Abstract
Background. No rehabilitation intervention has effectively improved functional use of the arm and hand in patients with severe upper limb paresis after stroke. Pilot studies suggest the potential for transcranial direct current stimulation and bilateral robotic training to enhance gains. Objective. In a double-blind, randomized trial the combination of these interventions was tested. Methods. This study randomized 96 patients with an ischemic supratentorial lesion of 3 to 8 weeks’ duration with severe impairment of motor control with a Fugl-Meyer score (FMS) for the upper limb <18 into 3 groups. For 6 weeks, group A received anodal stimulation of the lesioned hemisphere, group B received cathodal stimulation of the nonlesioned side for 20 minutes at 2.0 mA, and group C received sham stimulation. The electrodes were placed over the hand area and above the contralateral orbit. Contemporaneously, the subjects practiced 400 repetitions each of 2 different bilateral movements on a robotic assistive device. Results. The groups were matched at onset. The FMS improved in all patients at 6 weeks (P < .001). No between-group differences were found; initial versus finish FMS scores were 7.8 ± 3.8 versus 19.1 ± 14.4 in group A, 7.9 ± 3.4 versus 18.8 ± 10.5 in group B, and 8.2 ± 4.4 versus 19.2 ± 15.0 in group C. No significant changes between groups were present at 3 months. Conclusions. Neither anodal nor cathodal transcranial direct current stimulation enhanced the effect of bilateral arm training in this exploratory trial of patients with cortical involvement and severe weakness. Unilateral hand training and upregulation of the nonlesioned hemisphere might also be tried in this population.
Keywords
Introduction
A total of 80% of stroke survivors present with arm paresis whose severity is bimodally distributed. For the severely affected, nonfunctional upper extremity (UE), the prognosis to regain meaningful hand activity following conventional therapy is poor.1,2 Compensatory use of the nonaffected UE in the subacute phase may result in insufficient therapy intensity.
Robot-assisted arm training is a new approach to intensify UE rehabilitation. It has been effective, compared with conventional or sham therapy, on UE function in meta-analyses for impairment, but not for disability.3,4 A further option to improve UE outcomes after stroke is brain stimulation in combination with peripheral therapy, as a structured physiotherapy program, 5 electrical stimulation,6,7 or robot-mediated therapy.8,9
In primates, Plautz et al 10 have shown that coupled forced use of the paretic hand with implanted electrical stimulation to the ipsilesional M1 leads to significant behavioral improvements with large-scale expansions of the hand representation into areas previously representing proximal forelimb movements. Adkins-Muir and Jones 11 found similar results in lesioned rats. Among the 3 potential modes of central stimulation for patients, 12 transcranial direct current stimulation (tDCS), repetitive magnetic, and epidural stimulation, tDCS is the least invasive and expensive and thus the most viable in daily routine. Anodal stimulation facilitates and cathodal stimulation inhibits motor-evoked potentials (MEPs)13-15; facilitation can improve implicit motor learning in healthy subjects. Open studies and sham-controlled studies with single and repetitive tDCS sessions have shown its potential on motor, verbal learning, and attentional deficits in healthy and chronic stroke subjects.16-21 Cathodal stimulation aims to inhibit the nonlesioned hemisphere to address the issue of disturbed interhemispheric competition following stroke with the nonlesioned partially overriding the lesioned hemisphere. 22
Hesse et al 8 combined tDCS with robot-mediated bilateral arm therapy in subacute, severely paretic stroke patients without side effects. This pilot study found improved motor performance mainly in patients with a subcortical lesion. However, the number of patients was small and the protocol did not include a sham stimulation group. Robot-assisted bilateral arm training had shown efficacy in these patients. 23 This trial was powered to compare anodal and cathodal stimulation to sham stimulation combined with this bilateral robotic training in patients with a nonfunctional UE. Such patients do not meet the inclusion criteria for interventions that have helped less impaired patients, such as constraint-induced movement therapy and impairment-oriented training24,25 and often are given a lower priority for affected upper limb (UL) rehabilitation.
Methods
Participants
Over 3 years, 96 stroke patients from 3 centers participated in the double-blind randomized sham-controlled multicenter trial. Inclusion criteria were age 18 to 79 years, first supratentorial ischemic stroke with a stroke interval of 3 to 8 weeks duration, and participation in a comprehensive inpatient rehabilitation program. The patients were at least wheelchair-mobile, had a severe flaccid UL paresis with no (Medical Research Council Sum Score [MRC] 0) or minimal (MRC 1) volitional hand and finger extensor activity, 24 had a UL Fugl-Meyer motor score (FMS, range 0-66) <18, and were unable to transfer 3 wooden blocks from one compartment to the other in the Box and Block test. 25 They had no other neurological or orthopedic disease impairing the UL, understood the purpose and content of the study, and consented to participate in the study, as approved by the local ethical committee.
