eleven RCTs involved repeated rehabilitation sessions in clinical settings, while 2 tested the use of feedback from wearable devices during daily life.34,39 Two studies compared the effects of different feedback conditions on motor learning over multiple practice sessions, but did not measure clinical outcomes and were not designated as RCTs.44,45 The total sample size of studies with between-group designs ranged from N = 11 to 45.
Thirteen studies used within-participants designs to assess short-term (within a single session) changes in movement outcomes under different feedback conditions.46-58 Six of these studies were designated crossover design RTCs, including 2 pilot studies.49-52,56,57 Two studies were case series with sample sizes of n = 2 and 5.46,48 One study tested a clinical rehabilitation program under different feedback conditions using a within-participants reversal design involving 90 sessions. 59 The sample sizes of studies using within-participants designs, excluding case series, ranged from N = 5 to 43.
Clinical Characteristics of Participants
Stroke-Induced Lesions
Two studies only included participants with ischemic stroke,48,53 14 included participants with both ischemic and hemorrhagic stroke,32,34,35,39-41,43,44,52,55-59 and 13 did not specify the type of stroke.30,31,33,35,36,38,42,45,46,49-51,54 Twenty-two studies did not specify the vascular territory of stroke. Studies that did describe the affected vascular territories included patients with middle cerebral artery,41,48,49,53,58 anterior cerebral artery, 58 anterior circulation,34,59 posterior circulation, 34 and lacunar34,59 strokes. Seven studies in-cluded patients with both cortical and subcortical lesions,30,31,33,41,42,44,48,58 and the remaining 22 studies did not report whether lesions were cortical or subcortical. No study investigated whether the extent, location, or type of lesions were related to the effects of feedback.
Stroke Duration
Four studies included patients with acute stroke (duration less than 2 weeks),34,37,49,51 12 studies included patients with early subacute stroke (duration between 2 weeks and 3 months),32,34,37,38,43,48,49,51-53,55,58 10 studies included patients with late subacute stroke (duration between 3 and 6 months),30-32,37,38,43,46,51,52,55,58 and 20 studies included patients with chronic stroke (duration greater than 6 months).30,31,33,35,36,38-42,44,47,48,50-54,56,57,59 One study did not specify the duration of stroke. 45 No study analyzed whether stroke chronicity was related to outcomes.
Cognitive Impairment
Twelve studies excluded participants with cognitive function below a threshold determined by Mini Mental Status Examination (MMSE) or Montreal Cognitive Assessment (MoCA) tests.32,33,35-37,40,44,46,49,52,53,59 Thirteen studies excluded participants with aphasia,32,35,36,40-43,45,49,51,52,58,59 and 9 studies excluded participants with neglect.35,37,44-46,52-54,59 Only 2 studies included measures of cognitive function in their analysis. Cirstea et al 31 reported that greater cognitive impairment in verbal memory, planning, and mental flexibility were associated with worse motor learning. 31 This association was only present for learning mediated by verbal feedback regarding their upper limb movement patterns, but not learning through visual feedback regarding movement accuracy. Quattrocchi et al 44 controlled for cognitive function by including cognitive test scores as covariates in their statistical analyses but did not report on the effects of these variables.
Effects of Feedback
Clinical Outcomes
