Abstract
Introduction
Recent studies have shown that physiotherapy can be a useful adjunct to the medical management of patients with Parkinson disease (PD). 1 Its main goals are to preserve the patient’s motor function and functional independence. There are 2 distinct but complementary approaches to achieving these goals. 2 The first one includes physical practice to enhance muscular strength, endurance, balance, coordination, and flexibility, whereas the second approach is to instruct patients in using compensatory attentional strategies. PD patients benefit from a wide range of attentional strategies, including the use of external cues, 3,4 breaking long or complex movement sequences down into components, 5 and mental rehearsal of movements. 6
Motor imagery (MI), which can be defined as mental rehearsal of a motor act in the absence of overt motor output, is a training technique that has been used by athletes for years. 7 Recently, it has also gained attention as a promising additional rehabilitation method for patients with neurological disorders. Most evidence is found in stroke patients, 8,9 in whom it was shown that MI practice may lead to improvements in arm function and activities of daily living. 10,11 To date, however, it remains unclear whether patients with PD derive similar benefits from MI training. An important underlying question is whether these patients are still able to imagine movements because there are theoretical reasons to assume that imagery may be affected in PD. Nigrostriatal dopaminergic deficiency of PD could affect imagery performance because basal ganglia were shown to be activated not only during physical execution but also during imagery of movements. 12-14 Studies on the effect of basal ganglia dysfunction on MI, however, are scarce and have reported largely inconsistent results. 6,15 At the neural level, some imaging studies showed altered activation during MI in patients with PD in comparison to controls, 16,17 whereas others reported no significant differences when the patients were in the on phase of their medication cycle. 18 A behavioral study by Frak et al 19 indicated that PD affected the patients’ imagery of a grasping task, whereas physical movement was spared. Dominey et al 20 and Helmich et al, 21 on the other hand, reported that the MI deficits in PD parallel those observed during physical movement. The question remains whether these abnormalities reflect a lack of imagery quality or merely an adaptation of preserved MI to the reduced physical motor abilities of PD patients.
Furthermore, studies on MI often did not assess imagery ability at all or only assessed limited aspects. 22-25 However, it is of crucial importance to thoroughly evaluate PD patients’ MI ability because deficiencies likely reduce potential benefits of MI practice. Because MI is a complex and covert process, evaluating its different aspects is challenging. Currently, no standard clinical test battery is available for this purpose. Therefore, we assembled a comprehensive clinical MI test battery evaluating various aspects of MI, such as accuracy, vividness, and temporal organization as well as several types, including visual and kinesthetic and implicit and explicit MI. The present study compared MI ability in PD with age-matched controls and explored its relationship with motor and cognitive disease characteristics in PD.
Materials and Methods
Participants
14 patients with PD (9 men; 59.1 ± 9.6 years) and 14 age-matched healthy controls (8 men; 61.1 ± 6.6 years) participated. The diagnosis of PD was made by a medical doctor using the Brain Bank criteria. 26 All patients were in Hoehn and Yahr (H&Y) stages 1 to 3, as evaluated during the on state of their medication cycle. Motor status in “on” was assessed using the Unified Parkinson’s Disease Rating Scale (UPDRS) part III. Exclusion criteria were: Mini-Mental State Examination (MMSE) score <24, severe tremor, neurological comorbidity, unpredictable motor fluctuations, severe orthopedic problems of the upper limb, and treatment with deep brain stimulation. Patient characteristics are detailed in Table 1. All participants in this study were right handed as measured by the Edinburgh Handedness Inventory Questionnaire. 27 Besides the MMSE, a detailed cognitive screening was performed by means of the Scales for Outcomes in Parkinson’s Disease-Cognition (SCOPA-COG). The study was conducted in accordance with the ethical standards laid down in the 1964 Declaration of Helsinki and was approved by the Committee for Ethical Considerations in Human Experimentation of the Katholieke Universiteit Leuven. All participants gave written informed consent before taking part in the study.
