Abstract
Objective. This study investigated the course and relationship between investigator-determined and patient-reported level of independence within the first year after spinal cord injury (SCI). The authors examined variables that contributed to these scores. Methods. In this observational cohort study, 73 patients with traumatic SCI were evaluated at 1, 3, and 6 months (and 40 subjects at 1 to 12 months). The investigator-determined independence was quantified using the Spinal Cord Independence Measure (SCIM). The subjective, patient-reported independence was determined by asking how their general restrictions influenced everyday life activities. Several variables were used to explain these 2 scores. Results. The SCIM score was higher than the patient-reported independence and improved significantly more over time (up to about 70/100 at 12 months), whereas the perceived independence remained below 50/100. The correlations between the 2 measures were at most moderate (rs ≤ 0.51), but in general somewhat higher for subjects with tetraplegia. Age and muscle strength predicted the SCIM score well. No variable predicted the patient-reported level of independence. Conclusions. Investigator-determined and patient-reported outcomes can differ considerably and evolve differently. A patient-reported outcome measure may not detect actual functional improvement. It is likely that changes in patient-reported outcomes are influenced by many factors in addition to those associated with functional recovery, including psychological factors.
Keywords
Introduction
The need for functional outcome measures for spinal cord injury (SCI) is urgent to be able to test biological interventions that may move from the bench to bedside.1-4 The clinical field is paving the way by determining the psychometric characteristics of these outcome measures and by making recommendations.5,6 Although a primary outcome measure is needed to determine the number of patients required for trials, 7 current achievements suggest that a test battery is required to assess outcome at the several domains suggested by the International Classification of Functioning, Disability and Health (ICF).
At the domain of body functions and body structures, changes in sensory–motor function are preferably measured by the “International Standards for Neurological and Functional Classification of Spinal Cord Injury” according to the American Spinal Injury Association (ASIA). 8 At the domain of activities, capacity tests assess the highest level of functioning possible in a standardized environment, whereas performance measures assess the level of functioning in the everyday life environment. The Spinal Cord Independence Measure (SCIM)9-11 is recommended 5 to evaluate activities of daily life (ADL) and independence.
An ongoing debate in this field is about the importance of patient-reported outcome measures in clinical trials. For certain studies, patient-reported functional outcomes may be less appropriate. For example, in an early phase I/II trial where safety, tolerability, and proof of mechanism are the main focus, the patient’s perception of pain might be relevant, but the patient’s perceived interference with ADL is not an issue. A phase III trial, however, aims to demonstrate an improvement in functional activities and personal independence, which could be scored by investigator-determined “objective” outcome measures, such as the SCIM. However, the argument that what ultimately matters is the patient’s perception about functional improvement cannot be easily disregarded, especially without knowing the relationship that exists between these measures.
In the field of SCI, such knowledge is missing, as both measures have been used for different studies. For example, investigator-determined measures of performance have been applied to evaluate the influence of the patient’s age12,13 or the efficacy of rehabilitation intervention. 14 Patient-reported outcome measures, however, have been applied to address the influence of negative symptoms after SCI such as pain15-17 or depression.18,19 The aim of this study was therefore to investigate the relationship between investigator-determined and patient-reported levels of independence within the first year after SCI. Based on a previous study, 20 we hypothesized that patient-reported interference should relate well to bladder and bowel dysfunction in paraplegic subjects and to upper extremity function in tetraplegic patients. Finally, we explored whether the investigator-determined and patient-reported independence scores could be explained by similar factors. We emphasize that this latter analysis was not intended to be comprehensive and rather aimed at providing initial input for upcoming studies.
Methods
Persons With Spinal Cord Injury
This observational cohort study was performed within an international collaborative effort, the European Multicenter Study for Human Spinal Cord Injury (EM-SCI). 21 In this network, demographics and outcome measures are assessed at fixed time intervals after SCI; within the first 15 days postinjury, at 1 month (16-40 days), and 3 months (70-98 days), 6 months (150-186 days), and 12 months (300-400 days). Patients with reduced capabilities for cooperation or for giving consent, peripheral nerve lesions above the level of injury or brain injuries were not included. In December 2006, a subproject was started to evaluate several negative symptoms, quality of life measures and patient-reported outcomes. This study is based on these data. The study protocol was approved by the local institutional review board. Participants gave written, informed consent.
