Abstract
Objective. To investigate short- and long-term effects of intensive rehabilitation on ataxia, gait, and activities of daily living (ADLs) in patients with degenerative cerebellar disease. Methods. A total of 42 patients with pure cerebellar degeneration were randomly assigned to the immediate group or the delayed-entry control group. The immediate group received 2 hours of inpatient physical and occupational therapy, focusing on coordination, balance, and ADLs, on weekdays and 1 hour on weekends for 4 weeks. The control group received the same intervention after a 4-week delay. Short-term outcome was compared between the immediate and control groups. Long-term evaluation was done in both groups at 4, 12, and 24 weeks after the intervention. Outcome measures included the assessment and rating of ataxia, Functional Independence Measure, gait speed, cadence, functional ambulation category, and number of falls. Results. The immediate group showed significantly greater functional gains in ataxia, gait speed, and ADLs than the control group. Improvement of truncal ataxia was more prominent than limb ataxia. The gains in ataxia and gait were sustained at 12 weeks and 24 weeks, respectively. At least 1 measure was better than at baseline at 24 weeks in 22 patients. Conclusions. Short-term benefit of intensive rehabilitation was evident in patients with degenerative cerebellar diseases. Although functional status tended to decline to the baseline level within 24 weeks, gains were maintained in more than half of the participants.
Introduction
Task-oriented practice such as constraint-induced movement therapy, robotics, and gait training may improve motor function of patients presenting with hemiparesis after stroke.1-3 On the other hand, few strategies for the neurorehabilitation of cerebellar ataxia have been reported. 4 Especially in patients with degenerative cerebellar diseases, there are at least 2 essential questions regarding the benefit of rehabilitation. The first is whether patients can learn or relearn motor sequences required for activities of daily living (ADLs) because the cerebellum plays a crucial role in motor learning. 5 Several studies have demonstrated that patients with cerebellar damage show impaired motor learning, including adaptation learning6-11 and motor sequence learning.12-15 The cerebellum is also involved in shifting movement performance from an attentionally demanding state to a more automatic state. 16 Thus, it is likely that such impaired motor learning may influence the effect of rehabilitation on motor function and ADLs in patients with cerebellar damage. Indeed, impaired motor learning correlated with reduced rehabilitation gains in ataxic patients with infratentorial stroke. 17 It is also unclear whether the impaired motor learning is compensated by repeated practice for balance, gait, and ADLs. Ilg and colleagues 18 reported that continuous coordination training for 4 weeks improved motor performance and reduced ataxia symptoms in patients with cerebellar ataxia. The effect lasted for 8 weeks with a self-directed home exercise program.
The second question is whether functional gains can be sustained over several months after a rehabilitative intervention has a significant short-term effect on functioning. Consolidation and retention of learned motor skills could be hampered by progressive cerebellar degeneration. Ilg et al 19 also observed that improvements in motor performance and achievements in ADLs persisted 1 year after a 4-week intensive coordinative training, which was followed by a home training program. More detailed analyses for the serial changes of outcome measures might be helpful in optimizing a home program to maintain functional status.
We designed a randomized controlled trial to assess the effect of intensive rehabilitation in a relatively large number of patients with degenerative cerebellar diseases. In this study, patients received a greater dose of rehabilitation (2 hours per day for 4 weeks) compared with rehabilitation provided in prior reports. We evaluated the functional status at 0, 4, 12, and 24 weeks after the intervention. We hypothesized that ataxia and its related disability would be improved after focused inpatient rehabilitation and the gains maintained over several months.
