Abstract
Background. Although inpatient rehabilitation may enhance an individual’s functional ability after stroke, it is not known whether these improvements are accompanied by an increase in daily use of the arms and legs. Objectives. To determine the change in daily use of the upper and lower extremities of stroke patients during rehabilitation and to compare these values with that of community-dwelling older adults. Methods. A total of 60 stroke patients underwent functional assessments and also wore 3 accelerometers for 3 consecutive weekdays on admission to rehabilitation and 3 weeks later prior to hospital discharge. The number of steps and upper-extremity activity counts were measured over the waking hours and during daily use for occupational therapy (OT) and physical therapy (PT) sessions. Healthy older adults (n = 40) also wore 3 accelerometers for 5 consecutive days. Results. Stroke patients demonstrated a significant increase in mobility function, and this was accompanied by an increase in daily walking over the entire day as well as in PT. However, increases in daily walking were found predominantly in patients who were wheelchair users (and not walkers) at the time of admission. Control walking values (5202 steps) were more than 17 times that of stroke patients. Despite significant improvements in paretic hand function, no increase in daily use of the paretic or nonparetic hand was found over the entire day or in PT. Conclusions. A disparity between functional recovery and increases in daily use of the upper and lower extremities was found during inpatient stroke rehabilitation.
Introduction
The most common symptom poststroke is hemiparesis. 1 Physical therapy (PT) and occupational therapy (OT) focus on reducing the motor impairments early after stroke while enhancing the individual’s functional ability, but few studies have objectively quantified the upper and lower extremity activities performed by individuals who are undergoing rehabilitation.
Hundreds of repetitions of movement are required for learning and cerebral plasticity to occur.2,3 Observational analysis of typical stroke inpatient activities has provided useful information on the content of activities within therapy as well as during nonstructured time through the rest of the day. Lang et al
4
observed 312 inpatient and outpatient therapy sessions of PT or OT and reported on average 32 repetitions of upper-extremity movement and 367 lower-extremity steps per therapeutic session. Bernhardt et al
5
observed 58 acute stroke patients between 8
Thus, the data from observational analyses suggest that the number of repetitions, as well as the amount of physical activity (PA), is low during inpatient stroke rehabilitation. Instrumented monitoring of PA can provide continuous recording for a single session or even over several days to provide a more accurate quantification of activity. For example, Smith et al 8 used a positional activity logger and found that inpatients with stroke spent a median of 1.3 h/d standing upright, which was substantially less than that of the community group (5.5 hours). Accelerometers are a valid and reliable tool to objectively measure the amount of daily PA of individuals with stroke. 9 This technique can provide a continuous measure of daily use of the lower 9 and upper extremities. 10 In addition, long recordings of accelerometer activity can provide a picture of daily use of the upper and lower extremities during therapy as well as nonstructured time.
The first purpose of our study was to determine the for any change in daily use of the upper and lower extremities of patients with stroke during inpatient rehabilitation. The second aim was to compare the amount of daily use of the upper and lower extremities of these patients prior to returning home and compare this with the activity levels of older community-dwelling adults.
Methods
This study was approved by the local university and hospitals ethics board, and all participants provided informed consent.
Population
Participants were consecutive adults admitted to 2 inpatient rehabilitation centers within 60 days of sustaining a stroke as confirmed by CT or MRI. Patients were excluded if they were younger than 19 years old, had a significant musculoskeletal condition within the past 6 months, had a neurological condition other than stroke, or if they had communication or cognitive difficulties that prevented them from providing informed consent. When the patient could not speak English, we used a translator to assist with the assessment. In addition, a control sample of community-dwelling, healthy older adults was recruited through advertisements posted in community and shopping centers and via snowballing techniques. Inclusion criteria of the control group were right-handed as assessed by the Edinburgh Handedness Inventory, 11 full use of the upper extremities, and independent in activities of daily living. We selected right-handed individuals because up to 90% of the general population is right-handed. Exclusion criteria were a neurological condition or a significant musculoskeletal condition.