Assessment of Daily Life Activities and Independence
The investigator-determined or “objective” level of performance of ADL and independence was scored by the SCIM. The SCIM contains 3 subcategories: (a) self-care (eg, bathing, grooming), maximal score 20; (b) respiration and sphincter management, maximal score 40; and (c) mobility (eg, transfers as well as mobility indoors and outdoors), maximal score 40. The maximum score, indicating best performance, is 100. The SCIM has been revised twice since its introduction, 9 namely in 2001 (SCIM II 10 ) and in 2007 (SCIM III 11 ). In these revisions, single items were changed or deleted, but the category and total sum scores remained the same and the results are highly reliable.10,11 As we switched within the EM-SCI at a certain point from SCIM II to SCIM III, some persons in this study were initially, and therefore also repeatedly, assessed with the SCIM II, whereas others were scored with the SCIM III.
We used the question “How would you score your general restrictions on your everyday life” as an indicator for the patient-reported or “subjective” level of restrictions on ADL and independence. The scale varied between 0 (no restrictions) to 10 (major restrictions). To simplify the interpretation, we made the score comparable to the SCIM: patient-reported independence score = (10 − original score) × 10. A score of 8 (rather large restrictions) thereby resulted in a subjective independence score of 20, indicating little patient-reported independence. A score of 0 (no restrictions) resulted in 100, indicating maximal subjective independence.
Assessment of Neurological Impairment
The neurological assessment was performed in accordance with the International Standards for Neurological and Functional Classification of Spinal Cord Injury, 8 providing information about the completeness of the lesion, the sensory, motor, and neurological level of the lesion, as well as motor and sensory scores. Subjects are categorized into 5 ASIA Impairment Scale (AIS) subgroups: AIS A, sensory–motor complete; AIS B, motor complete, sensory incomplete; AIS C, sensory–motor incomplete, more than half of the key muscles below the neurological level have a muscle grade <3; and AIS D, sensory–motor incomplete, at least half of the number of muscles below the neurological level have a muscle grade ≥3. In AIS E patients, no impairment can be demonstrated. To ensure high examination quality, the EM-SCI investigators were trained to standardize the examination techniques, whereas the AIS classification was performed by a computer algorithm. 22
Statistics
Simple linear relationships were quantified using Spearman’s correlation coefficient (rs). Differences between investigator-determined and patient-reported independence scores were calculated using a 2-way mixed model analysis of variance for repeated measures. Pairwise comparisons were adjusted using Bonferroni’s correction. Multiple linear regression analyses were applied to determine what factors might influence investigator-determined and patient-reported independence scores. A backward model was used. The probability for entering the model was set at .05 and for removal at .10.
Results
Six-Month Cohort
At the time of analysis, 159 of 171 patients who participated in the subproject had a traumatic SCI. Selected were SCI subjects with complete SCIM and subjective independence scores at 1 and 6 months. Of the 73 participants included, 5 had no complete assessment at 3 months. The patients were on average (mean ± SD) 46 ± 19 years (range, 13-83 years) and 14 patients were female. The lesion was in the cervical (n = 39), high thoracic (T1-T6, n = 11), low thoracic (T7-T12, n = 10), and lumbar (n = 12) regions, with lesion data missing for 1 patient (32 paraplegic and 41 teraplegic patients). In all, 31 participants were classified as AIS A, 6 as AIS B, 9 as AIS C, and 27 as AIS D. The participants were admitted for rehabilitation between April 2007 and January 2009 in the following German centers: Halle (n = 22); Heidelberg (n = 1); Hessisch-Lichtenau (n = 5); Karlsbad-Langensteinbach (n = 7); Murnau (n = 14); Ulm (n = 10), and in 1 Swiss center at Zurich (n = 14). The average length of stay was 22 ± 14 weeks (data of 24 patients were missing, partly because of still being an inpatient).
Changes in Investigator-Determined and Patient-Reported Independence
The SCIM score improved over time and specifically between 1 and 3 months (see Table 1 and Figure 1). Between 3 and 6 months, the SCIM score did not change (P = .075).
