Abstract
Background. Nigrostriatal dopaminergic loss is a hallmark of Parkinson’s disease (PD) pathophysiology, leading to motor Parkinsonism. Different intervention protocols have shown that motor and cognitive functions improvement in PD occur via the modulation of distinct motor and cognitive pathways. Objective. To investigate the effects of two motor training programs on the brains’ functional networks in PD patients. Methods. Thirty-seven PD patients were prospectively studied. All enrolled patients underwent either treadmill training (TT) (n = 19) or treadmill with virtual reality (TT + VR) (n = 18) for 6 weeks. Magnetic resonance imaging (MRI) scans (3 T) acquiring 3-dimensional T1-weighted and resting-state functional MRI (rs-fMRI) data sets were performed at baseline and after 6 weeks. Independent component analysis (ICA) was conducted, and functional connectivity (FC) changes within large-scale functional brain networks were examined. Results. In both groups, significant post-training FC decrease in striatal, limbic, and parietal regions within the basal ganglia network, executive control network, and frontal-striatal network, and significant FC increase in the caudate, and cingulate within the sensorimotor network (SMN) were observed. Moreover, a significant time × group interaction was detected where TT + VR training had greater effects on FC levels in the supplementary motor area (SMA) and right precentral gyrus within the SMN, and in the right middle frontal gyrus (MFG) within the cerebellar network. These FC alterations were associated with improved usual and dual-task walking performance. Conclusions. These results suggest that TT with-and-without the addition of a VR component affects distinct neural pathways, highlighting the potential for beneficial neural plasticity in PD. Such distinctive task-specific pathways may foster the facilitation of interventions tailored to the individual needs of PD patients. Registered at Clinicaltrials.gov number: NCT01732653.
Introduction
Parkinson’s disease (PD) is a progressive neurodegenerative disease that manifests in various motor, autonomic and cognitive deficits including slowness in movements, postural instability, gait impairments, orthostatic hypotension, and cognitive decline.1-3 Gait impairments in PD include reduced gait speed and stride length, increased stride asymmetry and exaggerated variability of gait; changes that have been associated with a higher risk of falls. 4
A growing body of evidence indicates that specific cognitive subdomains such as attention and executive functions contribute to gait performance and mobility, especially in the presence of aging and neurodegeneration.5,6 Therefore, theoretically, interventions that target motor and cognitive functions to augment automaticity and/or efficiency of cognitive reliance will likely improve functional capacity and gait. In the V-TIME study, 7 we applied a 6-week multimodal treadmill training program augmented by virtual reality (TT + VR) to target both cognitive and motor aspects of mobility in PD patients. Several aspects of cognitive function and mobility improved in both groups; however, more positive benefits were observed in the TT + VR group. In particular, compared with patients in the active control group who trained on treadmill treatment (TT) only, those who underwent TT + VR showed a 50% reduction in fall rates, in addition to improvement in executive functioning tasks.7,8
In a previous task-based functional magnetic resonance imaging (fMRI) study, we explored the patterns of change in brain activation pre- and posttreatment using a motor imagery paradigm in PD patients, who were trained using TT alone or TT + VR. Results showed that the different motor training protocols targeted distinct brain regions. Post-training, participants in the TT + VR group showed lower activation in the right inferior frontal gyrus while those in the TT group demonstrated lower activation in the left cerebellum and left middle temporal areas. Hence, the combined motor-cognitive training apparently led to more efficient activation and decreased reliance on frontal regions. 8
Resting-state (rs)-fMRI allows for the investigation of the brains’ functional networks, that are, spatially distributed, yet anatomically and functionally connected brain regions. 9 Recently, rs-fMRI has emerged as a sensitive tool for monitoring disease progression and treatment outcomes in many neurological conditions as well as in PD.10,11 The quantified strength of the connection between regions may reflect areas that are more frequently mutually activated. Several studies have probed the modulation of resting functional networks in PD patients with advancing disease stages as well as under various treatment strategies.12-15
In this study, we sought to extend earlier fMRI findings, 8 and investigate patterns of functional connectivity (FC) in large-scale, task-negative functional networks in the PD patients trained for 6 weeks in either TT or TT + VR motor training as part of the V-TIME study. 16 We hypothesized that TT training with the addition of the VR component would lead to the modulation of FC of brain regions within functional networks related to high order aspects of gait, reflecting the multidimensionality of the administered interventions.
Methods and Materials
Study Participants
In this sub-study of the V-TIME project, 16 N = 42 PD patients were enrolled if they met the following inclusion criteria: (1) idiopathic PD patients aged 60 to 85 years diagnosed according to the UK Brain Bank criteria, (2) Hoehn and Yahr (H&R) stages I - III, (3) treatment with anti-Parkinsonian medication, (4) stable medication for the past 1 month, (5) ability to walk unassisted for 5 minutes, (6) adequate hearing and vision, and (7) at least 2 fall episodes in the previous 6 months prior to enrollment. Exclusion criteria were (1) MRI contraindications, (2) psychiatric comorbidity, (3) clinically significant cognitive impairment (Mini Mental State Examination [MMSE] <24), (4) a history of a neurological disorder aside from PD, (5) orthopedic problems, (6) unstable other medical condition, or (7) excessive head motion during MRI scan (>3 mm average frame-wise displacement). The study was approved by the institutional review board (IRB) at Tel Aviv Sourasky Medical Center. All included participants gave their informed written consent.
