Abstract
Keywords
Introduction
Huntington disease (HD) is a chronic neurodegenerative disorder affecting the striatum, resulting in widespread loss of medium spiny projection neurons, which constitute the majority of the population of striatal cells. 1 The use of embryonic striatal grafts in the treatment of HD is currently at a trial stage within Europe and the United States, with preliminary results indicating an overall positive reduction of negative symptoms in some but not all patients. 2-6 Experimental rodent models of HD, using excitotoxic lesions of the striatum, display anatomical, 7 behavioral, 8,9 and physiological 10,11 correlates, which share a symptomatic similarity with the human disorder. 12
Since the initial observations 4 decades ago, 13 a growing body of evidence has emerged that environmental enrichment can cause lasting changes in anatomical, physiological, and cognitive functions. 14-17 Environmental enrichment is defined as measures taken to enhance the sensory and motor experiences of an organism. Though studies involving enrichment do not conform to any form of standardized protocol, all studies use manipulations of the animals’ housing environment in order to increase social interaction, exploratory behavior, play, activity, and exercise levels. 14 Common environmental manipulations taken in order to enrich the environment that animals inhabit include increasing the number of animals housed in a single cage, increasing the cage size, and introducing many novel items. 14 Remarkably, such subtle environmental manipulations can have an impact on neuronal development and a range of functions.
Rosenzweig et al 13 demonstrated that environmental enrichment produces both structural changes in the brain, such as increased cortical thickness, and morphological changes, including increased soma size, dendritic branching, and density of dendritic spines. 13 More recently, it has been demonstrated that environmental enrichment can also have a positive impact on a number of cognitive and motor tasks, 18-20 with the observed enhancement of behavioral performance considered to be dependent on morphological, physiological, and biochemical changes induced by the enriched environment. Furthermore, it has been shown that environmental enrichment can have a direct impact on neuronal transmission 21,22 and can facilitate the induction and expression of synaptic plasticity within the hippocampus. 23,24
The adult brain is at its most plastic during development and following injury, and it has been known for more than 2 decades that environmental enrichment can enhance the integration and function of cell transplants in animal models of a range of neurodegenerative diseases. 14 Within rodent models of HD, environmental enrichment has been shown to increase behavioral performance on tasks that rely on the striatal hemisphere controlled by the graft. 15,25-27 Furthermore, it has been demonstrated that grafts in animals housed in enriched environments receive denser innervation from the host dopamine projection system 25 and increased levels of brain-derived neurotrophic factor (BDNF). 25 Similar increases in BDNF levels have been seen in both control animals 28 and HD transgenic mice, 29-31 where it has been shown to slow disease progression. 29-31
Though it has been clearly demonstrated that environmental enrichment can influence graft-mediated behavioral performance, anatomical characteristics, and biochemical properties, it is still unclear whether enrichment can influence physiological properties such as synaptic plasticity expressed from striatal grafts. In a previous study, we have demonstrated physiological plasticity at the host-graft within striatal grafts, 10,11 which we hypothesize provides the cellular mechanism for graft-induced recovery of motor learning. In the present study, we demonstrate that environmental enrichment increases the expression of BDNF, chances of successful expression, and levels of expression of host-graft corticostriatal long-term potentiation (LTP). This suggests that environmental enrichment can facilitate the functional integration of transplanted striatal cells into the adult host circuitry.
Materials and Methods
Animals
A total of 60 (Table 1) male C57BL/6 mice (Harlan, UK) were used as control animals and hosts for transplantation. Animals were housed in a temperature- and humidity-controlled (21°C and 50%, respectively) room, with a 12-hour light and 12-hour dark cycle (lights on during the day) and were given free access to food ad lib. Experiments were undertaken in accordance with the UK Animals (Scientific Procedures) Act 1986.
