Abstract
Background. The optimal physical therapy dose in acute stroke care is unknown. The authors hypothesized that physical therapy would be significantly different between treatment arms in a trial of very early and frequent mobilization (VEM) and that immobility-related adverse events would be associated with therapy dose. Methods. This study was a single-blind, multicenter, randomized control trial. Patients admitted to a stroke unit <24 hours of stroke randomized to standard care (SC) or intervention, SC plus additional early out-of-bed therapy (VEM). Timing, amount, and type of therapy recorded throughout the trial. Adverse events were recorded to 3 months. Results. A total of 71 patients (SC n = 33, VEM n = 38) received 788 therapy sessions in the first 2 weeks of stroke. Schedule (hours to first mobilization, dose per day, frequency and session duration) and nature (percentage out-of-bed activity) of therapy differed significantly between groups (P ≤ .001 for all components). Mobilization was earlier, happened on average 3 times per day in those receiving VEM, with the proportion of out-of-bed activity double in VEM session (median SC 42.5%, VEM 85.5%). SC consisted of 17 minutes of occupational and physiotherapy per day and was the same between groups. Number of immobility-related adverse events 3 months poststroke was not associated with therapy dose or frequency. Conclusions. The authors detailed usual care and intervention therapy provided to patients from admission to 14 days after stroke. The therapy schedule was markedly different in the intervention arm, but whether this schedule reduces complications or improves outcome is unknown.
Introduction
The optimal schedule and content of physical therapy for people with stroke, particularly in the acute phase of care, is unknown. In the postacute rehabilitation phase, more therapy accounts for better outcomes without a clear ceiling effect.1,2 Although many guidelines recommend starting out-of-bed activity “early” (early mobilization), they do not specify how soon after onset or how much therapy is best, largely because the supporting evidence to guide recommendations is insufficient.3-5 Some authors propose waiting 3 days before getting stroke patients out of bed 6 whereas others mobilize within 24 hours of stroke onset with positive outcomes. 7 The uncertainty surrounding early mobilization, combined with the potential benefits of this broadly applicable, early intervention, make an ideal target for clinical trials.
AVERT (A Very Early Rehabilitation Trial) Phase II was a single-blind randomized controlled trial investigating the safety and feasibility of a protocol promoting earlier and more frequent mobilization. In this study “mobilization” was defined as activity out of bed and the intervention, delivered in addition to usual care, commenced within 24 hours of stroke onset. We briefly summarized previously that intervention group participants received more total therapy, earlier, than controls. 8 In this further analysis of feasibility, we explore the nature and schedule of therapy delivered to both groups. We hypothesized that (a) the intervention group received a higher dose of therapy per day via more frequent sessions than controls and (b) the amount of usual care therapy did not differ between groups. In addition, having detailed all complications experienced by participants in this trial, 9 we explored the relationship between dose of therapy and immobility-related adverse events in particular. An estimated 85% of patients experience complications during their hospital stay. 10 Complications of immobility may account for 51% of deaths in the first 30 days after ischemic stroke, with 62% in the first week. 11 Early mobilization may influence immobility-related complications. Consequently, our final hypothesis was that (c) a higher dose of therapy would be associated with fewer adverse events.
Methods
The study was conducted at 2 large teaching hospital acute stroke units. Both institutional ethics committees approved the study.
Participants
Patients with first or subsequent stroke were eligible if they were older than 18 years, admitted within 24 hours of symptom onset, could react to verbal commands, systolic blood pressure 120 to 220 mm Hg, oxygen saturation >92%, heart rate 40 to 100 beats per minute, and a temperature <38.5°C. Patients with premorbid disability (modified Rankin Scale [mRS] >3), early deterioration, direct admission to intensive care, progressive neurological disorder, acute coronary syndrome, severe heart failure, lower limb fracture, requiring palliative care, thrombolysis treatment, or other trial participation were ineligible.
Randomization and Blinding
Stratification was by stroke severity and site, with mild (National Institutes of Health Stroke Scale [NIHSS] <8), moderate (NIHSS = 8-16), and severe (NIHSS > 16) strata. 12 Opaque envelopes concealed allocation. Assessors were blind to group and patients consented to participate in a study comparing 2 different rehabilitation programs with no detail provided. 8
Intervention
Participants received usual standard care from ward staff. We defined standard care as all usual therapy provided to patients as part of routine practice. No standardized protocols guided standard care (SC). Intervention participants received very early mobilization (VEM) by AVERT physiotherapists and nurses in addition to SC. The frequency, dose, and content of care varied according to physical ability as detailed in an intervention protocol. Dose monitoring and compliance for this study has been described previously. 13 In brief, VEM participants mobilized within 24 hours of symptom onset, thereafter the emphasis of VEM was to help the patient to sit out of bed, stand upright, and perform functional activities at intervals throughout the day. VEM continued until day 14 or discharge, whichever was sooner.