Evolution of Various Measures From 1 to 6 Months After Spinal Cord Injury
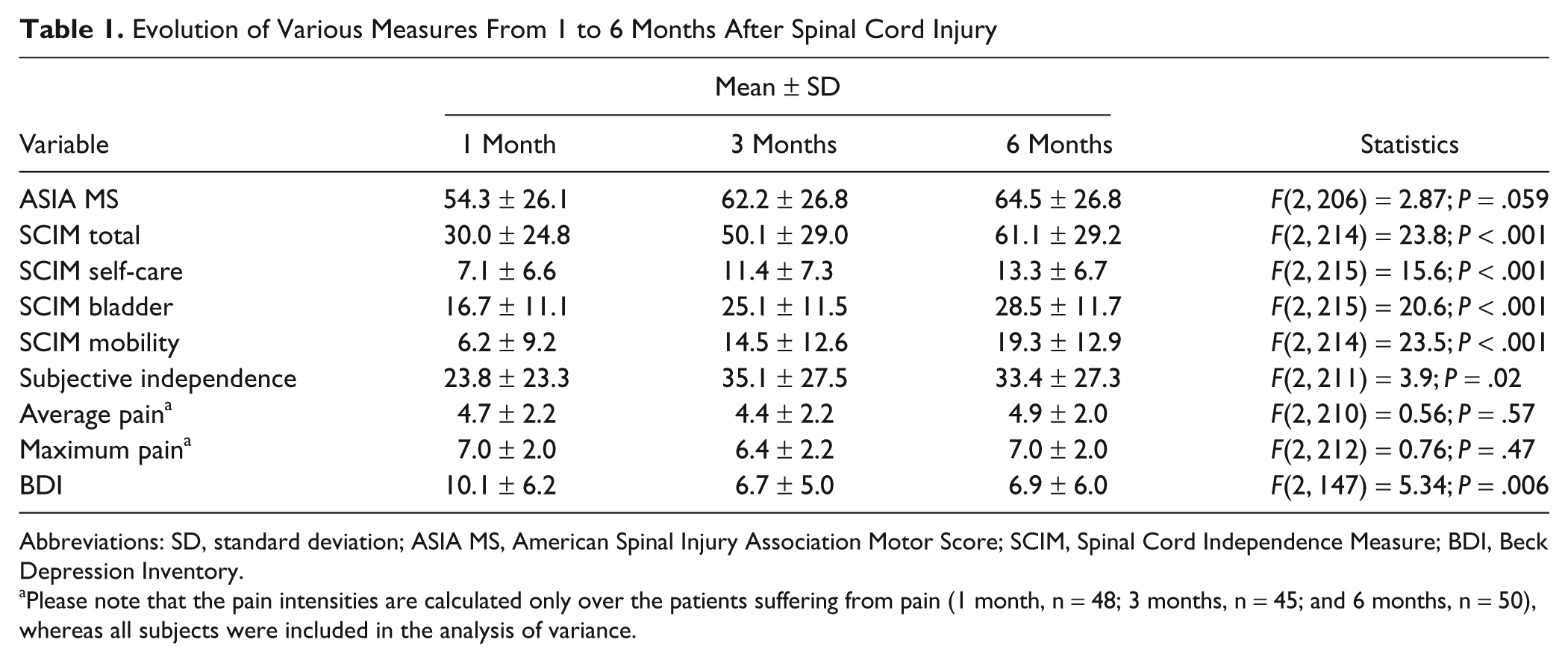
Abbreviations: SD, standard deviation; ASIA MS, American Spinal Injury Association Motor Score; SCIM, Spinal Cord Independence Measure; BDI, Beck Depression Inventory.
Please note that the pain intensities are calculated only over the patients suffering from pain (1 month, n = 48; 3 months, n = 45; and 6 months, n = 50), whereas all subjects were included in the analysis of variance.

Objective versus subjective independence after spinal cord injury
A similar course was observed for the patient-reported independence (see Table 1 and Figure 1). It improved between 1 and 3 months, after which it remained unchanged at 6 months (P = .082).
The investigator-determined independence was larger compared with the patient-reported independence at all time points (difference objective − subjective score = 6.1 ± 25.7 [1 month], 16.1 ± 28.8 [3 months], and 27.7 ± 30.2 [6 months], one-sample t tests; Figure 1). The differences changed significantly over time—F(2, 210) = 10.6, P < .001—specifically between 3 and 6 months (P = .049) and less between 1 and 3 months (P = .12).
Relationships Between Investigator-Determined and Patient-Reported Independence
Interestingly, single participants with a relative good investigator-determined level of independence reported poor to absent patient-reported independence, especially at 6 months after SCI. Overall, the relationships between the independence scores were moderate (Figure 1 and Table 2). Patients with tetraplegia showed higher correlations than those with paraplegia (Table 2). With respect to our hypotheses, we found no consistent findings that correlations between self-reported independence and self-care were higher in tetraplegic patients (compared with other SCIM categories) or that correlations between self-reported independence and bladder and bowel function were higher in paraplegic patients.