Methods
Participants
Patients with isolated cerebellar ataxia caused by degenerative cerebellar diseases were recruited to the study because interpretation of rehabilitation outcome might be confounded by the presence of the pyramidal and extrapyramidal signs, peripheral neuropathy, and other neurological deficits. Specifically, we enrolled patients with spinocerebellar ataxia type 6 (SCA 6), 20 SCA 31, 21 and idiopathic cerebellar ataxia (ICA) presenting with pure cerebellar ataxia. Diagnosis of these patients was based on positive results of DNA analysis for SCA 6 and SCA 31 and negative results of DNA analysis as well as no family history of cerebellar ataxia for ICA. To be eligible for the study, patients had to be able to ambulate 10 m independently or with the assistance of 1 person. Exclusion criteria included history of stroke, other neuromuscular diseases and dementia, complications including severe infection, deep vein thrombosis, orthopedic diseases causing pain during gait, unstable angina and cardiopulmonary disease causing dyspnea during exercise, and recent history of surgical intervention, including coronary artery bypass graft. Patients were recruited from outpatient clinics of neurology located in the central areas of Japan from September 2007 to August 2009. This study was approved by the local ethical committee of each participating facility, and written informed consent was obtained from each patient. This study is registered with UMIN clinical trials registry, number UMIN000000824
Randomization
Patients were randomly assigned to the immediate or delayed-entry control group by using sealed envelopes chosen by a person who was not involved in this study. Patients in the immediate group received 4-week inpatient rehabilitation after 1 baseline assessment and those in the delayed-entry control group received the intervention after 2 baseline assessments performed at 0 and 4 weeks. Thus, patients in the delayed-entry control group served as controls in assessing the short-term effect of rehabilitation. Long-term follow-up data were collected from the patients in both groups up to 24 weeks after the intervention. Thus, the short-term effect was assessed as a randomized controlled trial and the long-term effect as an observational study (Figure 1).
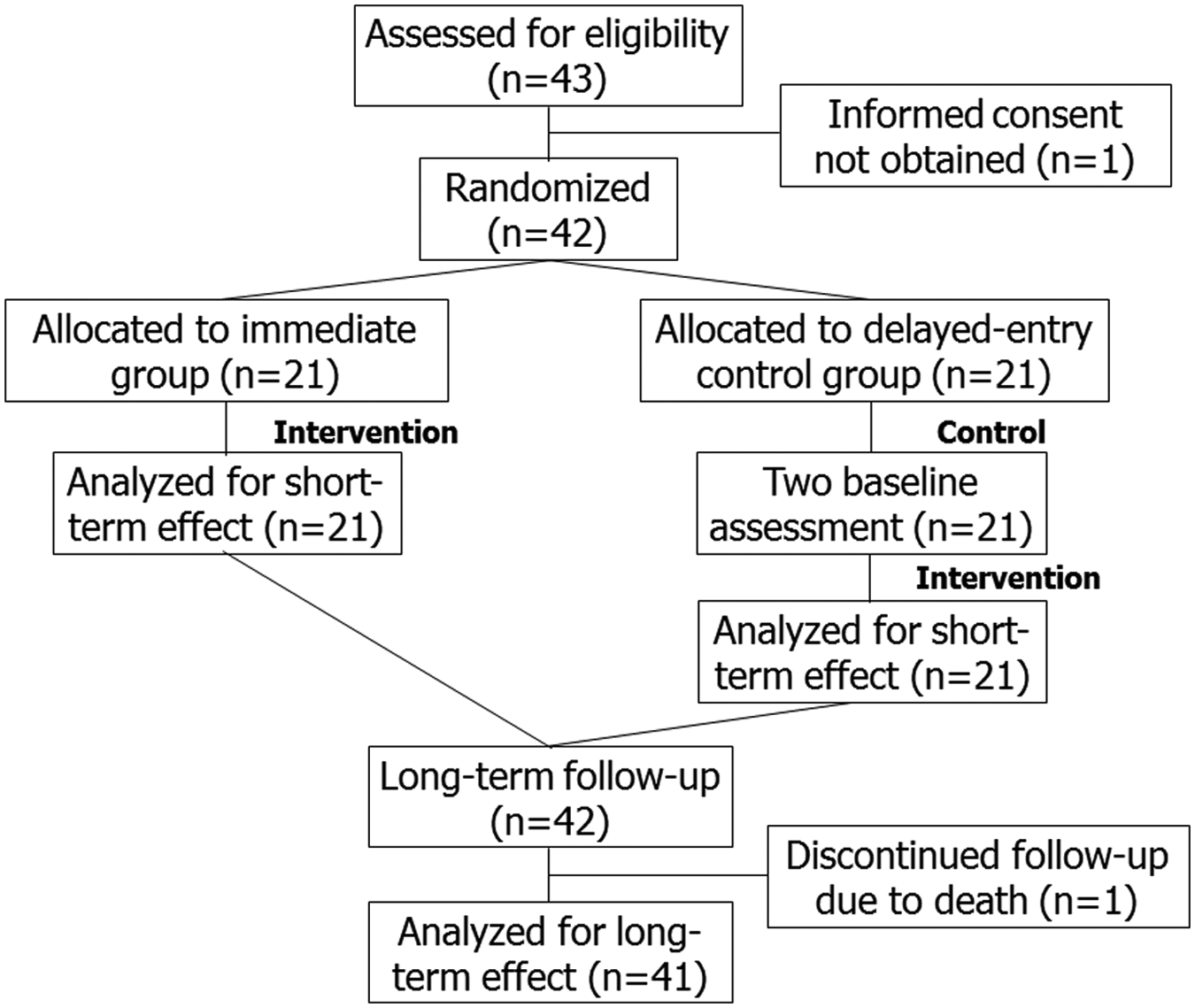
Trial profile.