Exclusion criteria were preceding epileptic seizures, an electroencephalogram with elevated cortical excitability, sensitive scalp skin, metallic implants within the brain, and previous brain neurosurgery. Antidepressants (selective serotonin reuptake inhibitors), benzodiazepines, and pregabaline could be prescribed but not anticonvulsant or neuroleptic medications.
Sample Size Calculation
The sample size was calculated to detect minimal statistical differences between the groups under the assumption of an effect size = 1 of Tend − T0, which equals standard deviations. We assumed a power of 80% and an adjusted α of 0.025 for the main outcome variable (differences Tend − T0). The sample was calculated to be 29 patients in each group. We estimated a dropout rate of 10% and therefore enrolled 32 patients per group.
Allocation
Following a telephone call, an independent person randomly allocated eligible patients to 1 of the 3 groups by drawing a lot out of an envelope containing 96 lots, indicating A, B, and C. He then informed the locally responsible person about the group assignment and the study started the next day.
Intervention
Each session lasted for 20 minutes every workday. During each session, the patients practiced with the arm robot (AT) and simultaneously received the tDCS (verum or sham) also for 20 minutes. The intervention period lasted 6 weeks, that is, a total of 30 sessions in each patient. tDCS was applied via saline-soaked surface sponge electrodes (35 cm2), connected to a constant current stimulator (Siemens Therapie, Neuroton 827, Munich, Germany). In group A patients (anodal group), the anodal electrode was placed over the presumed hand area of the lesioned hemisphere (C3 position according to the 10-20 system) and the cathodal electrode was placed above the contralateral orbit (C4 position). In group B patients (cathodal group), the cathodal electrode was placed over the presumed hand area of the nonlesioned hemisphere (C3 position) and the anodal electrode was placed above the contralateral orbit (C4 position). In group C patients (sham group), the positions of the electrodes either followed that of group A or group B patients; the order changed consecutively from patient to patient. The panel of the stimulator was invisible for the patients, in group A and B the intensity was set to 2 mA and in group C to 0 mA.
The AT was performed with the robot-assisted Bi-Manu Track (Reha-Stim Bi-Manu Track, Berlin, Germany), described in detail elsewhere. 28 It enabled the bilateral mirror-like practice of a forearm prosupination or a wrist extension flexion, the change of the movement direction required tilting the device and exchanging the handles. The amplitude, speed, and resistances could be set individually. Within one 20-minutes session, both cycles were trained for 10 minutes. Initially, the patients practiced each cycle 200 times in a passive manner, to be followed by an active–passive mode, that is, the nonparetic was driving the paretic extremity for another 200 times. If possible, the paretic extremity had to overcome an initial isometric resistance actively. The patients sat in a comfortable, height-adjustable armchair with the back supported.
The ongoing comprehensive rehabilitation program consisted of 45 minutes of individual physiotherapy sessions every workday, 30 minutes of individual occupational therapy 4 times a week, both following an eclectic approach, and daily ergometer training. The therapy primarily aimed at the restoration of mobility and daily living competence, stressing the compensatory use of the nonaffected UE. Specific exercises for the affected UL, compromising approximately 10% of the total therapy time, included tone-inhibiting maneuvers, bilateral tasks, and facilitation techniques. When possible, the paretic hand was trained to be used in daily activities. The therapists responsible for the comprehensive rehabilitation program were blind with respect to the group assignment of each individual patient.
Assessment
The primary outcome was sensory and motor integrity and the degree of synergy involved in executing movements, assessed by the Fugl-Meyer motor assessment score (FMS, 0-66, 0 = no movement, 66 = full motion). 25 Sensitivity of the FMS to change in the severely affected patients was demonstrated to be good by Filiatrault et al. 29 Platz et al 30 showed that the FMS was more sensitive to therapeutic change early after stroke than the Action Research Arm Test in stroke patients with severe arm paresis. To ensure blinded evaluation of the FMS, videos of the assessment, where the patients sat on a chair and a mirror was placed 45° behind them, were sent to an experienced therapist off site.
Secondary outcome parameters were UL muscle strength, muscle tone, the Barthel Index to assess the competence in basic activities of living, and the Box and Block test to assess UL function.24,25 Muscle strength was assessed by the MRC (0-5, 0 = plegic, 5 = full power). A sum score of 3 proximal (shoulder abduction, elbow flexion, and extension) and 5 distal (flexion and extension of the hand and fingers, and thumb flexion) muscle groups (0-40) was calculated. The muscle tone was assessed by the modified Ashworth score (0-5, 0 = no increase, 5 = affected part rigid in flexion or extension). A sum score of 2 proximal (shoulder abduction, elbow extension) and 3 distal (extension of the wrist, fingers, and of the thumb) passive movements (0-25) was calculated. The Box and Block tested the transfer of as many wooden blocks as possible with the affected hand from one compartment to the other within 1 minute. The test was rated positive when the patient could transfer at least 3 blocks, because 1 or 2 blocks could be accomplished by chance and with the help of flexor spasticity. Two experienced physiotherapists, blinded with respect to group assignment, assessed the secondary parameters together. The measurement points were T0 (study onset), Tend (end of the 6-week intervention), and Tfollow-up (3 months after Tend). The blinding was maintained at all measurement points.