Relationships Between the SCIM Total and Category Scores and the Subjective Independence Score a
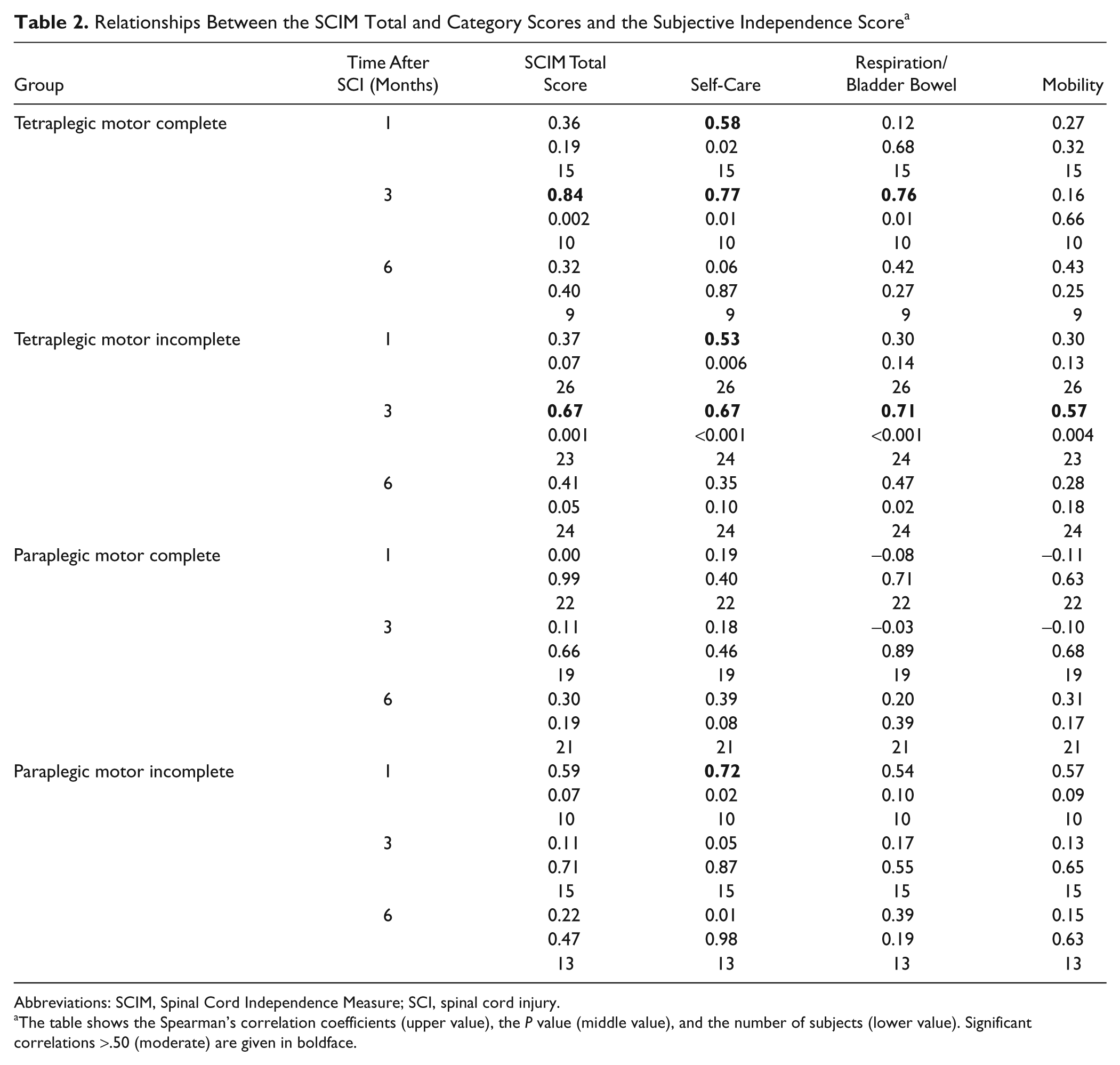
Abbreviations: SCIM, Spinal Cord Independence Measure; SCI, spinal cord injury.
The table shows the Spearman’s correlation coefficients (upper value), the P value (middle value), and the number of subjects (lower value). Significant correlations >.50 (moderate) are given in boldface.
Factors Influencing Investigator-Determined and Patient-Reported Independence
One important predictor for the objective level of ADL performance after SCI appears to be the ASIA motor score, as higher muscle strength correlates with better ADL performance.23,24 The average ASIA motor score improved but not significantly (Table 1). Another predictor is age, which is known to have a negative effect on ADL.12,13
Furthermore, patient-reported interference is influenced by pain or depression. Pain was quantified by rating the average and maximum pain intensity during the last week on an 11-point numerical rating scale (see Table 1). Depression severity was scored using the Beck Depression Inventory (BDI). 25 The BDI consists of 21 symptoms and attitudes and the intensity of each item is rated from 0 to 3. The total score varies between 0 and 63 and cutoff scores represent none or minimal depression (<10), mild to moderate (10-18), moderate to severe (19-29), and severe depression (30-63). The BDI has been proven useful in the SCI population having a good internal reliability and being most frequently applied. 26 In the 6-month cohort, the BDI score was significantly higher at 1 month compared with that at 3 months (P = .01) and 6 months (P = .02; see Table 1). The severity of the BDI at each time point is also presented in Figure 2.