Instruments
Accelerometers (Actical, Mini Mitter Co, Bend, Oregon) were used to quantify daily upper and lower-extremity use—one on each wrist to measure upper-extremity movement and one accelerometer on the hip to measure lower-extremity movements and steps walked. The accelerometer is small (28 × 27 × 10 mm3), light (17 g), waterproof, and sensitive to 0.05 to 2.0 g-force. It has a frequency range of 0.3 to 3 Hz, samples at 32 Hz, and then rectifies and integrates acceleration over 15-s epochs as activity counts. The Actical accelerometer detects acceleration in all 3 planes but is more sensitive in the vertical direction. When worn on the hip, it has a valid step count function. 12 The intrainstrument and interinstrument reliability of the Actical accelerometer was found to be superior to the Actigraph and RT3 accelerometers. 13 When worn on the hip, the Actical accelerometer has been found to be valid and reliable for detecting free-living PA in individuals with chronic stroke. 9 When worn on the wrist, the daily Actical accelerometer activity counts have been shown to correlate with hand dexterity (measured by the Box and Block Test) in older adults. 10 In addition, daily Actical wrist activity counts have been found to have moderate correlations with brain activation during a grip task in stroke patients. 14 Finally, studies using other types of wrist accelerometers have reported moderate to strong correlations between accelerometer readings and self-report measures of arm activity (r = 0.93) 15 or upper-extremity function (r = 0.40-0.91).16,17
A number of assessments were collected at baseline to provide a description of the participants. General cognitive ability (Mini-Mental State Examination) 18 was assessed and a screening test for unilateral visual neglect (BIT Star Cancellation Test) 19 was administered (neglect was defined if less than 45/54 stars were identified 20 ). Depressive symptoms were obtained using the Center for Epidemiologic Studies Depression Scale, 21 which is a reliable and valid screening tool for assessing depressive symptoms in individuals with stroke. 22
Clinical assessments were used to determine the level of motor and functional ability of the upper and lower extremities and were undertaken at both assessment 1 (on admission) and assessment 2 (3 weeks later). The Fugl-Meyer Motor assessment 23 (upper-extremity scale) was used to assess the motor impairment of the paretic upper extremity, and its validity and reliability have been well established in stroke.23,24 Scores range from 0 points (no active movement) to 60 points (full active movement). The Action Research Arm Test 25 is a reliable and valid measure for function of the paretic upper extremity of individuals with stroke 26 and measures the quality of movements such as grip, grasp, pinch tasks, and gross movements. Scores range from 0 points (a nonfunctional hand) to 57 points (a fully functional hand). The Berg Balance Scale 27 was administered to assess the ability to maintain balance while performing 14 functional tasks and is a reliable and valid measure of balance in stroke. 28 Scores range from 0 points (poor balance) to 56 points (excellent balance ability). Mobility was determined by using 2 walking tests: 10-Meter Walk Test 29 (gait speed in m/s over 10 m) and the Six-Minute Walk Test (6MWT, distance walked in meters over 6 minutes). 30 These measures are valid and reliable for use with individuals with stroke. 31 In addition, the functional ability of the patients performing basic activities of daily living was assessed using the Functional Independence Measure (FIM), 32 which has been found to be reliable and valid when used with individuals with stroke.
Procedure
Using triaxial accelerometers, the total amount of daily use of the upper and lower extremities was measured in addition to the amount of daily use performed during therapeutic OT and PT sessions. Measurements were undertaken over 3 days on admission to rehabilitation (assessment 1) and, 3 weeks later, prior to discharge (assessment 2). Eligible stroke patients were approached by a site coordinator during the first week of their rehabilitation. Starting on the following Monday, patients were given 3 accelerometers to wear for 3 days. They were allowed to remove the accelerometers during the night. Once removed, the nursing staff assisted the patients in putting on the accelerometers in the morning, and the site coordinator visited the patients each day to ensure that the accelerometers were worn correctly. During this 3-day period, the patient’s therapists filled in a log regarding the start and end time of each PT and OT session. The accelerometers were removed on the following Friday, and accelerometer readings were downloaded from 3 full days (Tuesday, Wednesday, and Thursday). The clinical assessments were then undertaken, and 3 weeks later, this procedure was repeated (see Figure 1).
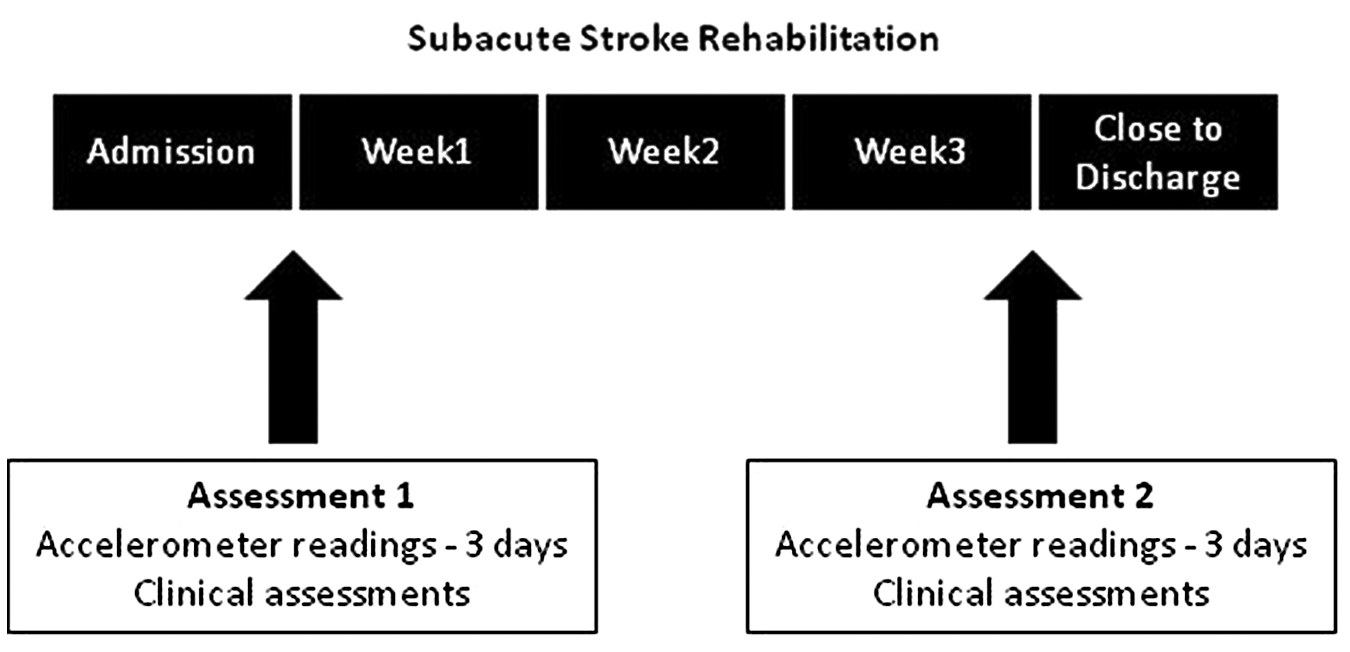
Data collection of the inpatient sample during stroke rehabilitation.