Patient Characteristics
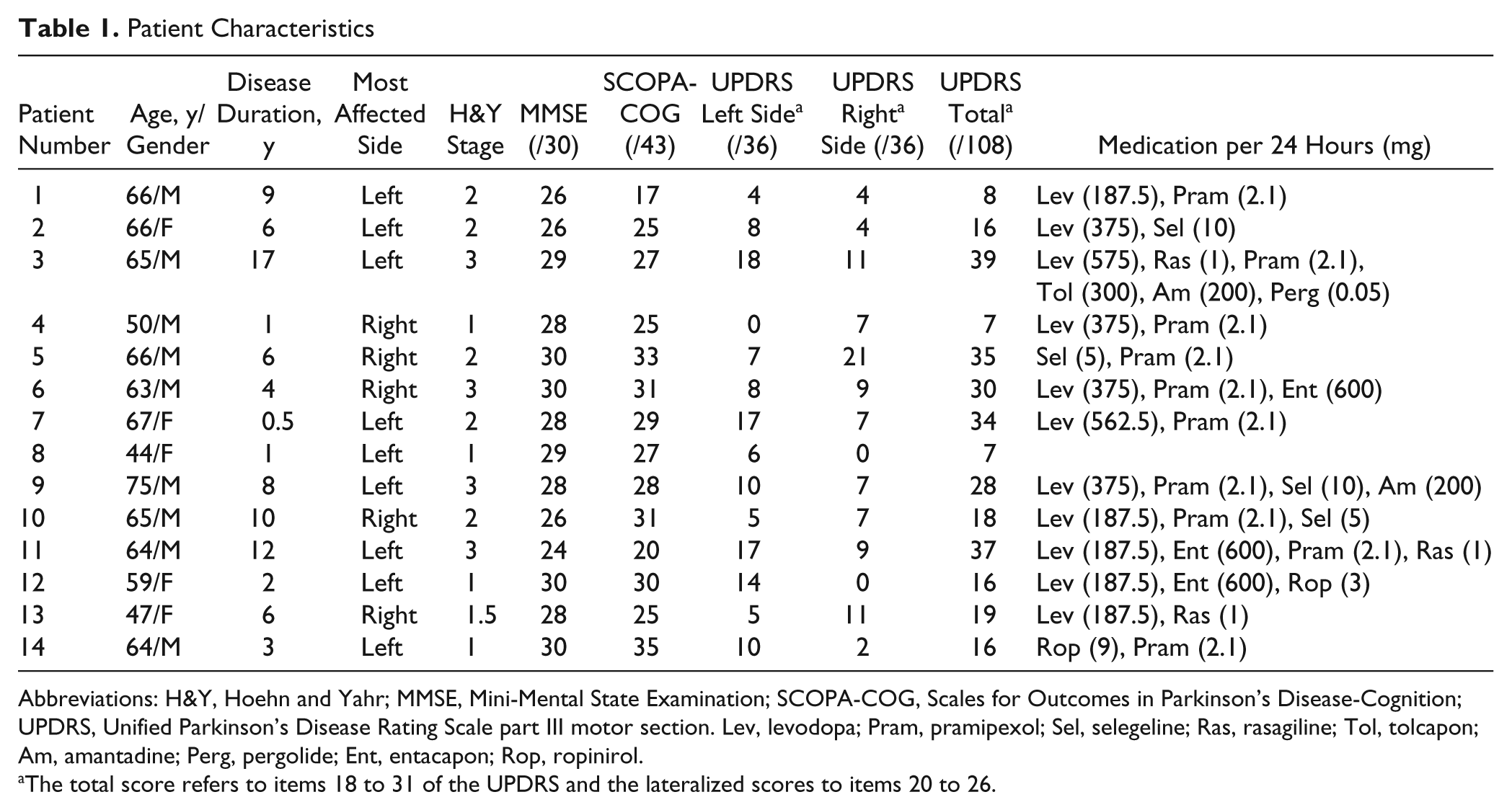
Abbreviations: H&Y, Hoehn and Yahr; MMSE, Mini-Mental State Examination; SCOPA-COG, Scales for Outcomes in Parkinson’s Disease-Cognition; UPDRS, Unified Parkinson’s Disease Rating Scale part III motor section. Lev, levodopa; Pram, pramipexol; Sel, selegeline; Ras, rasagiline; Tol, tolcapon; Am, amantadine; Perg, pergolide; Ent, entacapon; Rop, ropinirol.
The total score refers to items 18 to 31 of the UPDRS and the lateralized scores to items 20 to 26.