Statistics
An intention-to-treat analysis was performed, in the event of a dropout assessment, if not possible, the last available score was continued. To test for group differences at baseline, at the end of the study, and after follow-up, we used appropriate nonparametric methods, for example, Kruskal–Wallis test. All data were interpreted quantitatively (median, interquartile range, means, and standard deviation)
For the primary variable, the ΔFMS, we calculated intraindividual differences Tend − T0 and Tfollow-up − Tend. Bonferroni adjustment α for the 2 measure points was made (P < .025).
For the Box and Block test, responders were defined as patients who could transport at least 3 blocks within 1 minute. Between-group differences were calculated with the help of a one-sided exact Fisher test (P < .05). For the other secondary variables, muscle power, tone sum scores and Barthel Index, improvements over time and in between-group differences at Tend and Tfollow-up were calculated with the help of appropriate nonparametric tests (P < .025). SPSS software program version 19.0 was used.
Results
The distribution of the stroke subtypes according to the Oxford Stroke Scale 31 and baseline studies were not different. All but one patient in the anodal group completed the intervention. Follow-up data were obtained in all but 4 patients in group A, 3 patients in group B, and 4 patients in group C. Thus, 85 patients completed the whole study (Figure 1). Relevant side effects did not occur; an uncomfortable tingling under the electrodes (n = 12, evenly distributed in all 3 groups), and transient headaches (n = 4, known history of headaches) did not interrupt the intervention. At study onset (T0), the clinical data and the outcome parameters did not differ between the 3 groups (Table 1).
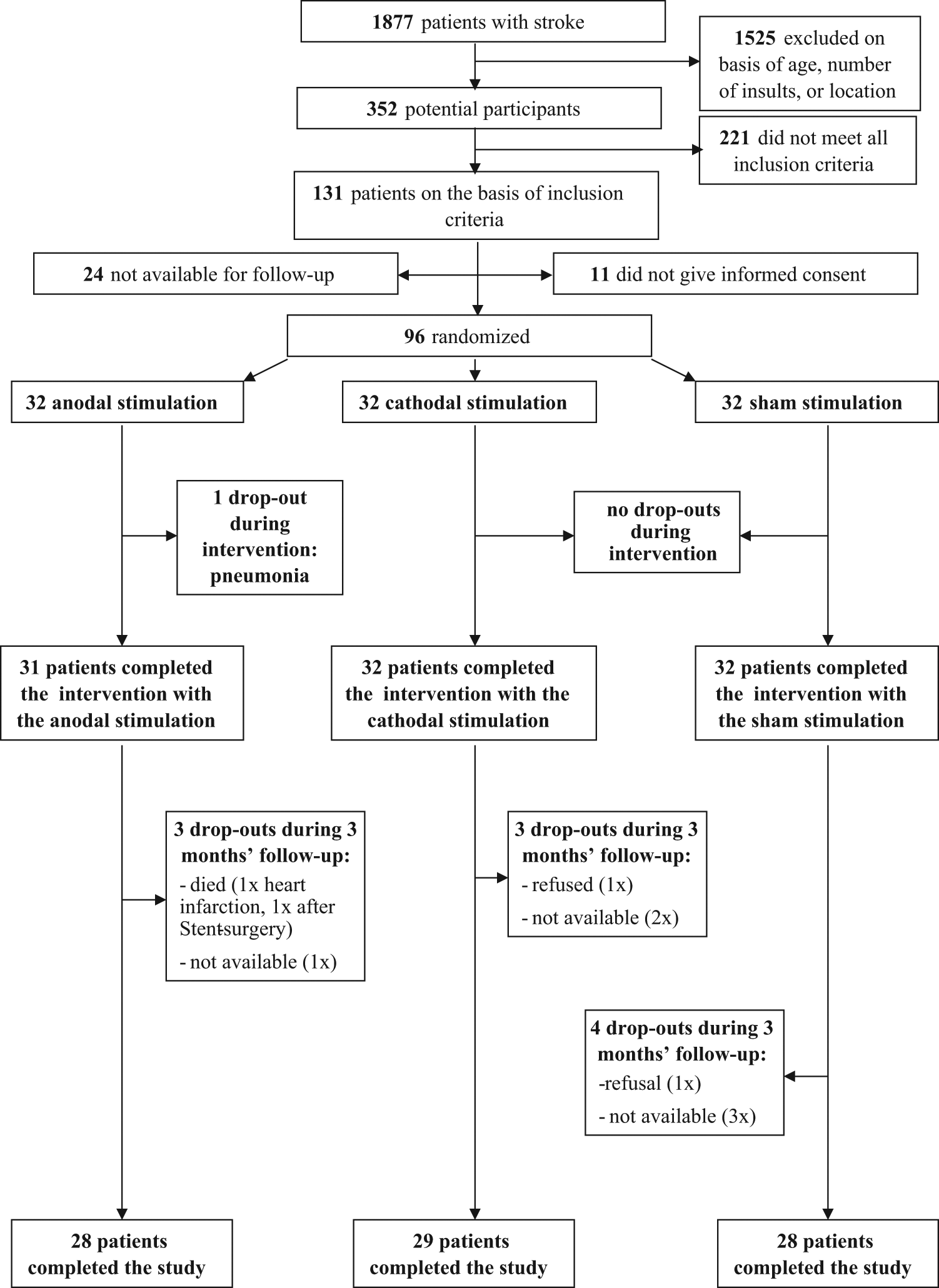
Flowchart of the trial.
