Abstract
Keywords
Introduction
Current debate centers on whether therapeutically induced improvements in upper-extremity (UE) function in persons poststroke reflect acquisition of compensatory movement strategies or restoration of more normal movement patterns. 1 Restoration involves the reappearance of motor patterns present prior to stroke, whereas compensation involves the appearance of new motor strategies, including substitution with different, atypical components. 1 Although compensatory movements may enable task performance in the short term, these may be detrimental in the long term by contributing to problems such as pain and reduced range of joint motion. 1 Moreover, the use and reinforcement of atypical movement components may interfere with attainment of normal UE motor patterns and thus limit genuine recovery. Compensatory strategies may also have a detrimental psychosocial impact. Both the self-perception and appearance to others of aberrant movement contribute to depression and, ultimately, avoidance of using the impaired arm. 2 These problems underscore the need to discriminate between compensation and restoration in neurorehabilitation and develop interventions that promote restoration of motor function present prior to stroke.
Kinematic analysis of UE motor performance enables sensitive, quantitative, and reliable 3 assessment of abnormal movements 4 and thus may facilitate discrimination between compensation 1 and restoration of arm function poststroke.3,5 To date, the majority of UE kinematic studies in persons poststroke have been cross-sectional investigations of reaching3,5-13 or grasping tasks,9,14,15 which reveal slowness, spatial coordination deficits, temporal joint dyscoordination, and compensatory movements that interfere with normal performance.3,5-15
Investigations of intervention-related changes in UE motor performance using kinematics have focused on evaluating the effects of constraint-induced movement therapy7,16 and functional unilateral14,15,17,18 or bilateral 19 repetitive task training. These therapeutic approaches are believed to facilitate neural plasticity through repetitive execution of functional movements. Although weakness is recognized as a major factor contributing to disability poststroke,10,20,21 strengthening is not typically included as a neurorehabilitation technique. Historical clinical perspectives cautioned against high-exertion activities for neurological populations because they were assumed to increase spasticity and impair motor performance. 20 More recently it has been argued that remediation of impairments (ie, weakness) does not generalize to functional task performance. To date, only 2 studies have directly compared UE strengthening and functional task practice (FTP) with kinematics. 17 Both studies used resistive therapeutic bands and concluded that strengthening does not improve paretic UE motor function, but the methods did not ensure either sufficient intensity or progression of the strengthening activities to offer sufficient overload. 22 Additionally, the importance of positioning, specifically trunk stabilization, has been confirmed by the demonstration of greater improvements in UE movements following treatment activities performed with external trunk stabilization.15,23 These studies positioned participants in a standard chair, which may not provide optimal trunk stabilization and may even enable acquisition of compensatory movements.
Although there is now considerable evidence that systematic high-intensity progressive resistance training increases strength, improves activity, and produces behavioral improvements in the hemiparetic UE without increasing spasticity,24,25 the majority of the literature reports effects of combined strengthening and FTP. Therefore, the behavioral effects of resistance training alone remain unclear. We compared 2 forms of UE rehabilitation for persons poststroke: (1) FTP and (2) dynamic high-intensity resistance training or power training (POWER). Our primary aim was to compare these 2 interventions using a battery of standardized clinical evaluations and kinematics of goal-directed UE movements. This approach enabled concurrent investigation of changes in function, as understood by commonly used clinical tools, and movement strategies used by persons poststroke during reaching tasks. We hypothesized that following POWER, behavioral motor improvements would reveal restoration of motor patterns more similar to healthy individuals, whereas following FTP, behavioral changes would reveal compensatory movement strategies. Our secondary aim was to determine the effects of treatment order. We hypothesized that FTP preceded by POWER would reveal greater behavioral motor improvements because FTP would be more effective following restoration of neuromechanical function.
Methods
Participants
This study was a single-center, randomized controlled trial. Participants included 14 persons with UE hemiparesis poststroke meeting the following inclusion criteria: single, unilateral stroke within 6 to 26 months of enrollment (confirmed by diagnostic imaging); voluntary movement in the major shoulder and elbow agonists in the horizontal plane (eg, gravity eliminated) 26 ; and active wrist extension, thumb abduction, and extension of any 2 digits (Table 1). Exclusion criteria were the presence of significant UE joint pain, limitations in passive range of motion (ROM) or proprioception deficits at the elbow or shoulder joint, lesions involving the brain stem or cerebellum, cognitive deficits affecting the ability to follow 3-step commands, and conditions involving any unstable cardiovascular, orthopedic, or neurological impairment precluding exercise. All procedures were approved by the Stanford University panel on human subjects research.