Experimental Procedure
All patients were examined in the on phase of their medication cycle. To ensure that there was no medication wearing off during the session, items 23, 24, and 25 of the UPRDS were applied both at the beginning and end of the experimental session, which lasted approximately 2 hours. First, the Edinburgh Handedness Inventory Questionnaire, MMSE, and SCOPA-COG were administered to all participants, followed by the UPDRS part III Motor Section and H&Y staging in the patient group. Subsequently, all participants performed the test battery for imagery ability, consisting of 4 parts: the Motor Imagery Questionnaire–Revised version (MIQ-R), 23 Kinesthetic and Visual Imagery Questionnaire (KVIQ), 25 Chaotic Motor Imagery Assessment Battery (CMIA), 28,29 and an adapted version of the Box and Block Test (BBT). 30 All tests involved both body sides, except the BBT, which was administered only on the most affected side in the patient group and side matched in the control group. Participants were instructed to use a first person perspective during MI.
Motor Imagery Questionnaires
The MIQ-R
23
was used to assess visual and kinesthetic MI ability. The 8 items concerned gross motor function involving trunk, upper-, and lower-limb movements. Participants first executed the movement physically, then imagined doing this movement visually or kinesthetically, and finally scored their imagery vividness on a Visual Analogue Scale (VAS), ranging from 1 (
The KVIQ
25
was developed for assessing imagery ability in populations with restricted mobility, with all movements performed in a sitting position. After a demonstration by the experimenter, the participant was instructed to first physically execute and then imagine doing the movements. We used the long version (KVIQ-20) that comprises 10 visual and 10 kinesthetic items, involving both gross and fine motor control in the trunk and upper and lower limbs. Items were scored on a 5-point VAS, where 1 =
Chaotic Motor Imagery Assessment
The CMIA 28,29 is a test battery consisting of 3 different components offered in a fixed order. This assessment screens for chaotic MI, which can be defined as an inability to perform MI accurately or if having preserved accuracy, demonstrating temporal uncoupling.
Component 1: hand rotation
A total of 96 successive line drawings of a hand (48 left and 48 right) were shown to the participants on a computer screen in 4 different views (back, palm, ulnar, radial) and 12 different rotations (30° steps; Figure 1). 28,31 To present the pictures of the hand and record the response time, we used E-prime 2.0 software and a PST serial response box connected with a microphone that was put in front of the participant. Participants were asked to judge as quickly and accurately as possible whether it was a picture of a left or right hand by saying “left” or “right” in the microphone and this without moving or seeing their own hands, which rested on their laps with the palms down. The response time, measured in milliseconds, was defined as the time between the presentation of the line drawing on the screen and the moment that the answer was given verbally. Note that this procedure is different from the original description of the CMIA, 9 which applied a paper version of the test without measurement of response time. As try-out, 6 practice pictures were given, which were not included in the analysis. This task aimed to provide an objective measure of an individual’s implicit ability to perform MI.
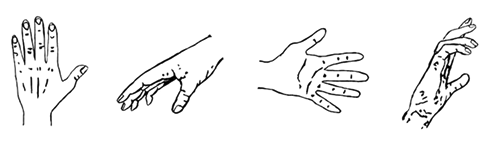
Examples of the line drawings used for component 1 of the Chaotic Motor Imagery Assessment Battery.