Clinical and Cognitive Assessments
All enrolled participants underwent general physical and cognitive assessments at baseline and after completion of the training protocol. All testing was done “ON” medication, approximately 1 hour after taking medication. The cognitive assessment included the Montreal Cognitive Assessment (MoCA) 17 and Color Trails Test (CTT) to evaluate executive function.
Gait and Balance Assessments
Gait was measured pre and post motor training using an electronic walkway with pressure sensors embedded in a carpet. Gait tests included 2 tasks, usual walking (UW) and dual-task (DT) walking while serially subtracting 3s from a 3-digit number. In each task, the subjects walked for 1 minute along a 30 meter walkway. To characterize the performance of usual and DT walking, gait, stride length, stride time, and cadence were analyzed. 18 Posture and balance were assessed using the mini-Balance Evaluation Systems Test (miniBESTest). 19
Training Protocols
The enrolled participants to the study were randomized to either the TT + VR (N = 22), or to the active control comparison TT only group (TT, N = 20). Both groups underwent 18 sessions (3 sessions/week × 6 weeks) as described elsewhere. 16
MRI Acquisition
Pre- and posttraining MR data sets were acquired from all patients using a 3 T Signa Excite (Signa Excite General Electric Healthcare) MR scanner with 8-channel phased-array head coil. At both time points, the MRI protocol included: a high-resolution Fast Spoiled Gradient Echo (FSPGR) 3-D T1-weighted sequence (TR [repetition time]/TE [echo time]/TI [inversion time] = 59/3.6/500 ms; FA [flip angle] = 8°, FoV = 256 × 256 mm; 162 slices, voxel size = 0.98 × 0.98 × 1 mm3), axial T2-weighted fluid attenuated inversion recovery (FLAIR) (TR/TE/TI = 9000/145/2100 ms; FA = 90°; FoV = 512 × 512; 36 axial slice; voxel size = 0.47 × 0.47 × 3.2 mm3), and 266 volumes resting-state fMRI data sets using single-shot echo-planar imaging (EPI) sequence (TR/TE = 3000/35 ms; FA = 90°, FoV = 64 × 64 mm; 30 axial slices with no gap, voxel size = 3.1 × 3.1 × 3.5 mm3).
MRI Data Processing
In total, N = 4 were excluded from the rs-fMRI analysis due to excessive head motion, resulting in N = 18 and N = 19 in the TT + VR and TT study groups, respectively. For rs-fMRI datasets, the following steps were applied: slice time correction, realignment to the first volume image in order to correct for head movement, normalization to MNI (Montreal Neurological Institute) using the forward transformation parameters obtained from DARTEL procedure, and smoothing using an 8 mm FWHM (full-width at half maximum) kernel. The denoising and filtering of preprocessed rs-fMRI data sets were analyzed using the CONN (v.17) toolbox (McGovern Institute for Brain Research, Massachusetts Institute of Technology, Cambridge; https://web.conn-toolbox.org/), where, linear detrending, temporal despiking, band-pass filtering (0.08-0.1 Hz) were carried out after regression. The subjects’ 6 head-motion parameters, white matter, and cerebrospinal fluid signal time course were entered as covariates of no interest in the regression model. CONN toolbox was used to identify brain networks of highly functionally connected regions. Here, group-level independent component analysis (ICA) with 20 independent components (IC) approach. Individual subject-level functional networks for each subject were obtained by GICA1 back-reconstruction. 20 The following networks were identified based on dice coefficients (ICC) for best spatial overlap with a default template provided by CONN. Additionally, a visual inspection of the obtained IC spatial maps was performed to for verification of spatial distribution and the identification of further networks: cerebellar network (CBL, independent component (IC)-2, r = 0.4), executive control network (ECN, IC-8, r = 0.2), lateral motor network (LMN, IC-13, r = 0.5), sensorimotor network (SMN, IC-14, r = 0.36), dorsal attention network (DAN, IC-17, r = 0.22), frontal-striatal network (FSN, IC-18), 21 and basal ganglia network (BGN, IC-20) 22 (see Figure 1).