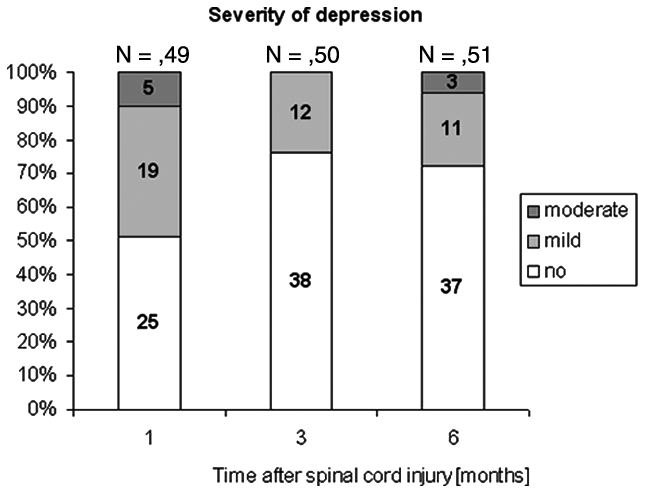
Severity of depression
A multiple regression analysis was performed to determine which of these factors contributed to the investigator-determined and patient-reported independence scores. The model was repeated for each time point (Table 3). When explaining the SCIM score, only age and the ASIA motor score remained in the model and the explained variance improved over time (Table 3). In contrast, the patient-reported independence score was more difficult to explain when using the same predictors. At 1 month, the ASIA motor score and the BDI score remained in the model, but the explained variance was low (Table 3). At 3 months, none of the variables remained in the model, whereas at 6 months, age, the ASIA motor score, and the BDI score remained in the model, but again the explained variance was low. We repeated all analyses with the maximal pain score with comparable results.
Multiple Regression Models Explaining Objective and Subjective Independence in Persons With SCI
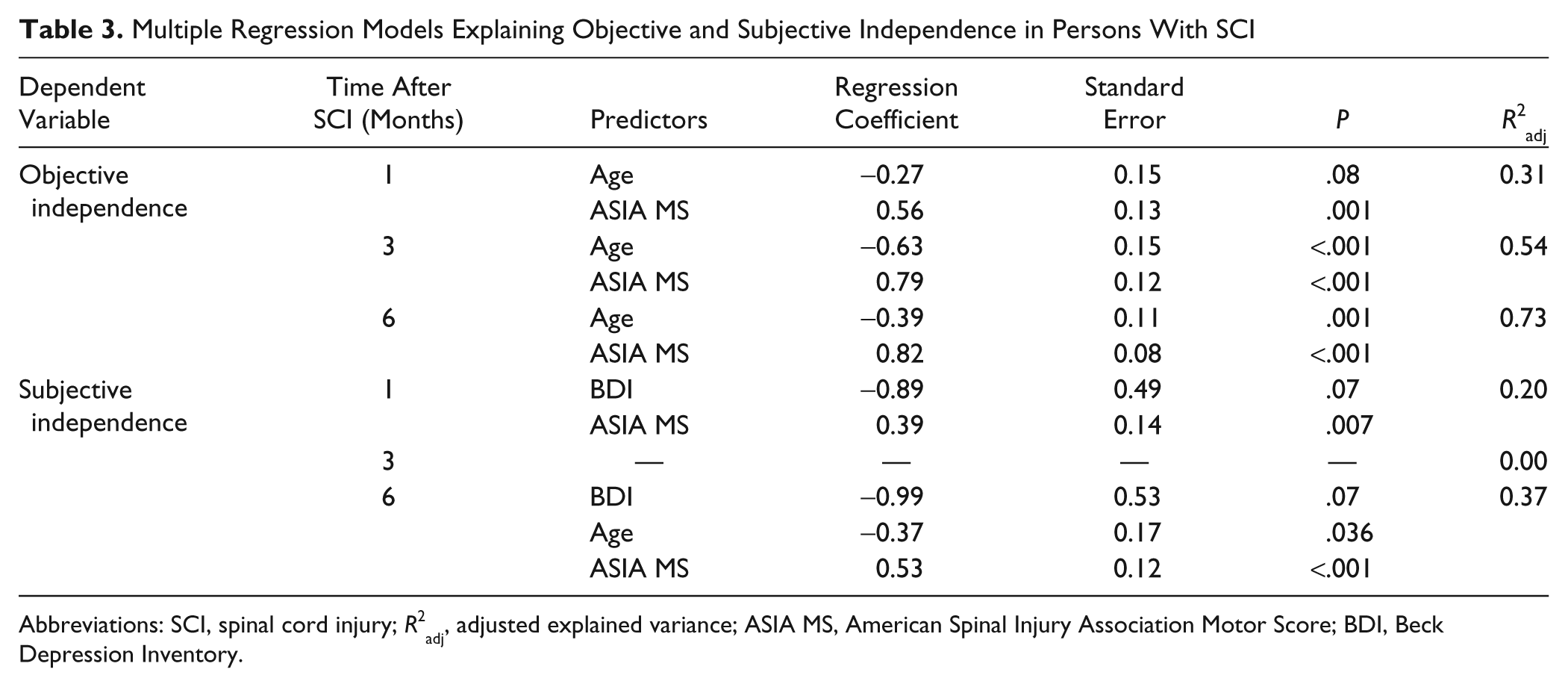
Abbreviations: SCI, spinal cord injury;
Twelve-Month Cohort
To verify whether these results might also apply at 12 months postinjury, we performed an additional analysis in a smaller cohort, who had at least complete assessments at 1 and 12 months. In short, of the 40 SCI patients (4 females), 5 had no complete assessment at 3 and/or 6 months. The participants were 44 ± 18 years old (range 19-76 years) and admitted for rehabilitation between April 2007 and January 2009 in 4 German centers (n = 32) and in Zurich (n = 8). The average length of stay was 20 ± 15 weeks (data of 10 patients were missing). A total of 15 subjects were classified as AIS A, 4 as AIS B, 7 as AIS C, and 13 as AIS D (1 missing); 22 were paraplegic and 18 tetraplegic. The ASIA motor score was 58.8 ± 21.0 at 1 month, 65.6 ± 23.0 at 3 months, 67.6 ± 23.3 at 6 months, and 70.1 ± 24.5 at 12 months; F(3, 145) = 1.67, P = .18.