For both the upper and lower-extremity activity, we calculated the mean daily use (activity counts for the upper extremity and steps for the lower extremity divided by the 3 days) for (1) an entire day, (2) a PT session, (3) an OT session, and (4) daily use not including the OT/PT sessions (total daily use minus the OT/PT sessions). For the upper extremities, we eliminated the activity counts of arm swing while walking (defined as 5 consecutive steps or more in 1 epoch), so that our measure reflected goal-directed hand use.
Controls wore 3 accelerometers for 5 consecutive days. The mean daily total number of steps/d was calculated in addition to the right and left upper-extremity activity counts. The activity counts were adjusted for the arm swing while walking in the same manner.
Data Analysis
Descriptive statistics of the demographic data in addition to the accelerometer readings were calculated. Paired t tests were used to assess changes in the clinical measures between assessments 1 and 2. Because patients unable to ambulate on admission may have different recovery, we divided the patients into 2 groups based on their admission walking ability (walkers and wheelchair users). Walkers were patients who were able to walk independently around the ward at admission. t Tests were used to assess the differences between the walkers and the wheelchair users for the clinical assessments. Because the distribution for the accelerometer readings was not normal, nonparametric statistics were used; the median and interquartile range were calculated for daily use, and Wilcoxon tests were used to assess changes in daily accelerometer use between assessments 1 and 2. This statistical procedure was applied to assess (1) steps within therapy, (2) steps outside of therapy, (3) upper-extremity activity counts within therapy, and (4) upper-extremity activity counts outside of therapy. Wilcoxon tests were also used to assess changes in daily use between assessments 1 and 2 for the subgroups (walkers and wheelchair users). SPSS (version 17.0; SPSS Inc, Chicago, Illinois) software was used to analyze the data.
Results
A total of 125 patients were admitted to 2 rehabilitation centers as a result of stroke from February 2008 to April 2009. Of these, 19 patients declined to participate in the study (mean age, 73.4 ± 9.0 years; 62% males); 68 adults were eligible and agreed to participate (mean age, 61.6 ± 13.3 years; 65% males); however, 4 patients (mean age, 73.2 ± 11.7 years; 1 man and 3 women) dropped out of the study before completing assessment 2, and 4 patients (mean age, 72.2 ± 4 years) were excluded because they did not have full accelerometer data (see Figure 2 for CONSORT flow diagram and Table 1 for patient description). In addition, 40 healthy older adults (20 men and 20 women) were recruited to participate in the study (mean age, 71.3 ± 3.8 years).