Total Number of Cases and Recordings Made
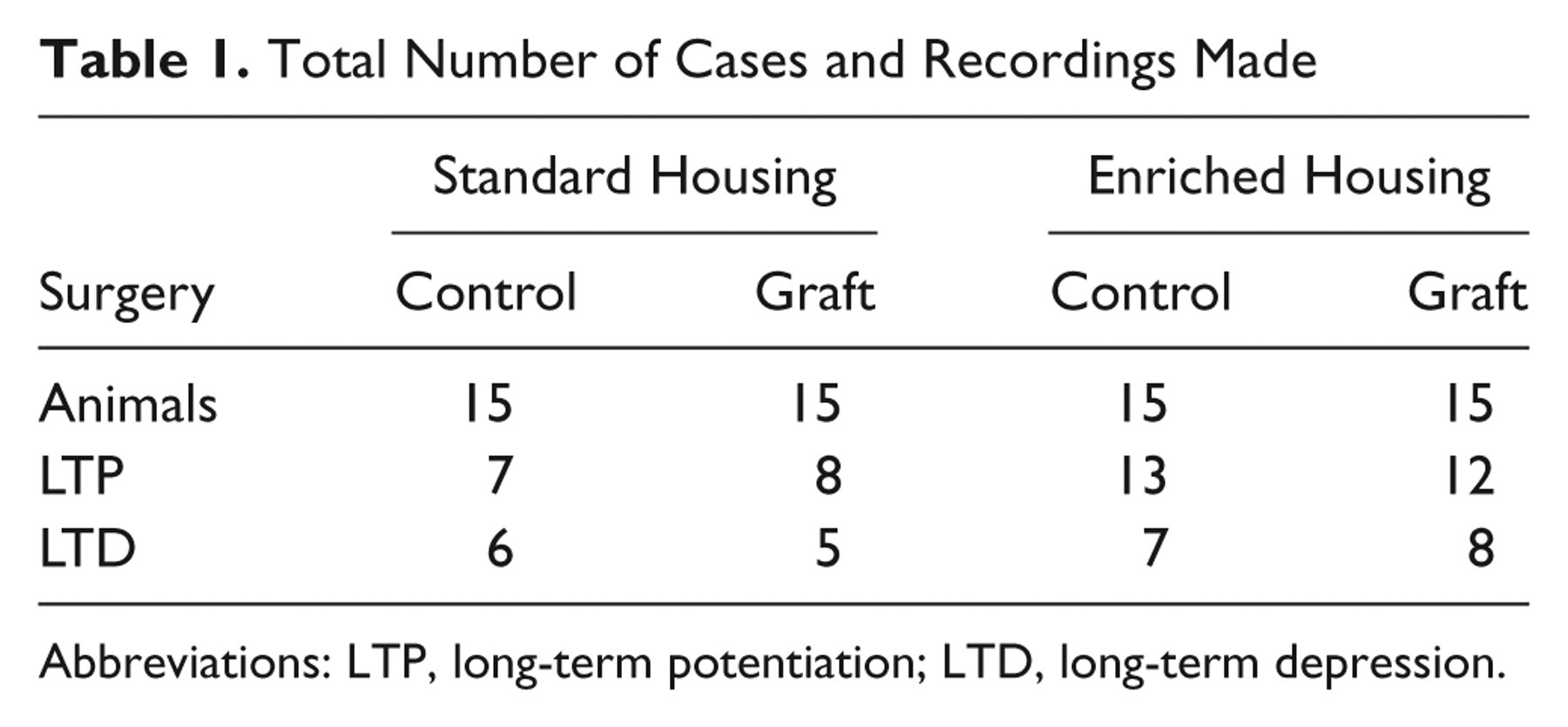
Abbreviations: LTP, long-term potentiation; LTD, long-term depression.
Graft tissue was collected from the PrP-L-EGFP-L transgenic mouse, 32 which exhibits endogenous expression of green fluorescent protein (GFP) under control of the PrP promoter in all cells. 32
Enrichment Protocol
On arrival, animals were assigned to the enriched or nonenriched housing groups. Animals housed within enriched environments were housed in groups of 6, within a 40 × 25 × 12 cm3 cage, equipped with a number of novel toys and environmental stimuli designed to induce increased social interaction and exploratory behavior (Bio-Serv, New Jersey; illustrated in reference 14). All enriched environment cages contained a horizontal mouse running wheel, designed to provide the animals with free access to exercise (Bio-Serv).
Animals housed within nonenriched environments were housed in groups of 2, within a 30 × 12 × 12 cm3 sized cage devoid of any environmental stimuli apart from the standard mouse bedding plus a single cardboard tube, as required under Home Office guidelines.
Lesion Surgery
Mice were anesthetized in an induction chamber by inhalation of anesthetic (Isoflurane, Abbott, UK), using O2 and NO as carrier gases, and placed in a stereotaxic frame (Kopf Instruments, Tujunga, California). Unilateral excitotoxic lesions of the dorsal striatum were made by injecting 2 × 0.24 mL of 0.12 M quinolinic acid (Cambridge Research Biochemicals, UK), dissolved in 0.1 M phosphate-buffered saline (pH = 7.4), at 2 depths within the same needle track in the left neostriatum. Each injection was administered over 1 minute via a 30 gauge stainless steel cannula connected to a microdrive pump, with a further 2 minutes for diffusion. The injection coordinates were as follows: A = +1.0, L = +1.8, V = 3.1 and 2.5, with measurements in millimeters anterior (A) to bregma, lateral (L) to the midline, and vertical (V) below dura, and the nose bar was set in the same horizontal plane as the interaural line. All animals received postoperative 5 mL glucose saline subcutaneously in the neck and 0.15 mL diazepam intramuscularly in the hind leg. For analgesia, 500 mg/L paracetamol was dissolved in drinking water for 48 hours postsurgery.
Harvesting of Embryos and Graft Tissue
Time-mated pregnant mice were killed at embryonic day (E) 13 by terminal anesthesia and decapitation. The uterine horns were removed and placed in 45 mL of dissection medium containing 95.7% Dulbecco modified Eagle medium, 2.2% of 30% glucose, 1.6% of 7.5% NaHCO3, and 0.5% 1M HEPES. Embryos were removed one at a time from the uterine horn for dissection in a sterile Petri dish. Crown–rump lengths were measured, and the embryos were then killed by decapitation. The brains were removed and the whole ganglionic eminence was dissected bilaterally according to a standard protocol. The tissue pieces from this were transferred to a sterile 1.5-mL Eppendorf tube containing dissection medium and kept on ice. Once the tissue pieces had settled, they were washed 3 to 4 times with sterile dissection medium, then incubated at 37°C for 20 minutes in 1.5-mL trypsin/DNase solution containing 0.1% bovine trypsin (Worthington, UK) and 0.05% DNase (Sigma, UK) in dissection medium. Following incubation, the trypsin/DNase solution was removed, and the tissue was washed 4 times with 200 µL of 0.05% DNase solution. The tissue was then mechanically dissociated using a Gilson pipette to obtain a cell suspension. Cell numbers and viability were calculated by the trypan blue dye exclusion method using a hemocytometer. Cell suspensions with viability of 95% or more were resuspended in DNase solution and used for the transplant procedure.