Rehabilitative Interventions
All patients were admitted to Morinomiya Hospital to receive 4-week inpatient rehabilitation, including 1 hour of physical therapy and 1 hour of occupational therapy per day on weekdays and 1 hour of either physical or occupational therapy on weekends. Each therapy focused on the improvement of ataxia and ADLs. Physical therapy emphasized improving postural balance and gait. The program included general conditioning; range-of-motion exercise for trunk and limbs; muscle strengthening; static and dynamic balance exercise with standing, kneeling, sitting, and quadruped standing; mobilizing the spine while prone and supine; walking indoors and outdoors; and climbing up and down stairs. The occupational therapy program emphasized improving ADLs and relaxation, hygiene, dressing, writing, eating, toileting, bathing, balance exercises, reaching, coordinative tasks of the upper limbs and trunk, and dual motor tasks such as handling objects while standing and walking. After discharge, patients were instructed to maintain a similar level of activity in their daily lives as they did before. Patients receiving home-based rehabilitation at baseline—usually 20 to 40 minutes of physical therapy per week—were advised to continue the program after discharge at the same frequency as they did before admission.
Outcome Measures
The primary outcome measures were Scale for the Assessment and Rating of Ataxia (SARA)22,23 and the Functional Independence Measure (FIM). 24 Secondary outcome measures included gait speed, cadence, functional ambulation category (FAC), 25 and number of falls. These were assessed by physicians or therapists who were familiar with the measures and were blinded to the group allocation in each participating facility.
Statistics
Although there were no available data for the effect of rehabilitation for degenerative cerebellar diseases, the average gains ± standard deviation of the FIM score in patients with chronic cerebellar stroke were approximately 7 ± 8 in our institute. Thus, we expected a 1-point improvement in 7 motor items (gait, stairs, transfer ×3, bathing, and toileting) of FIM related to the lower-extremity and balance functions. We postulated that the standardized effect size for short-term effect, that is, immediate gain after 4 weeks of rehabilitation, was 7out of 8 = 0.88. With an α value of .05 and β value of .20, the minimal sample size estimated was 36. 26 For the long-term outcome, each outcome measure was assessed before (pre) and immediately after (post) 4 weeks of intervention as well as at 4 weeks, 12 weeks, and 24 weeks after the intervention in both groups.
Statistical Analyses
For comparisons of demographic data and the baseline outcome measures between 2 groups, we used the unpaired t test, Wilcoxon rank-sum test, and the χ2 test as applicable.
The short-term effect of rehabilitation was evaluated by between-group differences for SARA, FIM, FAC, and number of falls using the Wilcoxon rank-sum test. For evaluation of the long-term effect on these measures, we relied on the Friedman test followed by the Wilcoxon signed-rank test for pairwise comparisons coupled with Bonferroni adjustment of multiple comparisons. For gait speed and cadence, we performed repeated-measure analyses of variance (ANOVAs) for the evaluation of short-term and long-term effects followed by the Bonferroni test for the adjustment of multiple comparisons. Statistical significance was set at P < .05 and P < .05 out of 4 for Bonferroni corrected data for multiple comparisons. Statistical analyses were performed using SPSS 18J (SPSS Inc, Chicago, Illinois).
Results
A total of 43 patients with degenerative cerebellar diseases were recruited. We could not obtain an informed consent from 1 patient. Consequently, 42 patients (20 women), including 20 patients with SCA 6, 6 with SCA 31, and 16 with ICA were randomized to the immediate or delayed-entry control group. All patients presented with pure cerebellar ataxia. None of the patients showed extrapyramidal and pyramidal signs, ophthalmoplegia, and peripheral neuropathy. Mean age ± standard error was 62.5 ± 1.7 years, ranging from 40 to 82 years old. The duration of the diseases was 9.8 ± 1.0 years, ranging from 1 to 30 years (Table 1).