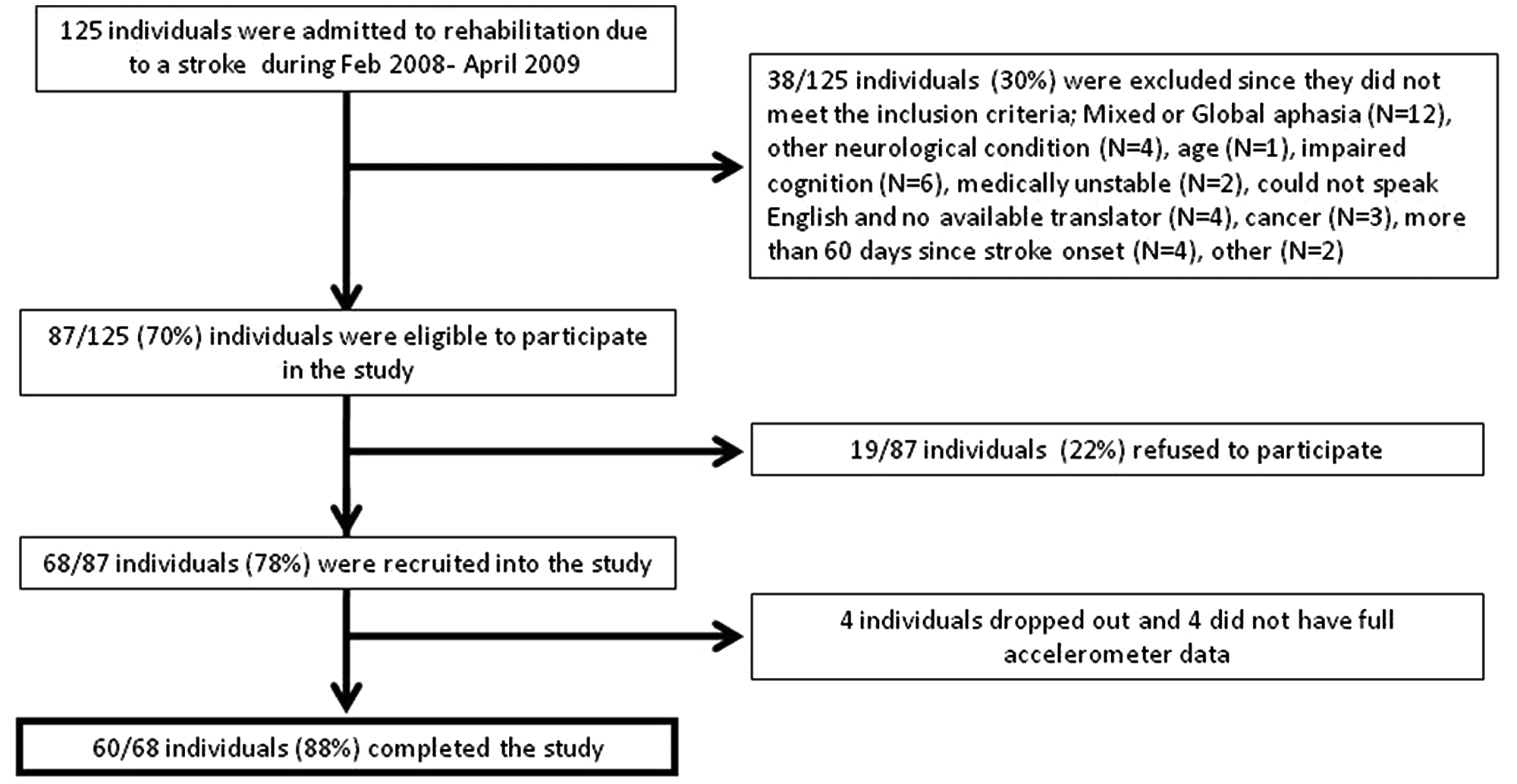
CONSORT flow diagram of recruitment to the study.
Demographic, Stroke, and Clinical Information of the Total Study Sample and the Sample Divided Into Walkers (n = 27) and Wheelchair Users (n = 33) on Admission
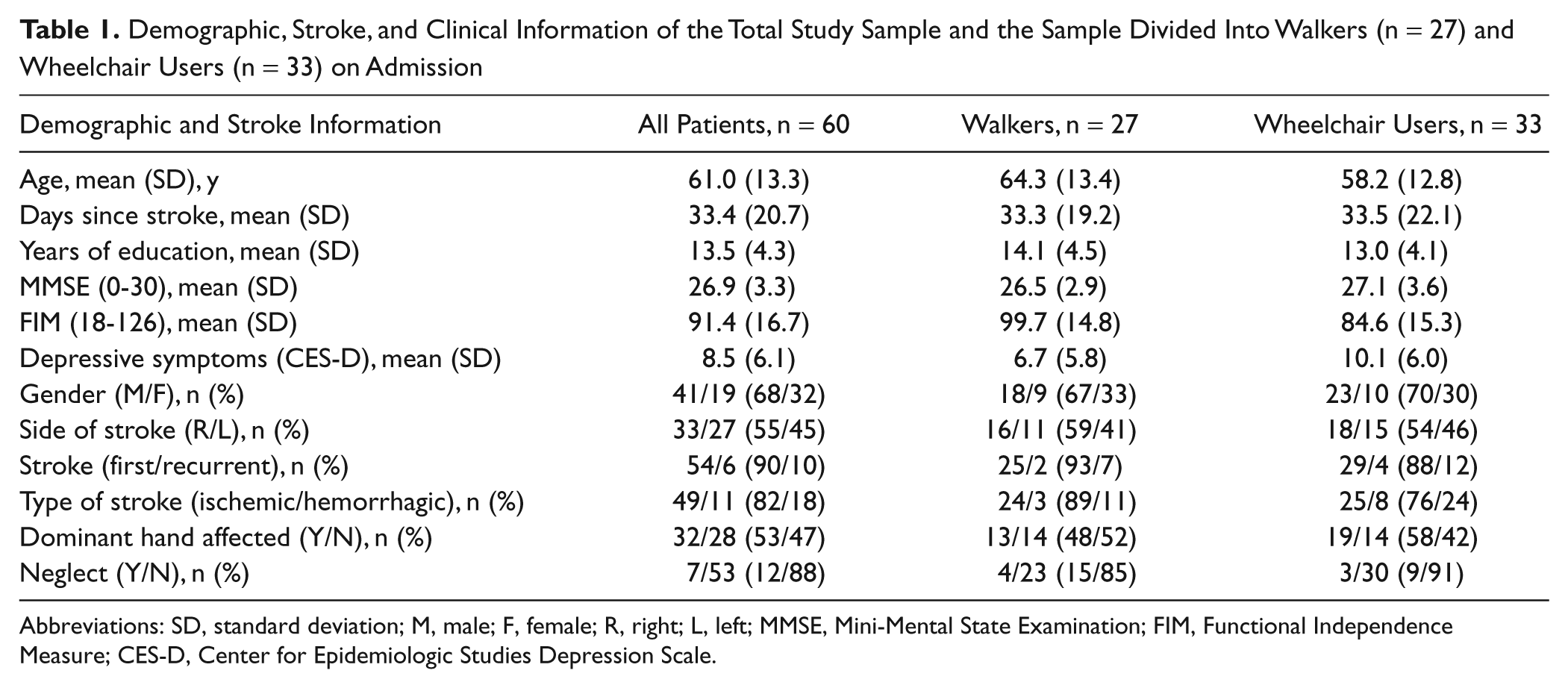
Abbreviations: SD, standard deviation; M, male; F, female; R, right; L, left; MMSE, Mini-Mental State Examination; FIM, Functional Independence Measure; CES-D, Center for Epidemiologic Studies Depression Scale.
Of the 60 patients with stroke, 27 were walking independently around the ward on assessment 1 (walkers), whereas 33 patients were using a wheelchair (wheelchair users). On assessment 2, 39% of the wheelchair users on assessment 1 had progressed to walking independently around the ward. On assessment 1, the scores for all the assessments of the walkers were significantly higher compared with those for wheelchair users (Table 2). The walkers were