Nine studies reported statistically significant effects of feedback on clinical outcome measures (Table 1). According to the international classification of Functioning, Disability and Health model (ICF), 8 studies reported effects at the level of body functions and structures,32,33,36,37,40,42,43,59 7 at the level of activities,32,33,37,38,40,43,59 and 3 at the level of participation.32,40,59
Effects of Feedback on Clinical Outcomes.
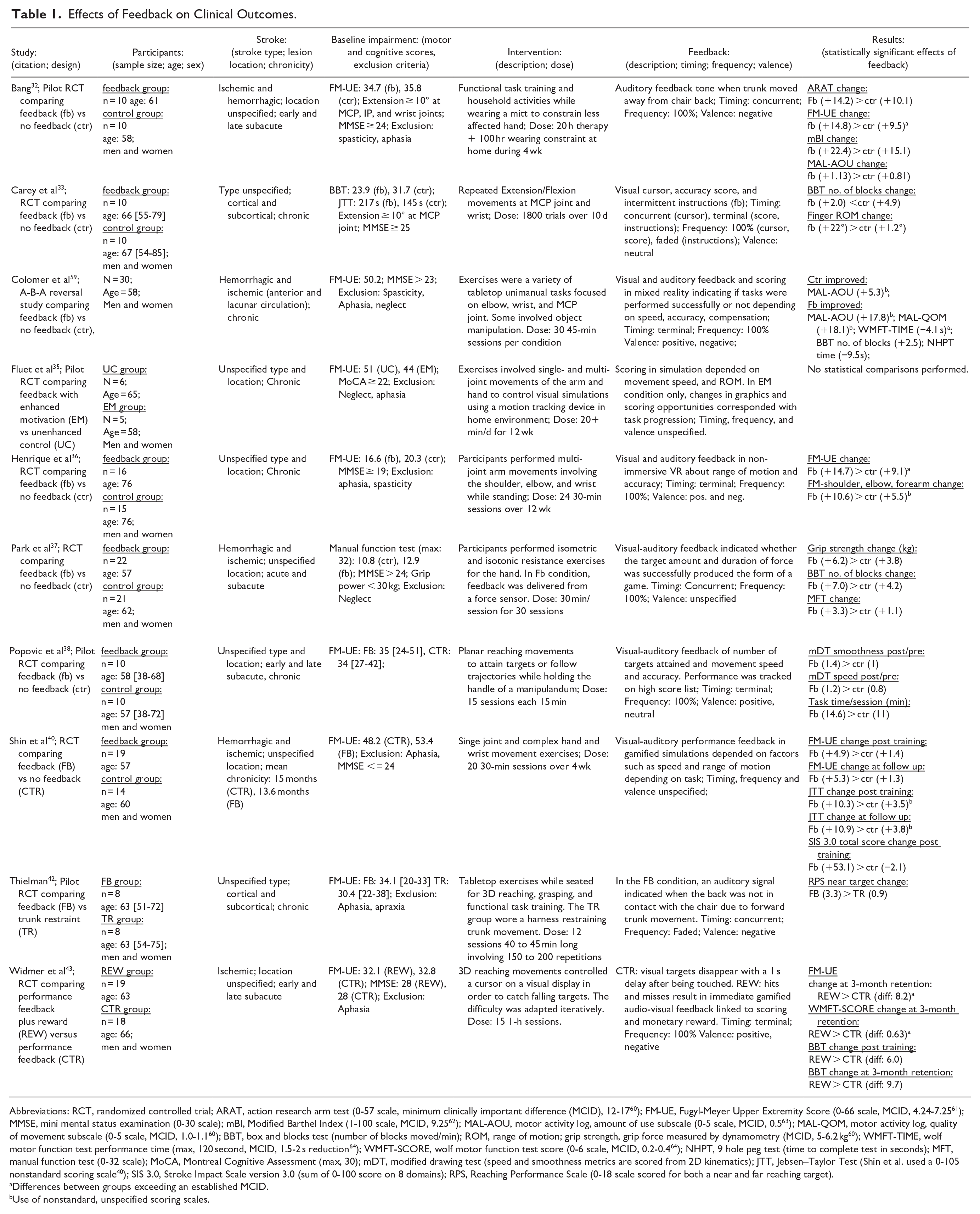
Abbreviations: RCT, randomized controlled trial; ARAT, action research arm test (0-57 scale, minimum clinically important difference (MCID), 12-17 60 ); FM-UE, Fugyl-Meyer Upper Extremity Score (0-66 scale, MCID, 4.24-7.25 61 ); MMSE, mini mental status examination (0-30 scale); mBI, Modified Barthel Index (1-100 scale, MCID, 9.25 62 ); MAL-AOU, motor activity log, amount of use subscale (0-5 scale, MCID, 0.5 63 ); MAL-QOM, motor activity log, quality of movement subscale (0-5 scale, MCID, 1.0-1.1 60 ); BBT, box and blocks test (number of blocks moved/min); ROM, range of motion; grip strength, grip force measured by dynamometry (MCID, 5-6.2 kg 60 ); WMFT-TIME, wolf motor function test performance time (max, 120 second, MCID, 1.5-2 s reduction 64 ); WMFT-SCORE, wolf motor function test score (0-6 scale, MCID, 0.2-0.4 64 ); NHPT, 9 hole peg test (time to complete test in seconds); MFT, manual function test (0-32 scale); MoCA, Montreal Cognitive Assessment (max, 30); mDT, modified drawing test (speed and smoothness metrics are scored from 2D kinematics); JTT, Jebsen–Taylor Test (Shin et al. used a 0-105 nonstandard scoring scale 40 ); SIS 3.0, Stroke Impact Scale version 3.0 (sum of 0-100 score on 8 domains); RPS, Reaching Performance Scale (0-18 scale scored for both a near and far reaching target).
Differences between groups exceeding an established MCID.
Use of nonstandard, unspecified scoring scales.
The types of movements performed in studies reporting effects on clinical outcomes reflected a large range of activities performed during rehabilitation. Movement types included 3-dimensional reaching,36,42,43 planar or linear reaching,38,59 isolated hand movements,37,40,59 reaching and grasping, 42 single joint movements,33,40,59 and complex functional tasks (eg, pouring water, folding, and combing hair).32,42 The information conveyed by feedback generally reflected the specific goals of these varying tasks. Feedback was contingent on movement accuracy,33,36,38,43,59 speed,38,40,41,43,59 range of motion,33,36,40 upper limb kinematics, 59 trunk kinematics,32,42 and force production. 37
The effect size of clinical benefits due to feedback was inconsistent; we only identified 4 studies reporting benefits of feedback exceeding established thresholds for minimum clinically important differences (Table 1). However, estimates of minimal clinically importance differences were not available for several measures. The variability in the effects of feedback is likely due to the fact that the studies identified here were small and highly heterogeneous in their interventions and clinical outcome measures
Performance
Five studies reported effects of feedback on motor performance (Table 2). We defined performance effects as immediate improvements in movement execution upon the introduction of feedback. Unlike motor learning, performance effects do not necessarily persist after the removal of feedback. 21 Four of the studies identified here used crossover designs with minimal washout periods, which should primarily measure transient effects of feedback.49,51,54,55
Effects of Feedback on Motor Performance.