Data Collected
The following demographic data were collected: age, gender, living arrangements before stroke, premorbid disability and type, side and severity of stroke, stroke classification, comorbidities, and length of stay (LOS). All AVERT and ward therapists, including occupational therapists, recorded therapy (minutes) immediately after each session using personal digital assistants (Palm Zire 21 Handheld). Therapy content was recorded under 10 prespecified categories; lying, bed activities, supported sit, sitting activities, sit-to-stand transfer, standing, early gait, advanced gait, upper limb training and other. This tool has been validated. 14
Outcomes
Schedule of therapy was of primary interest, best reflected by hours to first mobilization, total amount of therapy (minutes), dose per day (min/d), frequency (sessions/d), and duration of a session (minutes). The proportion of out-of-bed activity (%) helped describe the content of therapy.
The safety outcome was immobility-related adverse events (AEs) at 3 months poststroke. Immobility-related AEs included pressure sores, pain, deep venous thrombosis, pulmonary embolism, falls, urinary tract infection, pneumonia, chest infections, aspiration, and hypotension/ syncope. 9 AEs were extracted from medical records at 7 days, 14 days, and 3 months by the blinded assessor.
Data Analysis
This study was about therapy for mobility, so upper limb and “other” therapy were excluded from analyses. To calculate dose per day, we included all possible treatment days within the intervention period. As physiotherapists and occupational therapists only worked during weekdays, possible treatment days excluded weekends and holidays.
The prespecified therapy categories were grouped into 2 activity categories: (a) in bed activities—lying, in bed activities (eg, rolling) and (b) out-of-bed activities—sitting activities, sit-to-stand transfers, standing, early and advanced gait. Mobilization activities were out of bed. The percentage of time spent out of bed was calculated for each session and a per participant average determined.
Statistical Issues
Shapiro–Wilk tests were employed to assess whether the schedule of therapy outcome measures were normally distributed. They were not, so nonparametric tests were used to compare therapy data between groups (Mann–Whitney U test for continuous variables, Fisher exact test or χ2 test for categorical data). A Bonferroni correction was used to account for multiple tests on therapy data (significance level was α = .008). As stroke severity influences therapy, hours to mobilization, frequency, dose per day and out-of-bed activity are described grouped by NIHSS severity subgroups (mild, moderate, severe). Sample sizes were too small to justify subgroup comparisons. When examining whether SC was equivalent between groups, the nonparametric Van Elteren test was used with stroke severity as strata.
Previously, we found that longer LOS, increased age, and higher stroke severity were significantly associated with complications to 3 months 9 and that the interaction between LOS, stroke severity, and complications was complex. To further examine the relationship between total number of immobility-related AEs and therapy dose and to unpack these interactions, a model of count outcome was required. A fit analysis for models of count outcome using several standard criteria and fit measures was performed and the preferred model was the negative binomial regression model. Age, stroke severity, LOS, dose per day, and frequency of sessions were used as independent variables since they may influence immobility-related AEs. 9 Post hoc modeling was conducted (based on the negative binomial regression model results) to explicate the relationship between group allocation, LOS, and stroke severity. These results were used to predict expected numbers of immobility-related AEs for particular patient groups.