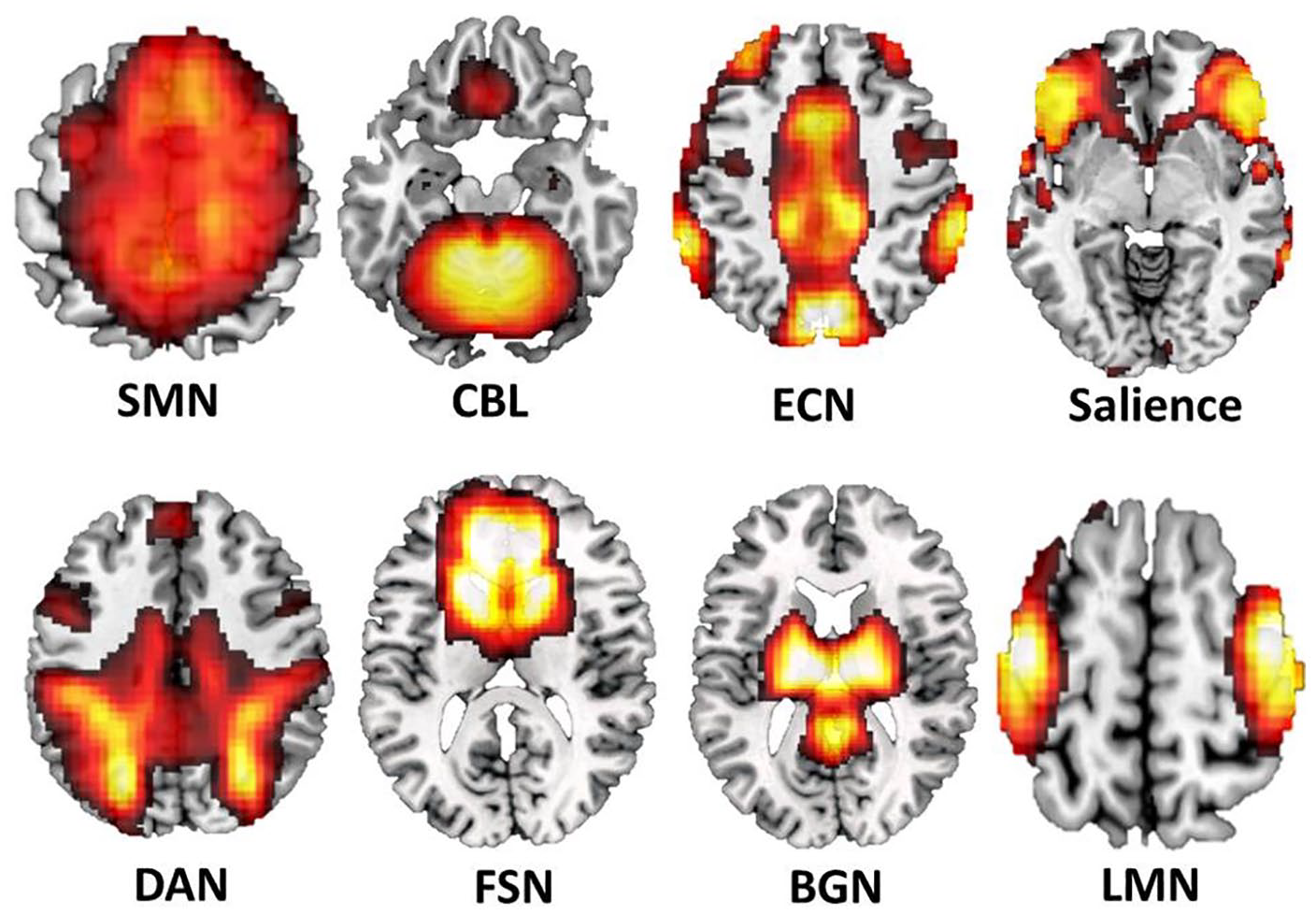
Spatial distribution of the investigated resting-state networks (RSNs). SMN, sensorimotor network; CBL, cerebellar network; ECN, executive control network; DAN, dorsal attention network; FSN, fronto-striatal network; BGN, basal ganglia network; LMN, lateral motor network.
Statistical Analysis
Statistical analyses of clinical and behavioural parameters were performed using Statistical Package for Social Science (SPSS v.25, IBM Corp). Gender differences among the groups (TT + VR vs TT) were assessed using a chi-square test. Two-sample t tests were used to evaluate differences between the groups for continuous measures at baseline. Two-way mixed-effects analysis of variance (2 × 2 ANOVA) model was performed to investigate the changes of the behavioural variables pre- and posttreatment in both study groups. P values <.05 were considered as a threshold for statistical significance.
In SPM, a flexible factorial model (2 × 2 ANOVA) was performed to investigate the patterns of treatment changes of FC within the networks on interest. The significance threshold of P < .001, FWE (family-wise error) adjusted for cluster size (FWEc) was adopted for the calculated t-maps. The peak maximum FC values within the detected significant clusters were extracted, and the relationship between FC levels within the detected significant clusters was further explored. The relationship between the overtime changes in FC levels (
Results
Demographics and Clinical Characteristics
Demographic and clinical characteristics of all study participants are presented in Table 1. At baseline, the groups were comparable in age, gender, Unified Parkinson’s Disease Rating Scale–III (UPDRS-III), levodopa equivalent daily dose (LEDD), gait speed, stride length, stride time, and cadence during both usual-and-dual task walking, as well as in miniBEST, MoCA, CTTa and CTTb (two-sample t test; P > .05 in all cases).
Baseline Demographic, Clinical, Gait, and Cognitive Characteristics of the Study Groups.