Clinical Data and Initial Assessment Scores (Means ± SD) for the 3 Groups
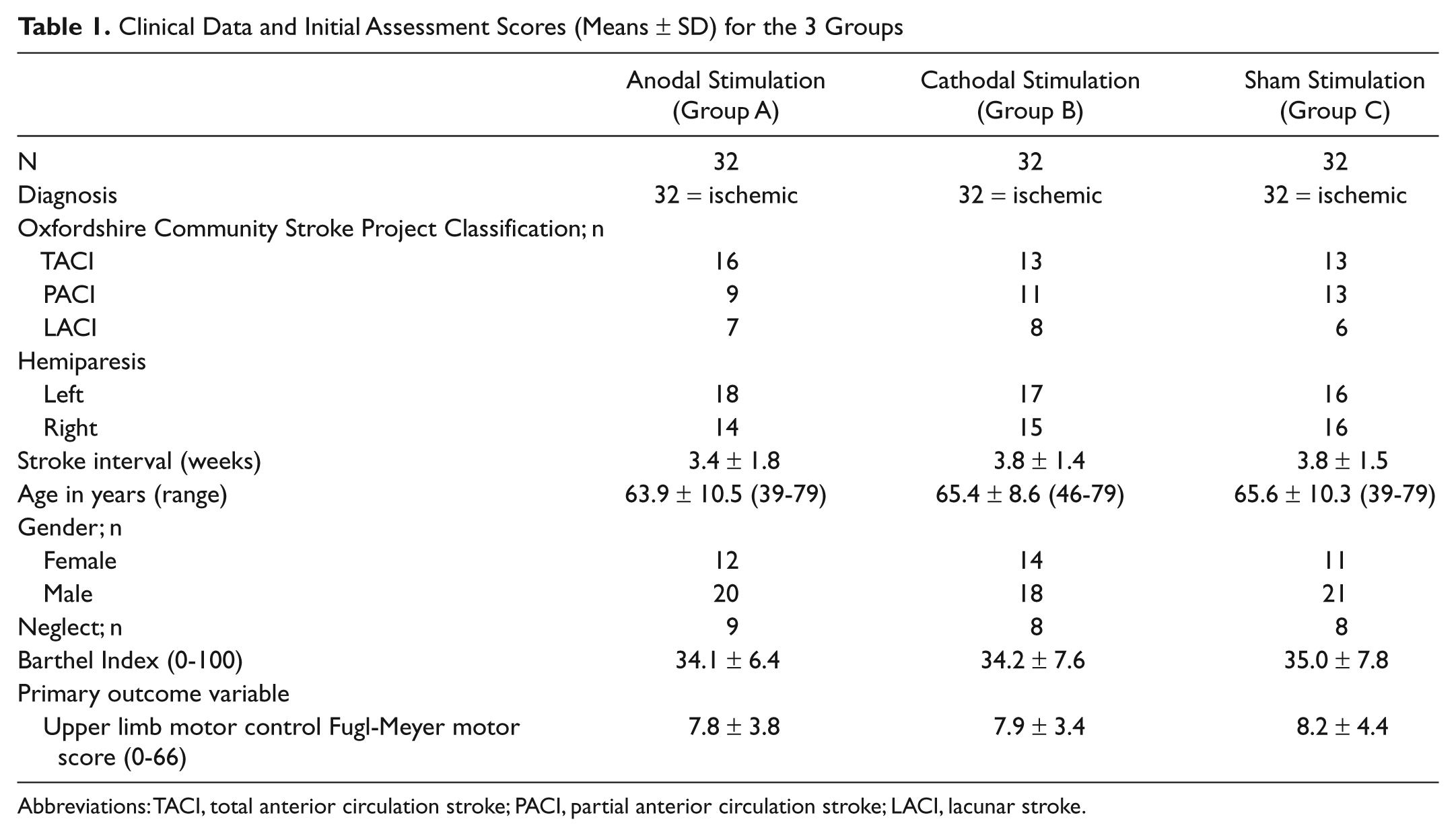
Abbreviations: TACI, total anterior circulation stroke; PACI, partial anterior circulation stroke; LACI, lacunar stroke.