significantly better functioning in basic activities of daily living (assessed by the FIM)—t(59) = 3.8, P < .001—had significantly better hand function (assessed by the ARAT [Action Research Arm Test])—t(59) = 3.5, P = .001—and walking ability (assessed by the 6MWT)—t(59) = 2.8, P = .007. The walkers also reported less depressive symptoms compared with the wheelchair users (see Table 1)—t(59) = −2.0, P = .04. However, there were no significant differences between the groups for age, days since stroke, number of years of education, and function of the nonparetic upper extremity (assessed by the Box and Blocks Test).
Clinical Assessments Administered on Assessments 1 and 2 for the Total Study Sample and the Walkers and Wheelchair Users
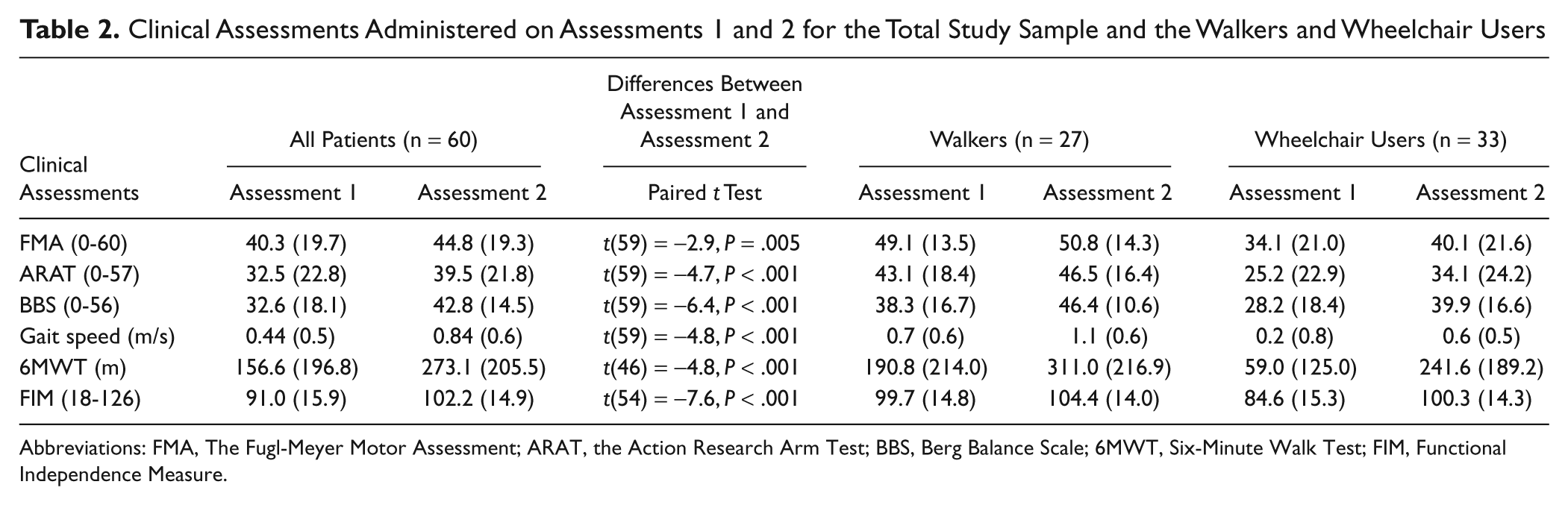
Abbreviations: FMA, The Fugl-Meyer Motor Assessment; ARAT, the Action Research Arm Test; BBS, Berg Balance Scale; 6MWT, Six-Minute Walk Test; FIM, Functional Independence Measure.