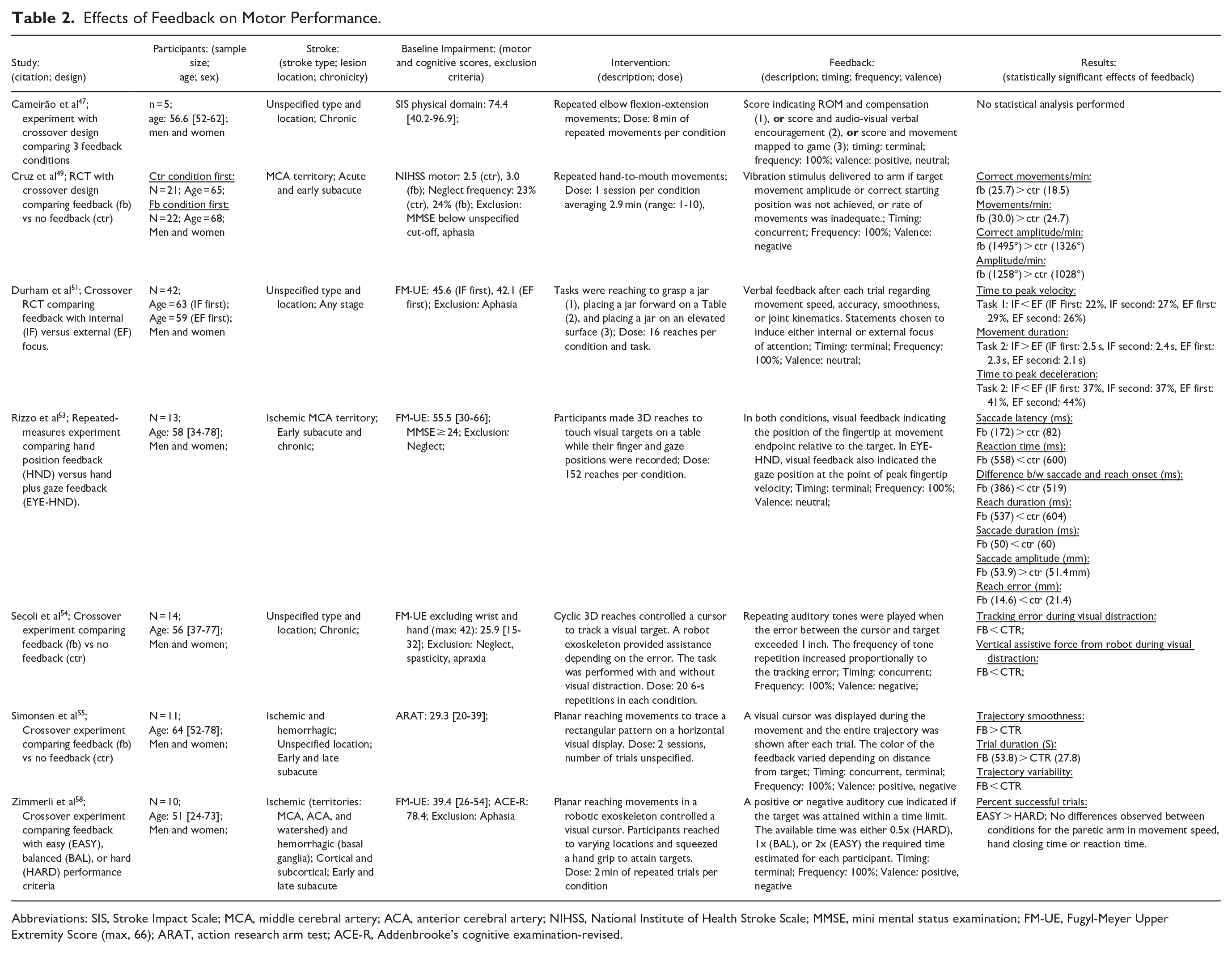
Abbreviations: SIS, Stroke Impact Scale; MCA, middle cerebral artery; ACA, anterior cerebral artery; NIHSS, National Institute of Health Stroke Scale; MMSE, mini mental status examination; FM-UE, Fugyl-Meyer Upper Extremity Score (max, 66); ARAT, action research arm test; ACE-R, Addenbrooke’s cognitive examination-revised.
Cruz et al 49 found that during a repetitive hand-to-mouth movement exercise, feedback dependent on the rate and amplitude of movements improved both of these factors, compared to no feedback. Durham et al 51 compared verbal feedback regarding various movement features that was formulated to induce either an internal or external focus of attention during reaching and grasping tasks. For example, participants were told to “think about lifting their arm up higher” (internal) versus to “think about being higher off the table” (external). Feedback targeting an external focus of attention improved movement duration and measures related to the velocity and acceleration profiles of reaching. Rizzo et al 53 found that the addition of feedback indicating gaze position during three-dimensional (3D) reaching improved movement accuracy, duration, and reaction time. Secoli et al 54 had patients perform 3D arm movements to track a visual target with and without visual distraction. They found that auditory feedback dependent on tracking error ameliorated reductions in performance due to distraction. Simonsen et al 55 found that visual feedback about the accuracy of 2D trajectory tracing improved the smoothness and variability of movements.