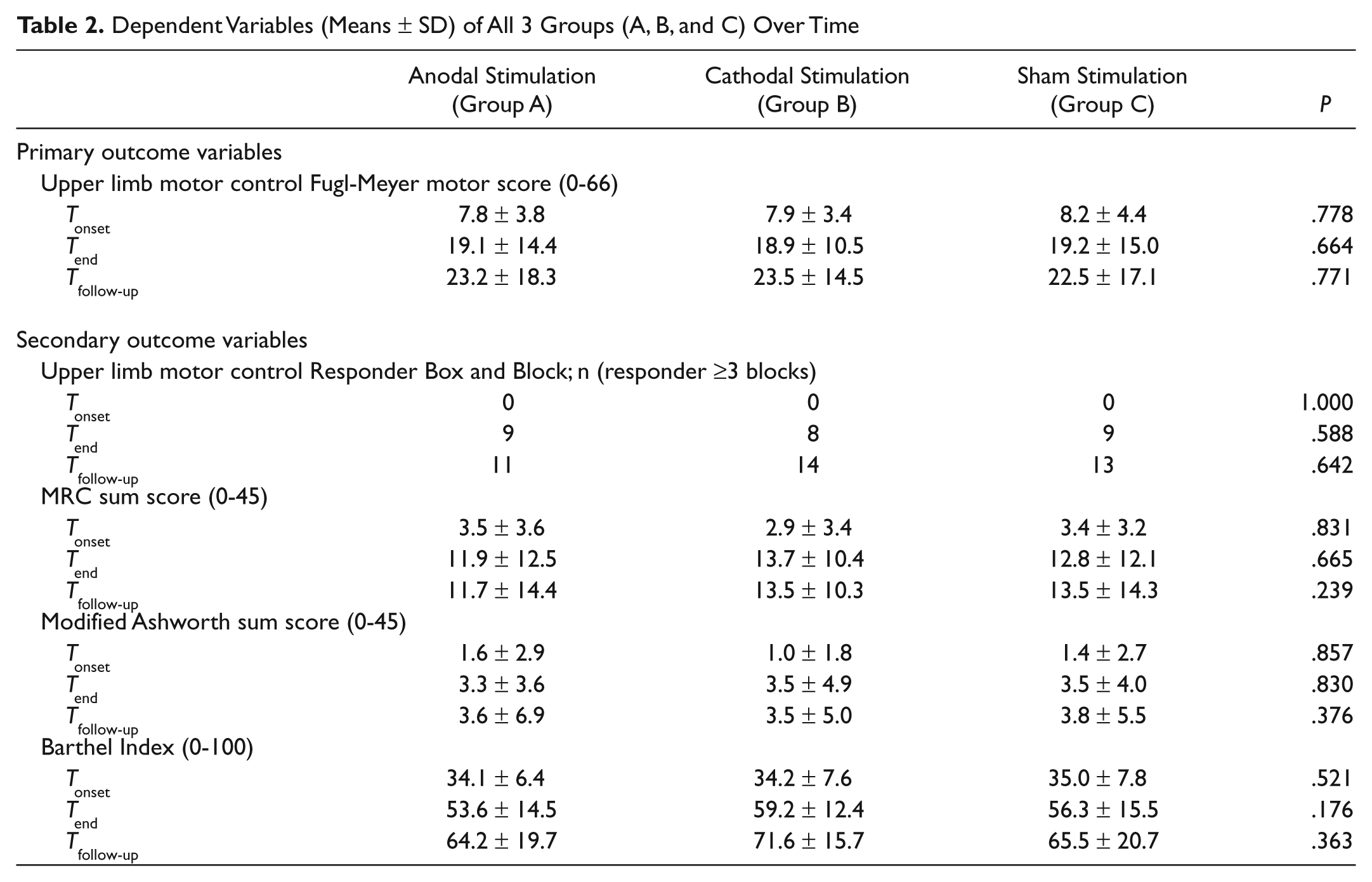
All patients improved their FMS scores significantly over time (P < .001), and between-group differences did not occur at any time (P > .025, Table 2, Figure 2). The same applied for the muscle strength, muscle tone, and the Barthel Index (P > .025, Table 2). The Box and Block test criterion of at least 3 wooden blocks being transferred within 1 minute, met 9 group A patients, 8 group B patients, and 9 group C patients at Tend; at Tfollow-up the number of patients were 11, 14, and 13, respectively, from groups A, B, and C. Neither the responder rate nor the absolute number of transferred blocks differed between the 3 groups (P > .025). The muscle tone increased evenly to a moderate extent in all 3 groups over time and no between-group differences occurred (P > .025).