Results
Table 1 summarizes the sample demographics of the 71 patients. More moderate/severe stroke patients were in the intervention group (60% vs 54%) but group differences were not significant for patient flow. 8
Baseline Characteristics
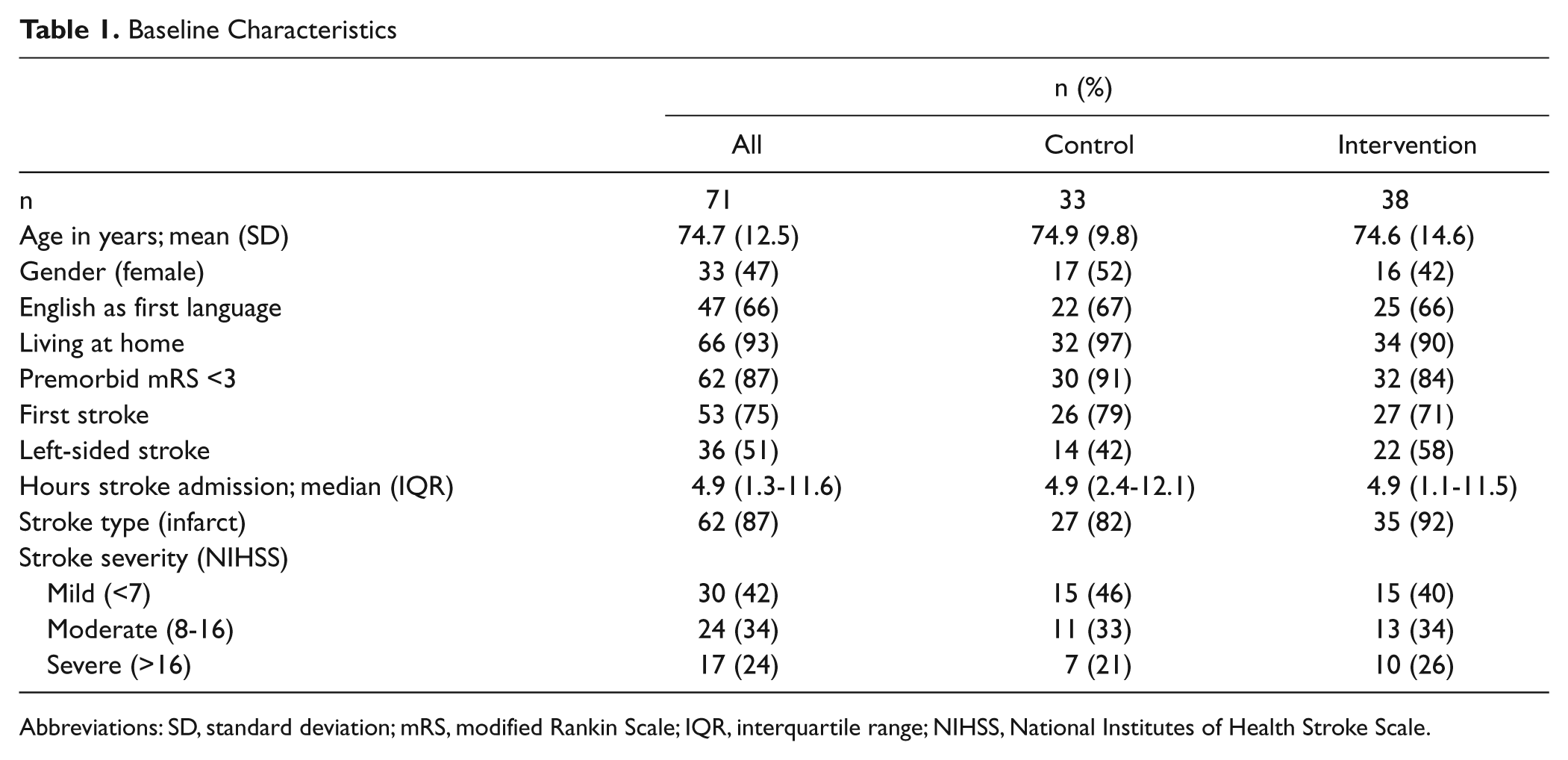
Abbreviations: SD, standard deviation; mRS, modified Rankin Scale; IQR, interquartile range; NIHSS, National Institutes of Health Stroke Scale.
Schedule of Therapy
Four participants did not receive any therapy; 3 were in the control group and were discharged within a week, the fourth person was in the intervention group and died within the first week. Five other participants died in the intervention period but received therapy. All were included in the analysis, in their allocated treatment group.
There were 788 therapy interventions delivered in this trial. Intervention group participants started mobilization earlier and received significantly higher amounts of therapy (min/d) than those in the control group (Table 2). VEM therapy accounted for 64% of total therapy received and made up two thirds of the total sessions. VEM sessions were only half the length of SC sessions. The proportion of out-of-bed activity in VEM sessions was twice as much as in SC sessions (VEM median 85.5%, interquartile range [IQR] 60.5-96.3; SC median 42.5%, IQR 22.3-64.0). All therapy variables (total minutes, dose, frequency, duration of a session, and proportion out-of-bed activity) were significantly different between the SC and VEM components of intervention care.