Overall, similar results were obtained as in the larger 6-month cohort. The SCIM score improved over time— F(3, 151) = 19.4; P < .001—and an improvement was observed between 1 month (34.8 ± 26.8) and 3 months (57.8 ± 26.3; P < .001). The SCIM score did not increase between 3 and 6 months (70.1 ± 22.8; P = .19) and between 6 and 12 months (72.7 ± 22.2; P = 1.0), although the 12-month score was higher than at 1 month (P < .001) and 3 months (P = .049).
The patient-reported independence did not change over time (1 month, 34.0 ± 26.1; 3 months, 48.0 ± 26.7; 6 months, 40.3 ± 28.7; and 12 months, 45.0 ± 28.9; F(3, 146) = 1.9, P = .14). The investigator-determined independence was larger compared with the patient-reported independence at 6 and 12 months (6 months, difference = 29.8 ± 30.4, P < .001 and 12 months, difference = 27.7 ± 32.8, P < .001). The differences changed significantly over time—F(3, 146) = 7.5, P < .001—however, pairwise comparisons showed only a trend between 3 and 6 months (P = .06).
Although the relationships between the SCIM score and the patient-reported independence score were somewhat smaller in this cohort (Table 4) compared with the 6-month cohort, the regression analyses resulted in similar models. At 12 months, again age and the ASIA motor score could explain the SCIM score well (
Relationships Between SCIM Total Score and Patient-Reported Independence Score (Smaller Cohort) a
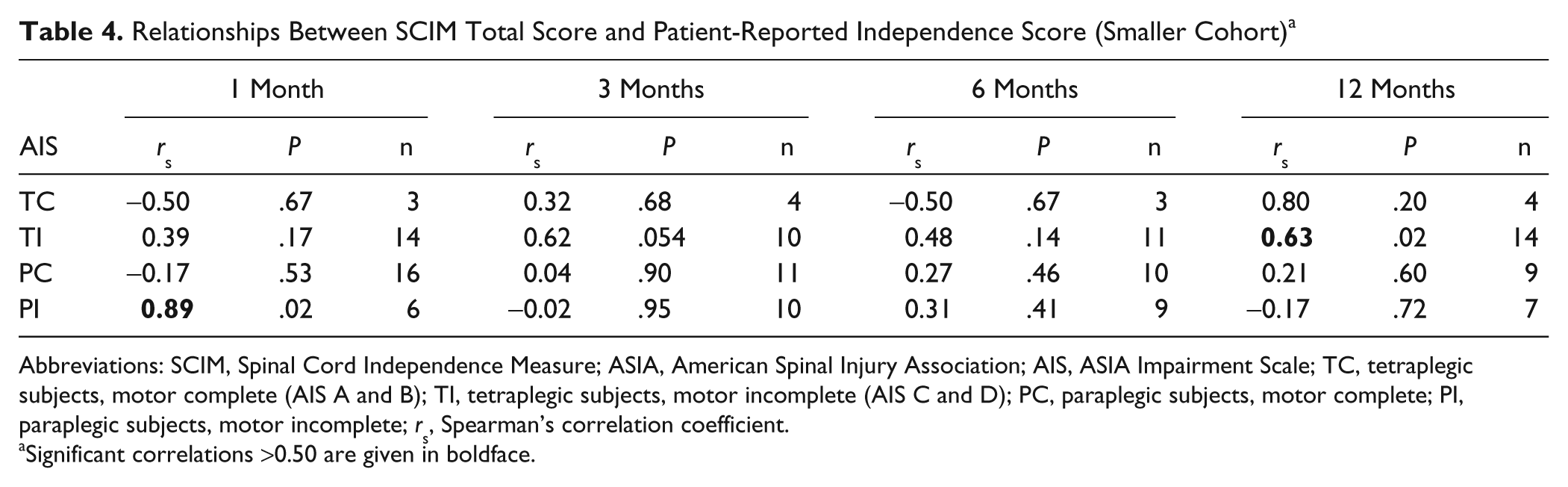
Abbreviations: SCIM, Spinal Cord Independence Measure; ASIA, American Spinal Injury Association; AIS, ASIA Impairment Scale; TC, tetraplegic subjects, motor complete (AIS A and B); TI, tetraplegic subjects, motor incomplete (AIS C and D); PC, paraplegic subjects, motor complete; PI, paraplegic subjects, motor incomplete; rs, Spearman’s correlation coefficient.