Baseline Characteristics of Patients With SCD
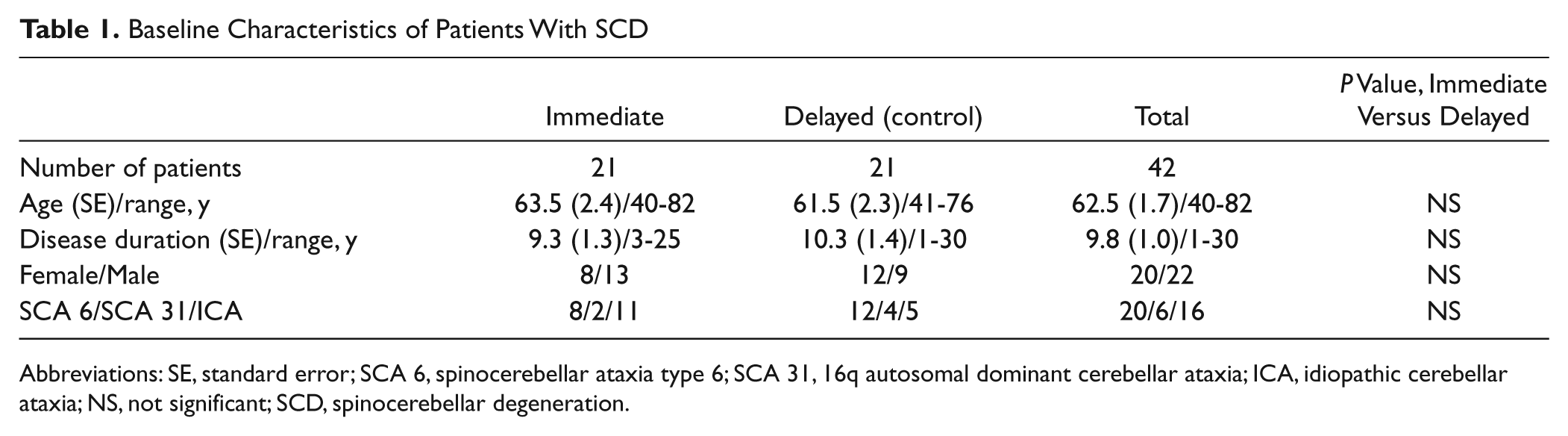
Abbreviations: SE, standard error; SCA 6, spinocerebellar ataxia type 6; SCA 31, 16q autosomal dominant cerebellar ataxia; ICA, idiopathic cerebellar ataxia; NS, not significant; SCD, spinocerebellar degeneration.
Short-Term Effect of Intensive Rehabilitation
A total of 21 patients were randomized to the immediate group and 21 to the delayed-entry control group. Demographic data and the baseline outcome measures of the 2 groups were comparable (Tables 1 and 2). The immediate group showed significantly greater improvement after 4 weeks of rehabilitation in SARA (P < .001), total and motor scores of FIM (P < .01), FAC (P < .05), and number of falls (P < .01) compared with the delayed-entry control group (Table 2). In terms of subscores of SARA (Table 2), improvement of truncal ataxia (gait, stance, and sitting) was more prominent than that of limb ataxia (finger chase, nose–finger test, fast alternating hand movements, and heel-shin slide). Similarly, the immediate group showed significantly greater improvement in gait speed (F1,40 = 7.5; P < .01) and normalized gait speed (F1,40 = 7.5; P < .01) compared with the delayed-entry control group, as indicated by significant Time × Group interactions.