Therapy Delivered to Groups
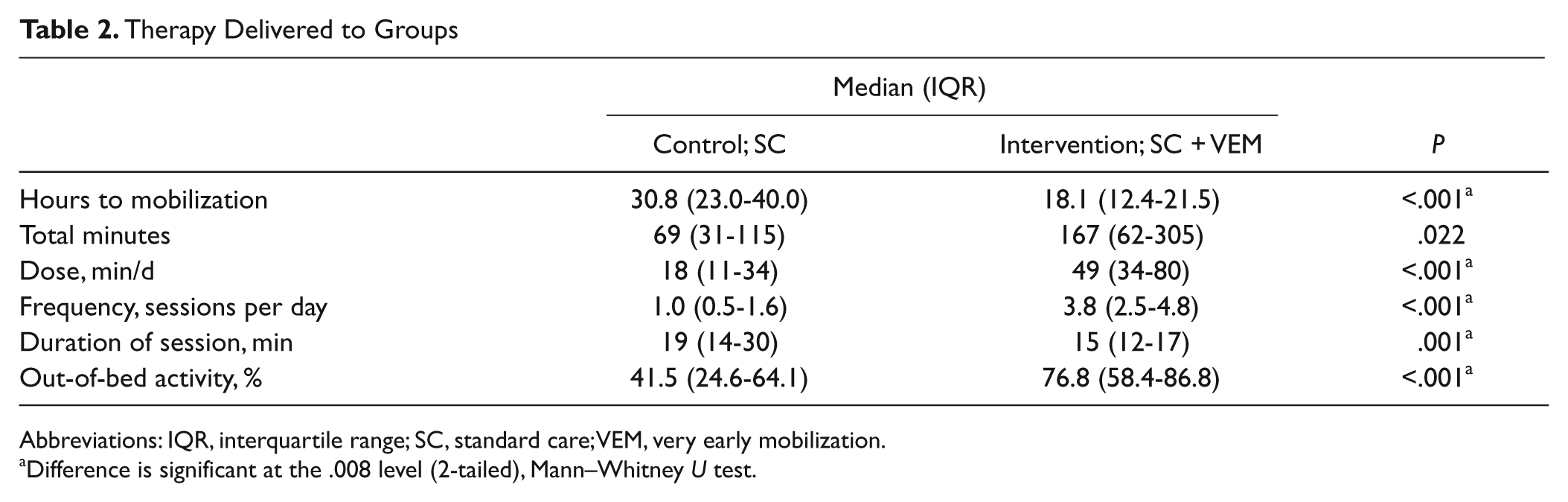
Abbreviations: IQR, interquartile range; SC, standard care; VEM, very early mobilization.
Difference is significant at the .008 level (2-tailed), Mann–Whitney U test.
Therapy according to severity subgroups is shown in Figure 1. These reflect overall group differences, with the exception that the proportion of out-of-bed activity per session in the severe subgroup was similar between groups (but more frequent in VEM).
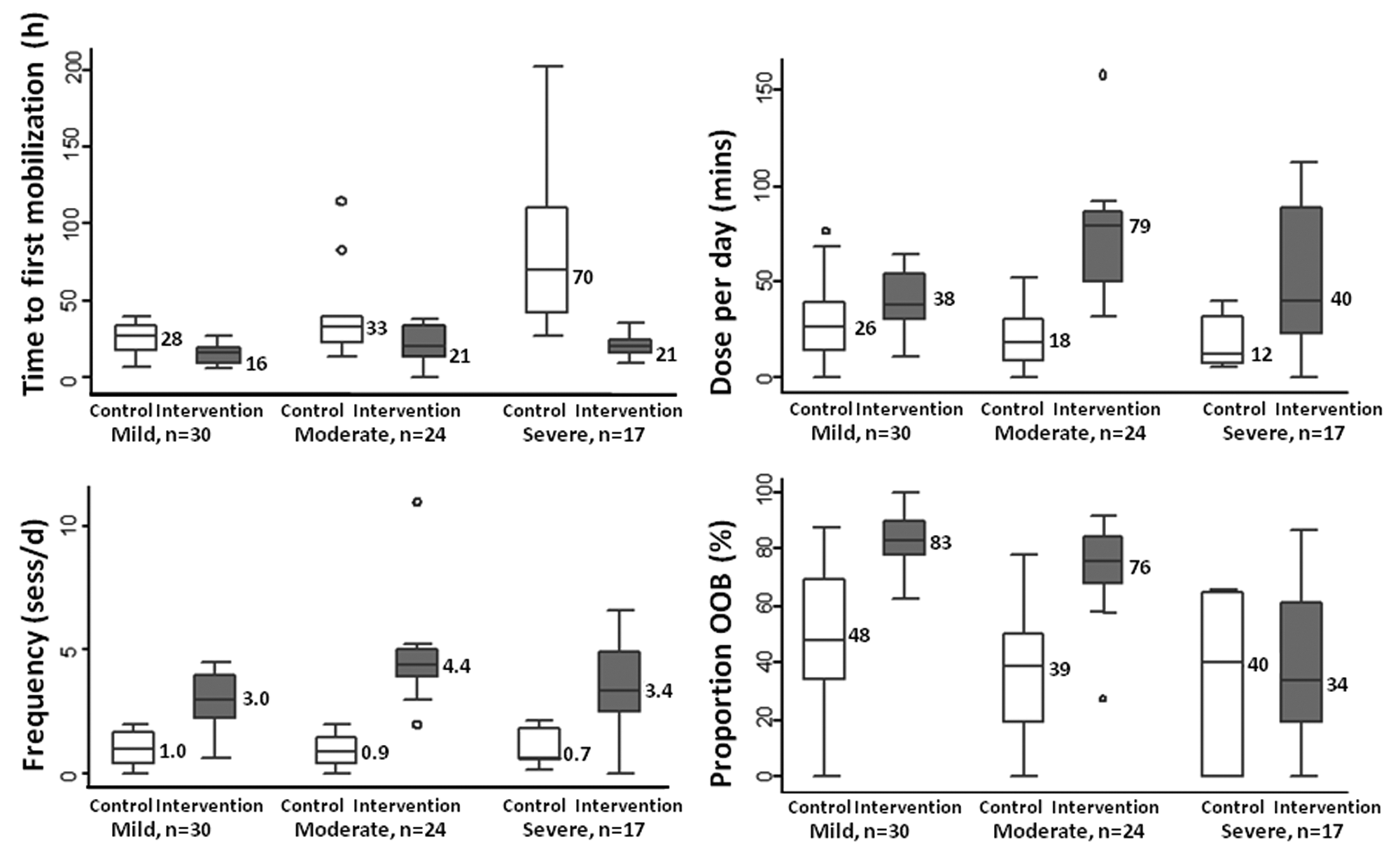
Box plots of therapy characteristics by stroke severity subgroup (mild, NIHSS < 7; moderate, NIHSS = 8-16; severe, NIHSS > 16). The box represents the middle 50% of the values, including the median. The bars show upper and lower 25% range of values, circles represent outliers.
Standard Care Component
There was no difference in the amount of SC therapy received between groups (control median, 18 minutes, IQR 11-34; intervention median, 16 minutes, IQR 8-25; Mann–Whitney U test P = .241). Van Elteren test indicated that, when stratified by stroke severity, the proportion of out-of-bed activity was similar in both groups (control 42%, IQR 25-64; intervention 43%, IQR 22-64; P = .602).
