Abstract
Background. Motor learning is essential to gain skills with neurorehabilitation. Objective. To investigate whether capacity for motor learning affects rehabilitation outcome and its relevant brain activation in ataxic patients with stroke. Methods. Twelve patients presenting with ataxia admitted for inpatient rehabilitation 2 to 3 months after infratentorial stroke and 6 control subjects performed 8 repetitions of 30-second pursuit rotor (PR) task. Cortical oxygenated hemoglobin (oxyHb) signals were measured using functional near-infrared spectroscopy. Results. Both patients and controls learned the PR skill, although the gains in PR performance were significantly lower in patients. In patients, the less learning significantly correlated with smaller rehabilitation gains assessed by the Functional Independence Measure. The Fugl-Meyer score for coordination and balance did not change. Center of task-related increase of cortical oxyHb signals shifted from the presupplementary motor area (preSMA) to the supplementary motor area (SMA) with task repetitions in controls but not in patients. Accordingly, serial changes of ratio of oxyHb increase in the preSMA to SMA (preSMA/SMA ratio) were significantly different between the groups. In patients and controls, gains in PR performance and changes of the preSMA/SMA ratio correlated. Conclusions. Impaired motor sequence learning by the PR task was correlated with reduced rehabilitation gains for ataxic patients with stroke.
Introduction
Repeated task-oriented exercise enhances functional recovery after stroke. In both human and experimental animal studies, augmented use of the paretic arm or forelimb induced use-dependent plasticity in the primary motor cortex (M1), as well as measurable improvement of real-world outcome.1-3
In the clinical setting, a dissociation between improvement of activities of daily living (ADLs) and reduction of motor impairment may occur. 4 This dissociation may partly be explained by patients’ capacity for learning of complex motor skills required for ADLs over repeated training sessions. Previous functional neuroimaging studies revealed a crucial role of the frontal cortex as well as the basal ganglia and cerebellum in motor learning. 5 To analyze brain activation associated with motor learning, most studies have used functional magnetic resonance imaging or positron emission tomography that requires subjects to minimize body motion for image acquisition. However, motor learning for ADLs requires postural control even during motor performance using the hand. We used functional near-infrared spectroscopy (fNIRS), which is suitable for measuring brain activities during dynamic motor behavior.6-8 We hypothesized that the capacity for motor learning affected rehabilitation outcome and motor network plasticity in patients with stroke. Since motor learning is expected to be impaired in patients with cerebellar ataxia, we have focused on patients with ataxia with minimal motor paresis and tested if improvement of ADLs is attributed to preserved capacity for motor learning.
Methods