Participant Characteristics a
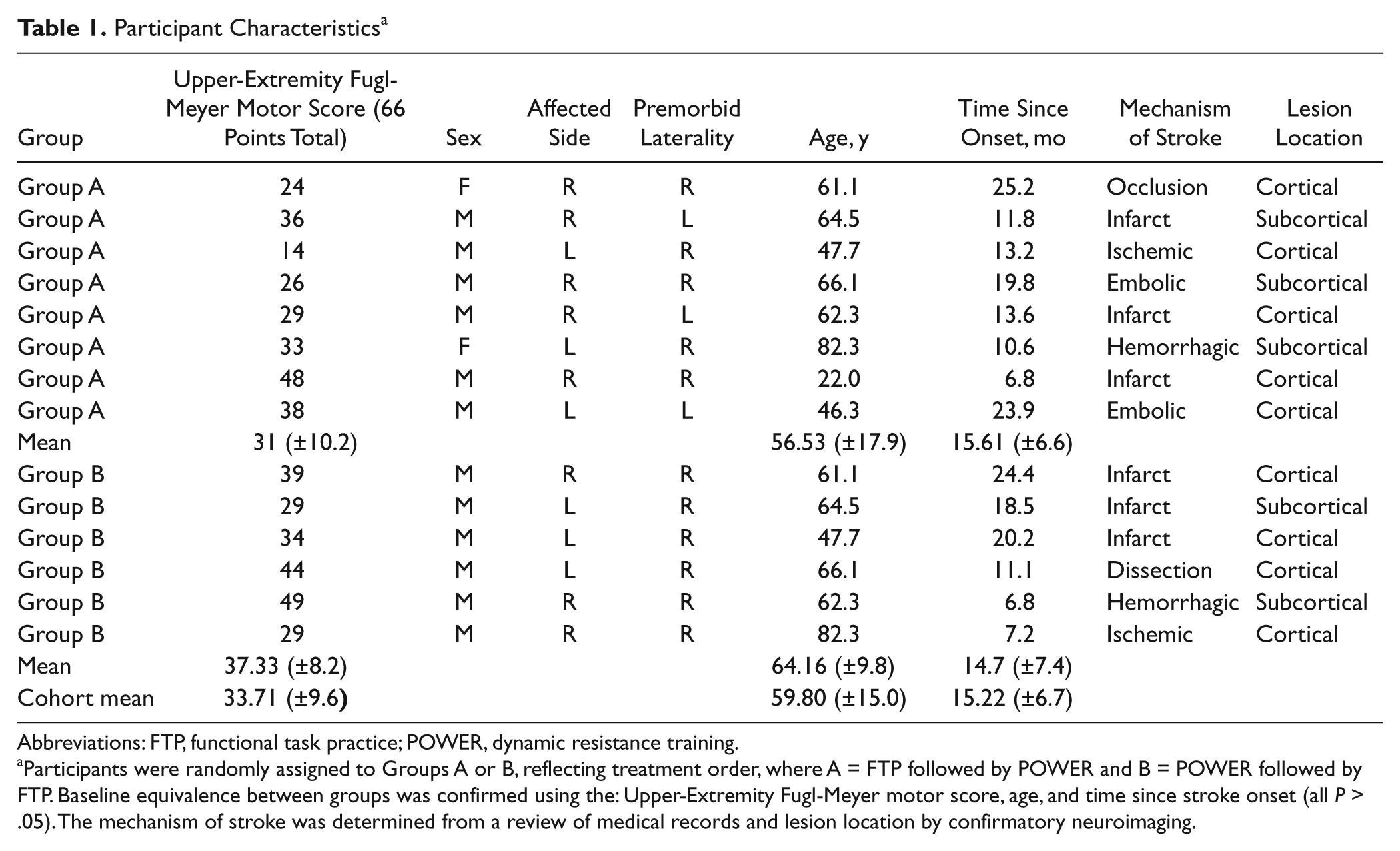
Abbreviations: FTP, functional task practice; POWER, dynamic resistance training.
Participants were randomly assigned to Groups A or B, reflecting treatment order, where A = FTP followed by POWER and B = POWER followed by FTP. Baseline equivalence between groups was confirmed using the: Upper-Extremity Fugl-Meyer motor score, age, and time since stroke onset (all
Procedures
After providing written informed consent, all participants were enrolled in a 2-stage crossover design (Figure 1). Participants were randomized to either (1) POWER or FTP, followed by (2) the alternate therapy. Each treatment block lasted 10 weeks and involved 30 sessions (ie, 3 sessions per week lasting 90 minutes each); thus, each participant received a total of 90 hours of one-on-one treatment with a licensed physical therapist. The treatment blocks were separated by a 2-week evaluation period. Clinical and kinematic assessments were conducted by blinded assessors: (1) at baseline and (2) following the first and (3) second treatment blocks. To ensure baseline equivalence between treatment orders, the shoulder/elbow components of the UE Fugl-Meyer Motor evaluation3,27 were used to identify higher (≥20/30 points) and lower functioning (<20/30 points) individuals. Separate randomization orders were prepared, allocated to sealed envelopes, and stored by the study coordinator in a locked drawer. Following baseline clinical assessment, the blinded evaluator drew a sequentially numbered sealed envelope from the appropriate grouping (ie, higher vs lower) and provided it to the treating physical therapist who broke the seal to reveal assignment to either treatment order A (FTP followed by POWER) or B (POWER followed by FTP). Participants were blinded to their randomization.