Transplant Surgery
Seven days postlesion, host mice were anesthetized with inhalation anesthetic and placed in a stereotaxic frame (as above). Each animal received 2 × 1 µL aliquots of graft suspension (approximately 200 000 cells per µL, 400 000 cells per graft), injected over 1 minute via a 10-µL Hamilton syringe at each of the 2 lesion injection sites A = +1.0, L = +1.8, V = 3.1 and 2.5. Following the injection, 2 minutes was allowed for diffusion before the syringe needle was withdrawn. All animals received postoperative care, as above.
Slice Preparation
Mice were killed by cervical dislocation and decapitated. The brains were quickly removed and placed into ice-cold artificial cerebrospinal fluid (aCSF; composition 126 mM NaCl, 2.5 mM KCl, 2.5 mM CaCl2, 1.3 mM MgSO4, 1 mM NaHPO4, 26 mM NaHCO3, and 11 mM
Electrode Placement
For all experiments, slices were viewed under 50× magnification using an upright microscope (Olympus, UK) under bright-field phase illumination for positioning the electrodes and under incident ultraviolet illumination to visualize fluorescent GFP expression in grafted cells.
Stainless steel monopolar stimulating electrodes were placed in the host corpus callosum 1 to 2 mm away from the graft site. Recording electrodes were produced from borosilicate glass capillaries (Harvard Apparatus, UK), with an external diameter of 1.5 mm and an internal diameter of 0.86 mm and pulled using a motorized micropipette puller (Sutter Instrument Co, Novato, California); they were then filled with 1 M KCl. The recording electrode was placed within the core of the graft region (region of dense GFP positive cells) in a ventral location to the stimulating electrode. Control recordings were made from the intact striatum (in the absence of lesions, and graft tissue was not present), with the electrode in a similar location in midstriatum and at a similar distance ventral to the stimulating electrode.
Stimuli and Recording
Stimulus patterns were generated using a Master 8 pulse generator (Intracel, UK) and a Neurolog stimulus isolator (Digitimer, UK). Responses were amplified using a differential amplifier (Warner Instrument Corp, Hamden, Connecticut) and Axoclamp (Axon Instruments, UK). Responses were monitored and stored for offline analysis using custom software written in LabView (National Instruments, Austin, Texas) running on a Macintosh G4 computer.
The initial slope of the field excitatory postsynaptic potential (fEPSP) was used as the measure of synaptic strength. Once a response had been gained, the slice was submitted to a 30-minute rest period where the slice was stimulated every 5 to 10 minutes until the fEPSP slope had stabilized.
Experimental Protocol
Initially, an input/output (I/O) curve was computed. The fEPSP slope was measured across a stimulus intensity of 1 to 10 mA, in steps of 1 mA. Four stimuli were given every 10 s for each current intensity. Following I/O curve measurement, the slices were allowed a 5-minute rest period. A stimulus intensity that evoked roughly 60% of the maximum response size was selected, and stimuli were delivered to the slice once every 20 s until a period of response stability was seen, defined as a <5% response size change over the 20-minute period.
Long-term depression (LTD) was expressed from both grafted and control tissue via high-frequency stimulation (HFS), under physiologically normal conditions (aCSF containing magnesium ions). The HFS comprised θ-burst stimulation in 6 trains, each of 10 bursts at 5 Hz and each burst consisting of 4 pulses at 100 Hz, with a pulse width of 0.4 ms and an intertrain interval of 20 s. LTP was expressed from both grafted and control tissue, via the same HFS protocol but in the absence of magnesium ions from the aCSF solution. 11,33 Following tetanus, slices were stimulated at baseline levels (0.05 Hz), and recording continued for a further 65 minutes. Successful expression of synaptic plasticity was defined as a greater than 5% change in response size, lasting at least 60 minutes post HFS.
Brain-Derived Neurotrophic Factor ELISA
Three animals from each group (Standard Control, Enriched Control, Standard Graft, Enriched Graft) were sacrificed via cervical dislocation and decapitated, and their brains were quickly removed and submersed in ice-cold aCSF containing 1 mM kynurenic acid. Brains were hemisected, and samples of the cortex, striatum, hippocampus, and cerebellum were taken and stored at −80°C in preweighed Eppendorf tubes. The tubes containing the tissue samples were weighed to determine the weight of the sample. The levels of BDNF present in the tissue were ascertained by following the protocol for the ELISA (enzyme-linked immunosorbent assay) method using
Data Analysis and Statistics
Physiological data were normalized to the baseline levels, defined as the average response size over the 20 minutes of baseline stimulation. The normalized values following tetanus were expressed as a percentage of the baseline response and averaged into 2-minute bins. Results are presented as the mean result gained ± the standard error of the mean.