Overall, the existing literature has shown that feedback can lead to performance improvements in the speed, accuracy, and vigor of reaching movements, indicating that it could potentially increase movement quality during therapy. However, it isn’t clear whether the magnitude of these improvements is of clinical importance. Notably, Cruz et al 49 reported that feedback increased the rate of movement repetitions by over 5 repetitions/minute, which could translate to hundreds of additional repetitions per session. The studies reviewed here focused almost entirely on proximal arm function during reaching; we didn’t identify any studies showing that feedback can lead to performance improvements other areas such as manual dexterity or the production of effort during resistance training.
Motor Learning
Four studies reported effects of feedback on motor learning, which we defined as improvements in task performance through repetitive practice that were assessed post-practice in the absence of feedback (Table 3). Three of these studies involved extensive training of 3D reaching movements. Maulucci and Eckhouse 45 tested the effects of feedback indicating when the hand deviated from an ideal trajectory, compared to no feedback. They found that feedback resulted in improved adherence to the ideal trajectory and reduced hand oscillations. Cirstea and Levin, 30 Cirstea et al 31 found that feedback about extension of the shoulder and elbow joints led to improvements in the range of motion and coordination of these joints, while feedback about the location of the hand at movement endpoint led to improved endpoint precision. Subramanian et al 41 had participants train with feedback that depended on movement speed, accuracy, and trunk displacement. Feedback was either delivered in a physical environment or was coupled with salient, gamified stimuli in virtual reality. They found that shoulder range of motion improved only when feedback was delivered in the virtual environment. Quattrocchi et al 44 investigated the effects of feedback linked to monetary reward and punishment, compared to neutral feedback, when participants learned to compensate for mechanical perturbations during 2D reaching. Both reward and punishment enhanced learning, while only reward benefited the retention of learning.
Effects of Feedback on Motor Learning.
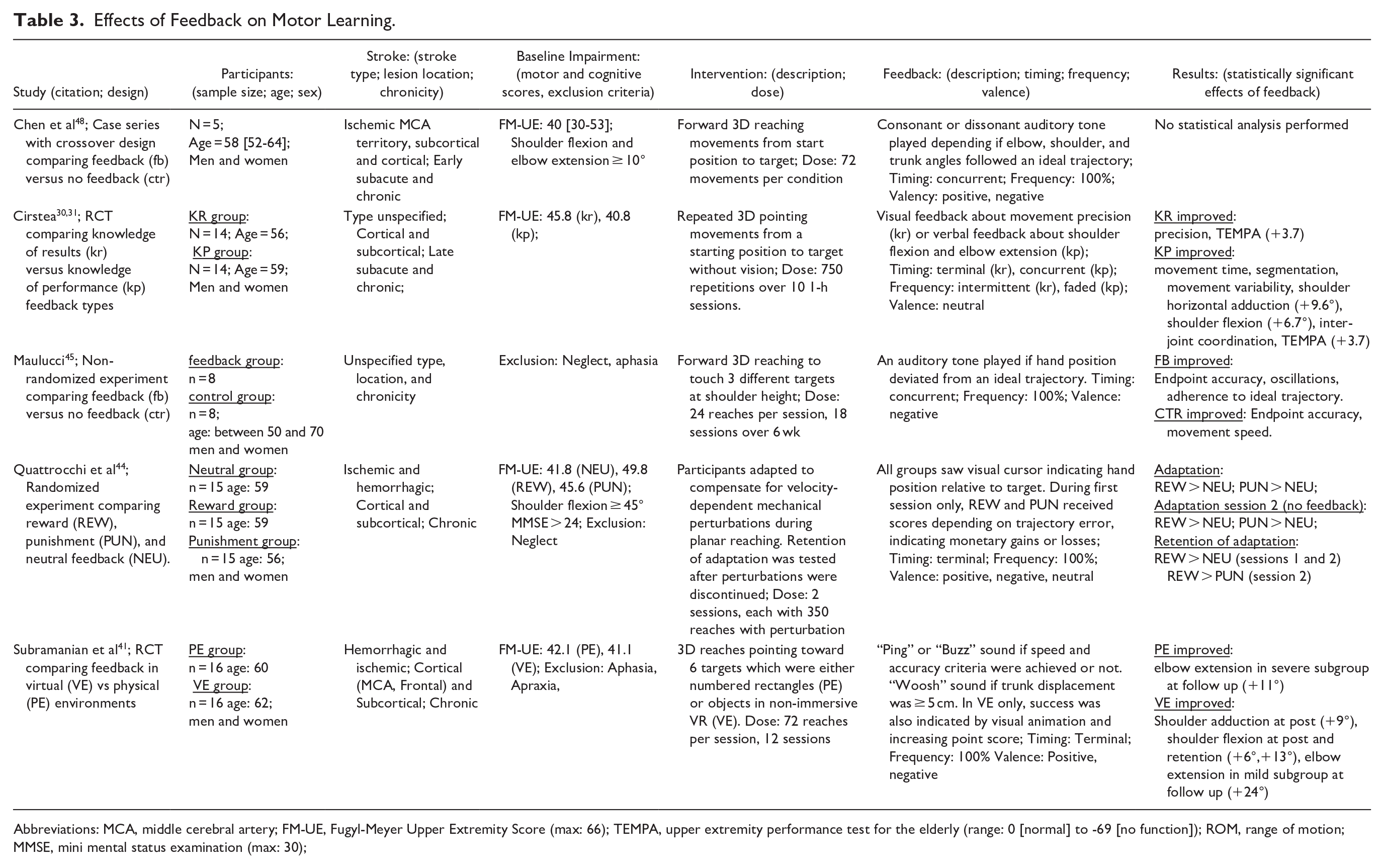
Abbreviations: MCA, middle cerebral artery; FM-UE, Fugyl-Meyer Upper Extremity Score (max: 66); TEMPA, upper extremity performance test for the elderly (range: 0 [normal] to -69 [no function]); ROM, range of motion; MMSE, mini mental status examination (max: 30);
In summary, the studies reviewed here found that feedback can improve learning of reaching movements both in terms of endpoint control and in the kinematics of the shoulder and elbow joints, depending on which aspect of movements are conveyed by feedback. Moderate improvements were reported for shoulder adduction, shoulder flexion, and elbow extension due to training with feedback, ranging from 9 to 9.6°, 6 to 13°, and 24°, respectively.30,31,41 Effect sizes were unfortunately not reported for other motor learning outcomes. Studies also found that the addition of salient and rewarding elements to feedback can lead to additional benefits. As was the case for motor performance, there were no studies reporting effects of feedback on motor learning for tasks focused on distal arm function.