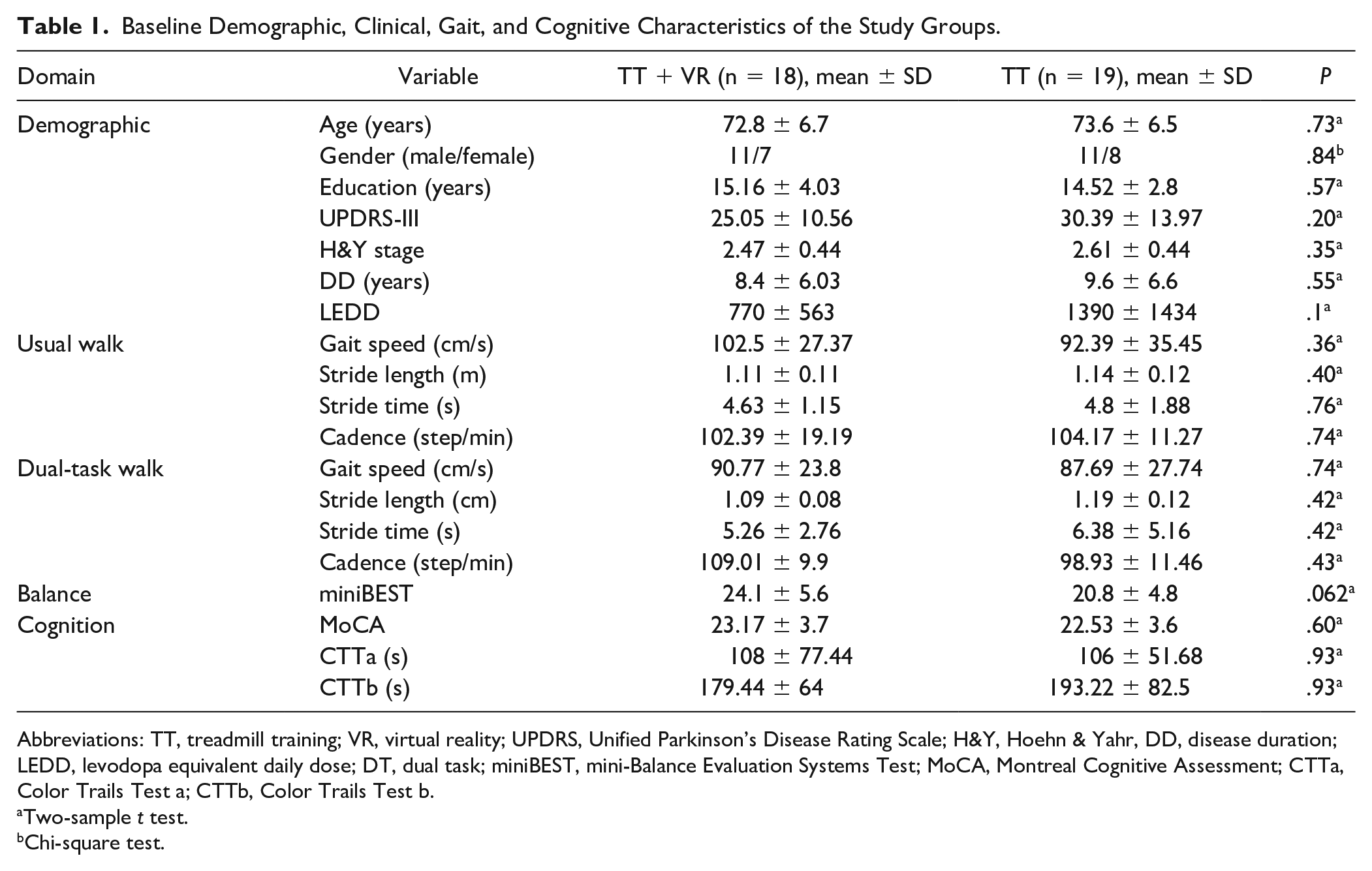
Abbreviations: TT, treadmill training; VR, virtual reality; UPDRS, Unified Parkinson’s Disease Rating Scale; H&Y, Hoehn & Yahr, DD, disease duration; LEDD, levodopa equivalent daily dose; DT, dual task; miniBEST, mini-Balance Evaluation Systems Test; MoCA, Montreal Cognitive Assessment; CTTa, Color Trails Test a; CTTb, Color Trails Test b.
Two-sample t test.
Chi-square test.
Training Effects on Gait and Cognitive Measures
Pre-and-post training performance scores in gait and cognitive function of both groups are detailed in Table 2. The TT + VR group showed an overall improvement in gait speed, stride length, and cadence performance during UW, as well as balance and cognitive test performance as reflected by CTTb. On the other hand, the TT group demonstrated improvement in UW gait speed, stride time and cadence, as well as in DT walk gait speed, balance, and CTTb. ANOVA revealed significant group main effect for cadence in DT, F(1, 34) = 5.5, P = .028, such that the TT-VR group showed significantly higher mean cadence in dual task compared to the TT group over the investigated time period. Moreover, a significant time × group interaction was observed for miniBEST balance scores, F(1, 34) = 4.91, P = .033, indicating that the effects of training type was greater in the TT group compared with TT + VR group on balance performance.