Data from electrophysiological experiments were analyzed offline by multifactorial analyses of variance using the Genstat statistical package (v.10.1; VSN International, Hemel Hempsted, UK), with post hoc tests correcting for multiple comparisons as appropriate. To measure the chance of successfully expressing synaptic plasticity, the total number of animals, slices, and cases of successful synaptic plasticity expression (Table 1) were compared using a χ2 test, using the Genstat statistical package (v.10.1; VSN International, Hemel Hempsted, UK).
Data from the BDNF assays were subjected to log transformation in order to normalize the variance between groups prior to multifactorial analysis.
Results
Extracellular in vitro slice recordings were obtained from a total of 60 animals across 2 surgical groups—control and graft—and 2 enrichment paradigms—enriched and standard housing (Table 1).
All control recordings were obtained from animals that had no lesion or graft surgery performed on them. Responses evoked from the control striatum appeared consistent with those previously reported. 11,34
A total of 30 mice were transplanted with embryonic striatal tissue. Following preparation for in vitro recording, grafted cells could be identified under fluorescent illumination in corticostriatal slices from all 30 cases.
Effect of Enrichment on Synaptic Plasticity from the Control Striatum
The effect of environmental enrichment on LTP and LTD recorded from the normal striatum in control animals is illustrated in Figure 1. Under physiologically normal conditions, HFS resulted in the expression of LTD from animals housed in both enriched and standard environments with no significant difference in the level of depression observed between the 2 housing groups (Figure 1C;
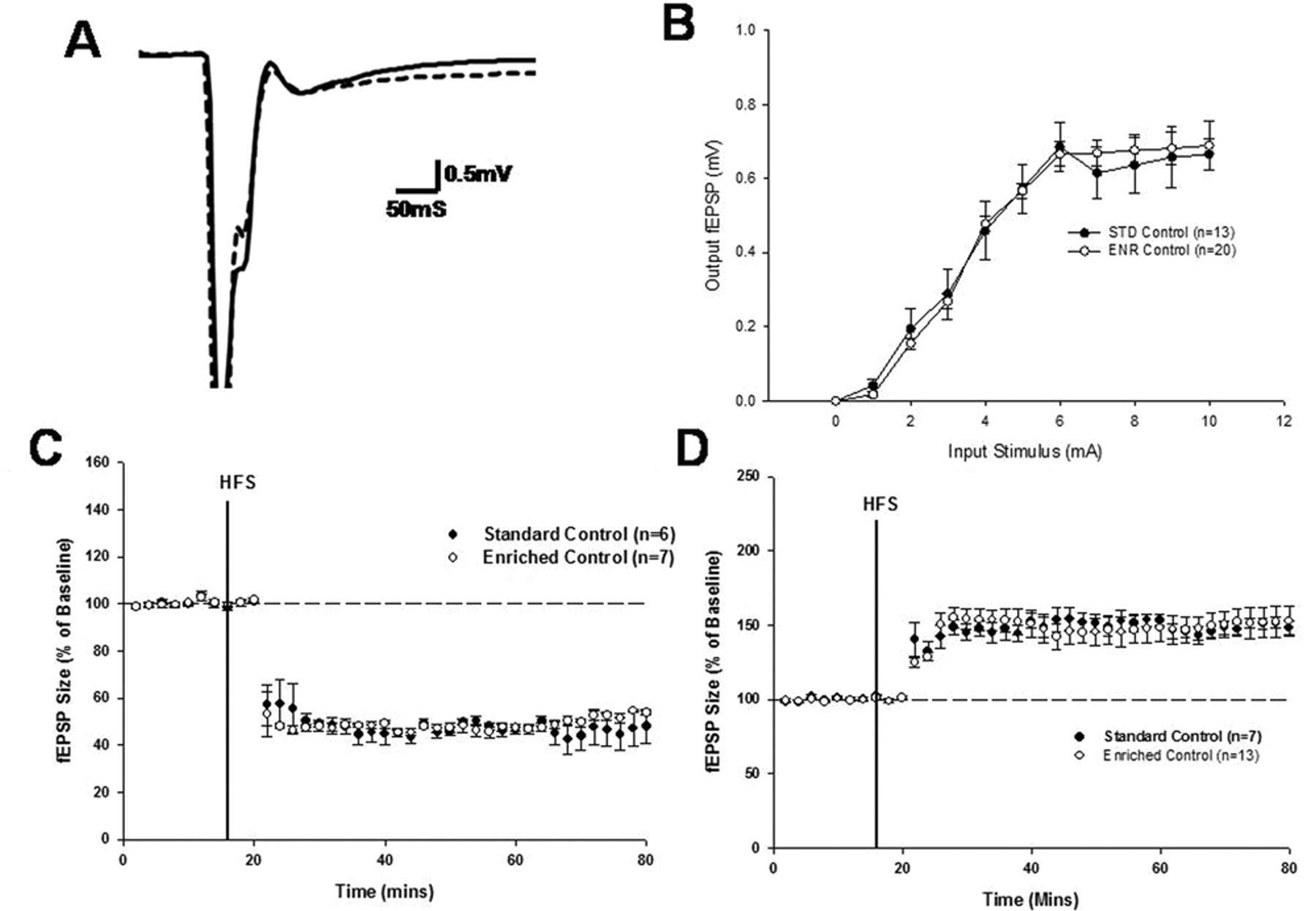
The effect of environmental enrichment on synaptic plasticity within the normal striatum: A, representative fEPSP trace from standard and enriched control recordings (bold line, standard recording; dashed line, enriched recording. B, input/output curve generated during enriched and standard recordings. C, expression of LTD in standard control and enriched control recordings. d, expression of LTP in standard control and enriched control recordings. Abbreviations: fEPSP, field excitatory postsynaptic potential; LTD, long-term depression; LTP, long-term potentiation; HFS, high-frequency stimulation; STD, standard housing; ENR, enriched housing.