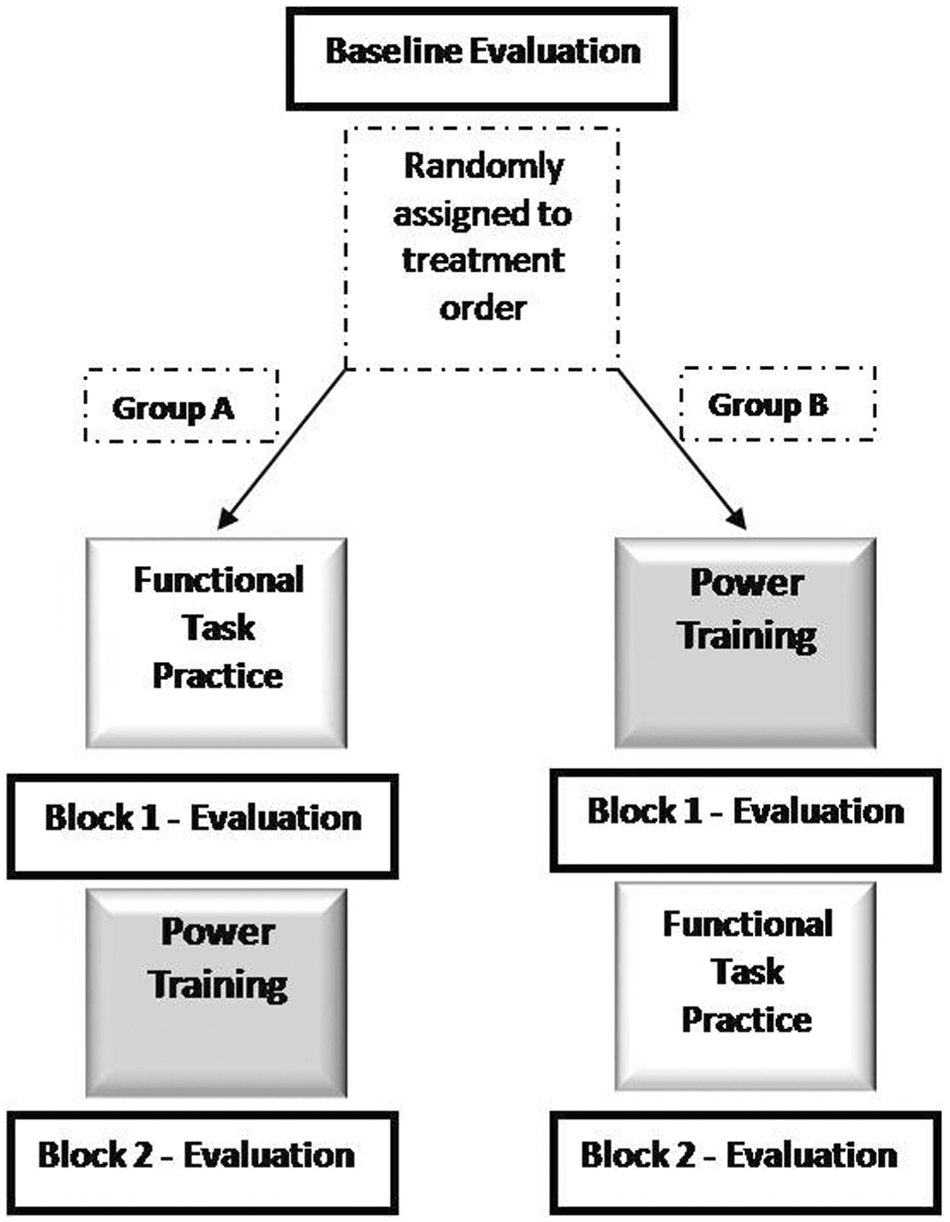
Summary of the cross-over design of the study. Participants were evaluated three times: at baseline and after each treatment block. The treatment order for Group A was FTP followed by POWER and for Group B POWER followed by FTP.
Measures
Clinical assessments
Clinical outcomes were assessed using valid and reliable tools for stroke that include all levels of the International Classification of Functioning, Disability, and Health 28 : the Modified Ashworth Scale, 29 Fugl-Meyer Upper-Extremity Motor Score (UEFMMS) 30 and the shoulder/elbow portion of the UEFMMS (30 points), 27 and European Stroke Scale 31 to characterize impairment; the Chedoke-McMaster Hand and Arm Inventory (CMHAI) 32 to characterize activities; and the Reintegration to Normal Living index 33 to characterize participation.
Kinematics of functional reach to grasp
Kinematics were obtained during performance of self-paced functional reach to grasp. Participants were seated in a straight-back chair with the paretic UE resting on the ipsilateral thigh, the shoulder in neutral flexion/extension and internal rotation, and the elbow at 75° to 90° of flexion with the wrist resting in pronation and were instructed to reach, grasp, and retrieve a full soda can positioned on a table top at 80% of arm’s length (ie, “Coke can” task 34 ). Two trials were obtained.
A 7-camera Motion Capture System (Qualisys North America Inc, Charlotte, North Carolina) recorded (120 Hz) displacements of 16 reflective markers, which were used to reconstruct 3-dimensional movements of the arm, forearm, and trunk. During dynamic trials, markers were positioned at C7, the sternal notch, right and left acromions, the lateral epicondyle, radial and ulnar styloid processes, the third dorsal metacarpal phalangeal joint, and the dorsal interphalangeal joint of the index finger, and triads were placed on the superior thirds of the upper arm and forearm. Static trials included an additional marker on the medial epicondyle to create the kinematic model. A 2.2-cm wide piece of reflective tape on the superior border of the soda can identified the reaching target.
Therapeutic Intervention
The treatment algorithms have been previously described in detail, 35 in which the 2 interventions were combined in a “hybrid” therapy. Here, all participants received both forms of interventions in separate bouts to allow comparison of individual treatment effects. FTP involved practice of functional tasks using a progression of 6 therapeutic goals and 9 activity categories. Specific therapeutic tasks were chosen from these activity categories based on participant-specific goals and baseline functional level and practiced on a structured rotation within the framework of the overriding therapeutic goals. POWER involved 5 reciprocal UE movements—shoulder abduction/adduction, shoulder flexion/extension, shoulder external/internal rotation, transverse plane elbow flexion/extension, and wrist flexion/extension—which were trained using a dynamometer (Biodex System 3.0 Pro). Custom-designed attachments to enable participants with impaired grasp to engage the dynamometer were used by all participants. POWER involved a standardized progression in which movement speed and the number of training sets (ie, 10 repetitions each) were adjusted to maintain a constant period of active muscle work 22 within each session. Both the FTP and Power programs are elaborated in Figure 2.