Short-Term Effect of Inpatient Rehabilitation a
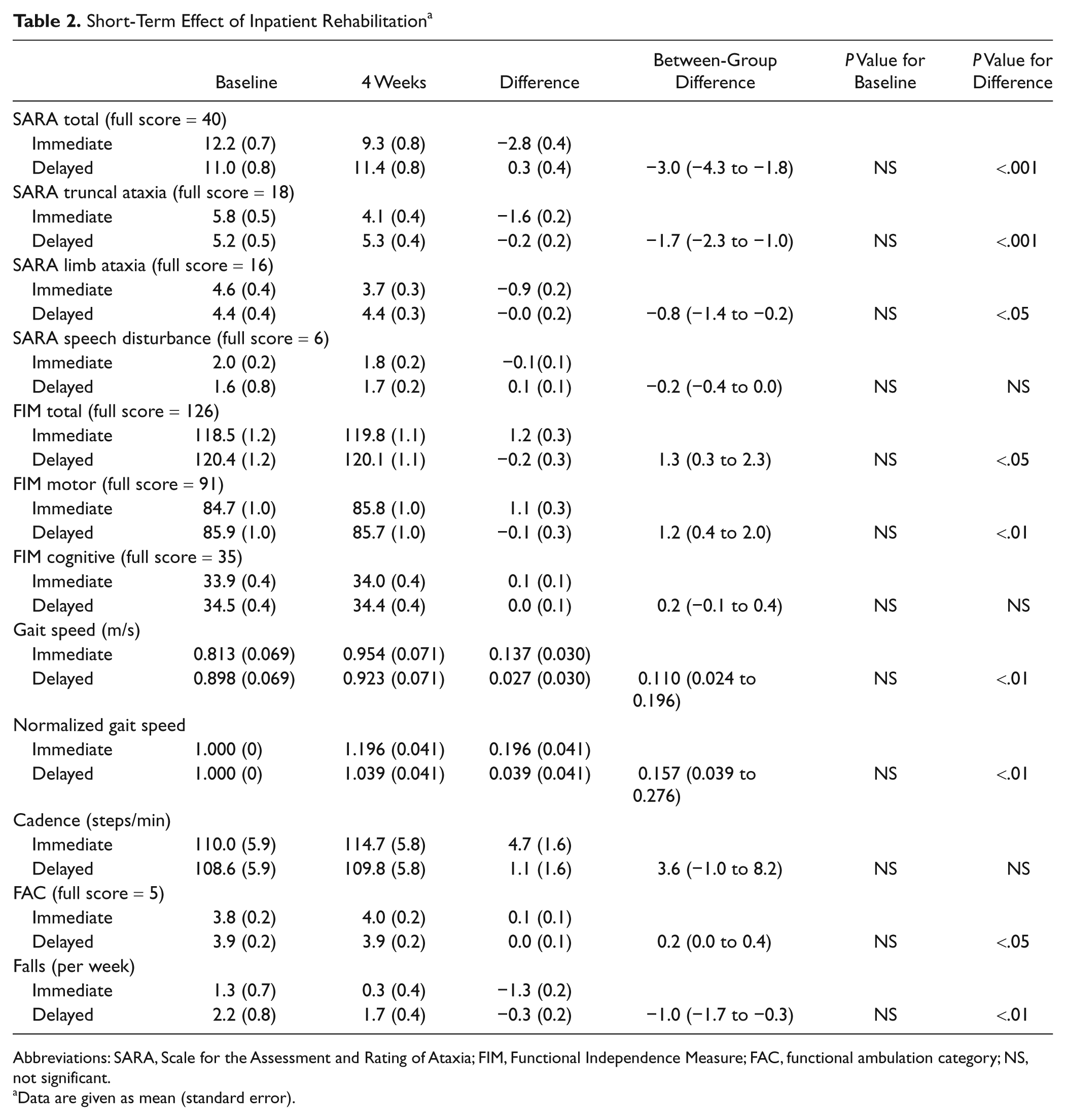
Abbreviations: SARA, Scale for the Assessment and Rating of Ataxia; FIM, Functional Independence Measure; FAC, functional ambulation category; NS, not significant.
Data are given as mean (standard error).
Long-Term Effect of Intensive Rehabilitation
One patient died because of cerebral hemorrhage at 17 weeks after the intervention, and the long-term effect was analyzed in 41 patients in both groups. The Friedman test showed significant effects of rehabilitation on SARA, total FIM, motor subscore of FIM, FAC, and number of falls. Similarly, there were significant main effects for time in gait speed and normalized gait speed (Table 3). The gains in these measures attenuated during the follow-up period. For SARA, gait speed, and FAC, post hoc testing showed that the gains were still significant at 12 weeks after the intervention. Normalized gait speed maintained improvement even at 24 weeks. However, FIM was less sensitive in detecting changes in our patient group with mild to moderate ataxia, although the improvement was significant at 4 weeks after the intervention. For falls, we did not observe any sustained improvement. There was no significant interaction between time and diagnosis in each measure, indicating that there was no difference in the time course of functional status among different types of degenerative cerebellar diseases.