Participants
A total of 12 patients with ataxia after infratentorial stroke and minimal hemiparesis participated in this study after transfer from an acute hospital for postacute inpatient rehabilitation. Clinical characteristics are shown in Table 1. Ataxia was assessed by the upper extremity coordination and the balance subscores of the Fugl-Meyer scale (3.5 ± 2.0, normal = 6; 8.7 ± 2.0, normal = 14, respectively), representing mild to moderate ataxia.
Characteristics of Ataxic Patients With Stroke and Controls
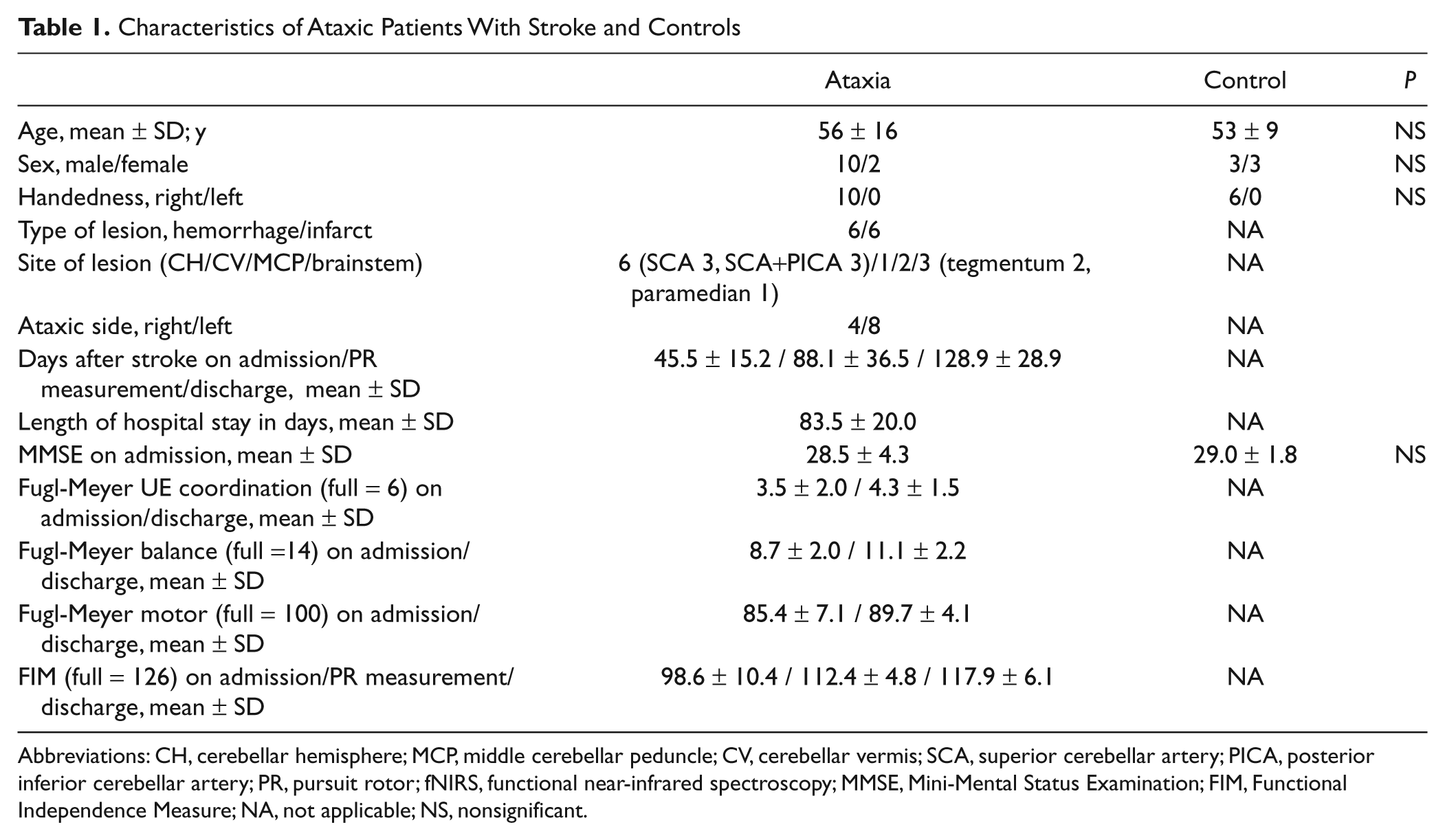
Abbreviations: CH, cerebellar hemisphere; MCP, middle cerebellar peduncle; CV, cerebellar vermis; SCA, superior cerebellar artery; PICA, posterior inferior cerebellar artery; PR, pursuit rotor; fNIRS, functional near-infrared spectroscopy; MMSE, Mini-Mental Status Examination; FIM, Functional Independence Measure; NA, not applicable; NS, nonsignificant.
Patients underwent 1 hour of physical and occupational therapy on weekdays and 1 hour on weekends, focused on improvement of ataxia and ADLs. Physical therapy included general conditioning, range of motion, muscle strengthening, static and dynamic balance exercise, walking, and climbing up and down stairs. Occupational therapy included hygiene, dressing, writing, eating, toileting, bathing, balance exercise, reaching, and coordinative tasks of upper limbs and trunk.
We assessed capacity for motor learning using a pursuit rotor (PR) in the patients and 6 right-handed healthy controls. During the PR performance, cortical activation patterns were monitored using an fNIRS system. The baseline assessment included the PR task and fNIRS as well as the Functional Independence Measure (FIM) at a mean 88.1 ± 36.5 days after stroke onset. FIM was also assessed at discharge. We tested for significant correlations between the gains in PR task prior to rehabilitation and FIM scores at entry and discharge. The FIM gains were calculated by subtracting FIM scores at the time of PR measurement from discharge FIM scores. This study was approved by the institutional review board and written informed consent was obtained from each participant.
Pursuit Rotor Task
Details of the PR task have been described. 8 Subjects sat in front of the PR instrument (TKK2110, Takei Co, Niigata, Japan) and were instructed to keep the tip of a metal stylus on a round target (11 mm in diameter) of the disc (105 mm in radius) rotating counterclockwise at 21 rpm. The distance from the shoulder and the edge of the PR instrument was set at 50 cm. Patients performed PR task with the nonataxic followed by the ataxic hand; controls performed it with the right followed by the left hand. No one had experienced the task before the experiment. Each subject performed a 30-second task alternating with 30 seconds of rest for 8 cycle repetitions. The PR task requires motor control of shoulder–elbow coordination and postural control but no need for precise control of finger movements. Capacity for motor learning was assessed by the gains of the contact time of the stylus on the target during 8 cycle repetitions.