Overview of the therapeutic interventions. Consistent with our previous work, the FTP program addressed six global therapeutic goals, delineated in Table, A. Therapeutic activities were developed on the basis of the current therapeutic goal, the nine activity categories (listed in the sub-table, B), the participant’s functional ability and his/her personal goals. Each 90-minute treatment session involved 15 minutes of stretching and warm up, followed by practice of activities in each of the 9 categories for 8minutes each. Specific examples of activities for high and low functioning individuals are provided in A. To assure consistency across all participants, treatment progressed to the next therapeutic goal on the timeline specified, the nine activity categories were presented in rotation, and timing of practice within each category was adhered to. During POWER training, participants performed both concentric and eccentric actions of: shoulder flexion, shoulder abduction, shoulder external rotation, elbow flexion and extension, wrist flexion and extension. Over the course of the 10 week intervention, movement speed ranged from 30 to 90 degrees per second for eccentric and from 30 to 180 degrees per second for concentric actions. Each bar in sub-plot C represents one set of 10 repetitions at the criterion speed noted on the Y-axis. A primary goal of power training is to improve the capacity for force production in dynamic conditions. Therefore, the training prescription was progressed through advancement of the criterion movement speed. Because the neuromuscular active state differs with movement speed, the number of sets was adjusted to maintain a consistent work:rest ratio across the 10 week program. Referenced to Week 1 (3 sets of each exercise: 30o/s, Con 60o/s, Ecc 30o/s), the work:rest averaged 1.07 (±.08, range 0.97-1.27). Each set involved 10 repetitions.
Kinematic Analysis
Marker data were identified using Qualisys Track Manager (Version 2.1, Qualysis, Gothenburg, Sweden), modeled using Visual3D (Version 4.00.19, C-Motion, Germantown, Maryland), and kinematic trajectories were analyzed using custom-written Matlab (Version 7.0, The Mathworks, Natick, Massachusetts) scripts. Kinematic data were low-pass filtered (12 Hz cutoff) using a bidirectional fourth-order Butterworth filter. The start of movement (SOM) was defined as the first point at which the velocity of the marker on the third metacarpophalangeal joint exceeded 5% peak velocity and the end of movement (EOM) as the last point at which the velocity of this marker fell below 5% of the peak. 10