Immobility-Related Adverse Events
By 3 months poststroke there were 92 immobility-related AEs reported. In all, 22 control participants (66.6%) and 18 intervention participants (47.7%) experienced at least one immobility-related AE, but counts were similar (control n = 48; intervention n = 44). Falls were most common (34% of AEs). 9 The regression analysis (Table 3) shows the relative contribution of each variable to the percentage change in expected count of immobility-related AEs. An extra day of hospital stay accounted for a 7.3% increase in AEs. Longer LOS and, to a lesser extent, older age were associated with higher numbers of immobility-related AEs. Therapy (minutes or sessions) did not have a significant influence.
Contribution of Age, Stroke Severity, LOS, and Therapy to Percentage Change in Expected Count of Immobility-Related Adverse Events
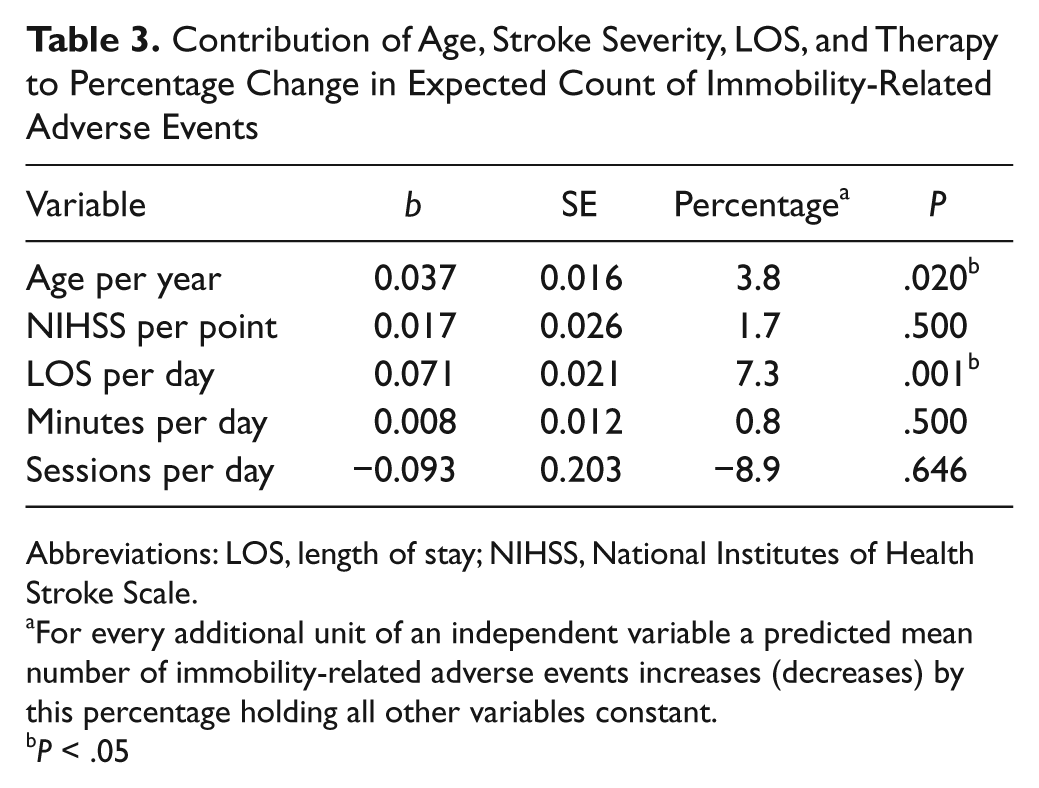
Abbreviations: LOS, length of stay; NIHSS, National Institutes of Health Stroke Scale.