Functional Near-Infrared Spectroscopy
Cortical activation during the PR task was assessed by task-related changes in hemoglobin oxygenation using an fNIRS system as reported previously.6-8 The system (OMM-2001, Shimadzu, Kyoto, Japan) comprised 12 near-infrared light sources (780, 805, and 830 nm) and 16 detectors resulting in 42 channels. Data sampling rate was 5.3 Hz. For anatomical information, the location of the optodes was marked with a 3-dimensional digitizer (FASTRAK; Polhemus, Colchester, Vermont). After calculating the midpoint of the neighboring light source and detector on the skull surface, the location of fNIRS channels on the cortex were estimated using the balloon inflation method. 9 Following this, anatomical normalization to the Montreal Neurological Institute standard template 10 was performed using a 12-parameter affine transformation. The estimated locations of fNIRS channels on the cortex were also transformed using the affine transformation matrix. These procedures were performed by the Fusion software (Shimadzu, Kyoto, Japan). We assumed that cortical representation of the fNIRS channels was similar to that reported in the prior study (Figure 1). 8 fNIRS topographic map was split into 9 regions of interest 11 according to functional anatomy of the frontal lobe, including the sensorimotor cortex (SMC), supplementary motor area (SMA), presupplementary motor area (preSMA), dorsal premotor cortex (PMC), and dorsolateral prefrontal cortex (PFC; see Figure 1). A standardized determination for optode location supports the validity of allocated regions of interest used in this study, provided that the spatial resolution of fNIRS imaging is a few centimeters. 12 Cortical activation was assessed as task-related increases in oxygenated hemoglobin (oxyHb) levels (mM cm) based on the modified Beer–Lambert law for highly scattering media 13 since oxyHb was more sensitive to task-related changes than deoxyHb in our experimental setting.6-8 To compare the amount of regional activation among 8 cycles of PR task, each regional activation was defined as average values of task-related oxyHb increases (ΔoxyHb) in the channels each region of interest, that is, ΔoxyHb = oxyHb changes during the task period − oxyHb changes during the rest period. For quantitative analyses for serial changes of regional activation, oxyHb signal values were normalized to those of the first cycle to circumvent influence of differential path length factors14,15 among different regions and subjects.
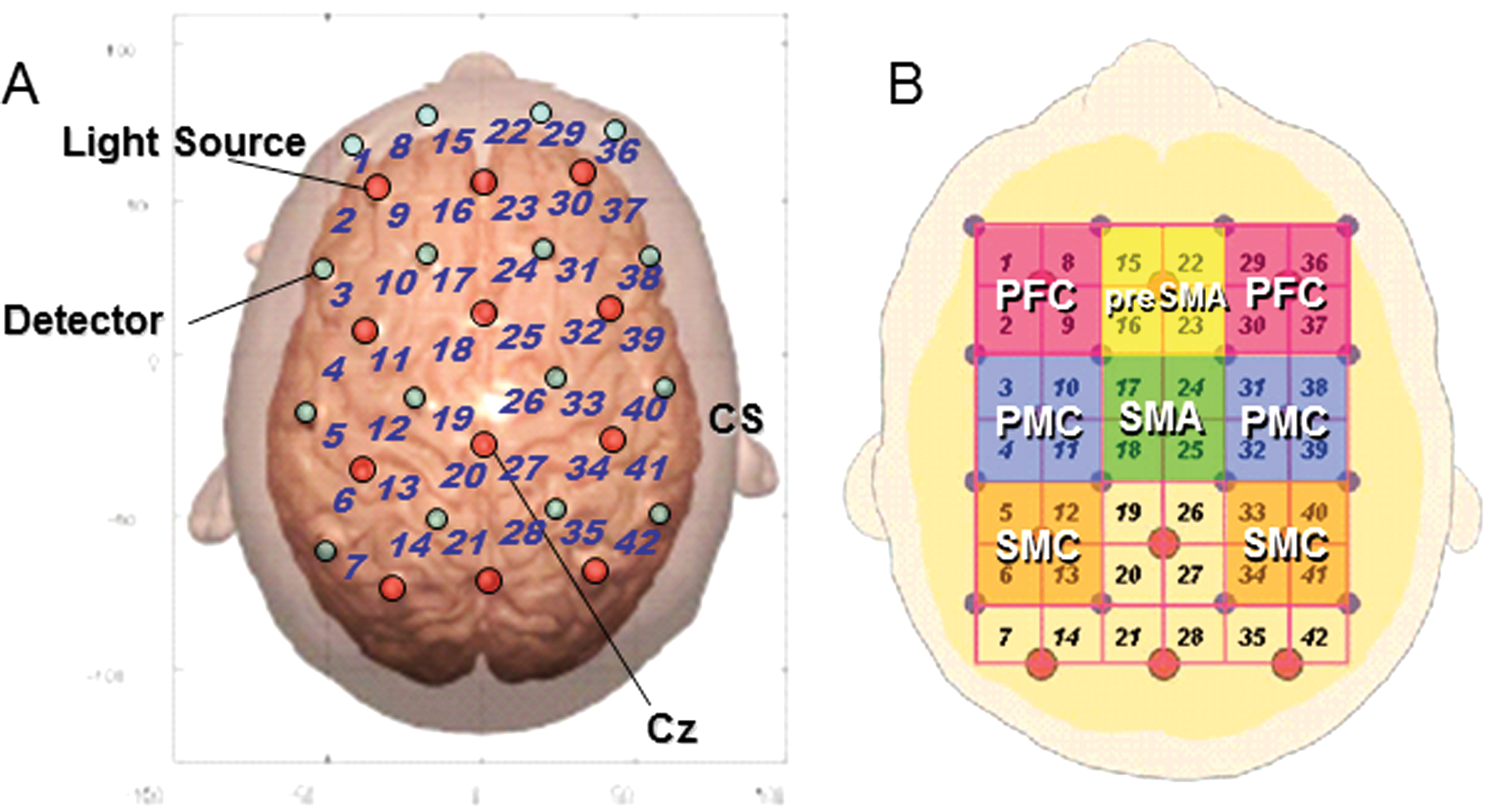