The following parameters were calculated from the kinematic trajectories: (1) mean velocity, to quantify movement speed; (2) reach-path ratio and submovements, to quantify movement accuracy; (3) time to peak hand velocity, time to maximum shoulder flexion, and time to maximum elbow extension, to quantify motor coordination; and (4) shoulder and elbow ROM and trunk displacement, to quantify movement execution. Movement time was defined as the time between SOM and EOM. Maximum hand velocity was defined as the maximum tangential linear velocity of the marker on the third metacarpophalangeal joint and mean velocity as the average of the hand’s speed during the entire movement. Submovements were defined as the number of peaks in the hand velocity profile as defined by SOM and EOM, described above.5,8,10,36,37 Time to peak hand velocity, time to maximum shoulder flexion, and time to maximum elbow extension were defined as the time between SOM and maximum hand velocity, maximum shoulder flexion, and maximum elbow extension, respectively. ROM was calculated for shoulder flexion and elbow extension as the difference between the maximum and minimum joint angle achieved. Trunk displacement was defined as the sagittal plane displacement of the sternum marker.
Statistical Analysis
Statistical analysis was performed with Matlab. Data were tested for normality using the Kolmogorov-Smirnov test. Kinematic data revealed normally distributed data (
Three sets of comparisons were performed for both clinical and kinematic data: the first 2 evaluated treatment-specific changes between FTP and POWER, whereas the third tested the treatment order (A, FTP followed by POWER, vs B, POWER followed by FTP). The full set of comparisons included the following:
The primary treatment effect, characterized by comparing block 1 change scores between FTP and POWER, was evaluated.
To probe for a potential period effect, the difference in magnitude of block 1 and block 2 change scores was compared within each treatment order (ie, POWER − FTP for order A versus FTP − POWER for order B). If FTP and POWER produced equivalent effects, this comparison would be nonsignificant because both between-block differences would reveal a potential period effect. However, a nonzero difference between orders A and B would reveal additional information regarding differential treatment effects for FTP versus POWER. 38
The effect of treatment order was determined by comparing the overall change between baseline and end of block 2 (ie, sum of block 1 and block 2 change scores for each group (order A vs order B).
Clinical Data a
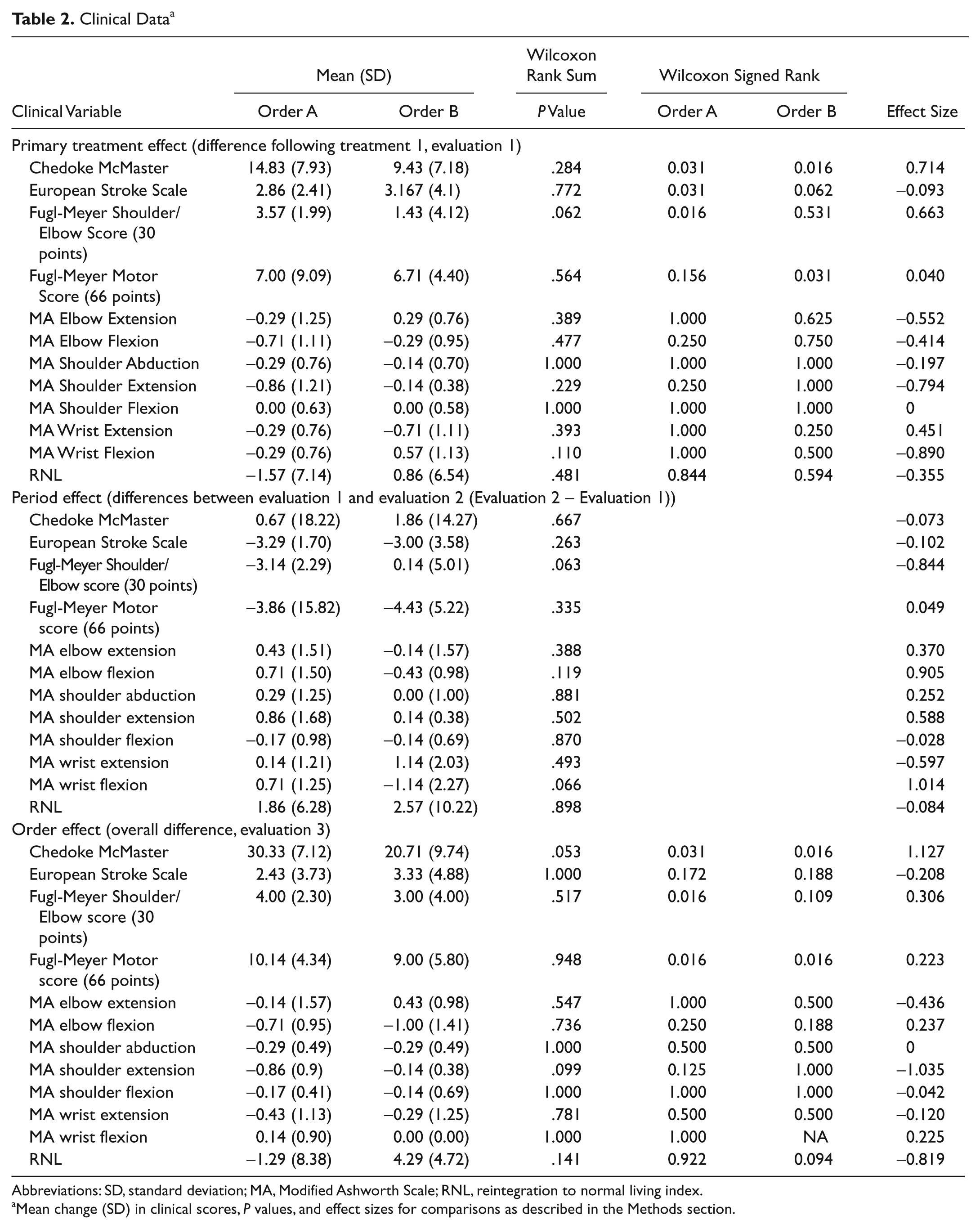
Abbreviations: SD, standard deviation; MA, Modified Ashworth Scale; RNL, reintegration to normal living index.
Mean change (SD) in clinical scores,
Kinematic Data a
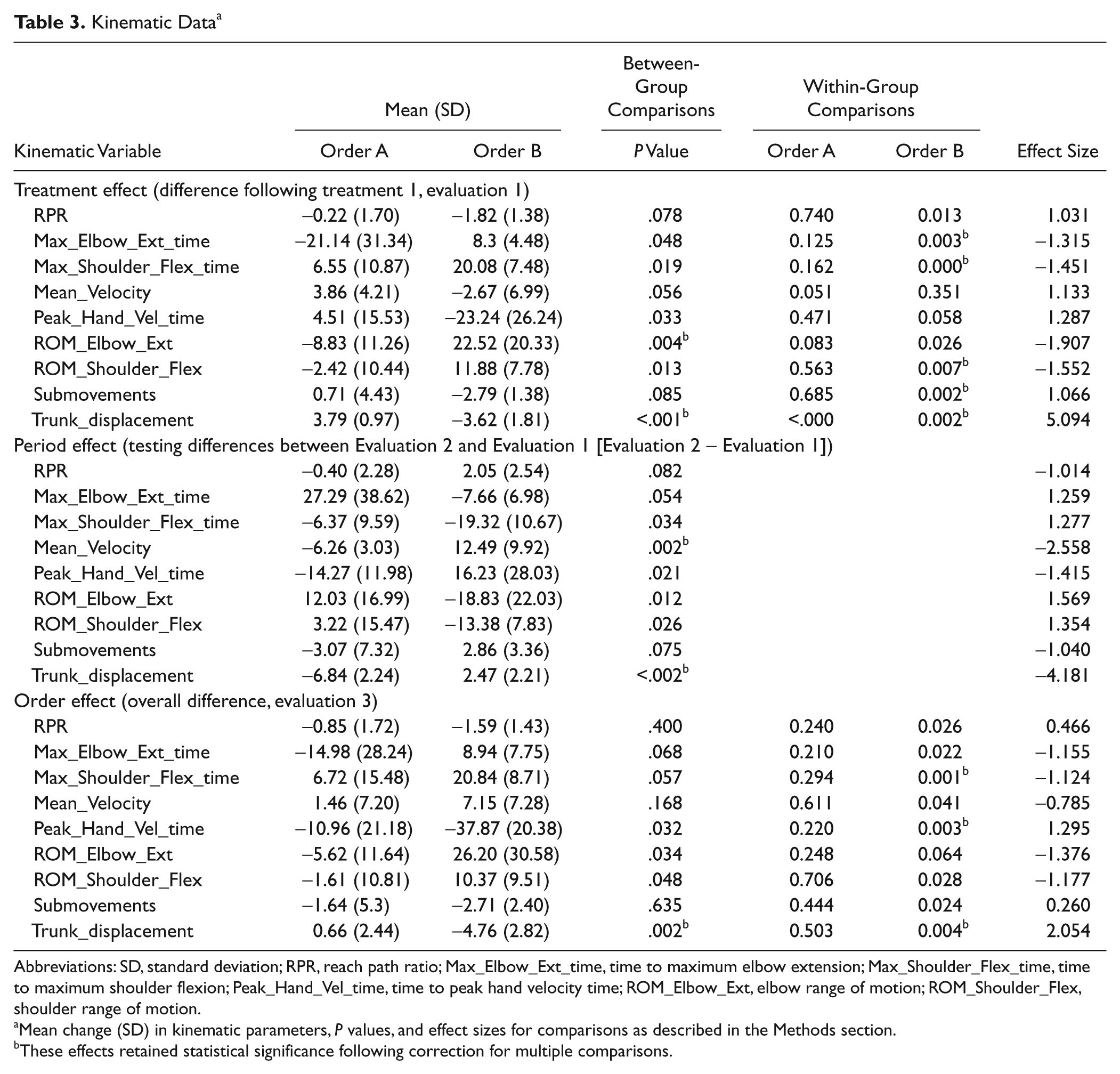
Abbreviations: SD, standard deviation; RPR, reach path ratio; Max_Elbow_Ext_time, time to maximum elbow extension; Max_Shoulder_Flex_time, time to maximum shoulder flexion; Peak_Hand_Vel_time, time to peak hand velocity time; ROM_Elbow_Ext, elbow range of motion; ROM_Shoulder_Flex, shoulder range of motion.