As previously demonstrated in the normal striatum
11,34
and in embryonic striatal grafts,
10,11
removal of magnesium from the aCSF perfusate solution resulted in the preferential expression of LTP following HFS. A similar level of LTP was recorded from animals irrespective of whether they had been housed in enriched or standard environments (Figure 1D; Groups,
Effect of Enrichment on Synaptic Plasticity From Striatal Grafts
As reported previously, we were able to record both LTD and LTP from grafted striatal neurons in response to tetanic stimulation of host cortical afferents (Figures 2C and 2D, respectively). Under physiologically normal conditions, HFS resulted in the expression of LTD of a magnitude that was comparable to that seen in the normal striatum (Figure 1C) and that did not differ between animals housed in the enriched and standard environments (Figure 2C;
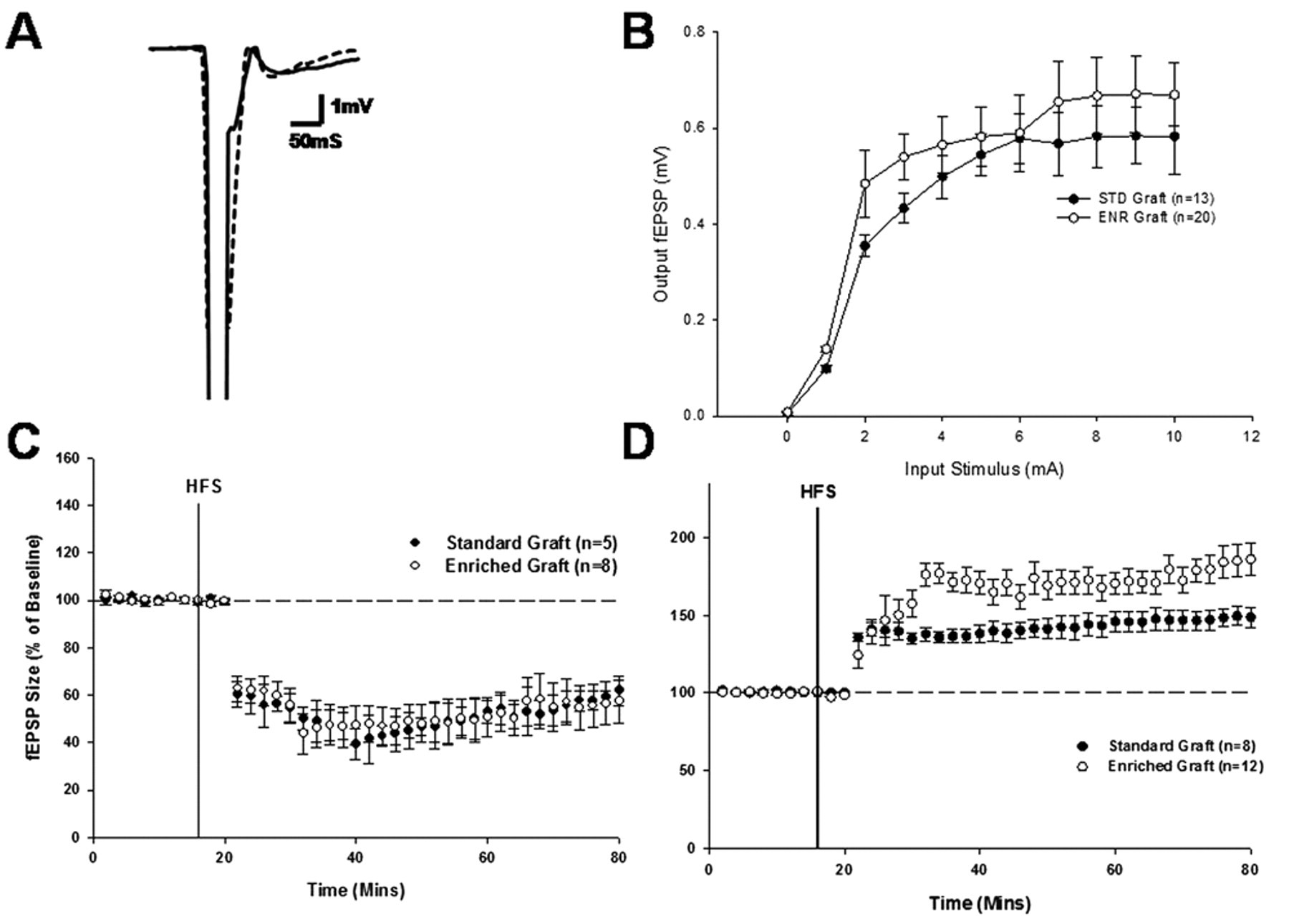
The effect of environmental enrichment on synaptic plasticity within the grafted striatum: A, representative fEPSP trace from standard and enriched graft recordings (bold line, standard recording; dashed line, enriched recording). B, input/output curve generated during enriched and standard recordings. C, expression of LTD in standard graft and enriched graft recordings. D, expression of LTP in standard graft and enriched graft recordings. Abbreviations: fEPSP, field excitatory postsynaptic potential; LTD, long-term depression; LTP, long-term potentiation; HFS, high-frequency stimulation; STD, standard housing; ENR, enriched housing.