Experimental setting of optodes. A, Location of optodes exposed to the brain surface of the standard brain. A total of 28 optodes, comprising 12 light source fibers (red) and 16 detectors (blue), were arranged on the scalp that enabled 42-channel measurement. We defined the fNIRS channels as the midpoint of the corresponding light source–detector pair. The optodes were fixed on the skull using a custom-made cap covering an area of 13 × 15 cm2 over the frontoparietal cortices after setting interoptode distance to 3.0 cm. Cz position of the international 10-20 system was the marker for ensuring replicable placement of the optodes. The anatomical location of the MRI surface images overlaid on the normalized brain surface. B, The channels covering SMC, SMA, preSMA, PMC, and PFC. The left and right lateral SMC were covered by channels 5, 6, 12, 13 and 33, 34, 40, 41, respectively; SMA by channels 17, 18, 24, and 25; preSMA by channels 15, 16, 22, and 23; the left and right PMC by channels 3, 4, 10, 11 and 31, 32, 38, 39, respectively; and the left and right PFC by channels 1, 2, 8, 9 and 29, 30, 36, 37, respectively. Abbreviations: fNIRS, functional near-infrared spectroscopy; MRI, magnetic resonance imaging; CS, central sulcus; SMC, sensorimotor cortex; SMA, supplementary motor area; PMC, dorsal premotor cortex; PFC, dorsolateral prefrontal cortex.
Statistical Analyses
Changes of PR performance across the task cycles were analyzed using 2-factorial repeated-measures analysis of variance (ANOVA) with task cycle (1 to 8) as within-subject factor and group (right in controls, left in controls, ataxic side in patients, and nonataxic side in patients) as between-subject factor. Serial changes of ΔoxyHb with cycle repetitions in each region were also evaluated using 2-factorial (task cycle and group) repeated-measures ANOVA. Correlations between performance gains and changes of oxyHb signals in the PR session, FIM scores at the time of PR measurement and at discharge as well as their gains were analyzed using Spearman rank correlation coefficient. Bonferroni test was used as post hoc test. Statistical significance was set at P < .05 after correction for multiple comparisons if applicable.
Results
Pursuit Rotor Performance
Pursuit rotor performance improved with cycle repetitions (F7,224 = 67.7, P < .0001) both in controls and patients with a significant cycle × group interaction indicating delayed motor learning in patients (F21,224 = 4.9, P <.0001, Figure 2A). Overall performance was significantly better in controls with right (P < .001) and left hand (P < .005) than in patients with the ataxic (P < .001/<.005) and nonataxic hand (P < .005/.05). Mean ± SD gain was 14.6 ± 2.9 with right hand, 11.4 ± 2.9 with left hand in controls, 7.6 ± 2.7 with nonataxic hand and 5.6 ± 3.4 with ataxic hand in patients. There was no significant correlation between PR gains and coordination or balance subscores of Fugl-Meyer scale.