The results of the Fugl-Meyer score (FMS, 0-66) over time for all 3 groups (anodal, cathodal, and sham stimulation) at study onset, study end, and at follow-up.
Dependent Variables (Means ± SD) of All 3 Groups (A, B, and C) Over Time
Furthermore, the FMS scores and number of responders in the Box and Block test of patients with a pure subcortical lesion and of patients with a cortical involvement were calculated for each of the 3 groups separately (Table 3). Within the cathodal group, the patients with a pure subcortical lesion improved to a significantly larger extent during the intervention than those patients with a cortical involvement (P = .0014, Figure 3). In the other 2 groups, no significant difference could be detected. Further statistical subgroup analysis did not detect an influence with regard to age, lesion size, and spasticity scores.
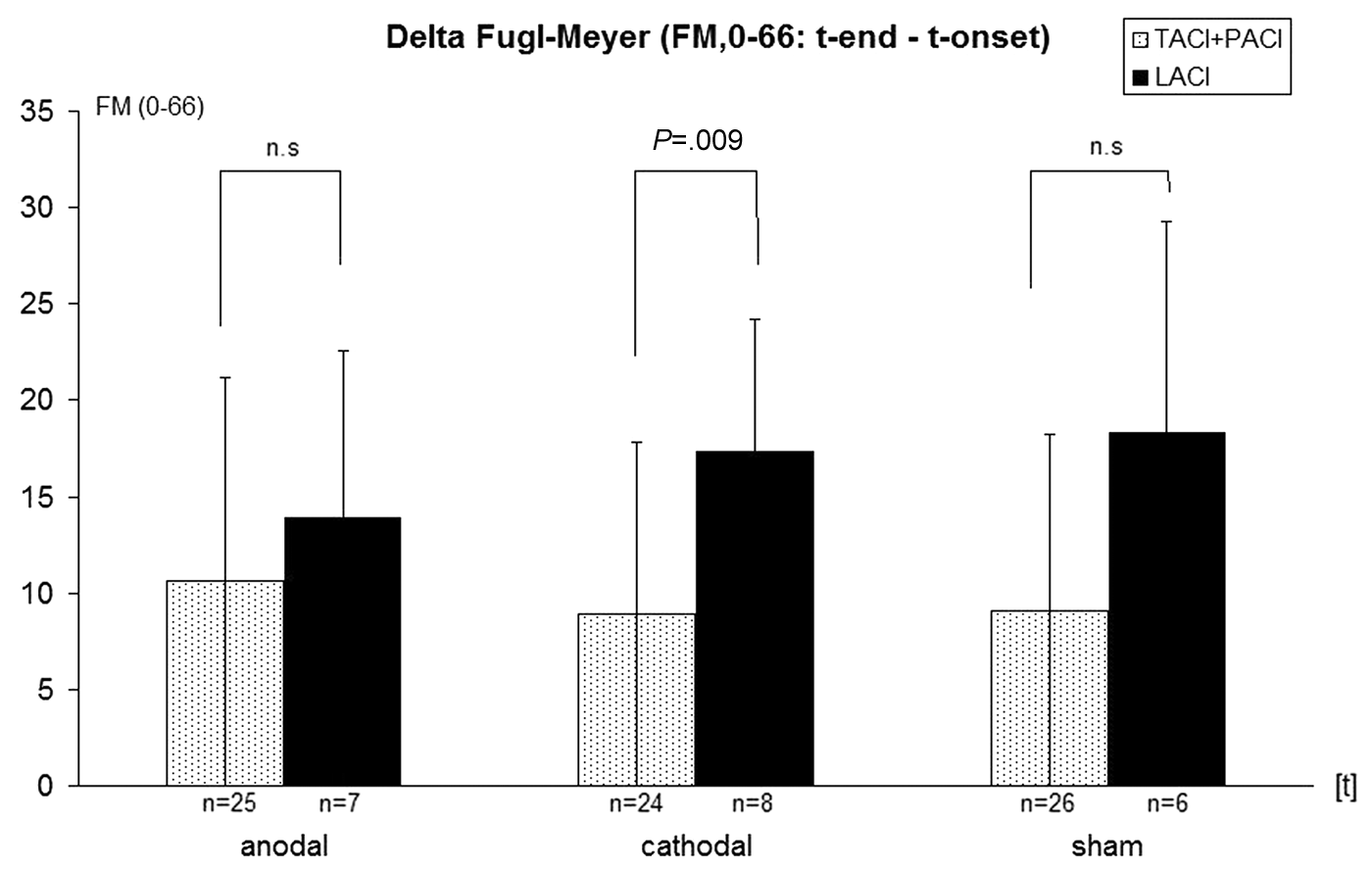
The improvement over time (Tend − T0) for each group in the Fugl-Meyer score additionally differentiated by the location of the lesion. Significant difference (P = .009) in the cathodal group for the patients with a subcortical lesion only.
