Abstract
Background. Rapid rate magnetic stimulation of human sacral nerve roots can reduce stress incontinence. Objective. To test stimulation for urgency incontinence due to lumbosacral plexopathy. Methods. The authors studied patients with intractable neurogenic urinary dysfunction due to lumbosacral nerve injuries. Eligible patients were randomized to receive either real or sham repetitive lumbosacral magnetic stimulation (rLMS; 15 Hz with a total of 1500 pulses/session) for 10 sessions. Evaluation was performed before starting treatment, immediately after the 5th and 10th treatment session, and 1 month later, using rating scales to score urinary dysfunction and a visual analogue scale (VAS) to assess lower back pain. Posterior tibial nerve F-wave and H-reflexes were tested before and after treatment. Results. Both treatment and control groups were comparable for baseline characters and sphincteric disturbances. The mean number of voids and incontinence per 24 hours was significantly reduced in patients who received rLMS. The improvement was maintained 1 month after the end of treatment. Patients receiving real-rLMS also reported an improvement in VAS ratings. Improvement in F-wave and H-reflex latencies after real stimulation (P = .002 and P = .036, respectively) was found as well. Conclusion. rLMS at 15 Hz may improve urinary dysfunction secondary to lumbosacral nerve injury. Further trials can determine the profile of patients who may benefit and optimal stimulation parameters.
Introduction
Neuromodulation through sacral nerve stimulation (SNS) is a relatively new and promising treatment for intractable fecal and urinary incontinence that involves chronic electrical stimulation of the sacral nerve roots. 1,2 It is performed in 2 stages: an initial temporary test phase to assess symptomatic response (percutaneous nerve evaluation test with a stimulation period of 3 to 7 days) followed by insertion of a permanent implant in subjects deemed to have a satisfactory response. Improvement in the symptoms of urinary incontinence and fecal incontinence has been reported. 1-3 However, treatment is rather invasive and the outcome varies from patient to patient. In a systematic review, Brazzelli et al 4 reported that SNS is effective for reducing symptoms in patients with urge incontinence. Nevertheless, adverse events (pain and infection) occurred in about half of the implanted cases and surgical revision (replacement and repositioning of the implanted pulse generator) was performed in 33%. 4 The electrical device is also expensive. Manganotti et al 5 applied repetitive magnetic stimulation of the sacral roots for women suffering from stress incontinence and reported a short-term effect on aspects of the quality of life.
Electrical stimulation of the sacral nerves activates both sensory afferents and motor efferents; the latter produces contraction of the anal sphincter muscle, which leads to additional natural sensory stimulation that adds to the electrically induced afferent activity. Sheldon et al 6 reported that sacral nerve stimulation reduces cortico-anal excitability in patients with fecal incontinence. Repetitive high-frequency lumbosacral magnetic stimulation (rLMS) at 15 Hz was also found to change cortico-anal excitability 7 in healthy subjects and may therefore have a therapeutic effect in patients with sphincter dysfunction. We investigated whether high-frequency repetitive and consecutive rLMS can improve the urinary disturbances associated with lumbosacral plexopathy.
Methods
Patients with symptoms suggestive of overactive bladder (OAB) with lower urinary tract dysfunction secondary to lumbosacral plexopathy (LUTS) as documented by magnetic resonance imaging (MRI) were recruited from the outpatient Neurology and Rheumatology & Rehabilitation Clinics, Assiut University Hospital, Egypt. According to the International Continence Society (ICS) 2002, OAB is defined as the symptomatic diagnosis of urgency, with or without urge incontinence, and usually associated with frequency and nocturia. The ICS endorsement of the term OAB recognizes that patients with this syndrome are almost always treated by nonsurgical means on an empirical basis without urodynamic confirmation of the presumed diagnosis, detrusor overactivity. 8
A total of 33 patients were evaluated. In all, 26 patients were eligible (Figure 1). All participants had weakness of one or both lower limbs due to lumbosacral root damage. We included patients who were refractory to standard treatment (anticholinergic medication and behavioral treatment) and lived in nearby areas to be able to get regular follow-up. We excluded patients with severe weakness of both lower limbs (bedridden) with retention of urine or genuine stress incontinence, unstable cardiac dysrrythmia, epilepsy, and metals, or pacemakers.