Component 2: finger–thumb opposition accuracy
Participants had to perform an auditory paced (1 Hz) sequential finger-to-thumb opposition movement in a fixed order, always starting at digit 2 (index) and ending at digit 5 (little finger). This sequence was repeated until the auditory pacing stopped. At that moment, participants indicated at which finger they had arrived by touching the conforming finger. To monitor compliance, the length of the blocks was varied in a random order. Periods of motor execution were alternated with periods of rest in a block design. Once performance was executed well, the test was repeated using MI. As an additional outcome, originally not included in the CMIA, imagery vividness was measured by means of a 5-point VAS at the end of the test. On this VAS, a score of 1 =
Component 3: finger–thumb opposition speed
Participants performed an auditory-paced finger–thumb opposition sequence (identical to the one described in component 2) of which the rate was gradually increased (with an initial speed of 40 beats/min, increasing by 10 beats/min every 5s). They performed 3 trials of physical execution and 3 trials of MI. Participants indicated when they were no longer able to accurately perform the task, either physically or mentally, by saying “stop” in a microphone, which time point was recorded as the “break point.” Similar to component 1, E-prime 2.0 software and a PST serial response box connected with a microphone were used to record the response time. This test was based on Fitts law (ie, speed/accuracy trade-off) that applies to both MI and physical execution of movements. If their mean break point was greater for MI compared with physical execution, the cognitive strategy did not comply with the principles of motor control, and alternative strategies (such as counting) were assumed to be used instead of correct MI. Participants rated their imagery vividness on a VAS, ranging from 1 (
Box and Block Test
An adapted version of the BBT
30
was performed using wooden blocks of 2.5 cm2 and a wooden box that was divided into 2 equal parts by means of a partition with a height of 18 cm. The box was placed at the participant’s midline, with the compartment holding the blocks oriented toward the hand being tested. Unlike the original test, in which the number of transported blocks was counted within a time limit, the time needed to transport 20 blocks was measured. As such, we used a mental chronometry paradigm, a method that has been shown previously to be reliable for the assessment of MI performance.
32,33
PD patients performed the test with their most affected side, whereas the tested sides of controls were matched to avoid interference of hand dominance. All participants performed 1 practice trial in physical execution and MI conditions. During imagery, the box was removed from the patient’s sight. The test consisted of 3 trials of each condition offered in random order. Additionally, participants scored their imagery vividness on a 7-point VAS similar to that in the MIQ-R (ranging from 1 =