Fugl-Meyer and Number of Responders of the Box and Block Test Differentiated by Location of Lesion
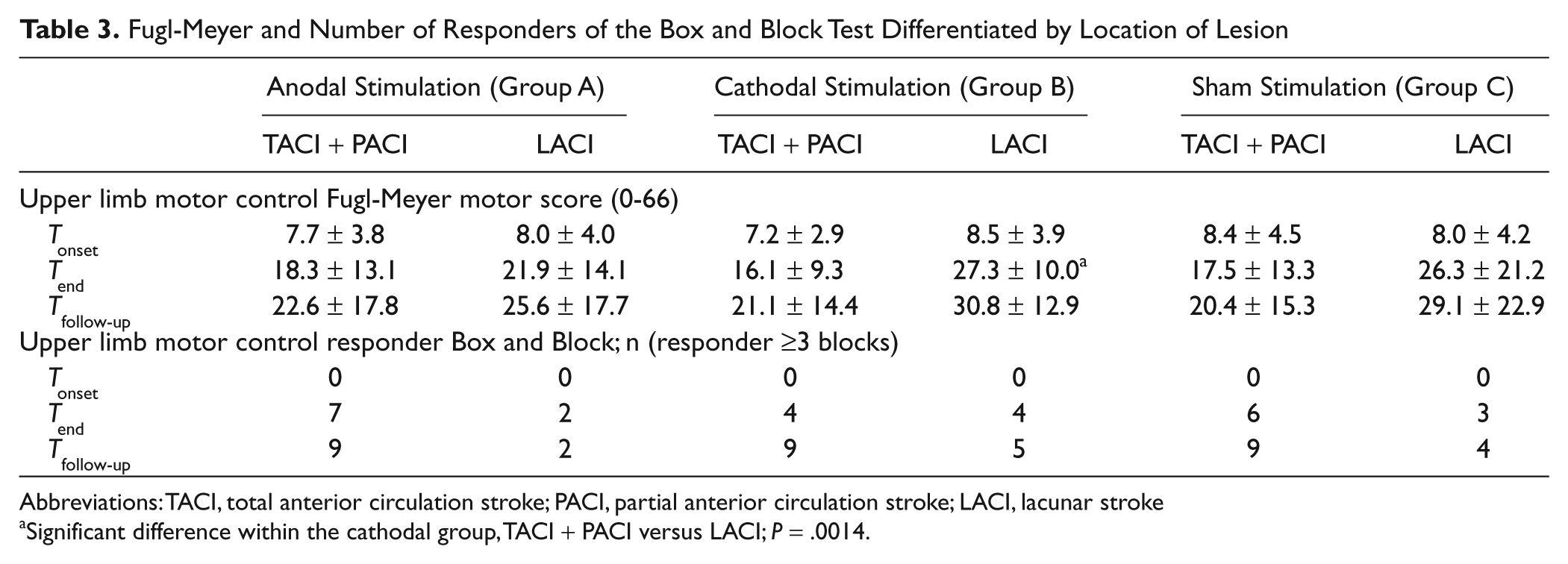
Abbreviations: TACI, total anterior circulation stroke; PACI, partial anterior circulation stroke; LACI, lacunar stroke
Significant difference within the cathodal group, TACI + PACI versus LACI; P = .0014.
Discussion
Contrary to the hypothesis, neither anodal stimulation of the lesioned nor the cathodal stimulation of the nonlesioned hemisphere potentiated the effect of the bilateral robot-assisted arm training. All patients significantly improved their UL function over time, but between group differences did not occur.
Several possibly confounding reasons may explain the lack of effect of the addition of anodal or cathodal tDCS stimulation. The majority of the patients (75% to 80%, distributed evenly in the 3 groups) presented either a large anterior circulation infarct with both cortical and subcortical involvement or a partial anterior circulation infarct resulting in a predominantly cortical involvement. Stroke patients with a subcortical infarct and thus intact cortical connectivity may profit more from the transcranial direct current stimulation. Preceding positive tDCS studies in subacute 8 and chronic stroke patients had focused on these subjects.16,19 In our trial, the FMS improvement of the patients with pure subcortical involvement in all 3 groups improved more, reaching a level of significance, without controlling for multiple comparisons, in the cathodal group (Table 3). Given the small number of patients with a pure subcortical lesion in all 3 groups and a similar trend in the sham group (Figure 3), conclusions must be drawn with caution. But one may speculate that cathodal stimulation of the nonlesioned hemisphere, aiming at the restoration of the excitatory imbalance between the 2 hemispheres after stroke, 32 needs continued evaluation.