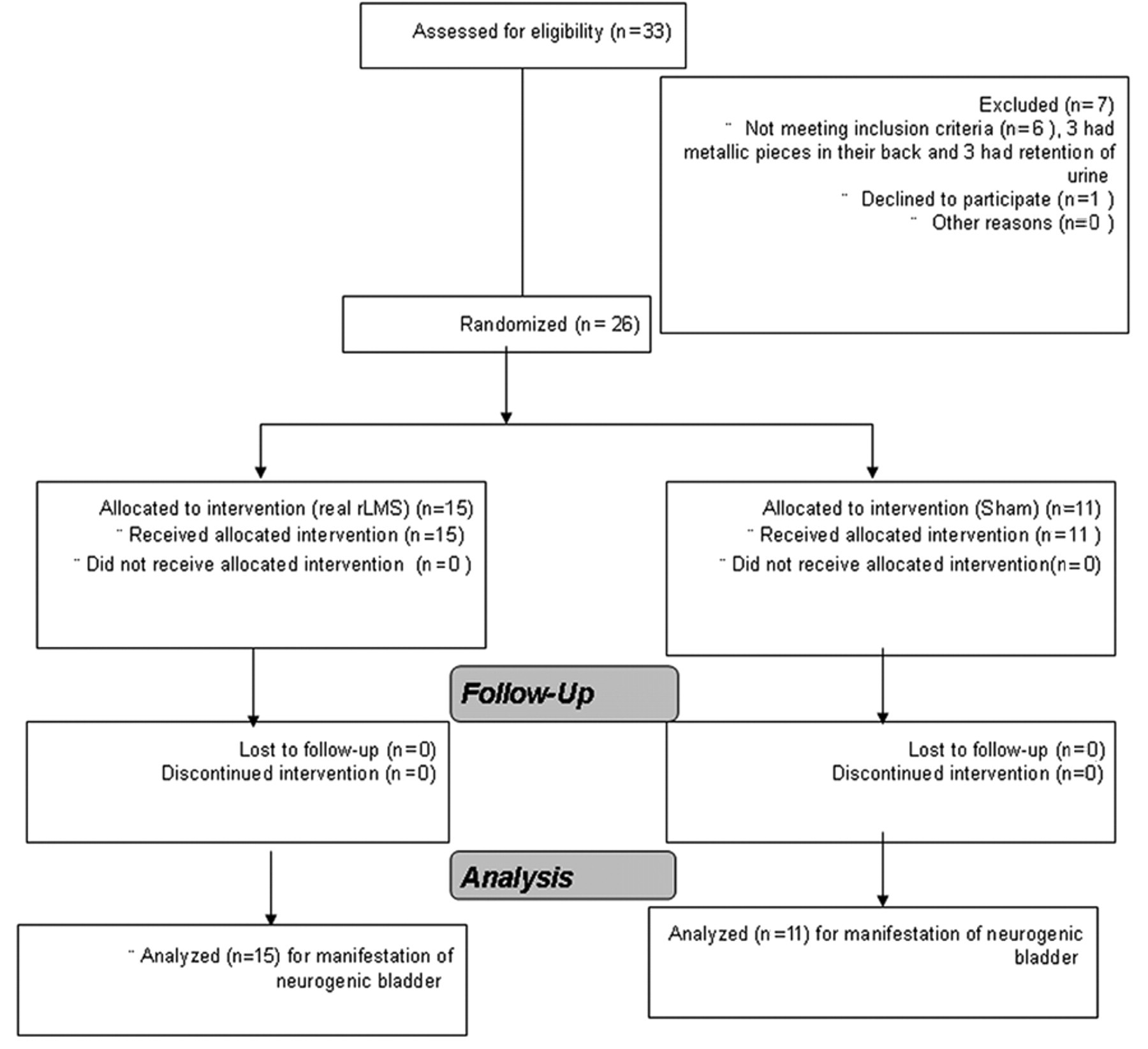
Study flow chart.
The Assiut Medical School Ethical Review Board approved the study. Written informed consent was obtained from all of the subjects after describing the nature of the intervention and the possibility of having sham stimulation.