Significant correlations >0.50 are given in boldface.
Discussion
We assessed the relationship between investigator-determined and patient-reported independence within the first year after SCI and explored factors that contributed to these independence scores. In general, the results of the 6- and 12-month cohorts were comparable.
Our first main finding was that the investigator-determined and patient-reported level of independence correlated at most moderately, and correlations were better in tetraplegic compared with paraplegic patients. In general, there appears to be a mismatch between these independence levels during the first year postinjury. Although the items of the SCIM are weighted according to the relevance for the patient, 9 especially in paraplegic subjects, relationships between investigator-determined and patient-reported independence were practically absent. Our hypothesis that in paraplegic patients the patient-reported independence score might correlate well with bladder and bowel function was not supported. Tetraplegic subjects, however, rate the recovery of hand function above anything else20,27 as they target an independent life. In these patients, the SCIM reflected better what the patients were aiming for, at least during rehabilitation at 1 and 3 months postinjury.
Another explanation for the mismatch between the investigator-determined and patient-reported independence could be that the question “How would you score your general restrictions on your everyday life” might have been interpreted by the SCI patients as a question about participation and not about activity. Indeed, even the ICF has difficulties in separating between activity and participation. Although such an interpretation might reduce the relationships, it could provide again an explanation why the relationships were poorer in paraplegic compared with tetraplegic patients. The SCIM is limited to the assessment of basic daily life relevant tasks. Paraplegic patients show a higher level of independent daily functioning compared with tetraplegic patients and, therefore, may be more interested in achieving functions that go beyond the level of functioning that the SCIM assesses, namely the participation in leisure and vocational activities, which are known to be important for the subjective well-being. 28 This could also explain why the relationship became worse in tetraplegic patients at 6 months postinjury, when many patients had finished their rehabilitation. At this stage, the improvements in upper extremity function might have reached a plateau and the tetraplegic patients might aim for more participative goals. Alternatively, at 6 months, the mobilization of psychological resources became poorer, as postulated in the “air bag effect.” 29
For future studies, we recommend to formulate questions that should assess patient-reported independence more specifically as activity questions (eg, “How would you score your restrictions on your daily activities in self-care, bladder and bowel management, and mobility?”). We cannot exclude that some patients might not have understood the patient-reported interference with ADL question well, or their attention became less during the 15-minute interview.
The second main finding was that the investigator-determined score could be explained by the small number of factors that we selected, whereas the patient-reported independence score was more difficult to explain. Initially, the motor score and age accounted for a small percentage of the investigator-determined independence measure, which can be explained by factors that can occur early during rehabilitation such as initial complications or cardiovascular instability. At 6 months after SCI, however, the estimation was much better and comparable to a previous study performed in persons 2 years after SCI. 30 We assume that the slightly smaller percentage at 12 months postinjury can be explained by other factors, such as supportive family members who take over daily life tasks of the patient. Indeed, most patients were at this time in their home environment.
Still, a perfect estimation was not achieved indicating that the motor score and age cannot replace the SCIM to assess performance. The lack of perfect agreement can be caused by various factors. For example, despite the high relevance of respiratory function, the International Standards do not include the testing of respiratory muscles. 31
In our study, age influenced objective independence negatively, which confirms earlier findings of a reduced recovery of ADL in older patients,12,13,32,33 although several studies found no influence of age.34-36
Although several studies have investigated the contribution of factors such as neurological impairment, disability, and pain for the quality of life or subjective well-being in persons with SCI,37,38 we are unaware of studies investigating the influence of pain and depression on functional outcome itself within the first year after SCI. The intensity of pain, however, did not contribute to the investigator-determined and patient-reported daily life performance. Although this is in line with a study showing that changes in pain interference status are unrelated to changes in self-reported handicap, 15 many studies report a negative influence of pain on ADL.17,38 As most of these studies were performed in community-based samples, we assume that this difference is the result of our-1 year time window. Persons with SCI undergoing rehabilitation are more likely to focus on functional improvement, whereas psychological and social factors become more important in the domestic environment 29 and might enhance the perception of pain.