As expected, the motor and functional abilities of the upper and lower extremities of the 60 stroke patients improved significantly over the 3-week period from assessment 1 to assessment 2 (see Table 2). When each group (walkers and wheelchair users) was assessed individually, the FMA score of the paretic hand of the walkers was the only measure that did not show significant improvement from assessments 1 to 2.
In general, a significant increase in the median values of the steps/d was seen (Table 3); however, these values were very low. For example, the 60 stroke patients walked a median of 176 steps on assessment 1 over an entire day and increased their steps significantly by 72% to a median of 302 steps/d on assessment 2 (Table 3); z = −2.2, P = .02. Control values (median of 5202 steps) were statistically greater (17 times more) than the values of that of stroke patients close to discharge; z = −6.6, P < .001. Walking during therapeutic sessions (combined OT and PT steps) accounted for 12% and 26% of the steps recorded over the entire day on assessment 1 and assessment 2, respectively.
Median (IQR) Number of Steps Walked by Healthy Older Adults, the Total Sample of Stroke Patients (n = 60), and the Stroke Subgroups for the Walkers (n = 27) and the Wheelchair Users (n = 33) on Assessment 1 and Assessment 2
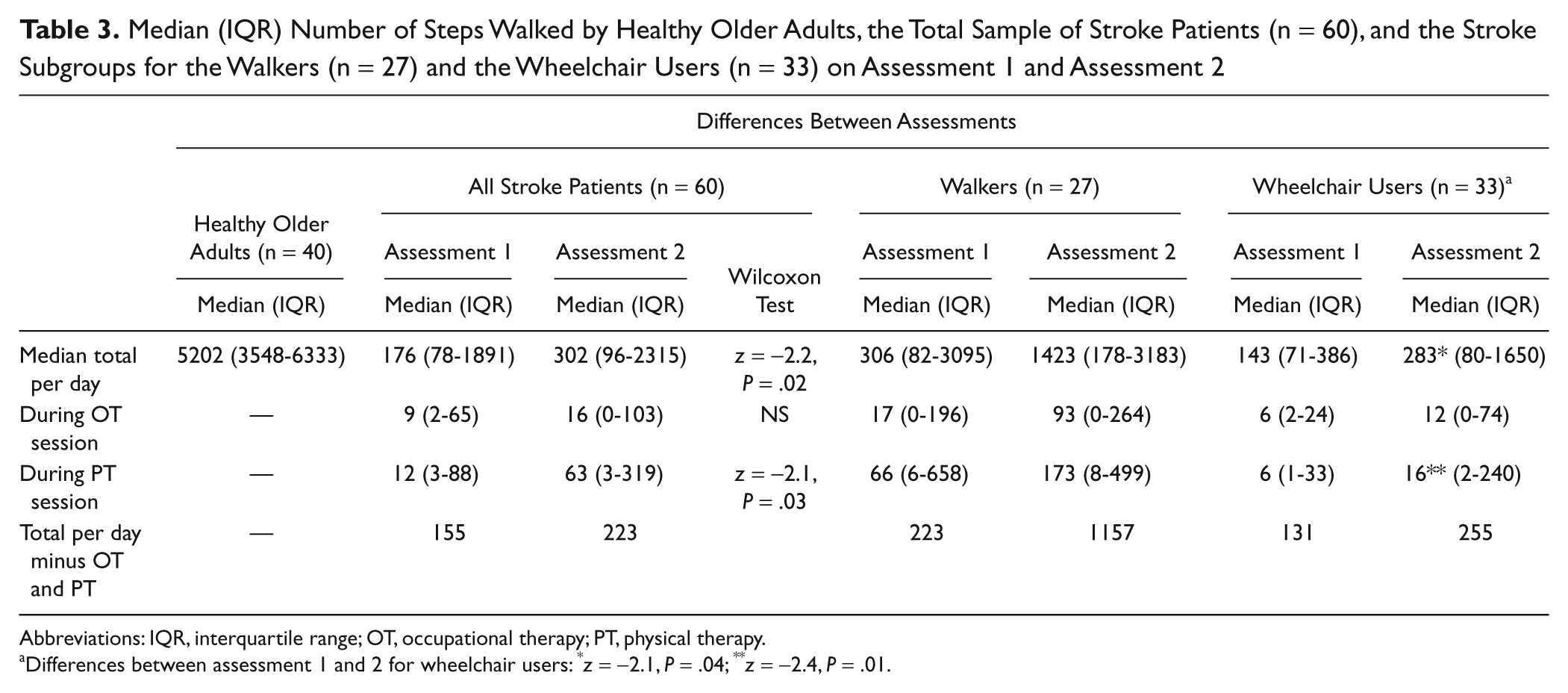
Abbreviations: IQR, interquartile range; OT, occupational therapy; PT, physical therapy.