Statistical Analyses
We calculated the mean score on the MIQ-R and KVIQ, subscores for the visual and kinesthetic items of the KVIQ and MIQ-R, accuracy and response time on CMIA-component 1, vividness and accuracy scores on CMIA-component 2, and vividness scores and mean duration on CMIA-component 3 and the BBT. After establishing normality assumptions for data distribution using the Shapiro-Wilk test, a repeated-measures ANOVA with group (PD, controls) as between-subject factor was used for the KVIQ, MIQ-R, hand rotation task, and BBT. As within-subject factor, imagery modality (visual, kinesthetic) was included for the KVIQ and MIQ-R, hand side (pictures of left and right hand) for the hand rotation task, and condition (physical execution, MI) for the BBT. For all significant effects at an α level of .05, post hoc Tukey HSD tests were applied. To study differences between the most- and least-affected body side of the patients, dependent
Results
An overview of the main group comparisons are given in Table 2.
Test Scores (Group Mean and Standard Deviation) per Variable
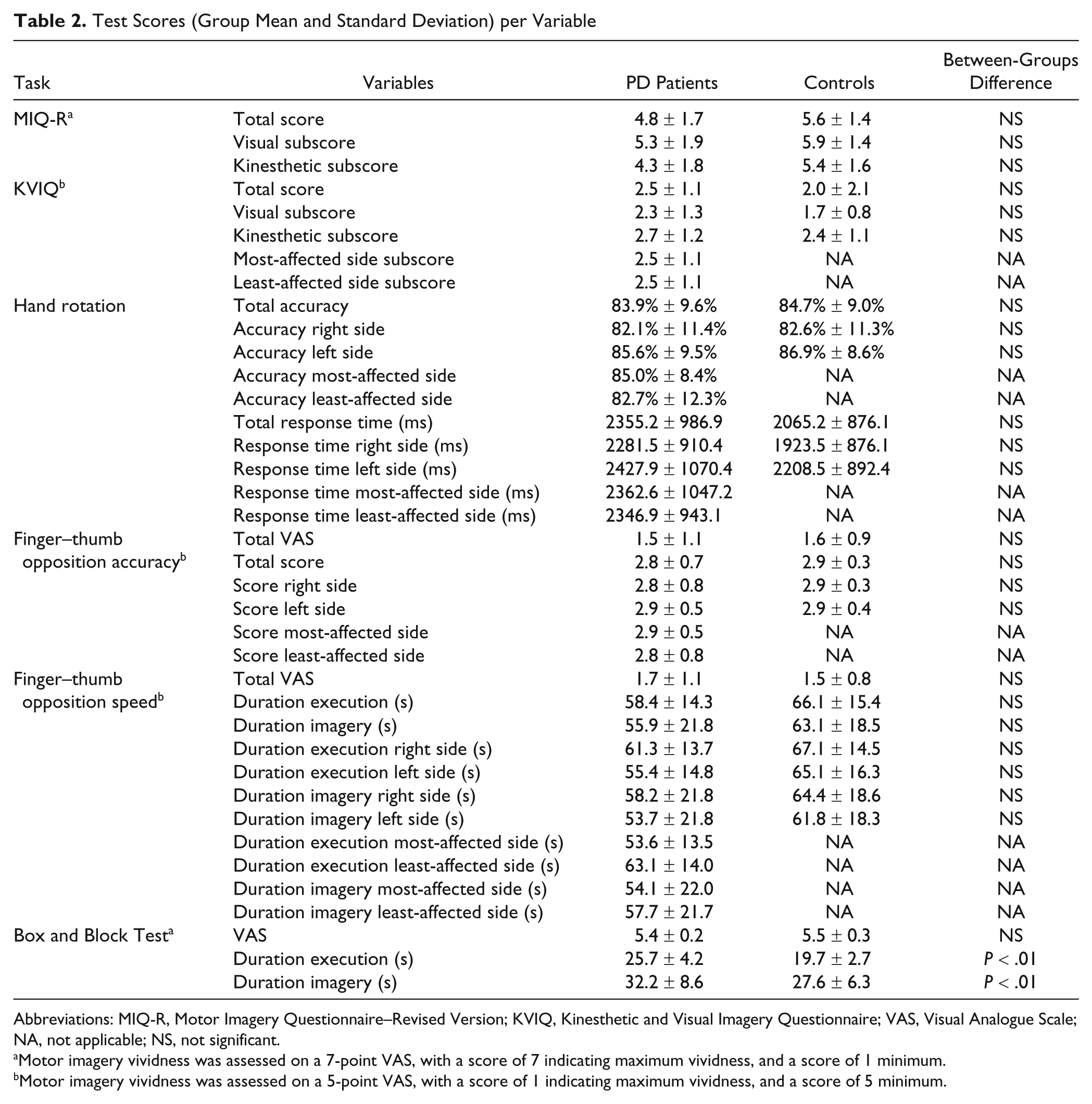
Abbreviations: MIQ-R, Motor Imagery Questionnaire–Revised Version; KVIQ, Kinesthetic and Visual Imagery Questionnaire; VAS, Visual Analogue Scale; NA, not applicable; NS, not significant.
Motor imagery vividness was assessed on a 7-point VAS, with a score of 7 indicating maximum vividness, and a score of 1 minimum.
Motor imagery vividness was assessed on a 5-point VAS, with a score of 1 indicating maximum vividness, and a score of 5 minimum.
Motor Imagery Questionnaires
Both on the MIQ-R
23
and KVIQ,
25
both groups rated their imagery ability as good (Table 2). For the KVIQ, a significant difference was found for modality: [
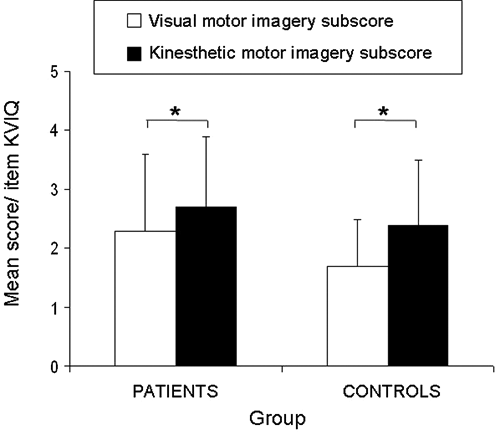
Vividness scores (group mean and standard deviation) on the visual and kinesthetic imagery subscales of the Kinesthetic and Visual Imagery Questionnaire (KVIQ); asterisks indicate significant differences, α < .05.
Chaotic Motor Imagery Assessment 28
Component 1
The patients had a mean accuracy score of 83.9% ± 9.6% and the controls a score of 84.7% ± 9.0%. Both in the PD and in the control group, 3 participants failed to reach the 75% limit that was used by Sharma et al. 28 The ANOVA for Group × Hand side showed no significant differences between groups both for accuracy and response time (Table 2). For hand side, however, a main effect was found for both variables. In both groups, pictures of the left hand were judged more accurately but significantly slower than pictures of the right hand. The within-group analysis in the PD group showed no differences between the patients’ most- and least-affected sides.