Mean change (SD) in kinematic parameters,
These effects retained statistical significance following correction for multiple comparisons.
Results
Individual participant characteristics are summarized in Table 1. At baseline, the mean age of the participants was 59.8 (±15.0) years (2 women); they were 15.22 (±6.7) months poststroke and had a UEFMM score of 33.71 (±9.6) points. Concealed allocation resulted in 8 and 6 participants randomized to treatment orders A and B, respectively. Clinical characteristics revealed no significant differences between groups at baseline.
Clinical Results
Results and effect sizes for all clinical measures are summarized in Table 2.
Treatment effect (FTP vs POWER): The primary treatment effect revealed a significant difference from 0 in the CMHAI for both treatments and in the Fugl-Meyer Shoulder/Elbow score for group A only. Between-group differences, approaching statistical significance, were revealed only in the Fugl-Meyer Shoulder/Elbow score. Following correction for multiple comparisons, no significant within- or between-group differences remained. Thus, based on clinical assessments, our results indicate that both groups improved without differential treatment effects.
Period effect (FTP vs POWER): Marginal between-group differences were revealed only in the Fugl-Meyer Shoulder/Elbow score and Modified Ashworth Scale for wrist flexion. However, following correction for multiple comparisons, no significant differences remained either within or between groups for any of the clinical scales.
Treatment order: The overall treatment effect for both orders A and B was significantly different from 0 in the CMHAI and UEFMMS. Marginal differences between groups were revealed only in the CMHAI. However, following correction for multiple comparisons, no significant differences were revealed either within or between groups for any of the clinical scales.
Kinematic Data
Results and effect sizes for kinematic variables are summarized in Table 3.
Treatment effect (FTP vs POWER): The primary treatment effect revealed significant differences from 0 in time to maximum elbow extension, time to maximum shoulder flexion, shoulder flexion ROM, submovements, and trunk displacement. Between-group differences were revealed in elbow extension ROM and trunk displacement (Figure 3) indicating that following block 1, POWER revealed greater improvements in elbow extension ROM and reduced trunk displacement. These comparisons retained statistical significance following correction for multiple comparisons.
Period effect (FTP vs POWER): Differential treatment effects were revealed in several parameters. Mean velocity and peak hand velocity time improved more after FTP, whereas maximum elbow extension time, maximum shoulder flexion time, elbow extension ROM, shoulder flexion ROM, and trunk displacement improved more after POWER. Following correction for multiple comparisons, between-group differences in mean velocity and trunk displacement retained statistical significance to reveal greater improvements in mean velocity following FTP, whereas trunk displacement improved more after POWER.
Treatment order effect: The overall treatment effect differed significantly from 0 in: time to maximum shoulder flexion, time to peak hand velocity, and trunk displacement. Between-group differences were revealed in trunk displacement, indicating that participants randomized to treatment order B (POWER followed by FTP) revealed greater improvements in trunk displacement (ie, reduced trunk displacement). These effects retained statistical significance following correction for multiple comparisons.
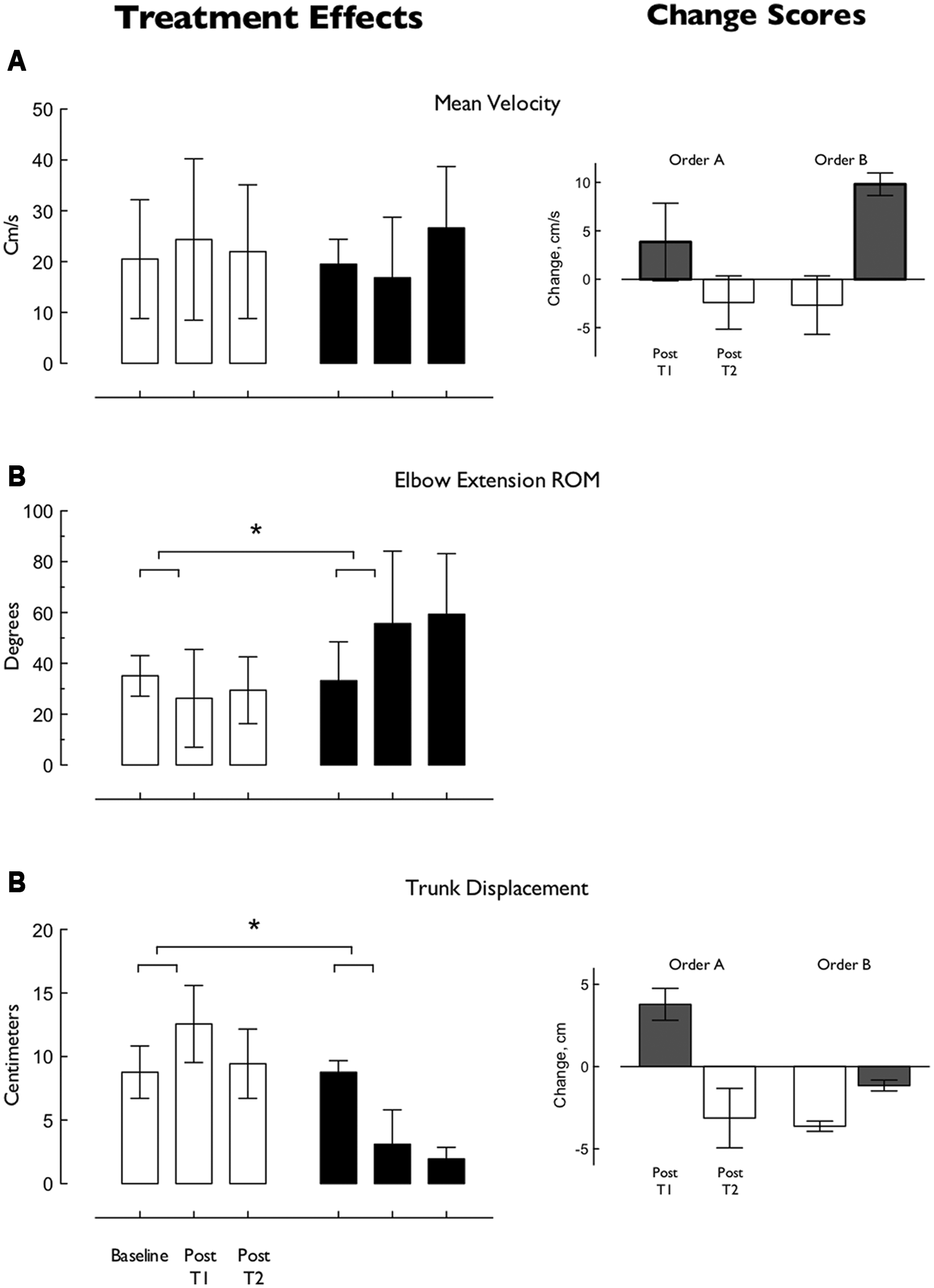