Effect of Enrichment on Hippocampal Synaptic Plasticity
Previous studies of the effects of environmental enrichment on synaptic plasticity have demonstrated a facilitation of LTP recorded from the CA1 region of the hippocampus. HFS of the CA1 resulted in the expression of LTP from animals housed in both enriched and standard environments (Figure 3C), but there was no significant difference in the level of potentiation observed in each housing group recording (
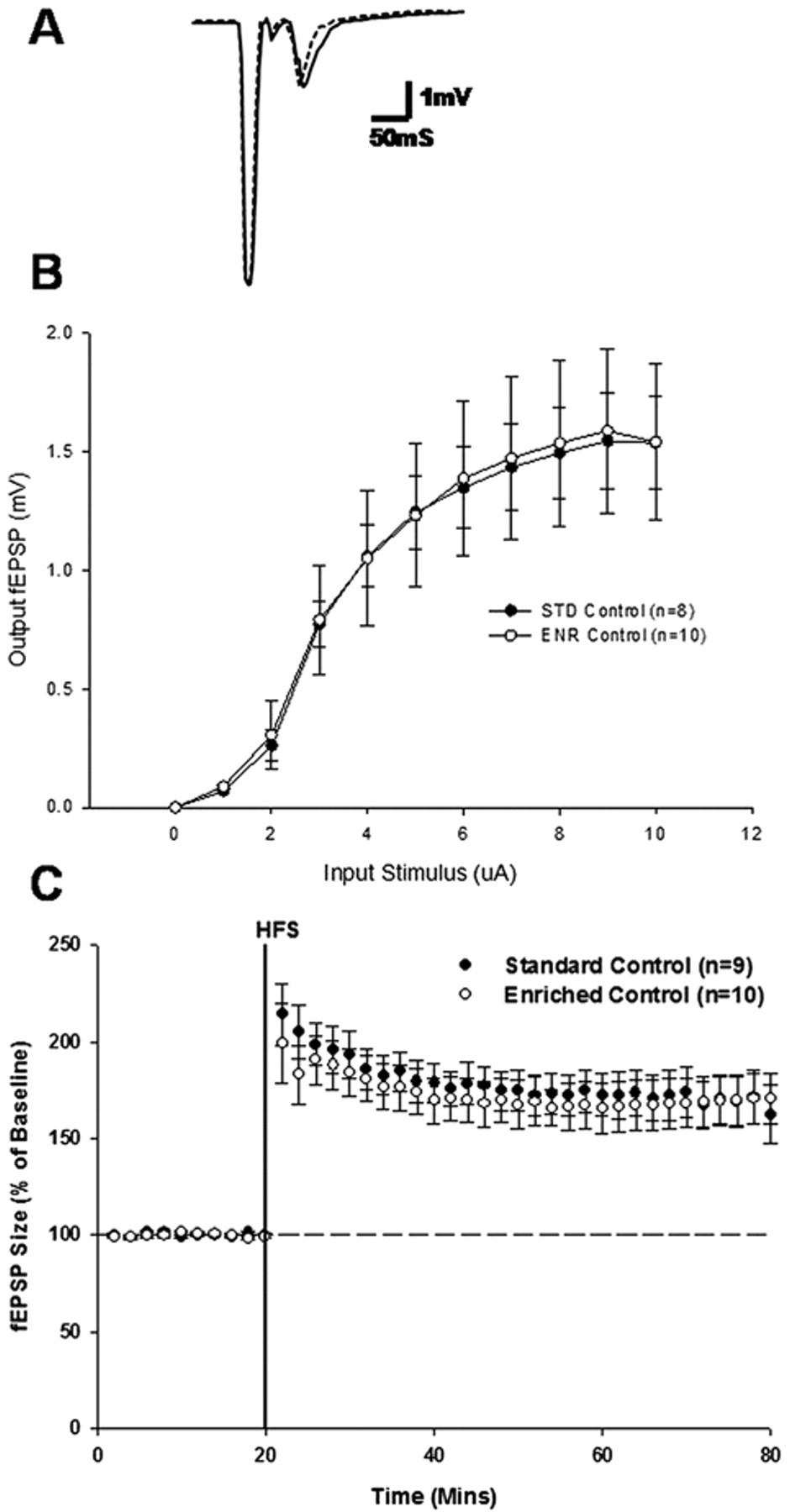
The effect of environmental enrichment on synaptic plasticity within the hippocampus. A, representative fEPSP trace from standard and enriched recordings (bold line, standard recording; dashed line, enriched recording). B, input/output curve generated during enriched and standard recordings. C, expression of LTP in standard control and enriched control recordings. Abbreviations: fEPSP, field excitatory postsynaptic potential; LTP, long-term potentiation; HFS, high-frequency stimulation; STD, standard housing; ENR, enriched housing.