Component 2
All patients except one and all controls scored 2/3 or 3/3 for both sides, indicating that they were able to perform the task accurately. This was also confirmed by the high vividness scores on the test (Table 2). Independent
Component 3
On average, both patients and controls could perform physical execution at higher speeds than imagery. All participants except patient 11 reported high vividness scores, without differences between groups.
Box and Block Test
No time data could be collected for patient 11 because he was not able to perform the imagery task. The ANOVA for Group (minus patient 11) × Condition showed main effects for group [
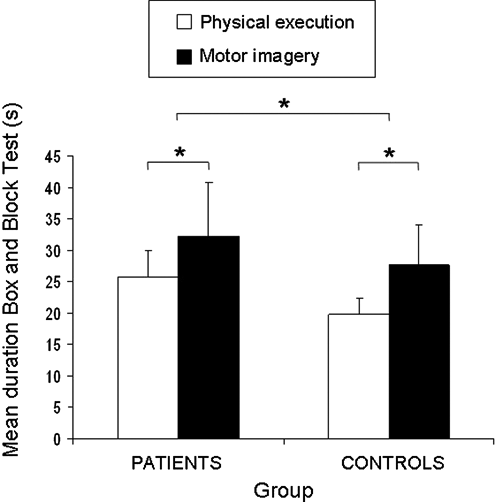
Duration (group mean and standard deviation) of the Box and Block Test per condition; asterisks indicate significant differences, α < .05.
Correlation Between Patient Characteristics and Imagery Tasks
There were no significant correlations between the patients’ disease characteristics and their imagery vividness and accuracy. Although the disease did not affect the patients’ imagery quality, it did, as expected, affect their movement speed, both during imagery and execution. This was reflected in significant correlations between disease duration, H&Y stage, and UPDRS motor score with the following temporal outcomes: (1) mean response time of the hand rotation task (ρ = 0.55 for disease duration, ρ = 0.56 for H&Y), (2) mean duration of physical execution of the BBT (ρ = 0.55 for disease duration (Figure 4A), ρ = 0.58 for H&Y), and (3) mean duration of component 3 (ρ = 0.72 for UPDRS, ρ = −0.61 for H&Y). The duration of physical execution of the BBT correlated in its turn significantly with the duration of imagining this task (ρ = 0.56; Figure 4B), confirming that both conditions were slowed to the same extent in the patient group. Notably, we also found a high correlation between the total score on the MIQ-R and the KVIQ (ρ = −0.90).
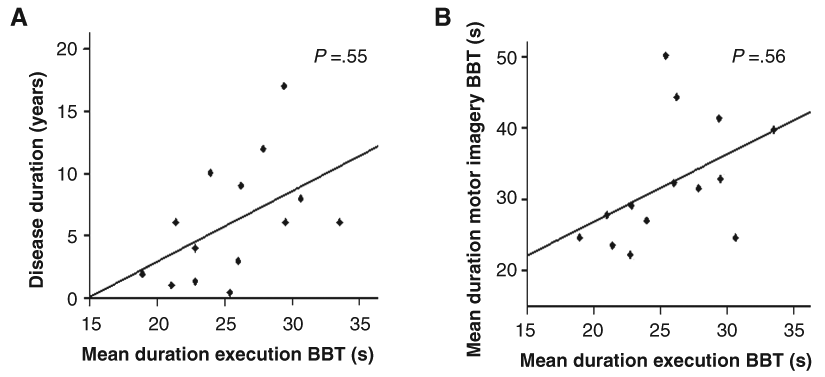
A. Correlation between disease duration and mean duration of physical execution of the Box and Block Test (BBT) for the PD patients. B. Correlation between mean duration of motor imagery and mean duration of physical execution of the BBT for the PD patients.
Discussion
MI therapy is a promising new tool in neurological rehabilitation. Evidence concerning its use for stroke patients is increasing substantially, 8,9 but studies on its application in patients with PD are still sparse and inconclusive. An important reason for this lack of studies is the uncertainty whether patients with PD are still able to use MI in an accurate way. Therefore, in the present study, we assessed the MI ability of 14 PD patients by means of an extensive test battery.