The tDCS treatment protocol with respect to electrode position, intensity, stimulation duration, and the combination with UL therapy followed recommendations at the time of planning the study. Recent research suggests alteration of the electrode positioning for dual hemispheric stimulation 33 and of the stimulation sequence, in that the robotic training would have followed the brain stimulation. 34
Patient selection and the mode of UL therapy are further issues. For this exploratory trial, 96 participants may be too small, since it was calculated on an assumed effect size of 1, which may be too big given the fact that all patients were in the period of spontaneous recovery and received training.
So far, positive reports following single and multiple tDCS interventions in chronic stroke patients have focused on moderately affected patients who were able to grasp and release objects with the paretic hand.7,16,19 Our patients had an initial mean FMS of <10. MEPs of the paretic hand muscles, regularly assessed in one center, were absent, and no patient could transfer a single wooden block in the Box and Block test. With no MEPs detectable, one cannot exclude that the stimulation site over the lesioned hemisphere in the anodal group was wrongly chosen because of plasticity-related shifts of cortical representation. For that kind of severely affected patient, Kwakkel et al 2 reported a probability of less than 5% of regaining meaningful hand activity 6 months later. In the present study, 25% to 30% of the highly paretic patients of all 3 groups became able to transfer a minimum of 3 wooden blocks in the Box and Block test within 1 minute at the end of the intervention, indicative of some possible ability to use the paretic hand in daily life.
The authors chose the bilateral UL therapy approach, allowing for 800 repetitions in a single session, because of its proven effectiveness in the cases of severely affected subacute stroke patients according to a previous RCT from the same group. 23 Functional images showed a strong activation of the lesioned hemisphere, particularly in the active–passive mode, supporting the intended facilitation of the lesioned side via intercallosal fibers. 35 However, the bilateral approach, aiming at an activation of the nonlesioned hemisphere, is a matter of constant debate given the current discussion on the disturbed interhemsipheral inhibition after stroke. 36
For healthy subjects, Bradnam et al 37 reported that cathodal stimulation of the left M1 area suppressed the contralateral and ipsilateral MEP of the right infraspinatus muscle following a unilateral preactivating task with the left hand, whereas a preactivating bilateral task did not, indicating a task-dependent modulation. Recently, Celnik et al 7 reported on the beneficial effects of combined unilateral peripheral nerve stimulation and brain polarization on performance of a motor sequence task, and Edwards et al 9 reported on a raised corticomotor excitability of the M1 forearm area following anodal tDCS sustained during unilateral robotic wrist therapy. Accordingly, one may speculate that a unilateral instead of a bilateral approach, which may have counteracted the intended inhibition of the nonlesioned hemisphere by the cathodal stimulation, 5 could have yielded a better result. However, these studies and most of the preceding ones were conducted in chronic and less severely affected patients, and had a small sample size.* Lindenberg et al, 38 for instance, included 20 moderately affected chronic stroke patients with an initial mean FMS of 38.2, and the mean improvement in the experimental group was 6.1. FMS points were measured after 5 consecutive sessions of bihemispehric tDCS. Present rehabilitation interventions in chronic patients with poor selective movements of the UL may not be robust enough to improve function.39-41
For severe arm paresis in the subacute stage, one may further speculate that upregulation of the healthy hemisphere might be an important strategy, so the combination of the robot training with anodal stimulation of the nonlesioned hemisphere might have been a better alternative. Repetitive magnetic cortical stimulation may be another option 42 as recent data showed a positive and sustained effect on the hand function in mildly affected subacute stroke patients. 43
Summary
We found no effect of brain polarization combined with UL repetitive practice on motor recovery in severely affected subacute stroke patients. On a secondary analysis of a small number of patients who received cathodal stimulation of the nonlesioned hemisphere, those with a pure subcortical lesion improved significantly more than patients with cortical involvement. Accordingly, the anatomic pattern, the severity of paresis, and the type of UL therapy—bilateral versus only the affected UL—are methodological issues to be addressed in future trials.
Footnotes
Acknowledgements
The authors are indebted to Dr M. A. Nitsche, Göttingen, for his kind help with the preparation of the article.
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article:
Reha-Stim, Berlin, holds the national patent of the robot applied in the study, the owner of the company is the spouse of the author SH.
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The Verein zur Förderung der Hirnforschung und Rehabilitation e.V. supported the TRAGAT study (NCT 00407667).