Action Selection
Five studies reported effects of feedback on action selection, which we defined as changes in the selection of particular movements or movement patterns (Table 4). Four studies reported that feedback resulted in immediate and short-term reductions in the use of compensatory movement patterns during repetitive, simple reaching tasks.46,50,56,57 Cai et al 46 found that compensatory trunk movements were reduced by audio-visual feedback delivered in non-immersive virtual reality, compared to a baseline condition with no feedback. Douglass-Kirk et al 50 tested the effects of musical feedback stimuli that stopped whenever compensatory movements of the trunk, shoulder, or scapula were detected. They found that feedback reduced compensation compared to a control condition with no feedback. Valdes et al 56 compared the effects of visual versus haptic feedback dependent on trunk displacement. Both types of feedback led to similar reductions in compensatory trunk movements. Similarly, Valdes and Van der Loos 57 found that combined haptic and visual feedback reduced trunk displacement, but that the addition of gamified scoring produced no additional benefits.
Effects of Feedback on Action Selection.
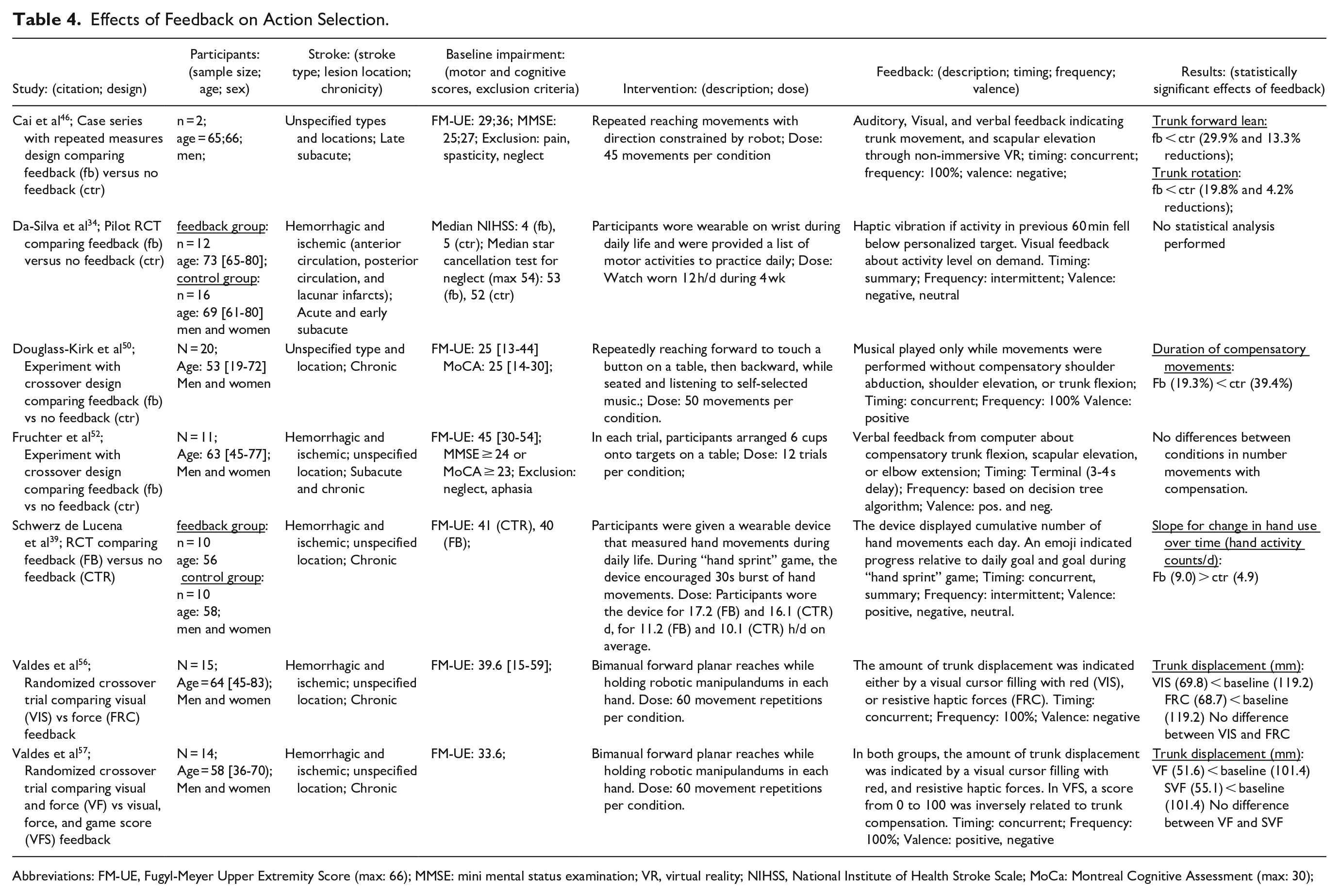
Abbreviations: FM-UE, Fugyl-Meyer Upper Extremity Score (max: 66); MMSE: mini mental status examination; VR, virtual reality; NIHSS, National Institute of Health Stroke Scale; MoCa: Montreal Cognitive Assessment (max: 30);
Overall, the studies reviewed here found that feedback consistently produced reductions in compensation with large effect sizes, with group average reductions in the duration and magnitude of compensatory movements ranging between about 40% to 50%. The effects of feedback on compensation did not depend on variations in the feedback such as the sensory modality or the addition of gamified scoring. Only 1 study, Fruchter et al, 52 did not find reliable reductions in compensation due to feedback. This study differed from the others in that it involved a task with more complex cognitive and motor demands. Furthermore, feedback was manually controlled by a therapist, which possibly resulted in delayed and imprecise feedback compared to automated systems.
Schwerz de Lucena et al 39 tested the effects of feedback from a wearable device designed to encourage use of the hemiparetic hand during daily life. They reported greater increases in the amount of hand use over time in a group receiving feedback, compared to a group wearing the device without feedback. However, they found no differences between groups in clinical outcomes nor hand use at a 3-month follow up assessment.
Characteristics of Feedback