Long-Term Changes of Outcome Measures After 4-Week Inpatient Rehabilitation in Patients With Degenerative Cerebellar Diseases a
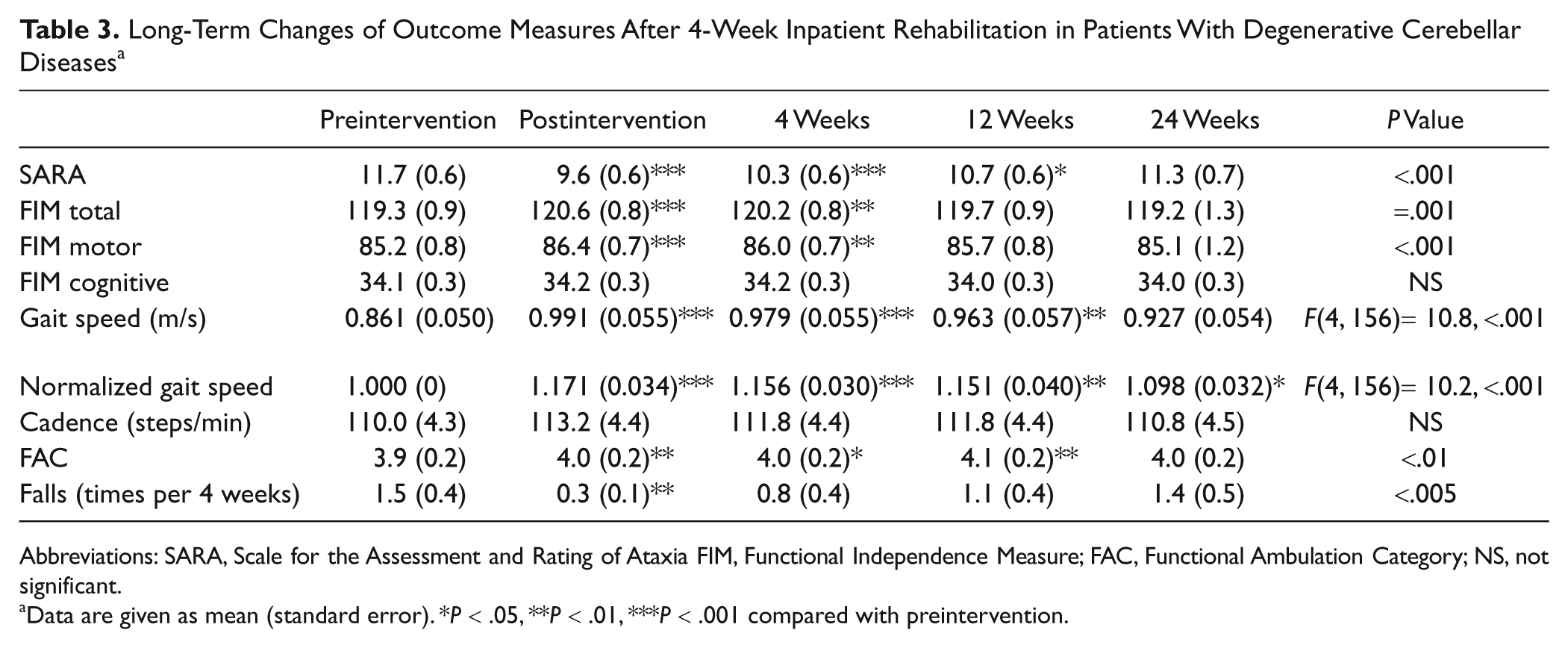
Abbreviations: SARA, Scale for the Assessment and Rating of Ataxia FIM, Functional Independence Measure; FAC, Functional Ambulation Category; NS, not significant.
Data are given as mean (standard error). *P < .05, **P < .01, ***P < .001 compared with preintervention.
In 22 patients, at least 1 item of the outcome measures was better at 24 weeks than at baseline, whereas all measures were lower at 24 weeks than at baseline in 19 patients. Thus, we compared demographic and clinical data, including type of disease, age, gender, duration of disease, SARA, FIM, gait speed, cadence, FAC, and number of falls between the groups with and without sustained improvement. A significant difference was seen only in the baseline SARA score. Patients with sustained improvement had less severe ataxia (lower SARA score) than those without sustained improvement (8.9 ± 0.7 vs 12.7 ± 0.9; P < .05). Similarly, the stepwise logistic regression model revealed that the baseline SARA score was a significant determinant (odds ratio = 0.797; 95% CI = 0.665-0.970; P < .05) for sustained improvement at 24 weeks after intensive rehabilitation. The receiver operating characteristic curve (ROC) analysis showed that the baseline SARA score was moderately predictive of the sustained improvement at 24 weeks (area under the ROC curve = 0.719; CI = 0.560-0.878; P < .05). The sensitivity and specificity were 59.1% and 68.4%, respectively, at the cutoff score of 9.75.