Intervention
Since no available assessment scale addresses frequency of voiding and incontinence, we devised the present rating scale to quantify the changes after rLMS. Each patient was asked about his or her urinary symptoms, including number of voids per day and night, number of leaks per day and night, severity of leaks (few drops, wetting innerwear, wetting outerwear), and timed voiding. After this initial evaluation, each participant was given a 24-hour urinary frequency volume chart to be filled at home and was asked to return it at each follow-up visit.
Each participant evaluated the degree of low back pain associated with the lumbosacral plexopathy from 1 to 10 (most severe) using the standard visual analogue scale (VAS).
The experimental group received biphasic rLMS using a Magstim Super Rapid (Magstim, Whitland, UK) stimulator connected to a 90-mm outer diameter figure-of-8 coil positioned in the midline over the sacral vertebrae (approximately 5 cm above the natal cleft, which approximates to the level of S2). Stimulation was delivered at 15 Hz (10 seconds on and 30 seconds off). To control for total energy delivered, we chose to limit stimulation to a total of 1500 pulses. The stimulation was repeated for 10 sessions, 5 sessions per week and 2 days off. This regimen was a practical schedule for patients.
The control group received sham rLMS stimulation using the same electrodes, the same session frequency, in the same setting, but the coil was tilted 90.
Neurophysiological Studies
H-reflex testing was performed in the prone position with the feet supported by a pillow under the ankles. The tibial nerve was stimulated in the popliteal fossa for 0.5 or 1.0 ms with the intensity increased gradually to construct a recruitment curve for the H-reflex and M-wave for the gastrocnemius-soleus muscle. Electromyogram (EMG) was recorded using Neuropack S1 (EMG/EP measuring system) equipment (model MEB-9400K, Nihon Kohden, Tokyo, Japan) from surface electrodes placed halfway between the popliteal crease and the proximal medial malleolus. Ten trials were recorded. The F-wave was recorded after supramaximal stimulation for 20 trials. A difference of 1.5 ms between the F-wave latencies in right and left legs was considered abnormal.
All participants maintained their medication all through the study and received conventional physical treatment. Bladder retraining was performed in all patients except those with complete retention of urine. First. we asked patients to report the maximum time they can keep dry without voiding or any urinary leakage. Patients were instructed to perform timed voiding starting with an interval set to half an hour less than the usual duration. The time between voids was then gradually increased until a normal interval of 3 to 4 hours was reached. In addition, pelvic floor exercise was provided. First, the patients were taught to recognize the pubococcygeus muscle. They were then told to control or squeeze the muscle for 10 seconds and then relax 10 seconds, repeating this maneuver 15 times at least 3 times daily. To help patient compliance with the required home exercise, each received an educational pamphlet and a diary denoting the adherence to the required number and time of the exercises prescribed.
Randomization
The patients were randomly assigned to 1 of the 2 groups according to a computer-generated random table to determine the allocation sequence. Serially numbered opaque closed envelopes were used for allocation concealment. Allocation was maintained. To ensure double blinding, the random allocation sequence was kept by one investigator different from the one who enrolled the participants or assigned them to the interventions. Moreover, a third investigator was responsible for following the patients up and for assessment.
Outcomes
After the 5th session, 10th session, and 1 month later, 24-hour urinary frequency volume charts were reviewed with patients to ensure that they were filled correctly. Assessment of the degree of pain during micturition was reevaluated using the VAS at each of the above visits. F-waves and H-reflexes were also reassessed. The primary outcome was the improvement of urinary disturbances (number of voids per day and night, number of leaks per day and night, severity of leaks ([few drops, wetting innerwear, and wetting outerwear]), and timed voiding). The secondary outcome was the degree of pain using the VAS, along with changes after the end of the sessions, including F-wave and H-reflex.
Statistical Methods
Urinary incontinence at baseline, on the 5th day, 10th day of treatment, and 1 month after the end of sessions were analyzed with repeated measures analysis of variance (ANOVA). The Greenhouse–Geisser correction for degrees of freedom was used when necessary to correct for nonsphericity of data.