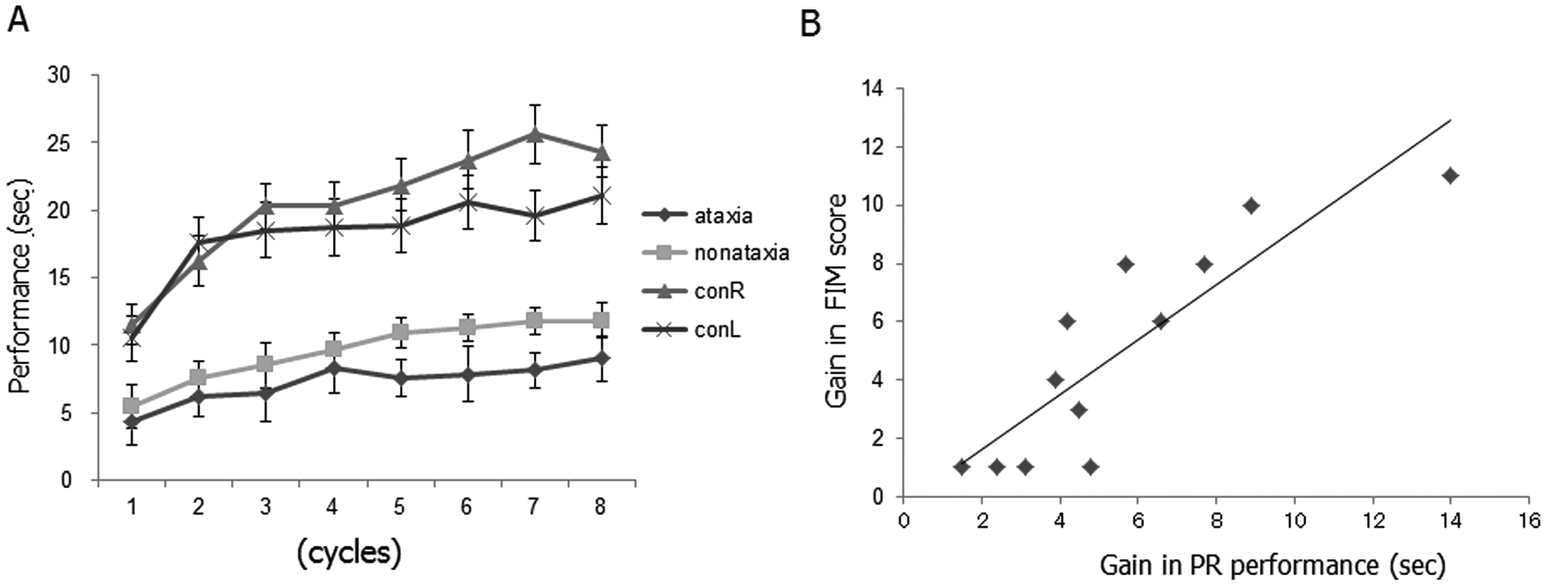
Pursuit rotor performance. A, Overall performance of pursuit rotor (F7,224 = 67.7, P < .0001) and gain (F21,224 =4.9, P < .0001) were significantly lower in ataxic patients than in controls. The gain of the PR performance was better in the following order: right hand in controls (conR), left hand in controls (conL), nonataxic hand in patients (nonataxia), and ataxic hand in patients (ataxia). Data are mean ± standard error. B, The gains of PR performance using the ataxic hand significantly correlated with the gains of FIM scores from the day of PR measurement to discharge (r = 0.861, P < .001). Abbreviations: PR, pursuit rotor; FIM, Functional Independence Measure.
Relationship Between Pursuit Rotor Performance and Rehabilitation Outcome
Functional Independence Measure scores significantly improved during inpatient rehabilitation (Table 1, F1,11 = 4882.6, P < .001). The gains of PR performance using the ataxic hand significantly correlated with FIM scores on discharge (r = 0.788, P < .005) and with the gains of FIM scores from the day of PR measurement to discharge (r = 0.861, P < .001) but not with the gains of FIM scores from admission to discharge (Figure 2B).
fNIRS Findings
In controls, cortical mapping based on oxyHb signal changes revealed that cortical activation centered in the preSMA in the early cycles and its center gradually shifted to the SMA in the late cycles as reported previously. 8 In ataxic patients with stroke, PR performance using the ataxic and nonataxic hand was associated with sustained activation of the prefrontal regions compared with controls (Figure 3). There was no difference in the serial changes of task-related oxyHb signals in the SMC, PMC, and PFC) between the groups. To quantitatively evaluate the shift of activation center from the preSMA to SMA, we calculated preSMA/SMA ratio of normalized oxyHb signals from each regions. 8 The smaller preSMA/SMA ratio indicates greater shift of activation center to the SMA. There was a significant main effect for group in preSMA/SMA ratio (Figure 4A, F3.40 = 3.4, P < .05). Post hoc test revealed a significant difference between controls and patients (P < .05). These indicated that the posterior shift of activation center in the medial frontal regions was less evident in ataxic patients.