Assessing the Levels of BDNF Following Enrichment
BDNF levels in the striatum, hippocampus, cortex, and cerebellum from the intact hemispheres of control and grafted rats housed in standard and enriched environments are shown in Figure 4. Environmental enrichment induced a significant increase in BDNF levels across all brain regions (
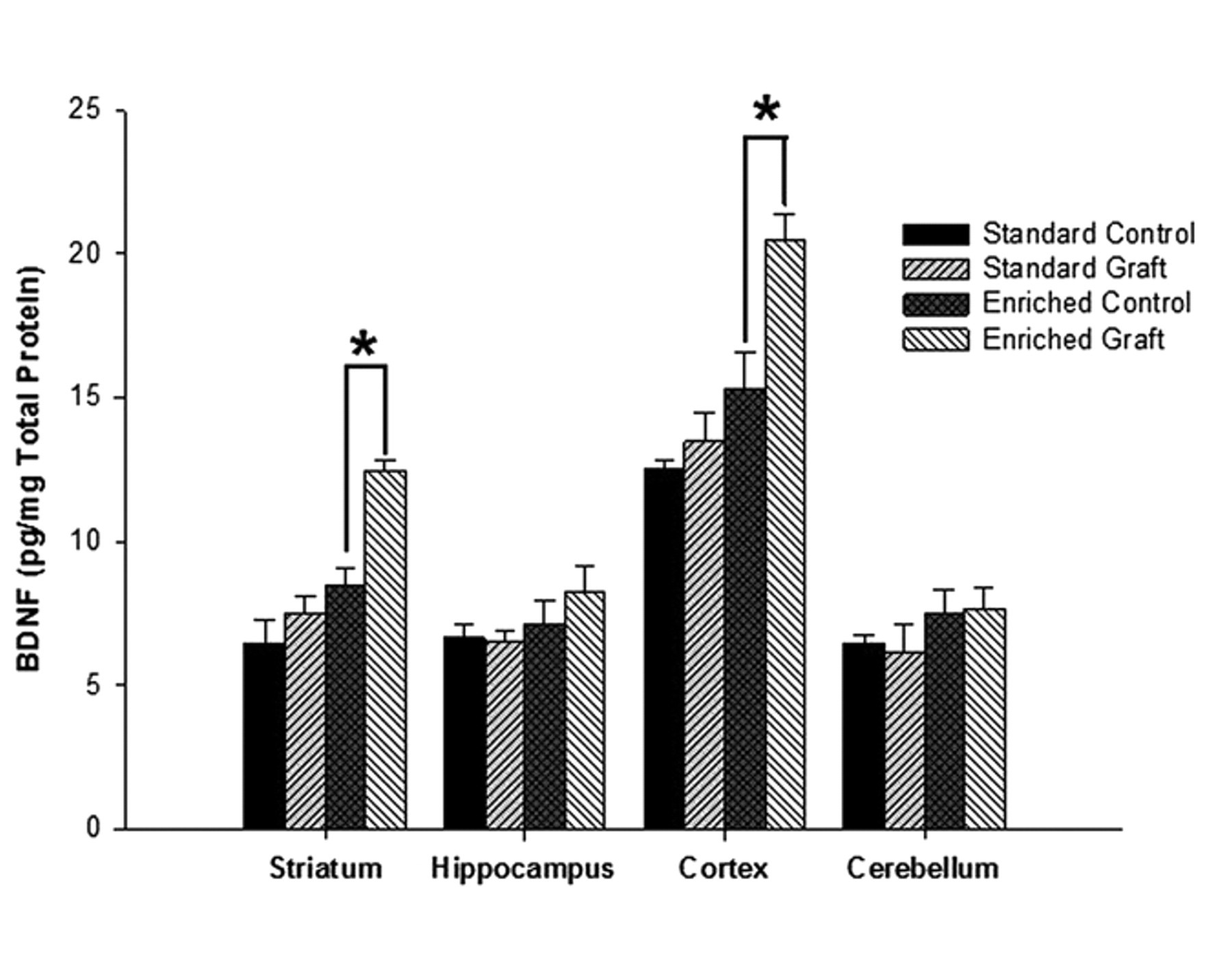
The effect of environmental enrichment on BDNF levels: histogram plot demonstrating the mean concentration of BDNF (pg/mg of total protein) in the striatum, hippocampus, cortex, and cerebellum samples collected from control and grafted animals housed in standard and enriched environments. Abbreviation: BDNF, brain-derived neurotrophic factor.