The results of the CMIA, which can be seen as an objective screening tool for MI ability, showed no differences between patients and controls, indicating that imagery accuracy is well preserved in the patient group and that it concurs with general motor control principles. Furthermore, the results of the questionnaires showed that the PD patients had preserved imagery vividness. These results were confirmed by a lack of significant correlations between disease parameters and outcomes of imagery accuracy and vividness. Similar to controls, patients reported more vivid sensations during visual than during kinesthetic MI. No differences were found between body sides, although all patients had asymmetrical PD.
Our results are encouraging with respect to the integration of MI therapy in rehabilitation. It was shown that cognitive control techniques help PD patients compensate for some of their motor problems. 2 MI might be advocated as an attentional strategy to prepare for upcoming functional movement sequences such as passing through a narrow door or walking between obstacles. The priming of the motor program beforehand may reduce the attentional load during actual motor performance. MI practice might also be used to adjust for motor impairments, such as decreased movement amplitude or speed. For example, a patient could imagine the rhythm of a metronome while walking or a floor marker to step over. This is supported by a recent intervention study of Tamir et al, 6 showing that a combination of MI and real practice may be effective in the treatment of PD, especially for reducing bradykinesia. An important advantage of MI therapy is that it can be used to practice potentially dangerous movements in a safe way, even without the need of a therapist. This makes it an extremely suitable technique for home-based exercises, which might, in combination with regular follow-up treatments, be an excellent way to maintain gains or prevent the progressive decline in motor performance. 34
Whereas no differences between PD patients and controls were found in imagery vividness and accuracy, the tests revealed a significant temporal difference in imagery between both groups. Significant correlations were found between disease parameters such as disease duration and H&Y stage and response time on the hand rotation task. Furthermore, PD patients were shown to be markedly slower on the imagery condition of the BBT than controls. This slowness of MI is in accordance with previous studies 20,21 and might reflect a temporal problem with MI. However, the fact that their physical execution was slowed to the same extent indicates that the slowness of MI rather reflects the bradykinesia inherent to PD than an inability to perform MI in a correct way. This hypothesis fits well within the motor simulation theory of Jeannerod, 35 which states that simulated actions are by nature similar to actual actions, with the exception that they are not overtly executed. Several previous studies have shown that the time taken to mentally represent a given movement closely mimics the duration of the same movement when it is physically executed. 33,22,36 MI also causes similar autonomic changes (eg, changes in heart rate and respiratory frequency) as physically executed movements 37 and seems to be constrained by the same motor laws that apply to physical execution of the movement, such as Fitts law. 38,39 Other striking parallels are the common neural substrate of executed and imagined movements and the fact that learning a motor task through MI practice produces cerebral functional changes similar to those after physical practice of the same task. 40,41
Remarkably, one of the patients in the present study was unable to imagine any body movement. Both motor and cognitive impairment were more severe in this patient than in most other patients. Although the correlation analysis did not confirm this at the group level, it is possible that his lack of imagery ability is related to his disease progression and its effect on cognitive functioning. 42,43 But in healthy persons also, there is a spectrum from very good to very bad imagers, with occasionally people reporting to be completely unable to imagine movements. Especially in elderly people, a decreased ability to imagine complex movements has been reported. 44,45 This might explain why in both groups, 3 out of 14 participants did not reach the postulated 75% score in the hand rotation task. However, alternative explanations for the divergent performance of these participants specifically on this task can be a problem with left–right distinction or the use of a different type of mental spatial transformation. 46 Although lack of imagery skills in one patient does not necessarily imply that PD pathophysiology impairs MI, it reaffirms the fact that a thorough evaluation of a person’s imagery ability at onset of MI practice is required. Furthermore, we should be aware that our findings can only be generalized to patients in the early- and mid-stages of the disease (H&Y 1-3) without additional neurological conditions or severe cognitive impairment. It remains to be investigated whether patients in later stages of the disease or those suffering from severe cognitive decline are as proficient at performing imagery as the present cohort. In addition, in the present study, patients were only assessed in the on phase of their medication cycle. As such, effects of the disease on imagery performance may have been masked by the medication. However, levodopa therapy only partially restores patterns of cortical activity associated with motor execution and motor preparation. 17,18