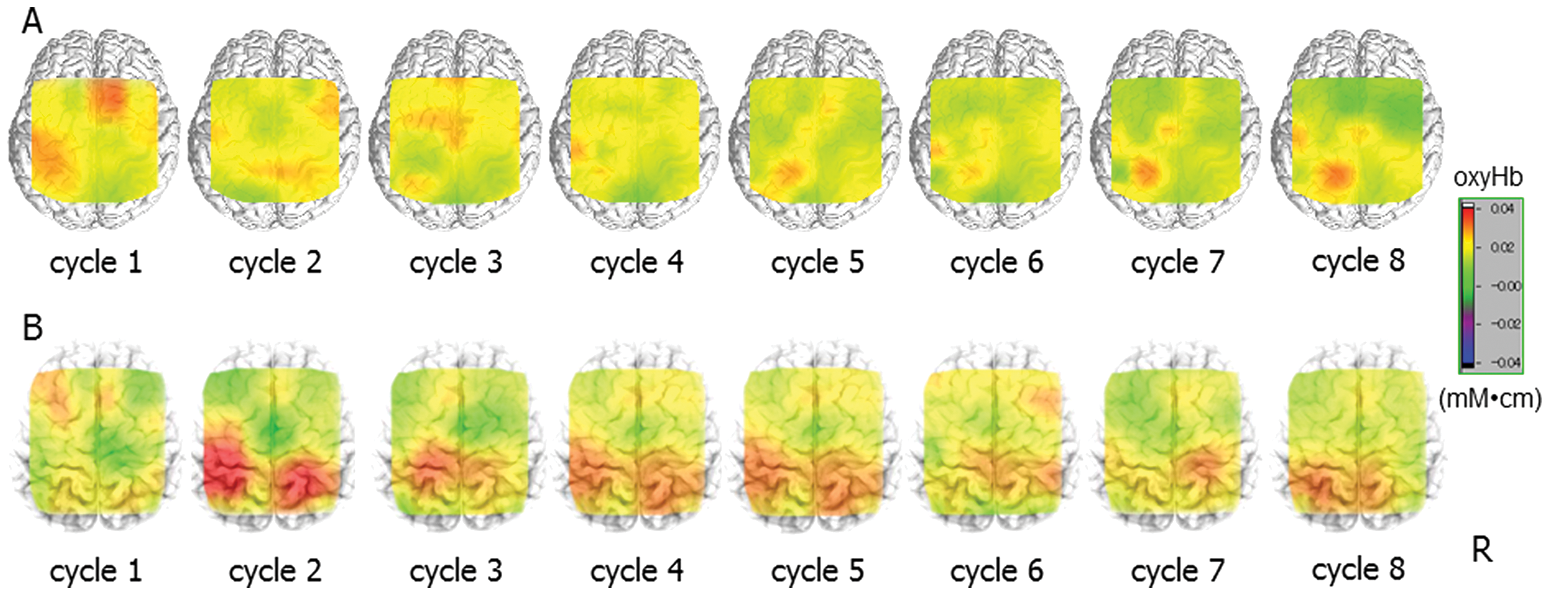
Changes of cortical activation patterns during PR performance. A, Task-related changes of cortical oxyHb signals in a control subject (45-year-old male). Serial changes of PR performance with the right hand were 17.1, 18.4, 23.1, 22.4, 24.3, 25.9, 26.7, and 27.5 seconds. There is a recruitment of preSMA in the early cycles and SMA in the late cycles. B, Task-related changes of cortical oxyHb signals in a patient (59-year-old male) 115 days after the onset of right cerebellar infarction. Serial changes of PR performance with ataxic right hand were 5.8, 9.4, 10.3, 10.8, 10.6, 11.5, 8.6, and 13.5 seconds. There is a sustained recruitment of preSMA, and shift of activation center from preSMA to SMA in the late cycles is less prominent. Abbreviations: PR, pursuit rotor; oxyHb, oxygenated hemoglobin; SMA, supplementary motor area.