Discussion
We show that 4 weeks of intensive rehabilitation significantly improves ataxia, ADLs, and gait in patients with degenerative cerebellar diseases. This suggests that encoding of motor learning for these skills is at least partly preserved in these patients because functional gains after rehabilitation correlated with capacity for motor learning in patients with cerebellar ataxia. 17 Although such motor learning depends on both cerebellar and basal ganglia systems, 5 our findings suggest that repetitive rehabilitative interventions focusing on balance, gait, and ADLs may improve impairment and its related disability in spite of considerable cerebellar dysfunction. Although the improvement of ataxia was prominent in the trunk, limb ataxia also showed a significant gain. This implies that the effect is not explained by promotion of physical fitness alone.
Previous studies demonstrated impaired motor learning in patients with cerebellar damage.6-16 However, most studies used a stimulus–response design in which the performance might be influenced by action-based working memory that maintains a stimulus–response association rather than a learning mechanism. 15 Indeed, patients with cerebellar degeneration showed improved sequence learning when the responses were directly cued. 15 Because motor sequences required for basic movements are given as direct instructions by therapists, this observation by Spencer and Ivry 15 might partially explain the functional gains after focused rehabilitation in our patients. For instance, therapists modified patients’ balance reaction directly by handling their trunk and then indirectly by providing verbal commands, so that they could avoid rigid axial control. These multimodal inputs, including adjusted visual, somatosensory, and vestibular information, may help patients learn balance skills more efficiently compared with self-practice that simply repeats a balance exercise. Another example might be a dual motor task such as handling objects while standing and walking to promote implicit motor learning.
Long-term follow-up revealed that the gains gradually attenuated as we hypothesized. Ilg and colleagues 18 reported that a less frequent intervention of 1 hour of physical therapy given 3 sessions per week for 4 weeks improved SARA and gait scores in patients with cerebellar ataxia but not with afferent ataxia. The effect lasted for 8 weeks with a self-directed home exercise program. Their observation is comparable with our findings that gains in SARA and gait speed were sustained for 12 weeks and 24 weeks, respectively. In addition to their findings, we showed that the SARA score declined to baseline at 24 weeks. There are at least 2 possible explanations for this decline. First consolidation of the encoded motor skill might be impaired because of cerebellar lesions. Second the progressive degenerative process might impede motor performance itself. Intermittent home-based therapy as well as systematic self-exercise may be necessary to preserve functional status after an intensive intervention. Indeed Ilg et al 19 observed that encouraged self-exercise led to sustained improvement of ataxia up to 1 year. It is also possible that intensive intervention once or twice a year might prevent functional declines in patients.
Although functional gains attenuated to the baseline level at 24 weeks after the intervention, studies regarding natural history of degenerative cerebellar diseases indicated that ataxic symptoms were progressive even in a half year. In patients with SCA 6, there was a 3.1 ± 4.3 point change in International Corporative Ataxia Rating Scale scores for 4 years in a retrospective study, and the change of SARA scores was 1.5 ± 2.0 per year in a prospective follow-up.27,28 Thus, no significant difference in functional status between assessment at the baseline and 24 weeks might be interpreted as an improvement. However, to elucidate if repeated rehabilitative intervention affects the natural history of degenerative disease, long-term evaluation over 10 years is necessary. 29
Of note, more than half the patients showed better functional status at least in 1 of the outcome measures at 24 weeks after the intervention. Subanalyses of the data revealed that the only factor contributing to sustained improvement was severity of ataxia. The ROC analysis suggested that patients whose SARA scores were less than 10 were likely to preserve their functional status. This finding is in accordance with the previous study reporting that the exercise training program significantly improved the neurological indices in a mild stage of SCA 2. 30 We observed no relationship between the time course of the functional status and disease type. Thus, patients with mild ataxia are more likely to benefit from focused rehabilitation regardless of disease type probably because of greater capacity for motor learning compared with those with moderate to severe ataxia.
Although the neural mechanisms underlying functional improvement after focused rehabilitation remain unknown, previous neuroimaging studies have suggested that cerebellar dysfunction might be compensated by neural networks other than the cerebellum. In patients with multiple system atrophy, hand movement induced greater recruitment of frontal mesial areas and superior parietal cortices. 31 Sustained recruitment of the prefrontal areas was associated with execution of pursuit rotor task 17 and treadmill walking 32 in patients with cerebellar stroke. Although there have been no longitudinal trials to determine which activation patterns are beneficial for functional improvement, repetitive practice may promote such compensatory mechanisms within the neural networks of motor control.