When investigating MI, one should keep in mind that both because of its covert nature and of the different aspects that this term embraces, we should be cautious about drawing firm conclusions about a person’s imagery ability. Previous studies in which MI ability was taken into account often only relied on questionnaires for its assessment. Although questionnaires contain very interesting information on participants’ imagery vividness, the received information remains subjective. In this respect, the evaluation of MI by means of mental chronometry can be considered as more objective. However, some have validly argued that the close temporal correspondence between MI and execution may be attributable to a participant’s tacit knowledge about the time it takes to actually execute the movement. 47 Evaluation methods using accuracy scores to evaluate MI performance are less subject to these shortcomings but have the limitation that we cannot exclude with certainty whether participants used alternative strategies, such as counting. A further shortcoming of most methods is that no fixed cutoff score is defined to determine if participants have sufficient MI ability to take part in MI therapy. Because each test evaluates a specific aspect or type of MI, we do not recommend excluding patients from MI therapy based on a deviating score on only one test. Therefore, in the present study, an extensive test battery, consisting of a combination of several distinct evaluation methods, is proposed to evaluate MI ability in detail. 48 This test battery requires only a limited amount of equipment and assessment time and provides the therapist with a broad picture of the participant’s imagery ability because several types of MI (eg, visual and kinesthetic MI), several types of movements (eg, gross and fine motor movements), and several MI aspects (eg, accuracy, temporal structure, and vividness) are addressed. Furthermore, the present test battery is mainly composed of objective screening instruments. Mental chronometry, as was used in the BBT, was previously shown to be a reliable method to assess the temporal structure of MI. 33 The CMIA is, in our opinion, the only existing test battery that evaluates in an objective way whether the participant’s imagery is accurate and agrees with principles of motor control. As such, the CMIA and BBT should be seen as complementary tests. Whereas the first one assesses MI accuracy and conformance to general motor laws such as Fitts law, the second one focuses on its temporal aspects. Another interesting distinction is that the BBT requires the participants to interact (or imagine interacting) with objects, whereas the CMIA does not. There is an extensive amount of literature showing differences in neural activation when actions are object related versus when they are not, and depending on the goal to be achieved. 49,50 Furthermore, we previously showed that MI is positively affected by external movement-related stimuli. 51 Therefore, we hypothesize that the presence of objects might be relevant when determining the ability of participants to perform MI within a rehabilitation context. However, this should be further investigated in future studies. To complete the test battery, the KVIQ and MIQ-R were added to also evaluate participants’ imagery vividness. Both questionnaires were shown to be highly correlated. We recommend the use of the KVIQ over the MIQ-R because it discriminates better between visual and kinesthetic imagery and is also appropriate for patients with movement limitations. However, because its administration is rather time-consuming (30-45 minutes), the MIQ-R can be considered if assessment time is very limited.
In the present study, we found that visual imagery is considered to be easier than kinesthetic imagery in both PD patients and controls. This observation is in accordance with previous studies in healthy participants 52 and neurological patients. 25 This suggests that when engaging individuals in imagery practice, it may be easier to first learn to use visual images and later incorporate kinesthetic images. The choice of which imagery modality to use might also strongly depend on the task to be learned. Féry 53 suggested that visual imagery is better for tasks that emphasize form, whereas kinesthetic imagery is better for tasks that emphasize timing or coordination of the 2 hands.
In conclusion, the present study showed that most patients with mid- and early-stage PD have preserved MI vividness and accuracy as measured by a clinical test battery covering different aspects of MI. Bradykinesia, which was present during physical execution, also occurred during imagery. We assume that this indicates an adaptation of imagery to the actual motor functioning of the patient rather than an inability to assemble an accurate mental representation of a movement. The results of the present study are promising regarding the implementation of MI training in the rehabilitation of patients with PD.
Footnotes
Acknowledgements
We thank all participants for their time and patience and Ir. M. Beirinckx and Ir. P. Meugens for their technical assistance.
The author(s) declared no potential conflicts of interest with respect to the authorship and/or publication of this article.
The author(s) disclosed receipt of the following financial support for the research and/or authorship of this article: Elke Heremans is a Research Assistant and Wim Vandenberghe a Senior Clinical Investigator at the Research Foundation – Flanders (FWO).