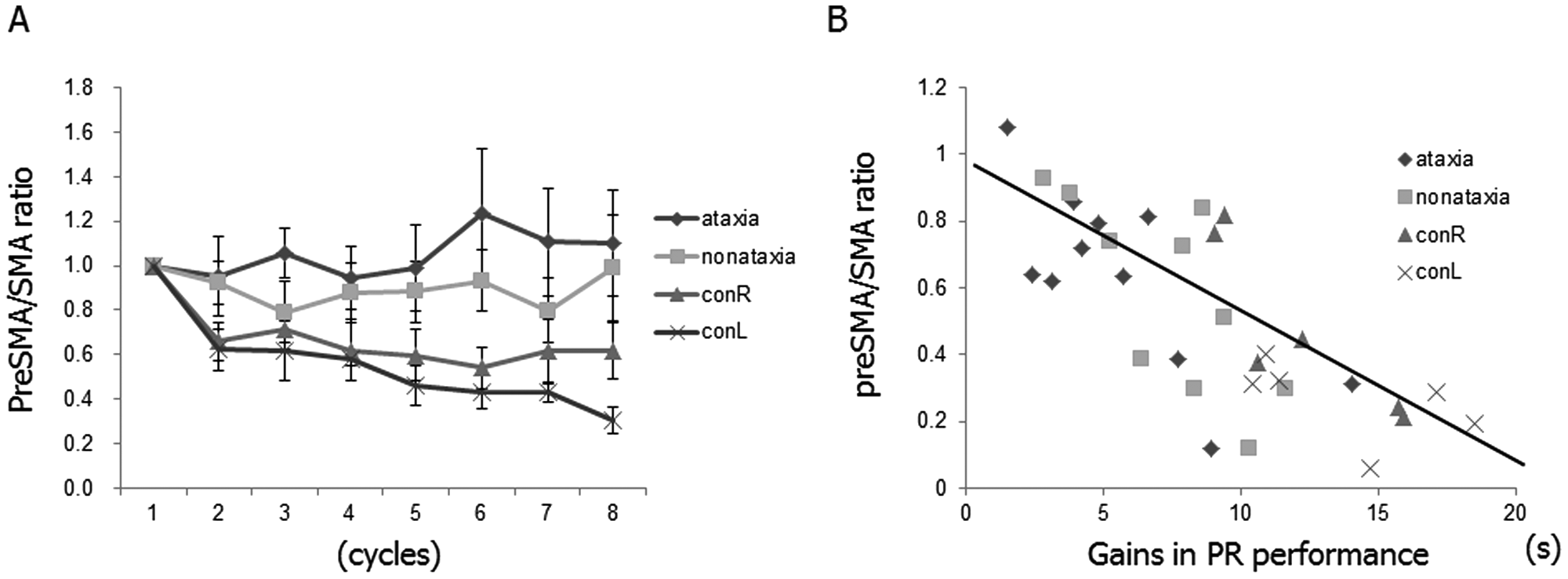
Serial changes in preSMA/SMA ratio of normalized oxyHb signals. A, The posterior shift of activation center in the medial frontal regions was less evident in ataxic patients. See text for details. Data are mean ± standard error. B, There was a significant correlation between the gains in PR performance and the relative shift of medial frontal activation during the task as evaluated by the smallest preSMA/SMA ratio (r = −0.747, P < .001). Abbreviations: SMA, supplementary motor area; oxyHb, oxygenated hemoglobin; PR, pursuit rotor.
Correlation Between Pursuit Rotor Performance and fNIRS Findings in Ataxic Patients
Since the gain in PR performance significantly correlated with final ADL status after inpatient rehabilitation, it was of interest whether serial activation patterns and the gains in PR performance were correlated. In both controls and patients, there was a significant correlation between the gains in PR performance and the relative shift of medial frontal activation as evaluated by the smallest preSMA/SMA ratio (r = −0.747, P < .001 for overall correlation, r = −0.655 for ataxic hand, r = −0.733 for nonataxic hand, r = −0.886, for right hand in controls, and r = −0.657, for left hand in controls, Figure 4B). In patients, the smallest preSMA/SMA ratio during PR performance using the ataxic hand also correlated with the gain of FIM score from the day of PR measurement to discharge (r = −0.650, P < .05). Thus, to test whether the PR gain or fNIRS measure was better at correlating with the FIM gain than baseline FIM, we performed a multiple regression analysis. The model (r 2 = 0.823) indicated that PR gain (84.7%, P < .05) significantly contributed to the FIM gain, but neither preSMA/SMA ratio nor baseline FIM reached significance.