Effects of FTP and Power on key kinematic parameters of functional reach-to-grasp. Plots in the left panel illustrate individual treatment effects. Data are mean (±SEM) of: mean velocity (A), elbow extension ROM (B) and trunk displacement (C) at baseline, and after first and second treatments. Group A (FTP followed by POWER) is represented in white, Group B (POWER followed by FTP) in black. Significant differences between treatments are revealed in elbow extension ROM (B) and trunk displacement (C). Plots in the right panel illustrate change scores (±SEM) after first and second treatment blocks and reveal significant order effects in mean velocity (Right, top row) and reduced trunk displacement (Right, lower row). Changes following FTP are illustrated in gray and following POWER in white. Overall changes in both mean reaching velocity and trunk displacement favor Order B (eg, POWER followed by FTP).
Discussion
Compensation Versus Restoration
Here, we investigated concurrent clinical and kinematic changes following 2 UE rehabilitation treatments—FTP and POWER training—with the primary aim of understanding whether improved UE function poststroke results from the use of compensatory movements or restoration of more normal movement patterns. As hypothesized, behavioral motor improvements (eg, kinematics) post-POWER reveal restoration of more normal movement function. In contrast, behavioral changes post-FTP reveal compensatory movement strategies. Whereas mean reaching velocity increased post-FTP, this apparent improvement involved concurrent reductions in shoulder flexion and elbow extension ROM and increased trunk displacement—changes indicating reinforcement of compensatory movement strategies. 8 Following POWER, participants increased shoulder flexion and elbow extension ROM, reduced associated trunk displacement, and also demonstrated greater improvements in time to maximum shoulder flexion and elbow extension, parameters contributing to normal interjoint coordination. As revealed by a shift toward normal across numerous kinematic parameters, 3 motor patterns more similar to that of healthy individuals were revealed following POWER. These behavioral manifestations can be attributed to restoration or true motor recovery.
Effect of Treatment Order
We addressed our secondary aim, understanding the effect of treatment order, using a crossover design. As hypothesized, our data reveal that POWER followed by FTP produced greater improvements, primarily significantly reduced trunk displacement, indicating a marked reduction in compensatory movements. Notably, this reduced compensation was accompanied by reappearance of normal patterns of shoulder and elbow movement present prior to stroke.
Use of Kinematics to Investigate Motor Control
It is important to note that clinical assessments of motor function revealed similar improvements after both POWER and FTP. Clinical scales focus on gross indicators of task accomplishment (ie, success or failure of task completion, assistance required, and time to task completion) but are unable to discern differences in actual movement performance. This focus on task completion ignores the fact that individual participants may adopt unique approaches. Indeed, comparable change scores may result from the use of wholly different movement strategies involving either the adoption of compensatory movements or the acquisition of normal movement patterns. In this light, the results of the present study emphasize the important contribution of kinematics 5 to understanding the effects and efficacy of neurorehabilitation. The goal of rehabilitation is not only to facilitate behavioral improvement at the level of task completion but to promote neural recovery and, ultimately, improve the individual’s quality of life. 40 Accurate evaluation of motor dysfunction is therefore fundamental to developing rehabilitation interventions with the capacity to produce neural recovery that manifests as improved behavioral function. In contrast to the clinical assessments, our results illustrate that UE kinematics3,5,10,17 discriminate between normal and compensatory movement strategies and therefore reveal the actual effects of rehabilitation interventions. Although the therapeutic goal in this study was not specifically to “train to normal,” 41 our kinematic data reveal differential intervention responses, with reacquisition of many features of normal movement following POWER.