Gait and Cognitive Performance Scores of Both Study Groups, Pre and Post 6 Weeks of Training. a
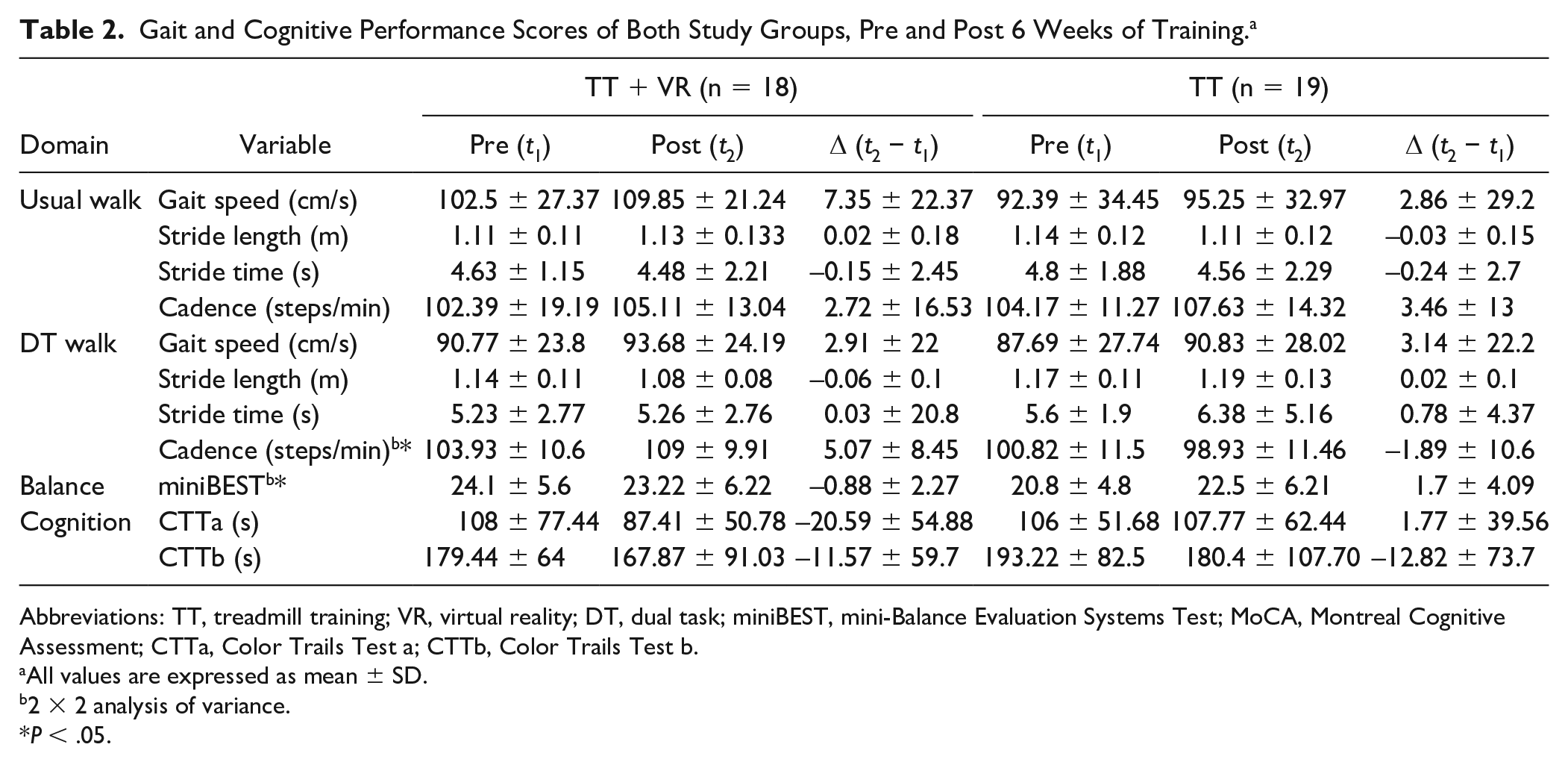
Abbreviations: TT, treadmill training; VR, virtual reality; DT, dual task; miniBEST, mini-Balance Evaluation Systems Test; MoCA, Montreal Cognitive Assessment; CTTa, Color Trails Test a; CTTb, Color Trails Test b.
All values are expressed as mean ± SD.
2 × 2 analysis of variance.
P < .05.
Training Effects on FC in Resting-State Networks in Both Groups
The results of the 2-way ANOVA based on the calculated FC maps of both training groups are summarized in Supplementary Table 1 and Figure 2. In the investigated networks, significant time effect was observed in the BGN, ECN, FSN, and SMN in both study groups. Specifically, post treatment FC decrease was observed in the left and right posterior mid-cingulate cortex (pMCC), and left precuneus within the BGN, in the bilateral thalamus within the ECN, and the bilateral cuneus and left lingual gyrus within the FSN network (P < .001, FWEc) (Figure 2A). Whereas, within the SMN, both groups demonstrated significant post treatment FC increase in the right angular gyrus, right caudate, and right anterior MCC (aMCC) (P < .001, FWEc) (Figure 2B). Moreover, significant time × group interactions where observed in the SMN as well as the CBL networks. Within the SMN network, TT + VR training showed to have greater effect on FC in the supplementary motor area (SMA), and right precentral gyrus (R PCG) compared with TT. Similarly, TT + VR training showed to have greater effects on FC levels in the right middle frontal gyrus (R MFG) within the CBL network (P < .001, FWEc) (Figure 2C). No significant effects were detected in the DAN, LMN, and salience networks.