There are several limitations in this study. We did not use video examination to ensure complete blinding, but the outcome measures were assessed by examiners who were not engaged in the rehabilitative intervention and were blinded to assignment. We could not examine the effect of a specific component of the interventions. Thus, a nonspecific effect of the intervention might have affected their functional improvement. However, it is unlikely that only a placebo effect affected the patients’ outcome because the gains were still significant at 12 weeks after the intervention. It is also possible that patients were motivated to increase their daily activities after the intervention. But this cannot be verified because of the lack of monitoring of home activities. Sustained improvement was less evident in ADLs as measured by FIM than in ataxic symptoms. One possible explanation is that there was a ceiling effect of this measure in patients who were nearly independent in ADLs. Although a decreased number of falls might be one outcome important in day-to-day life, the intervention did not reduce risk for falls. Environmental setting and practice at home might be more important than hospital-based rehabilitation. Finally, our analysis for subgroups of patients revealed that patients with mild ataxia most benefitted from intensive rehabilitation. An optimized intervention for those with moderate to severe ataxia has to be investigated. In addition, it will also be necessary to investigate if a combined intervention consisting of rehabilitation and neuromodulation, including brain stimulation 33 and neuropharmacological treatment with, for example, thyrotropin-releasing hormone, 34 may enhance functional outcome. 35
In conclusion, it is suggested that intensive and focused rehabilitation can at least partially overcome impaired motor learning in patients with degenerative cerebellar diseases. Further studies will be necessary to determine optimal dose, durations and intervals of therapies to sustain significant functional gains.
Footnotes
The following is the list of the Cerebellar Ataxia Rehabilitation Trialists Collaboration: Naonobu Futamura, Department of Neurology, Hyogo-Cyuo National Hospital; Yusaku Nakamura, Department of Neurology, Sakai Hospital, Kinki University Faculty of Medicine; Reika Wate, Department of Neurology, Kansai Medical University; Yoshio Ikeda, Department of Neurology, Okayama University Graduate School of Medicine, Dentistry and Pharmaceutical Sciences; Kenichi Yasui, Department of Neurology, Tottori University Graduate School of Medicine; Hiroo Yoshikawa, Department of Internal Medicine, Division of Neurology, Hyogo College of Medicine; Hiroshi Takada, Department of Internal Medicine, Division of Neurology, National Hospital Organization, Minami-Okayama Medical Center (present affiliation: Kinashi Obayashi Hospital); Michikazu Nakamura and Toshihiko Suenaga, Tenri Hospital; Makito Hirano, Department of Neurology, Nara Medical University (present affiliation: Department of Neurology, Sakai Hospital, Kinki University Faculty of Medicine); Yoshinori Kajimoto, Department of Neurology, Wakayama Medical University; Masaki Kondo, and Nagato Kuriyama, Department of Neurology, Kyoto Prefectural University of Medicine; Kazumasa Saigo, Department of Neurology, Kinki University Faculty of Medicine; Kengo Uemura, Department of Neurology, Kyoto University Graduate School of Medicine. This study is registered with UMIN Japanese clinical trials registry, number UMIN000000824.
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Dr Miyai is supported by Grant-in-Aid for “the Research Committee for Ataxic Diseases” of the Research on Measures for Intractable Diseases and The Research Grant (21B-9) for Nervous and Mental Disorders from the Ministry of Health, Labor and Welfare, Japan. Dr Hattori is supported by PREST from Japan Science and Technology Agency. Dr Sobue is supported by Scientific advisory boards for Kanae Science Foundation for the Promotion of Medical Science, Naito Science Foundation Funded by Ministry of Education, Culture, Sports, Science and Technology of Japan (#21229011, #17025020, #09042025), and Ministry of Welfare, Health and Labor of Japan; and Japan Science and Technology Agency, Core Research for Evolutional Science and Technology. Dr Ito is supported by Grant-in-Aid by Ministry of Education, Culture, Sports, Science and Technology (#22590955-00). Dr Nishizawa is supported by Grant-in-Aid for “the Research Committee for Ataxic Diseases” of the Research on Measures for Intractable Diseases by Ministry of Welfare, Health and Labor, Japan, and Grant-in-Aid by Ministry of Education, Culture, Sports, Science and Technology, Japan (#22249036). Dr Mihara reports no disclosures. Dr Hatakenaka reports no disclosures. Dr Yagura reports no disclosures.