Differences between assessment 1 and 2 for wheelchair users: *z = −2.1, P = .04; **z = −2.4, P = .01.
The median number of steps walked by all the patients during PT increased significantly from 12 to 63 steps per session over the 3-week period (z = −2.1; P = .03); however, this likely constituted only a few additional minutes of walking over the session. The steps taken during the OT sessions did not differ significantly from assessment 1 (median of 9 steps) to assessment 2 (16 steps). A significant increase from a median of 143 to 283 steps/d by the wheelchair users was found (z = −2.1; P = .04). The walkers on the other hand were not found to significantly increase the number of steps walked per day or during therapy sessions.
The daily use of the paretic upper extremity on assessment 2 was very similar to the daily use measured on assessment 1 (Table 4). The right and left upper-extremity values of the control group (all right-hand dominant) were similar, with only a 14% difference between sides. On assessment 2, the daily use of the paretic upper extremity was significantly lower than the daily use of both the right (z = −6.8; P < .001) and the left hands (z = −5.9; P < .001) of healthy controls, but the daily use of the nonparetic upper extremity was not significantly different from the daily use of either hand of the healthy controls.
Median (IQR) Daily Use (Activity Counts) of the Paretic and Nonparetic Upper Extremity (Assessment 1 and Assessment 2) of the Stroke Patients Compared With the Right and Left Hands of Healthy Controls
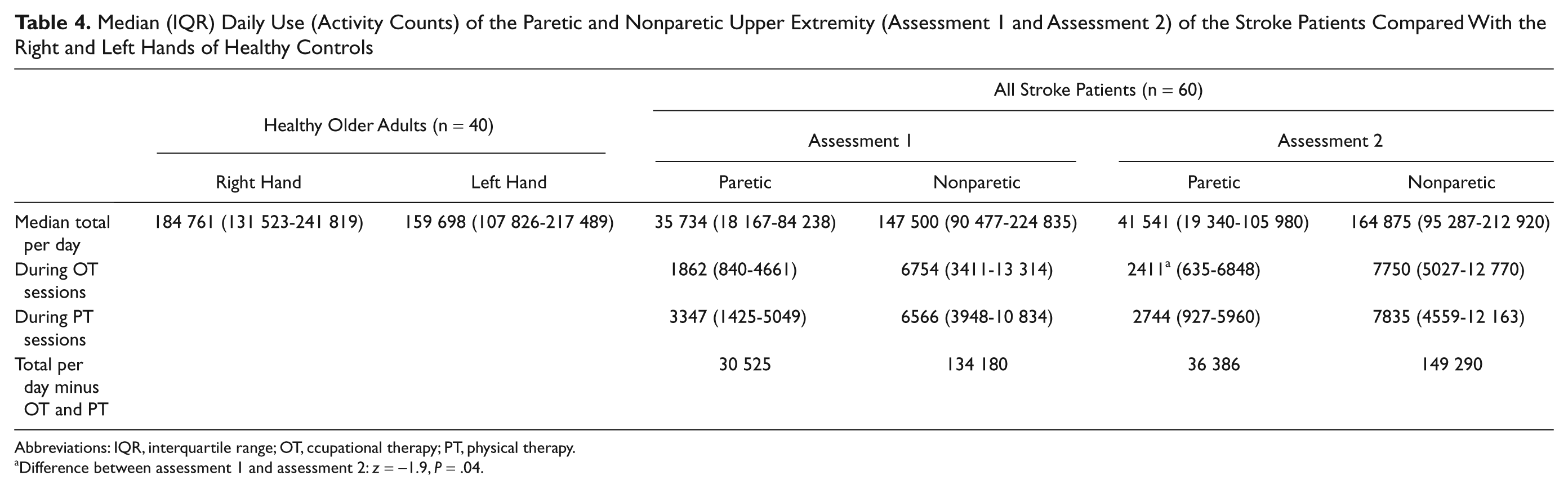
Abbreviations: IQR, interquartile range; OT, ccupational therapy; PT, physical therapy.
Difference between assessment 1 and assessment 2: z = −1.9, P = .04.
Upper-extremity daily use during therapeutic sessions (PT and OT combined) accounted for 14% and 12% of the use recorded over the entire day on assessment 1 and assessment 2, respectively (Table 4). Daily use of the paretic upper extremity in PT sessions decreased by 18% from assessment 1 to assessment 2, but this was not statistically different. There was a significant increase over the 3 weeks for paretic upper extremity daily use during OT from a median of 1862 to 2411 activity counts (z = −1.9; P = .04).
The daily use of the nonparetic upper extremity was about 4 times more than the daily use of the paretic upper extremity at both assessments 1 and 2. The paretic upper-extremity activity counts were one third to one half of the values for the nonparetic upper extremity during the therapy sessions.