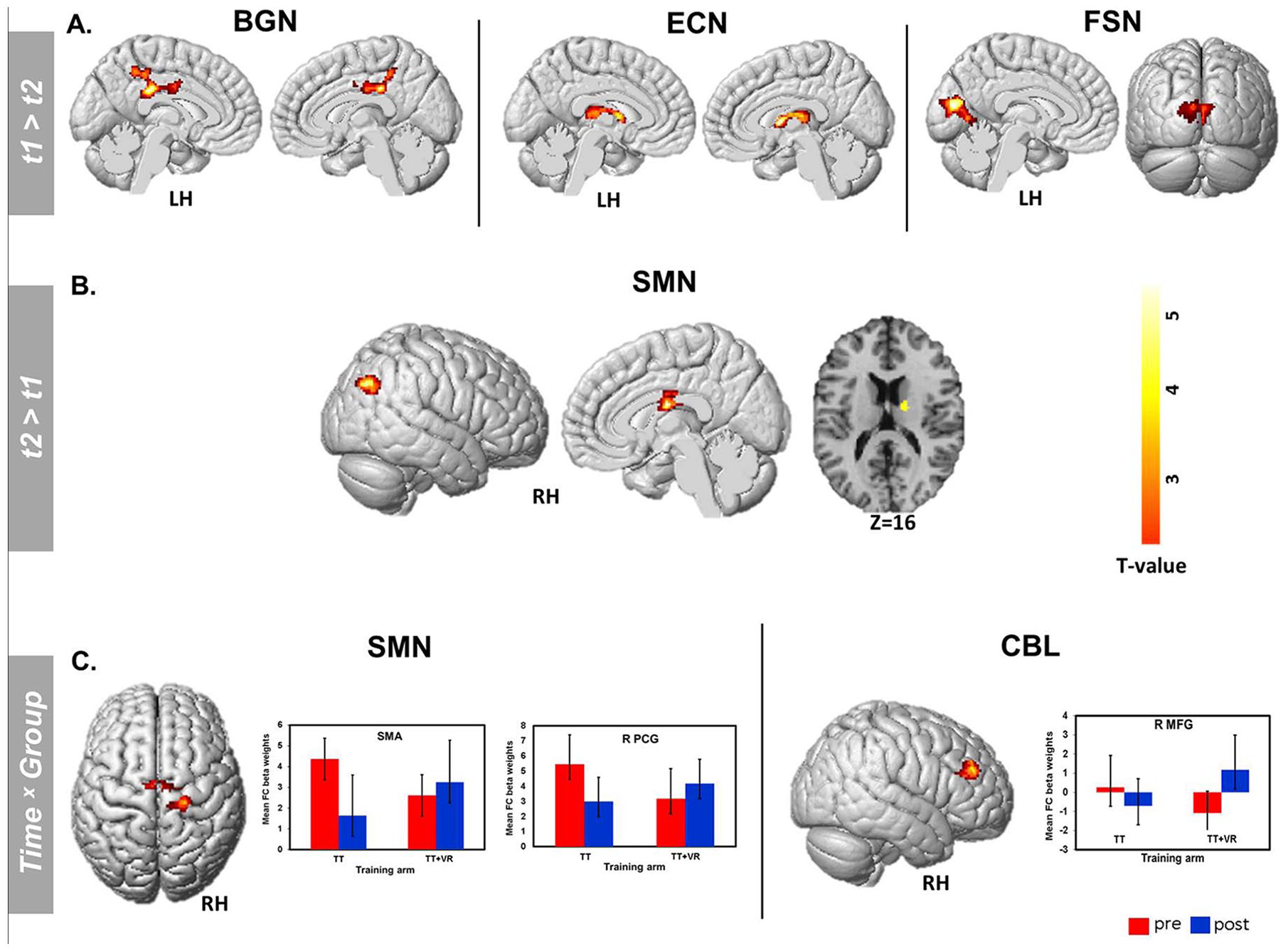
Training effects on FC within the investigated functional networks. (A) In both groups, a significant time effect was observed, in which, FC decrease was observed in the left and right posterior MCC, and left precuneus within the BGN, in the bilateral thalamus within the ECN, and the bilateral cuneus and left lingual gyrus within the FSN. (B) Within the SMN, both groups demonstrated significant posttraining FC increase in the right angular gyrus, right caudate, and right anterior MCC (P < .001, FWEc). (C) A significant time × group interaction was observed in the SMN as well as the CBL networks. Post training FC increase in the SMA, and right R PCG within the SMN, and in the R MFG within the CBL was observed in TT + VR group, whereas in the TT group, FC in these regions was observed to decrease post training (P < .001, FWEc).
Correlations Between Motor Performance and FC Levels
The correlation results between FC levels, balance, and gait performance scores are summarized in Table 3. In the TT group, the observed reduction of FC (ΔFC) in the R pMCC within the BGN was found to correlate significantly with the change (Δ) in UW gait speed (r =0.57, P = .02). In the ECN, ΔFC in L thalamus was associated with Δ stride time (r = 0.41, P = .046) in both treatment groups. In TT group, ΔFC in R thalamus was inversely associated with ΔDT gait speed (r = −0.53, P = .038). Moreover, in the TT + VR group, the following significant associations where detected between FC in regions within SMN and performance scores: ΔFC in the R angular gyrus, R caudate, and R MCC were found to be significantly associated with ΔUW stride length and ΔDT stride length (r = 0.65, P = .031; r = 0.61, P = .042; r = 0.7, P = .017, respectively). In the TT group, the reduced ΔFC levels in the R PCG within the SMN correlated inversely with the post training DT stride length performance scores (r = −0.6, P = .022). Moreover, ΔFC levels in SMA correlated positively with the Δ miniBEST scores (r = 0.53, P = .025). Finally, in the TT + VR group, post treatment FC levels in the R MFG within the CBL network correlated significantly with the ΔDT stride time (r = −0.76, P = .006). In both groups, no significant correlations were detected between the reduction of FC in the obtained clusters within the FSN and gait, and balance performance scores.