Discussion
Motor learning includes motor adaptation and motor sequence learning. 5 Motor adaptation refers to the ability to compensate for environmental changes such as external force. 16 Motor sequence learning is defined as learning of sequential motor actions. ADLs require motor sequence learning. The early phase of motor learning, that is, fast motor learning depends both on the corticostriatal and the corticocerebellar systems, including the frontoparietal cortex.5,17 The cerebellum is involved in processing feedback information, including visuomotor association and controlling real-time movements. 18 Thus, cerebellar damage can cause a deficit in motor learning. Indeed, patients with cerebellar lesions showed impairment in finger sequence learning 19 and in adaptation learning.20,21 We demonstrated that ataxic patients with stroke showed significantly smaller gains in a PR task compared with healthy controls for motor learning. The deficit in performance gain could be explained by the ataxia, rather than by a direct role of the cerebellum in motor learning. 22 We, however, found no relationship between the PR gains and degree of limb ataxia. Thus, it is likely that smaller PR gains are explained by impaired motor learning.
We also found that the degree of impaired motor learning correlated with functional outcome after focused inpatient rehabilitation. Significant correlations were found between PR gains and discharge FIM scores and between PR gains and FIM gains from the day of PR measurement to discharge (Figure 2B) but not between PR gains and FIM gains from admission to discharge. Since there was a chance that the rehabilitation altered these parameters before PR measurement, it may be that the capacity for motor learning as measured by PR gains correlated with the gains of FIM thereafter. This suggests that patients with greater capacity for motor learning showed more improvement in ADLs in response to further rehabilitation.
In the process of neurorehabilitation, patients relearn motor skills for ADLs by repeating sequential movements. Further practice may lead to the later stage of motor learning in which mainly the intact striatum is involved. 5 It is possible that dose-dependent effect of neurorehabilitation for stroke23,24 may be partly attributed to impaired motor learning since patients need to compensate for the impairment by repeating task-oriented practice.
Serial changes of preSMA/SMA oxyHb signal ratio showed a similar time course to those of PR performance as reported previously. 8 PreSMA may be more engaged in cognitive aspects of movements, whereas SMA is more involved in motor aspects of movements. 25 Similarly, preSMA was involved in the process of learning sequences, whereas SMA was active during the performance of the learned sequential movements. 26 Reduced preSMA activation coupled with improved motor skill may reflect a neurophysiological “pruning” mechanism. 27 Furthermore, preSMA is involved in conscious visuomotor association rather than sequence learning and the association may be replaced by the formation of sequential procedural memory outside the preSMA. 28 Thus, the relative shift of activation from preSMA to SMA might be attributed to improved visuomotor association and transition of PR performance from feedback to feed-forward control. In patients with cerebellar stroke, preSMA activation was relatively sustained even in the late cycles. This suggests that they continuously relied on visuomotor association to perform the task, although performing the PR task after removing visual control is needed to test this hypothesis. Furthermore the changes of preSMA/SMA ratio showed significant correlation with the PR gains in both controls and patients. The serial changes of activation patterns are likely to reflect the capacity for motor learning. Performance gain was a stronger marker than the activation change, however, during PR measurement and the baseline FIM in determining the FIM gain.
Although fNIRS cannot assess cerebellar activities, the shift of activation center from the preSMA to SMA could be a surrogate marker for fast motor learning and be useful for a neuro-feedback parameter of motor learning status. Since function of a particular brain region can be viewed in terms of the emergent properties of large-scale neural network interaction, 27 the preSMA/SMA ratio may reflect interaction of these brain areas during motor and cognitive operation of the motor learning task. Anatomical studies revealed the tight connection between the cerebellum and cerebral cortices, especially the frontal lobe. 29 The prefrontal cortex has close anatomical interconnection forming a closed-loop circuit with the cerebellum, specifically Crus II in monkey. 30 The primary motor cortex has interconnection with lobules IV to VI of the cerebellum, suggesting that these cerebellar loops are involved in cognitive processing as well as generation and control of movement. 30 A crucial role of the corticocerebellar system in motor learning is supported by an observation that functional recovery after cerebellar stroke was affected by supratentorial white matter lesions that might disrupt functional connectivity. 31
Although our results may provide further insight into the relationship between motor learning and functional outcome in patients with cerebellar damage, there are a number of limitations. We only tested the early stage of motor learning. It is also important to see how consolidation and retention of learned skills affect functional outcome. Our findings may be overstated, since we did exploratory analyses to find any significant correlation between motor learning and functional measures. It is unclear whether our findings can be generalized to functional recovery of hemiparetic stroke.
Conclusions
Impaired motor learning affected rehabilitation outcome of ataxic patients with stroke and cortical activation patterns in the medial frontal areas in the process of motor sequence learning.
Footnotes
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Dr Miyai is supported by Grant-in-Aid for “the Research Committee for Ataxic Diseases” of the Research on Measures for Intractable Diseases and The Research Grant (21B-9) for Nervous and Mental Disorders from the Ministry of Health, Labor and Welfare, Japan. Dr Hattori is supported by PRESTO from the Japan Science and Technology Agency.