Discussion
In the present study, we demonstrate that (1) environmental enrichment facilitates the chance of induction and level of expression of host-graft LTP, (2) environmental enrichment increases the levels of BDNF within the striatum and cortex of grafted animals, and (3) the enrichment-enhanced LTP is limited to grafted animals.
The mammalian brain is most plastic during development and following injury. Therefore, embryonic striatal grafts, which represent a situation where developing embryonic tissue is transplanted into the damaged adult nervous system, provide a strong target for the effects of enrichment. Studies of the effects of enrichment on graft function have demonstrated increased performance on motor and cognitive tasks that impinge on host-graft integration, 15,25-27 suggesting that the observed improvement following enrichment is dependent on the integration of transplanted neurons into the host circuitry. We have recently demonstrated that striatal grafts are capable of expressing bidirectional synaptic plasticity, consistent with that observed within the normal striatum, 11 and we have hypothesized that the expression of synaptic plasticity provides a cellular correlate for the phenomenon “learning to use the graft,” whereby striatal grafts provide the cellular substrate for new learning to take place. 35,36 However, in the present study, we demonstrate that environmental enrichment has little detectable effect on physiological plasticity in the normal striatum but enhances the functional plasticity of striatal grafts, manifesting as an increase in the chance of induction and level of LTP expressed. This suggests that enrichment further improves the functional integration of grafted cells into the host corticostriatal circuitry.
Previous studies have demonstrated that environmental enrichment can enhance behavioral performance on motor learning tasks under the control of the grafted striatum. 15,25-27 Although the mechanisms of this increased behavioral performance are not fully understood, enrichment can induce morphological changes in the grafted striatal cells, including increased cell size, spine density, dopaminergic innervation, and BDNF expression. 15,25-27 In the present study, we demonstrate that environmental enrichment facilitates the level of LTP expressed from striatal grafts while having no effect on the level of LTD.
Enrichment ultimately results in an improvement in performance on motor and cognitive tasks, 15,25-27 and such improvement correlates with changes in cellular morphology and levels of BDNF expression. 27 Our findings further demonstrate that enrichment increases the chance of induction and level of LTP expressed, which we hypothesize provides a link between the cellular correlates and observed motor and cognitive improvements. 15,25-27 However, it must be noted that additional studies in which this increased induction and level of LTP expressed is blocked, resulting in impaired behavioral improvement, would establish causality for this theory. The cellular mechanisms underlying enrichment-induced facilitation of graft LTP remain unclear, although a plausible mechanism is that enhanced sensory and motor experiences provided by the enriched environment can influence graft LTP via growth factor–mediated changes in cellular morphology and physiology.
Our results confirm earlier findings, in that enrichment results in the increased expression of growth factors such as BDNF. 15,25-27,29 31 Inhibition of BDNF has been shown to impair hippocampal LTP 37 ; thus, the observed higher levels of BDNF may act directly on transplanted striatal neurons, facilitating graft LTP. This enrichment-induced, BDNF-mediated effect may directly affect graft LTP because BDNF knockout mice demonstrate impaired LTP, 38,39 which is reversed by transfection with lentiviruses expressing BDNF. 38 Furthermore, spatial learning has been shown to activate BDNF expression 40 while inactivation of BDNF impairs spatial learning. 37 Also, mice lacking the BDNF receptor show impaired learning, 41 whereas mice overexpressing the BDNF receptor display improved cognitive performance. 42 However, it is important to note that enrichment causes activity-dependent changes in the morphology of transplanted cells, such as increased cell size, spine density, and dopaminergic innervations, 15,25-27 all of which would provide cellular mechanisms for the facilitation of LTP. Therefore, although the exact mechanisms linking increased levels of BDNF with facilitated expression of LTP are not fully understood, it is clear that the enrichment has positive effects on the expression and levels of both.
Whereas the enrichment paradigm used in this study showed no change in the level of LTP or LTD expressed from the normal striatum, environmental enrichment causes an increased chance of successful expression of LTP (Table 1) within the normal and grafted striatum. Similar findings have been found with regard to CA1 LTP. 23,24 It has been suggested that increased expression of synaptic plasticity may provide a physiological mechanism for the observed increases in behavioral performance following enrichment. 23 Therefore, though the enrichment paradigm used in the experiments presented here does not affect the level of corticostriatal LTP and LTD, it does result in an increased chance of induction of corticostriatal synaptic plasticity, which may have an effect on behavioral performance.
In conclusion, the results of this study clearly demonstrate that environmental enrichment can facilitate the chance of successful expression and level of graft LTP, thus suggesting that graft performance can be improved posttransplantation via environmental enrichment and that graft LTP displays a level of plasticity to cellular mechanisms. Therefore, to improve integration and graft performance, it is imperative to further understand, and identify, the cellular mechanisms underlying the enrichment effect. Furthermore, such potential cellular targets could be manipulated pretransplantation and posttransplantation to further increase the efficacy of graft function. As cell transplantation in HD moves toward the clinical trial phase, with potential as a future treatment, the cellular mechanisms underlying the enrichment effect need to be clearly understood in order to maximize the benefit of rehabilitation.
Footnotes
The author(s) declared no potential conflicts of interest with respect to the authorship and/or publication of this article.
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Medical Research Council (UK).