Results
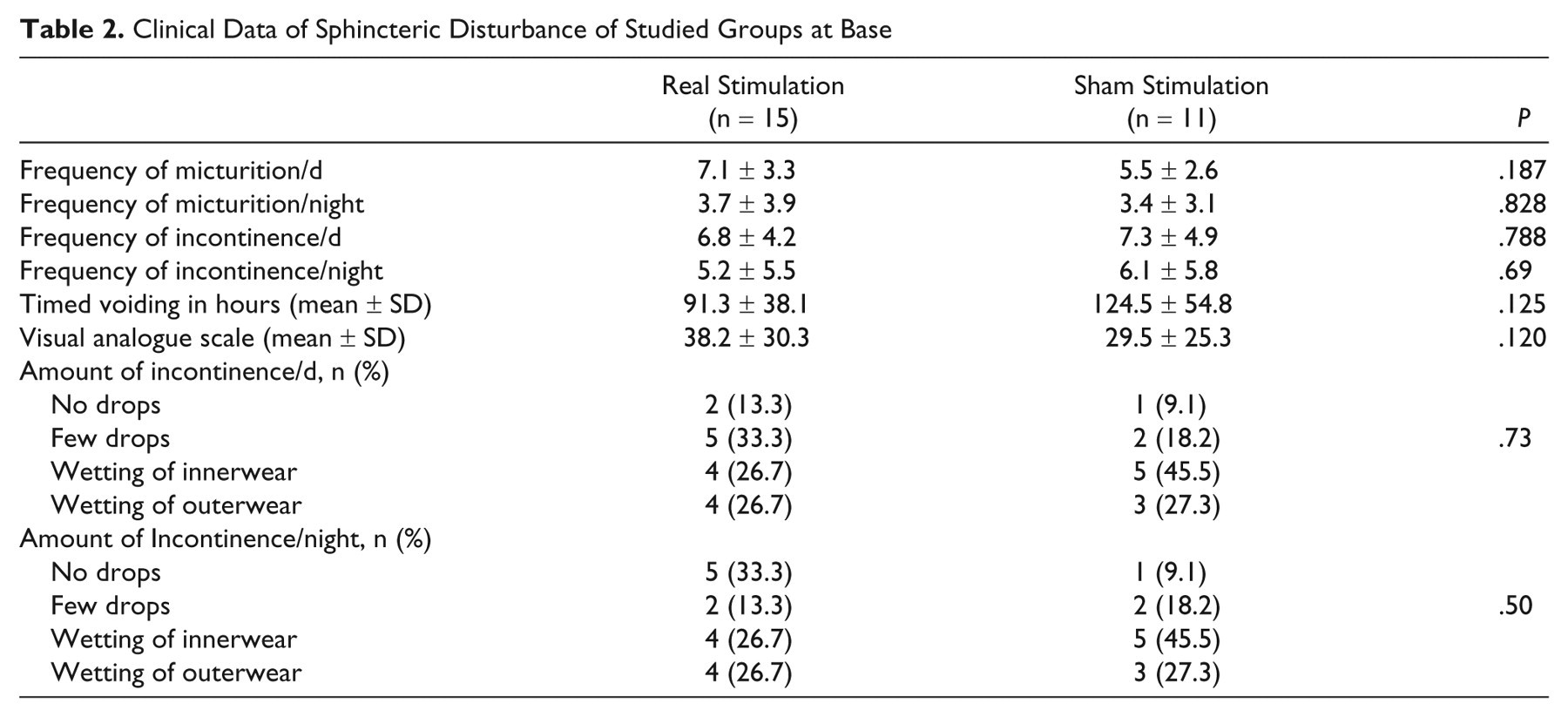
A total of 26 out of 33 patients with neurogenic bladder completed the study (19 males and 7 females) between March 2009 and December 2009 (Figure 1). Nine participants had history of trauma (falling from a height or direct trauma to the back in a car accident). All other patients had evidence of lumbosacral spondylosis, 5 of them had been operated with partial or no improvement of the condition (Table 1). All patients in the real and sham groups had high frequency of micturition and incontinence of urine through the day and night with no significant differences between groups at baseline assessment (Table 2).