Modality of Feedback
Sixteen studies included non-verbal auditory feedback,32,36-38,40-43,45-48,50,54,58,59 18 studies included non-verbal visual feedback,30,31,33,35-37,39-41,43,44,46,47,53,55-57,58 4 studies included haptic feedback,34,49,56,57 and 4 studies included verbal feedback either by therapists or automated systems.30,31,46,51,52 Only 1 study was designed to compare the effects of feedback with similar informational content delivered through different sensory modalities, finding that visual and haptic feedback produced similar reductions in compensatory movements. 56
Valence of Feedback
Nineteen studies included feedback indicating unsuccessful task performance (negative valence),32,34,36,39,41-46,48,49,52,54-59 14 studies included feedback indicating successful task performance (positive valence),36,38,39,41,43,44,47,48,50,52,55,57-59 and 8 delivered performance feedback with neutral valence, such as a point score indicating performance that was not explicitly framed as good or bad.30,31,33,34,38,44,47,51,53 Only 1 study directly compared feedback with positive versus negative valence, finding that while both positive (reward) and negative (punishment) feedback improved the acquisition of motor learning, only positive feedback improved retention. 43
Timing of Feedback
Fifteen studies involved continuous feedback concurrent with movements,30-33,37,39,42,45,46,48-50,54-57 and 15 studies involved terminal feedback at the end of movements.30,31,33,35,36,38,41,43,44,47,51-53,55,58,59 One study did not provide information about the timing of feedback. 40 Only 2 studies provided information regarding the delay of terminal feedback. Fruchter et al 52 reported that the delay was approximately 3 to 4 seconds. Widmer et al 43 intentionally imposed a 1 s delay in the control condition in order to reduce the efficacy of feedback, as it was not possible to eliminate performance feedback entirely in the task. No study specifically varied the timing of feedback while controlling for all other feedback characteristics.
Scheduling of Feedback
Twenty-two studies provided feedback, or a possibility of feedback, for every movement.32,33,36-38,41,43-51,53-59 Three studies provided intermittent feedback with reducing frequency over time (faded feedback).31,33,42 Two studies provided intermittent summary feedback regarding the amount of use of the upper extremity during daily life.34,39 In 1 study, a decision tree algorithm determined whether feedback was delivered after each movement. 52 Two studies did not provide information regarding the frequency of feedback.35,40 No study specifically varied the scheduling of feedback while controlling for other factors.