Similar reasoning may explain the lack of impact of the severity of depression on ADL, when assessed objectively. Depression is not an inevitable consequence following SCI. 39 Indeed, although the depression intensity did not change after 3 months postinjury, it was not until 6 and 12 months after injury that it influenced the patient-reported independence. Again, this might depend on the availability of coping strategies. 29 Depression may be more likely after discharge from the rehabilitation center, when patients are confronted with their disabilities in their own environment.40,41 As factors other than just functional recovery appear relevant for patient-reported independence, and investigator-determined independence was higher compared with patient-reported independence, long-term psychological coaching might prove useful in assisting the SCI patient to regain a positive perception.
Study Limitations
The present findings should be interpreted in light of methodological issues. (a) Whereas the “zero-independence” SCIM score is well defined, the patient-reported “zero-independence” may vary considerably among patients and tasks. Correlations might improve when the patient-reported zero-independence might become adapted according to the perceived full-restriction of each individual SCIM item. One possibility to do so could be to rank the criteria of each SCIM item from best to worst and ask the patient which criterion he considers full-restrictive. Then, the sum of the item score differences between the personal full-restricted score and the highest score could be correlated with the investigator-determined SCIM scores. (b) The use of (originally) a 10-point patient-reported scale versus a 100-point SCIM sum scale is likely to cause scatter and thus reduce the size of the correlation coefficients. However, this cannot be the sole explanation of the poor to moderate magnitude of the correlations. (c) With respect to the generalizability of our results, the patients who were included showed similar characteristics to the general population of SCI patients, 42 except for age, which was on average 12 years older as reported, and the cause (traumatic SCI), because of our inclusion criteria. (d) The finding that a significant increase in SCIM score was observed only between 1 and 3 months is not in line with previous studies that reported improvements up to 1 year after SCI,24,43 but can be explained by the small number of subjects and large between-subject variability, as AIS A to AIS D patients were included in these analyses. (e) We presented little information about the pain symptoms. However, the occurrence of pain in our sample appears representative, as the prevalence of 65% fits excellently with the mean prevalence of pain in SCI 44 and the average pain intensity is similar to previously reported levels. 45
Implications
Selecting the best primary outcome measure for experimental trials must take into consideration that the patient-reported and objective level of ADL performance and independence can differ to a large degree. Although the subjective perception of independence (ie, the domain of participation according to the ICF) might be the most valuable criterion for a patient who would participate in such a trial, we would disagree with its use as a primary outcome measure, as actual functional improvements might be masked by a patient-reported measure. The results of studies that used a patient-reported independence measure might be difficult to compare to studies that applied objective independence measures and vice versa. Studies that investigate the influence of negative symptoms, such as pain or depression, tend to assess interference on ADL, rather than use an investigator-determined measure reflecting disability. 17 Previous studies reported that pain-related interference can be distinguished from impairment-related interference. 46 We extended these findings to show that investigator-derived and patient-reported independence differ within SCI patients.
In conclusion, investigator-determined independence scores should be applied to document functional recovery during rehabilitation, whereas patient-reported independence measures could be useful to evaluate the impact of negative emotional symptoms on perceived independence and quality of life.
Footnotes
Acknowledgements
We thank the Neurosciences Center Zurich and EM-SCI study group, especially René Koller and Dr F. Röhrich. The following centers participated in this study: K. Röhl, MD, Berufsgenossenschaftliche Klinik Bergmanstrost, Halle (Germany); N. Weidner and R. Rupp, University Clinic of Heidelberg (Germany); R.D. Sambale, MD, Orthopädische Klinik Hessisch-Lichtenau (Germany); J. Harms, MD, SRH Klinikum Karlsbad-Langensteinbach (Germany); D. Maier, MD, Berufsgenossenschaftliche Klinik in Murnau (Germany); Y.-B. Kalke, MD, Rehabilitationskrankhaus Ulm (Germany); and finally, A Curt, MD, and M. Schubert, MD, Balgrist University Hospital, Zurich (Switzerland).
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article:
The study was supported by the International Foundation for Research in Paraplegia (IFP) and the International Spinal Research Trust (ISRT; Stage II of the Clinical Initiative: CLI006).