Demographic Data of Studied Groups at Baseline Assessment
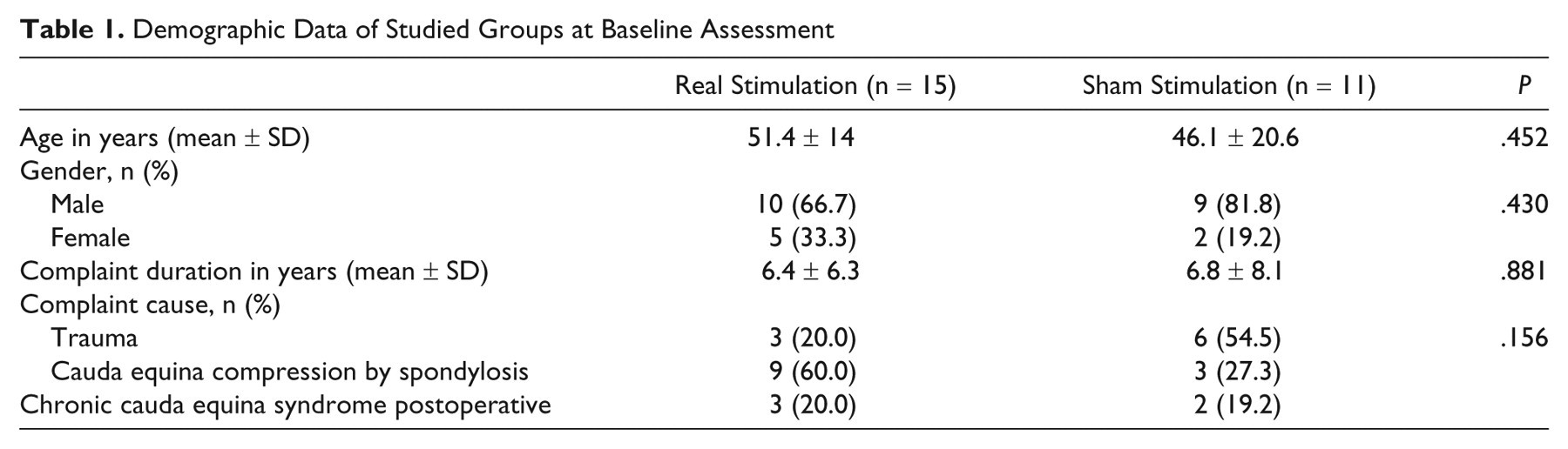
Clinical Data of Sphincteric Disturbance of Studied Groups at Base
All patients had evidence of lower motor neuron lesions due to injury of the lumbosacral roots of one or both lower limbs. In the real stimulation group, 14 (93.3%) patients had radicular sensory signs and 12 (80%) had motor weakness and wasting with diminished deep reflexes in one or both lower limbs. In the sham group, 10 (90.9%) had radicular sensory loss and 8 (72.7%) had motor weakness and wasting of the muscle of one or both lower limbs.
The mean F-wave latencies in both legs of patients with urinary incontinence (patients with retention of urine were excluded from the neurophysiological data) were significantly prolonged in the real group (34 ± 3.7 ms) and in the sham group (32.5 ± 4.3 ms) compared with healthy controls (27.9 ± 1.9 ms). Three patients in each group had absent F-waves.
In total, 14 of the 15 patients in the real group had abnormal H-reflexes (8 absent and 6 prolonged latencies in one or both lower limbs). In the sham group, 8 out of 11 patients had the same abnormalities with no significant differences between groups.