Elements of Additional Motivational Salience
Two studies used monetary reward linked to performance feedback,43,44 and 1 study used monetary punishment. 44 Eleven studies used gamified scoring.35-41,43,47,57,59 Five studies used virtual or augmented reality.36,40,41,46,59 Two studies delivered performance feedback in the form of musical stimuli.48,50 One study used social comparison tied to performance feedback in the form of a high score list. 38 Four studies directly compared feedback conditions with different elements of motivational salience that were otherwise similar, including monetary incentives,43,44 gamified scoring,41,43,57 and complex multisensory stimuli.41,43 These studies found that the addition of these motivational elements can improve both motor learning and recovery. While the addition of simple game scoring and virtual reality stimuli to feedback produced moderate learning-related improvements in reaching kinematics, 41 a more elaborate gamified experience coupled with monetary reward during reaching practice produced clinical gains well above established minimum clinically important differences. 43
Discussion
We identified 29 studies that tested the effects of feedback on upper limb motor outcomes in stroke. Nearly all (26/29) of the studies reviewed here were published after previous reviews on the topic.26,27 Most of the studies included in these previous reviews didn’t isolate the effects of feedback, as they lacked control conditions in which participants performed similar activities with different feedback, and therefore were not included in the current review.
Beneficial effects of feedback were reported across a range of endpoints in clinical trials spanning all 3 domains of the ICF model. These results are promising in that they indicate that feedback may have the potential to confer benefits on clinically meaningful and valid measures. However, the mechanisms by which feedback produced benefits were ambiguous in the majority of studies, as feedback during rehabilitation could improve coarse clinical measures in multiple, very different ways. For example, feedback could improve dexterity by enhancing the consolidation of motor skill learning between rehabilitation sessions,18,23 or it could lead to greater peripheral muscle adaptations by motivating increased effort during rehabilitation exercises. 65 Either mechanism could produce benefits in clinical assessments, which are often sensitive to both dexterity and strength. 66
Applications of feedback are likely to be more effective if their design and evaluation are targeted to specific neurobehavioral mechanisms. We found that besides clinical measures, the effects of feedback related to more specific behavioral measures could be classified according to 3 distinct concepts: motor learning, performance, and action selection. We mapped the current evidence for effects of feedback on upper extremity function post-stroke onto these distinct concepts in order to better understand what is currently known, and what questions remain outstanding.
Effects of Feedback on Motor Learning
The use of feedback to improve motor learning is often cited as a fundamental principle of neurorehabilitation.2,27,67 We identified 4 studies that reported effects of feedback on motor learning. Learning improvements due to feedback were mostly specific to the movement parameters that were relayed by feedback (ie, endpoint accuracy, 31 shoulder and elbow motion, 30 and movement trajectories44,45). Therefore, the information content of feedback should be carefully selected to convey aspects of movement that are clinically important. Ongoing work to establish valid and responsive metrics for quality of movement post-stroke may be informative in determining which parameters should be relayed to patients through feedback.68,69
A key question is whether feedback-mediated motor learning during specific tasks can generalize beyond the training context to produce meaningful reductions in impairment or activity limitations. Only 1 study identified here directly tested generalization. They found that improved movement kinematics due to feedback when training reaches toward 1 target generalized to untrained reaches toward a different target.30,31 However, this is a rather limited form of generalization, and its clinical significance is unclear. Two studies tested the effects of feedback on both motor learning and clinical outcomes. In both cases, feedback manipulations improved motor learning, but these improvements did not generalize to differences in clinical measures.30,31,41 We recommend that future work investigating the effects of feedback on motor learning post-stroke also includes generalization tests that approximate activities of daily living.
Effects of Feedback on Performance
The introduction of feedback can lead to immediate improvements in the vigor and accuracy of movements. 22 We identified evidence for feedback-mediated performance benefits on movement amplitude, 49 speed,49,51,53 accuracy,53,54 and smoothness. 55 As was the case for motor learning, performance tended to improve on variables that were directly related to the content of the feedback. Performance improvements during rehabilitation may benefit long-term outcomes. However, improvements may not persist beyond rehabilitation if performance becomes reliant on extrinsic feedback.11,12,70 Future work is needed to understand how short-term performance gains due to feedback during stroke rehabilitation translate to longer-term outcomes such as learning and recovery.
Effects of feedback on performance were mainly reported for simple feedback indicating movement execution relative to task goals, compared to no feedback.49,53-55 No studies tested whether elements of motivational salience coupled with feedback improved motor performance. It is well established that rewarding feedback can increase movement vigor by motivating physical effort.22,65,71 Rewarding features such as gamification are common in neurorehabilitation technologies, and may have the potential to promote increases in the intensity of rehabilitation, which is a key factor for recovery.72,73 Future research should therefore investigate whether elements of motivational salience tied to feedback can be designed to produce sustained increases in rehabilitation intensity.
Effects of Feedback on Action Selection
Feedback could promote recovery by encouraging stroke survivors to practice impaired movement patterns instead of compensatory ones. The studies reviewed here show that various types of feedback can effectively reduce the compensation during reaching.46,50,56,57 Additionally, 2 pilot RCTs reported that feedback designed to discourage compensatory trunk movements during rehabilitation improved clinical outcomes.32,42 Larger randomized controlled trials are needed to confirm whether feedback to reduce compensation during therapy can result in clinically meaningful enhancements of recovery.
Besides feedback, physical trunk restraint has often been used to prevent compensation during rehabilitation. 74 Although effective in preventing compensatory trunk flexion, physical trunk restraint doesn’t address other forms of compensation involving the scapula and shoulder. 74 Feedback could provide a more versatile solution as it could in principle be targeted to any compensatory movement pattern. Furthermore, the use of feedback could have lasting benefits if reductions in compensation are retained as long-term habits that generalize to activities of daily living. Future research is needed to test the retention and generalization of feedback-mediated reductions in compensation.