For every additional unit of an independent variable a predicted mean number of immobility-related adverse events increases (decreases) by this percentage holding all other variables constant.
P < .05
Post Hoc Modeling to Predict Adverse Events
We predicted the likely number of immobility-related AEs for an “average” patient of any given age, stroke severity, LOS, or amount of therapy received using the coefficients from the regression analysis (Table 3). For example, a “person” 74.7 years old, with a mean NIHSS score of 10.3, a 9.2-day LOS, and receiving 41.9 minutes per day of therapy in 2.5 sessions would likely experience on average 1.03 immobility-related AEs, with a 45% chance of having 0 AE, 28% chance of having 1 AE, and 14% chance of having 2 AEs. Given the importance of LOS, we calculated median LOS for the 6 trial subgroups (mild stroke–control, mild stroke–intervention, etc, Table 4).
Expected Count of Immobility-Related Adverse Events at 3 Months
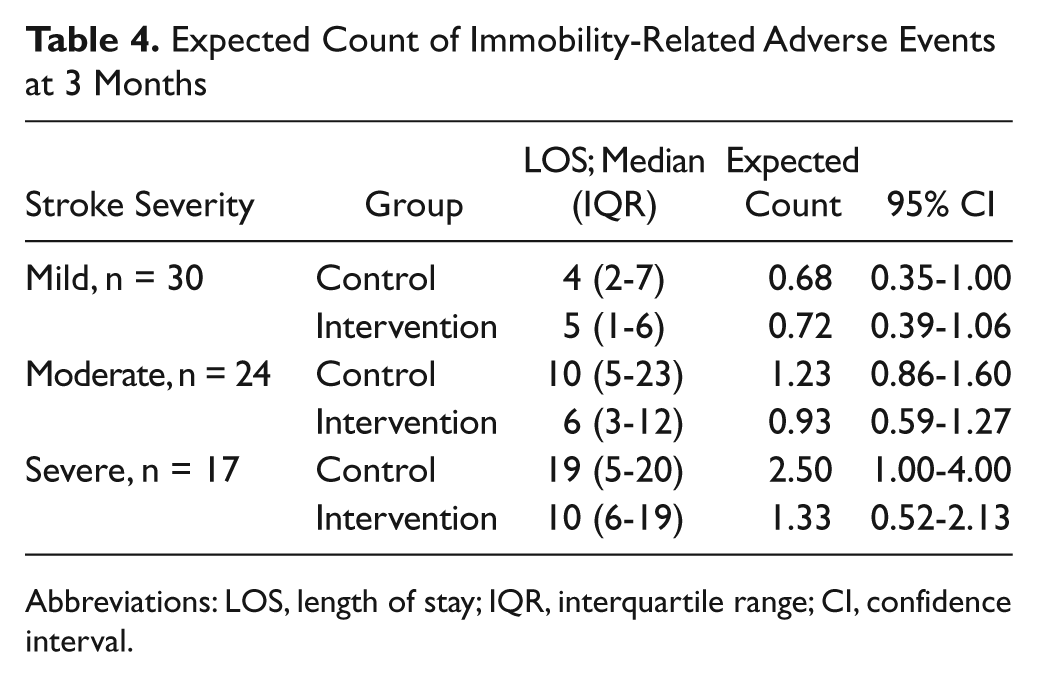
Abbreviations: LOS, length of stay; IQR, interquartile range; CI, confidence interval.