Correlations Between rs-fMRI FC Scores, and Gait and Balance Performance in Both Groups.
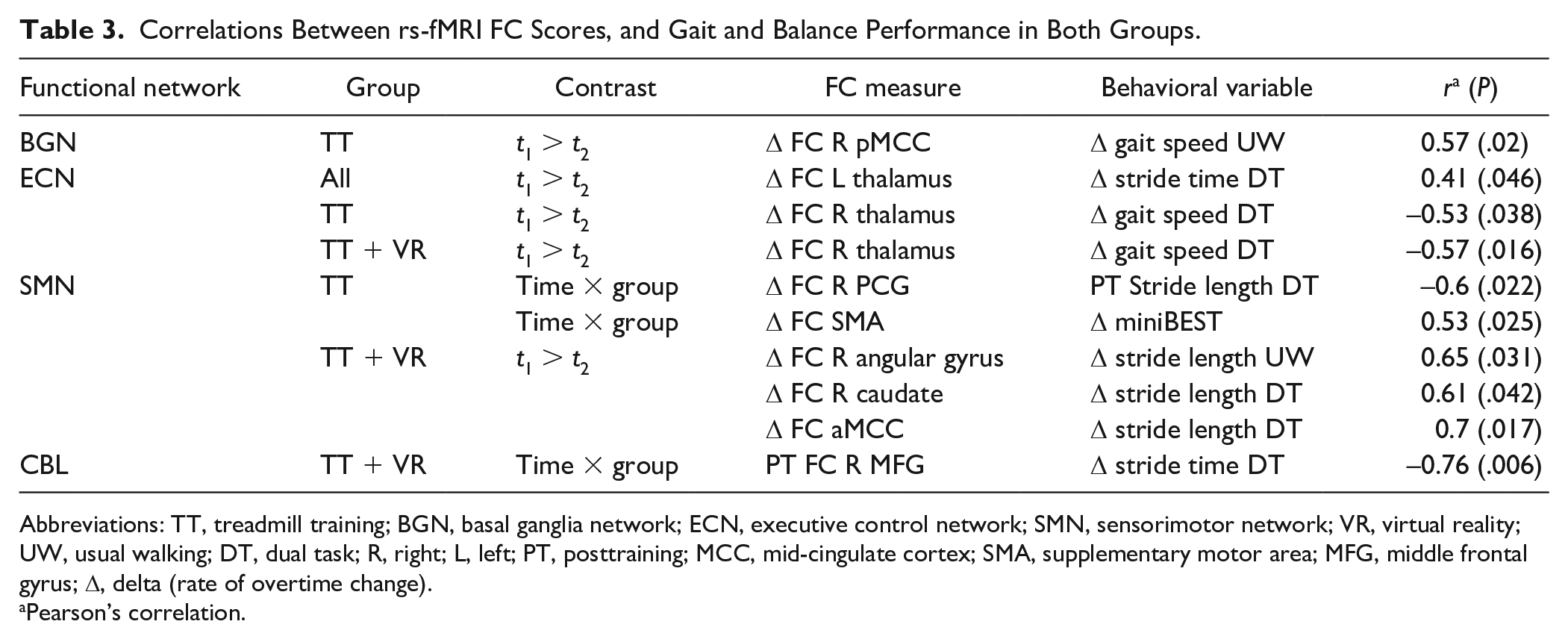
Abbreviations: TT, treadmill training; BGN, basal ganglia network; ECN, executive control network; SMN, sensorimotor network; VR, virtual reality; UW, usual walking; DT, dual task; R, right; L, left; PT, posttraining; MCC, mid-cingulate cortex; SMA, supplementary motor area; MFG, middle frontal gyrus; Δ, delta (rate of overtime change).
Pearson’s correlation.
Discussion
In this study, we investigated the effects of two training programs on the brains’ resting-state networks in PD patients. We analyzed large-scale functional networks in two groups of age- and gender-matched PD patients that underwent training either in TT + VR or TT programs. The results indicate that while both training programs resulted in similar patterns of FC decrease in brain regions within the BGN, FSN, and ECN networks, as well as FC increase in regions within the SMN; FC increase in the SMA and R PCG within the SMN, and R MFG within the CBL network were observed in the TT + VR training group only.
Decreased FC levels in dopaminergic mesocorticolimbic regions involved in multisensory executive and direction of motor functions including the thalamus, pMCC, and cuneus within multiple brain functional networks were observed as a result of both training programs. These patterns of FC decrease were found to be correlated with improvement in gait speed in both usual and dual-task walking, as well as dual task stride time. Additionally, both groups demonstrated post treatment FC increase in the angular gyrus, caudate, and the aMCC within the SMN, and this observed increase was associated with improved gait performance. These findings further emphasize the involvement of brain regions implicated in visuospatial processing, information integration and attention during walking and coordination of gait.23,24 Recent studies have highlighted the role of higher cortical brain regions during walking. For example, it was shown that increased activations in frontal, sensory, and motor brain regions in PD patients are associated with gait adaptability during treadmill walking. 25 Moreover, following gait and motor imagery exercises, Myers et al 26 showed that despite the motor imagery intervention, PD patients with freezing showed no improvements in gait, possibly due to the fact that FC levels in higher cortical regions remained lower compared to non-freezers.