Discussion
Our findings partially supported the hypothesis that daily use of the lower extremity increases over inpatient stroke rehabilitation. However, our results do not support the hypothesis that daily use of the upper extremity increases over inpatient stroke rehabilitation.
Patients who could already walk on admission (albeit slowly at 0.7 m/s) did not increase the daily practice of their walking over the 3 weeks during PT, OT, or during the rest of the day. During the PT sessions, all 60 patients walked a median of 63 steps per session near discharge, whereas the walker subgroup walked a median of 173 steps. This number of steps would constitute only a few minutes of daily ambulatory exercise, which seems insufficient for optimizing gait recovery or to obtain a cardiovascular training effect.
Our findings supported the second hypothesis that the amount of daily use of upper and lower extremities of the older community-dwelling adults was substantially higher compared with daily use of the stroke patients at discharge to the community. The median steps/d (5202) of our healthy controls fell within the expected range from population-based studies of older adults. 33 Most investigators consider the original recommendation of 10 000 steps/d to be unrealistically high, especially for older adults.34,35 Several investigators have suggested that older adults in their 70s should strive for 7000 to 8000 steps/d for health benefits,34-36 and Tudor-Locke et al 37 classified those walking fewer than 5000 steps/d as sedentary. Nevertheless, our patients with stroke are far below the number of steps/d that are likely required for health benefits and for coping with the everyday needs of independent community living.
Although the nonparetic upper-extremity activity counts (164 875) approached values for the right and left hands of healthy older adults (184 761 and 159 698 activity counts), the paretic upper-extremity counts were only 25% of those of the nonparetic side. Because individuals undergoing rehabilitation are encouraged to be independent in daily activities, it is evident that they are mainly using their nonparetic hand to do so. In an attempt to explain the limited hand use post hoc, we examined whether having a dominant versus nondominant hand affected following stroke influenced paretic hand daily use, but no significant effects were found.
The discrepancy between improvements in function and increases in PA participation can be framed within the International Classification of Functioning model. 38 The gap between the recovery of capacity (in our case, clinical measures of impairment and function) and the very limited improvement in performance (in our case, daily use of the upper and lower extremities) provides a useful guide as to what can be done to improve performance.
Outside of the PT/OT therapy sessions, the institutional setting may not be conducive to patients undertaking PA or exercise outside of their regular therapeutic sessions. 39 Space or exercise equipment may not be available and may restrict opportunities for walking or upper-extremity exercise. In addition, individuals may not want to undertake exercise because of fatigue, low self-efficacy, or learned nonuse of the upper extremity.40,41 In some cases, patients may not be able to undertake PA safely without supervision because of impaired cognition, impaired balance, or impulsive behavior. Furthermore, patients and their families may lack awareness of the activities they could be participating in outside of therapy sessions.
Attempts have been made to increase the amount of PA outside of formal therapy time by using pedometers, 42 through implementation of a home-work-based exercise program, 43 and by using inexpensive interactive video games such as the Nintendo Wii system. 44
Limitations
Although the Actical accelerometer is superior to most pedometers, which do not record any steps when individuals walk at slower speeds, this accelerometer underestimates steps at slower speeds (7% at walking speeds of 0.83 m/s). 12 For the walkers subgroup who progressed from 0.7 to 1.1 m/s, there is probably minimal underestimation, but for the wheelchair users who progressed from 0.2 to 0.6 m/s, the underestimation could be greater. In addition, we based our measure of lower-extremity PA on steps that would capture activity during walking, stepping, or transfers but would not capture tasks such as stationary cycling or leg exercises while supine or sitting. More so, for the upper extremity, we cannot determine what specific activities are done when the movement is registered. The Actical seems sensitive to typical upper-extremity movements (reaching and bringing hand to mouth). We have assumed that the arm activity counts primarily represent functional, purposeful movements. Although we have removed any arm swing from the arm activity counts, we cannot discount that other nonpurposeful movements were recorded. The accelerometers do provide a measure of use in one’s environment, unlike standard clinical measures. Despite older age, the adults in the control group demonstrated substantially higher activity levels compared with the inpatients.
In conclusion, this is the first study to quantify the change in daily use of the extremities during subacute inpatient rehabilitation. Actual use of the upper and lower extremities was low, with little change over 3 weeks of rehabilitation, despite significant improvement on the clinical measures. These quantitative values will inform other investigators who attempt to increase daily use through innovative interventions.
Footnotes
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article:
We acknowledge the support of BC Medical Services Foundation (BCM08 -0098 to J.J.E. and D.R.), postdoctoral funding (D.R.) from the Heart and Stroke Foundation of Canada, Canadian Stroke Network, Canadian Institutes of Health Research (CIHR)/Rx&D Collaborative Research Program with AstraZeneca Canada Inc., and career scientist awards (J.J.E.) from CIHR (MSH -63617) and the Michael Smith Foundation for Health Research.