Before predicting probabilities for specific patient types, we checked the model for all participants. Median scores for the 5 variables (age, NIHSS, LOS, dose per day, and session frequency) were calculated and the regression model was run with these medians. The final regression model was run again for stroke severity subgroups, including group allocation and stroke subgroup specific LOS (data from Table 4) to derive predicted probabilities of different AE counts for each subgroup (Figure 2). Moderate and severe stroke patients were predicted to have a lower probability of having more than one AE by 3 months if they had an “intervention” LOS instead of a “control group” LOS. Difference in LOS did not appear to be important in mild stroke patients. The expected count of AEs could also be predicted for a specific patient type (Table 4). For example, an average moderate stroke patient with a LOS of 6 days would likely experience one (0.93) adverse event by 3 months.
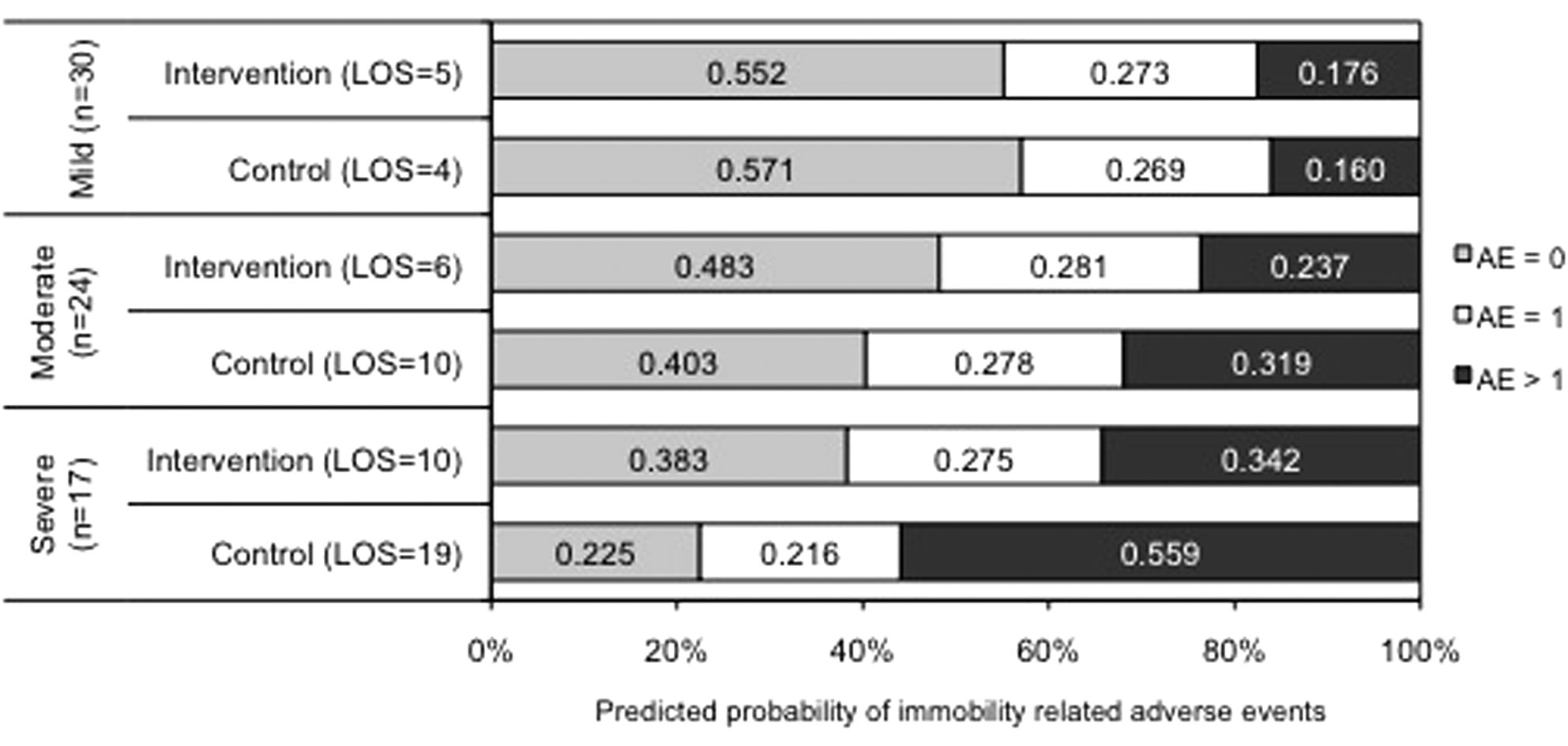
Predicted probabilities of immobility-related AEs for mild (NIHSS < 7), moderate (NIHSS = 8-16), and severe (NIHSS > 16) stroke cases.
Discussion
Adding an early mobilization intervention to SC effectively altered the schedule and nature of therapy delivered. Many aspects of the schedule of therapy were different between groups. Dose per day, frequency of sessions, and level of activity (proportion out-of-bed activity) were higher and duration of a session and time to mobilization were lower in the intervention group. These differences were consistent across the 3 stroke severity subgroups. Importantly, the SC delivered to both groups was consistent, indicating that it was feasible to add a more intensive training program on top of usual care without adversely affecting delivery of usual therapy.