Neuroplasticity and Specificity of Training
The capacity for neural plasticity after stroke is now well recognized and may include different degrees of physiological recovery. Current evidence suggests that in both animal and human models with and without stroke, neural recovery and reorganization of neuronal function are not only spontaneous processes but are strongly influenced and modulated by activity (eg, activity-dependent plasticity). 42 Because recent studies demonstrate that neural plasticity is task specific, 43 task-related practice is considered essential for driving neuroplasticity. 43 However, as Daly and Ruff suggest, “a central assumption of motor learning is that the neural structures controlling movement are required to adapt to constraints that are imposed by: the structure of the musculoskeletal system, the physical laws governing movement, and the impairments that are present.”44p2033 Building on these assumptions, the constraints imposed on the individual in a given motor task must be incorporated into a successful treatment plan. For example, an individual impaired by weakness may be unable to reach and grasp a soda can, and thus the performance (eg, completion) of this task would necessarily involve compensation with other body segments or use of motor strategies not typically involved in the movement. Rehabilitation using repetitive task practice would reinforce these abnormal movements, and the motor skill acquired would not be the one desired. Latash and Anson 41 have proposed that the choice of a particular movement pattern is based on priorities and that under atypical conditions (ie, structural or biomechanical changes within the neuromusculoskeletal system poststroke), the central nervous system may reweight its priorities for movement execution, leading to altered movement patterns. This perspective suggests that therapeutic approaches directed toward remediation of underlying impairments may reduce the need for reweighting movement priorities. Weakness is one of the most significant impairments poststroke.10,20,21,25 Results of the present study indicate that therapeutic intervention directly addressing weakness effectively restores motor control in the hemiparetic UE.
Our results also suggest that an effective therapeutic approach most likely involves multiple stages, each with a specific goal. The present study design reflects 2 such stages: (1) remediation of hemiparetic weakness by training the individual to recruit and control the requisite force for task execution, followed by (2) use of this enhanced neuromechanical capacity in repetitive practice of close-to-normal movements. Therefore, our results are consistent with contemporary principles of neuroplasticity. 43 First, force production is a neurological phenomenon, and there is considerable evidence that strengthening elicits profound adaptations at both supraspinal and spinal levels in nondisabled45-49 and neurologically impaired populations. 50 Moreover, the neural phase of strengthening is argued to involve motor learning.51,52 Second, weakness contributes to impaired movement patterns10,53,54 and functional motor performance, 55 whereas changes in critical movement parameters are demonstrated poststrengthening.56,57 Finally, repetitive practice of close-to-normal movements is an important aspect of training specificity. Our results suggest that functional outcomes are enhanced when therapeutic interventions train the capacity of the motor system prior to engaging in repetitive task practice.
Limitations
We acknowledge limitations of the present study. First, FTP did not provide trunk stabilization, whereas POWER provided some stabilization from the dynamometer chair and chest strap. Other authors have suggested 23 that implicit feedback provided by trunk stabilization may be influential in relearning of normal motor strategies. What is important in this regard is our use of a crossover design and its capacity to monitor differential treatment responses in the same individuals. Although all participants received both FTP and POWER, our results reveal a significant order effect when POWER preceded FTP. If the primary affordance of POWER training was the trunk restraint, it is unlikely to have been retained throughout the FTP treatment, which reintroduced the opportunity to use and practice with compensatory trunk movements. Second, although not an explicit limitation, both treatments were based on systematic approaches for intensity and progression; however, the physical and physiological demands of POWER were indeed more intense than those of FTP. Consistent with the literature, which indicates that better functional outcomes are associated with higher treatment intensity, 58 our data suggest that FTP in isolation may lack the requisite intensity to stimulate the appropriate neuroplastic processes underlying recovery of normal movement patterns. Third, heterogeneity in lesion location among our participants precludes our ability to make direct associations between lesion location and functional outcome. There is a need for further research to analyze the relationship between CST involvement and individual responses to interventions. Finally, our kinematic analysis is based on 2 trials of each movement. There is no consensus regarding the appropriate or optimal number of behavioral trials to sample. Some investigators capture multiple trials to understand the variability/consistency over repeated trials of a movement 5 ; however, in studying individuals with severely compromised motor function, our primary concern was to avoid inducing a confounding effect of fatigue.
By current standards of clinical rehabilitation in the United States, 60 sessions of one-on-one treatment delivered over 20 weeks may not readily translate to clinical practice. However, our findings offer an opportunity for reconsideration of appropriate rehabilitation practice models. It is important to note that participants in this study presented with lower functional status than in many currently reported studies,35,59 yet substantial UE improvements were revealed across levels of measurement. Our results suggest that our approach of 90-minute treatments thrice weekly for an extended period enables incorporation of progressive, incremental physiological and behavioral changes into movements encountered in the course of daily life. 60 An additional consideration for rehabilitation practice models is that the systematic power training component of this intervention could be incorporated in the outpatient or community settings, where multiple individuals can participate simultaneously.
Clinical Relevance
We directly compared 2 intervention approaches with the aim of demonstrating differential mechanisms of motor recovery. Our findings demonstrate that POWER promotes restoration of normal movement patterns, which may result from increased neural drive from the impaired hemisphere. The results demonstrate that it is possible to correct compensatory movement strategies in persons poststroke, such as those we studied, and confirm the lack of deleterious effects of high-intensity activities. Taken together, our results offer novel insight for identifying effective UE rehabilitation interventions that promote restoration of normal motor function. Further experimental studies are necessary to identify the physiological mechanisms that underlie restoration of normal movement.
Footnotes
Acknowledgements
We thank the following individuals for their contributions to this project: Jody M. Dozono, MPT, Kasthuri Veeragathan, PT, GCS, and Fayeza Mullamithawala, PT, for delivering the intervention protocols; Jeffrey Jaramillo, PT, MS, and Christine A. Dairaghi for conduct of patient assessments; and Dr James Cauraugh and Dan Neal for helpful comments on a previous version of this manuscript.
Authors’ Note
A portion of this work has been presented in abstract form at the 2009 International Society for Posture and Gait Research, Bologna, Italy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This material is based upon work supported in part by: the Department of Veterans Affairs Office of Research and Development Rehabilitation R&D Service - Merit Review #B3964R and Research Career Scientist Award (#F7823S) to CP and the VA RR&D Brain Rehabilitation Research Center of Excellence (#B6793C). MC is supported by a University of Florida Alumni Fellowship.