The fact that in both training groups similar patterns of post treatment FC decrease in BGN, FSN, and ECN networks were observed can be attributed to the use of the treadmill component, reflecting successful compensatory mechanisms involved in externally-guided rhythmic movements provided by the treadmill rhythm. The treadmill can act as an external cue by setting the walking pattern, surpassing the depleted striatal networks via the facilitation of higher motor and cognitive pathways that contribute to gait pacing, leading to improvements in gait in PD.8,27-29 Moreover, the post treatment FC increase observed in both groups in the caudate, MCC, and R angular gyrus regions within the SMN is indicative that TT also affects the higher order aspects of gait. In line with this, a previous study in PD patients demonstrated both cognitive and motor improvements following one month of treadmill training, arguing that this can effectively improve both cognitive and motor features in patients with PD. 30 This assertion can be further emphasized by the fact that the overtime FC change in these regions was found to correlate with both usual walking and dual task stride length in TT + VR group, where the addition of the VR component might have further augmented the modulation of FC within regions involved in the higher-order aspects of gait.
Most interestingly, a significant interaction was observed in both the SMN and CBL networks. In which, TT + VR demonstrated post training FC increases in the R PCG and SMA within the SMN network as well as in the R MFG within the CBL network were detected. The FC levels in the R MFG at follow-up were found to be associated with the change (over time) in DT stride time. Recently, the role of the fronto-cerebellar connections in gait have been implicated, highlighting its involvement in the allocation of attentional resources for maintaining automated motor sequences. 31 In the TT group, FC levels in the aforementioned regions within the SMN and CBL were reduced post training. The amount of change in FC from pre- to- post training correlated with DT stride length increase, as well as poorer balance performance. The role of SMA and prefrontal cortical regions has been implicated in high-order multisensory integration and postural stabilization during the execution of every-day routine motor tasks.32,33 For example, a positron emission tomography study including healthy participants, activation of SMA and bilateral motor cortices was observed when the participants simulated bicycling movements compared with rest condition. 34 Moreover, transcranial magnetic stimulation to SMA was found to be effective for motor signs improvements in PD.35,36
The main limitation of this study is the relatively small groups’ sizes, and therefore based on our findings we can only infer associations between training type and the patterns of FC changes within the different brain functional networks. Additionally, given that the patients’ population is relatively of older age, future trials should enroll larger and more diverse treatment groups as well as a control group of healthy individuals, to establish the distinct patterns of internetwork dynamics as a result of the different motor training regimens and their association patterns with age, motor, and cognitive performance in PD patients.
The present results demonstrate the beneficial effects of both motor training protocols on the modulation of FC in PD patients. In other words, while the TT led to reconfiguring of gait scheme, the addition of the cognitive component led to beneficial effects on FC in regions involved in the higher-order aspects of gait. Thus, emphasizing the importance of novel rehabilitation interventions for the modulation of distinct neural pathways tailored to the individual needs of the patients.
Supplemental Material
CONSORT_2010_Checklist_NNR_Droby_et_al – Supplemental material for Distinct Effects of Motor Training on Resting-State Functional Networks of the Brain in Parkinson’s Disease
Supplemental material, CONSORT_2010_Checklist_NNR_Droby_et_al for Distinct Effects of Motor Training on Resting-State Functional Networks of the Brain in Parkinson’s Disease by Amgad Droby, Inbal Maidan, Yael Jacob, Nir Giladi, Jeffrey M. Hausdorff and Anat Mirelman in Neurorehabilitation and Neural Repair
Supplemental Material
Flow_Diagram_Droby_et_al_NNR – Supplemental material for Distinct Effects of Motor Training on Resting-State Functional Networks of the Brain in Parkinson’s Disease
Supplemental material, Flow_Diagram_Droby_et_al_NNR for Distinct Effects of Motor Training on Resting-State Functional Networks of the Brain in Parkinson’s Disease by Amgad Droby, Inbal Maidan, Yael Jacob, Nir Giladi, Jeffrey M. Hausdorff and Anat Mirelman in Neurorehabilitation and Neural Repair
Supplemental Material
Suppl_Table_1 – Supplemental material for Distinct Effects of Motor Training on Resting-State Functional Networks of the Brain in Parkinson’s Disease
Supplemental material, Suppl_Table_1 for Distinct Effects of Motor Training on Resting-State Functional Networks of the Brain in Parkinson’s Disease by Amgad Droby, Inbal Maidan, Yael Jacob, Nir Giladi, Jeffrey M. Hausdorff and Anat Mirelman in Neurorehabilitation and Neural Repair
Footnotes
Acknowledgements
We wish to thank all participants who took part in this study, and all V-TIME project partners.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: A.D, I.M, Y.J, have nothing to disclose. Drs Mirelman, Hausdorff, and Giladi report having submitted a patent application on the use of virtual reality. The intellectual property rights for this patent application are held by the Tel Aviv Medical Center.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded in part by European Commission (FP7 project V-Time-278169).
Supplementary material for this article is available on the Neurorehabilitation & Neural Repair website along with the online version of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