Delivering the experimental intervention as planned is a challenge in rehabilitation trials. Detailing the planned intervention in objective terms and reporting the level of success of delivering this care is critical. 15 If therapy delivered to an intervention group does not differ from that delivered to controls, then “no effect” will be the finding. In this study, we aimed to deliver a higher therapy dose with greater frequency and double the intensity. Therapy was delivered as described in the protocol in most cases. This was expected because we monitored dose and provided feedback to trial therapists. 13 Failure to deliver intervention as planned represents an important feasibility issue.
This study provides novel data about the standard therapy delivered in stroke units from admission with stroke up to 14 days. So far, little is known about the therapy provided for stroke patients in acute care. Physiotherapy and occupational therapy delivered in acute stroke units have been determined from short observational studies in unselected groups. Therapy accounted for only 5% of the day and patients had low activity levels overall. 16 Patients in these units received on average 0.74 sessions of physiotherapy and 0.47 sessions of occupational therapy per day, with an average of 20 minutes per session (excluding upper limb therapy). The average SC dose of 17 minutes of all therapy in the present study was similar, but only 42% of therapy involved out-of-bed activity. The SC dose in this study is well below the level recommended in the review of effective stroke unit care 17 and is lower than that reported in a small sample of stroke patients who commenced training at around 8 days poststroke (32 minutes mobility-related training per day). 18 In contrast, our intervention dose of 49 minutes per day is comparable with the stroke unit in Trondheim, Norway that routinely provides rehabilitation starting <48 hours poststroke, with 2.1 sessions of physiotherapy per day, and sessions averaging 27 minutes (including upper limb training). 19
Our third hypothesis, that a higher dose of therapy would be associated with fewer adverse events, was not supported. The multivariate regression analysis showed that adverse events were not significantly related to daily dose or session frequency. However, as expected we found that length of hospital stay was strongly related to number of adverse events. 20 It is difficult to say anything about the influence of other variables given the dominant effect of length of stay. In a previous analysis, we examined factors associated with experiencing a stroke or immobility-related complication. Stroke severity and smoking history were associated with increased odds of experiencing a stroke-related complication, whereas older age and LOS were associated with experiencing immobility-related complications. 9 It remains unclear whether patients stay longer because of complications, or whether longer hospital stay (because of other factors, which may include more severe stroke) increases the risk of complications. Both are likely and interventions that reduce length of stay may help reduce immobility-related AEs. LOS did differ between groups in this study, with marked reduction in the LOS of patients with moderate and severe stroke in the intervention group. If the intervention made an important contribution to reducing LOS (which would be a reasonable expectation) then we need to account for this effect in future analyses of data from the phase III trial currently underway. The modeling presented here should be considered an illustration of the approach that may be necessary to adequately explore the relationship between early intervention and AEs in the larger study.
We acknowledge the limitation of a small sample size. However, phase II trials allow us to explore both the feasibility of intervention protocols and planned analyses for future trials. We have shown that asking busy clinicians to follow an intensive treatment protocol on top of SC was feasible, even for patients with severe stroke. We have also detailed the standard physical therapy delivered to acute stroke patients. The stroke units in this study had an average physiotherapist–patient ratio of 1:11, occupational therapists–patient ratio of 1:12, and nurse–patient ratio of 1:4, which is probably inadequate to allow routine delivery of the experimental intervention. At present, there are no evidence-based recommendations concerning therapist staffing levels in acute stroke units. Staffing levels should be driven by the amount of therapy that needs to be delivered to patients to improve outcome, but we are still some way from knowing what that should be.
Determining whether the therapy dose successfully delivered in this pilot study is sufficient to reduce disability and hasten walking recovery will make an important contribution to the evidence base. What is clear from this study is that the term “very early mobilization” is simplistic and fails to capture the early, intense, and ongoing nature of the intervention being tested. This complex nature will be encapsulated in an “AVERT score” that is currently being developed. The score will be validated when phase III is complete.
Footnotes
Acknowledgements
We gratefully acknowledge the support of the clinical staff at the Austin Hospital and St. Vincent’s Hospital who participated in this trial and Dr Janice Collier of the National Stroke Research Institute.
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
This trial was supported by grants from the National Heart Foundation (G04M 1571), Affinity Health and the Austin Hospital Medical Research Fund. Our institute also received operational infrastructure support from the Victorian State Government. Dr Bernhardt received fellowship support from the National Health and Medical Research Council (Australia) and the National Heart Foundation of Australia over the course of this study.