Two studies involved feedback from wearable devices to promote increased use of the affected upper extremity during daily life. One was a pilot study that did not report any statistical analysis of its outcomes, and the other found that feedback resulted in modest increases in hand use over time that did not correspond to any benefits on clinical outcomes. Therefore, it is not yet clear whether feedback can promote recovery post-stroke by promoting increased use of the more affected limb.
Do the Characteristics of Feedback Influence Motor Outcomes Post-Stroke?
The sensory modality, valence, timing, and scheduling of feedback varied widely across studies. However, the heterogeneity in interventions and outcomes makes it difficult to draw any conclusions regarding the effects of these feedback characteristics. Only a few studies performed direct, controlled comparisons between different types of feedback. The addition of monetary rewards, gamified scoring, and complex multisensory sensory stimuli coupled with positive performance feedback was shown to benefit multiple forms of motor learning and clinical outcomes of rehabilitation.41,43,44 These findings suggest that the well-established benefits of reward during motor learning in healthy populations also extend to stroke survivors, and that these benefits can potentially be leveraged to improve the effectiveness of rehabilitation.
One study found that motor performance was improved when verbal feedback was formulated to promote an external, rather than an internal, focus of attention. This is in line with extensive research indicating that motor performance and learning is facilitated by a focus on movement outcomes, rather than the details of movement execution. 75 A unique problem in stroke, however, is that the most efficient way to achieve successful movement outcomes is generally to use compensatory movement patterns that are counterproductive to recovery in the long term. 25 Indeed, another study found that patients learned to improve reach precision when given feedback about the movement outcome, but that quality of movement only improved through feedback about movement execution.30,31 An optimal strategy may be to first use feedback to train the desired patterns of movement execution to a high level of automaticity, so that the focus of attention can then be shifted toward the movement outcome.
Do Clinical Characteristics Interact With the Effects of Feedback?
The processing of motor feedback depends on specific neural structures as well as cognitive and perceptual capacities that may be compromised to varying degrees by stroke.9,10,76-78 The majority of studies excluded participants with cognitive impairments that are common in stroke survivors, limiting the generalizability of the results. Future work is needed to better understand how specific cognitive impairments or patterns of brain damage might impair the use of feedback, and how feedback can be designed to overcome these potential barriers.
Neural Mechanisms of Reward-Based Motor Learning
In theories of biological reinforcement learning, the production of rewarded behaviors is thought to be strengthened through neuroplasticity in the striatum and frontal cortical areas mediated by the release of dopamine. 79 Dopamine signaling is both necessary and sufficient for various forms of motor learning and motor cortical plasticity.81-87 Rewards linked to good motor performance have been shown to improve both the rate and retention of motor learning,18-20,87 possibly by affecting activity in cortico-striatal network.24,89 Interestingly, positive performance feedback alone may act as a form of intrinsic reward which promotes both motor learning and neural activation in reward processing brain regions.7,90-92 Future research should ask whether the benefits of performance feedback and reward during stroke rehabilitation depend on cortico-striatal networks, and whether they are impacted by damage to these regions. 93
Next Steps
Although the results reviewed here show that performance feedback is promising as a tool to improve recovery, larger randomized controlled trials are clearly needed. However, we suggest that it may be too early to embark on such studies, as a systematic framework for designing feedback systems is still lacking. This reflects a general lack of clarity on the “active ingredients” of rehabilitation. The field should first seek to identify the mechanisms of training-induced recovery, then ways to formulate feedback that increase the exposure to “active ingredients” of therapy through its effects on motor learning, performance, and action selection. Subsequently, it will be necessary to systematically vary the characteristics of feedback in controlled studies to determine how its effects can be optimized. Finding new strategies to enhance the rewarding effects of performance feedback may be particularly important.
Limitations of the Existing Evidence
The clinical trials reviewed here mostly had small sample sizes and were highly heterogeneous in their treatment protocols and outcome measures. Conclusive evidence for clinical benefits related to feedback during therapy will require larger trials and/or more standardized treatment and study protocols.
Conclusions
Feedback can affect motor performance, motor learning, and action selection post-stroke in ways that may benefit rehabilitation outcomes. The inclusion of more granular behavioral measures in clinical trials could lead to a better understanding of how these specific processes relate to the effects of feedback on clinical endpoints. The use of salient and rewarding stimuli coupled with positive performance feedback may benefit motor learning and recovery post-stroke. Otherwise, there is very limited direct evidence to support the use of specific feedback characteristics at this time, and more research is needed in this area.
Footnotes
Author Contributions
Dimitrios J. Palidis: Conceptualization; Formal analysis; Investigation; Methodology; Supervision; Writing—original draft; Writing—review & editing. Zoe Gardiner: Investigation; Writing—review & editing. Amelia Stephenson: Investigation; Writing—review & editing. Kevin Zhang: Investigation; Writing—review & editing. Jill Boruff: Investigation; Methodology. Lesley K. Fellows: Conceptualization; Project administration; Supervision; Writing—review & editing.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: DJP was supported by a postdoctoral fellowship award from the Canadian Institutes of Health Research.