No side effects were observed during or after rLMS in any of our patients. Figure 2A shows the mean frequency of micturition per 24 hours (day + night) in the 2 groups during the course of treatment. A 2-way ANOVA on the daytime data (main factors of “real vs sham” and “time of assessment”) showed a significant time × group interaction (F = 5.8, df = 1.4 (30.4), P = .014). Follow-up 1-way ANOVA for each group separately showed that patients who received real-rLMS (15 Hz) had decreased frequency over the course of treatment (F = 20.7, df = 1.2 (15.6), P = .0001) whereas no changes occurred in the sham group (F = 3.9, df = 1 (9), P = .08). Similar effects were observed for decrease in frequency of micturition at night: 2-way ANOVA (F = 5.79, df = 1.38 (30), P = .014) and 1-way ANOVA for each group separately with real-rLMS (F = 11.4, df = 1.35 (17.6), P = .002) and sham (F = 2.9, df = 1.48 (13.3), P = .099)
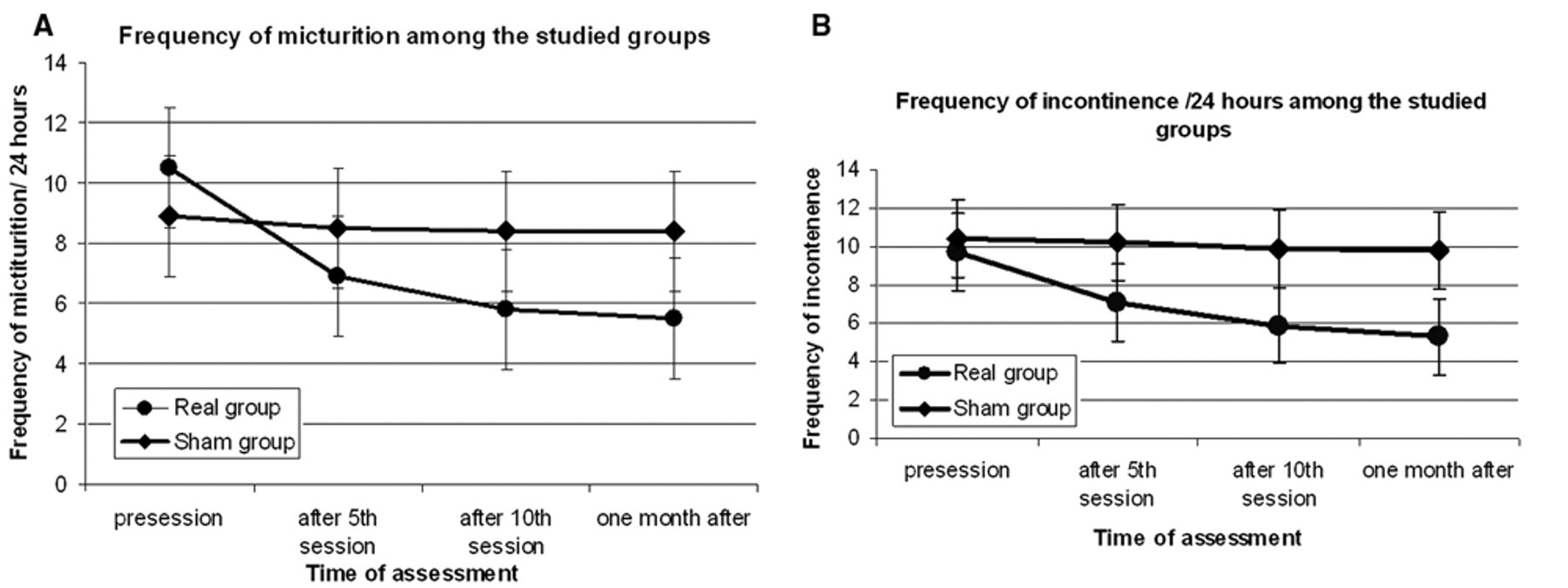
Changes in mean number of frequency (A) and incontinence (B) during 24 hours (day + night) among the real and sham groups along the course of stimulation. The first assessment was immediately prior to commencing repetitive trans-sacral magnetic stimulation (rLMS) treatment (pre), the second (post 5) was immediately after the end of the 5th and 10th session of rLMS, and then 1 month after. As the data show, the mean number of frequency and incontinence/day and night of the patients who received real-rLMS (15 Hz) decreased more over the course of the treatment than those who received sham-rLMS.
Figure 2B shows the mean frequency of incontinence per 24 hours (day + night) in the 2 groups during the course of treatment. Two-way ANOVA for the score during daytime favored real stimulation (F = 14.4, df = 1.3 (27.4), P = .0001). One-way ANOVA showed that the patients who received real-rLMS (15 Hz) had reduced incontinence (F = 23.8 df = 1.3 (15.3), P = .0001) with no change in the sham group (F = 3.3, df = 1.1 (10.2), P = .09). Similar effects were observed for night frequency of incontinence: 2-way ANOVA (F = 3.66, df = 1.16 (24.2) and P = .050) and 1-way ANOVA for each group separately with real-rLMS (F = 6.7, df = 1.1 (13), P = .019) and sham (F = 2.9, df = 1.1 (9.9), P = .11).
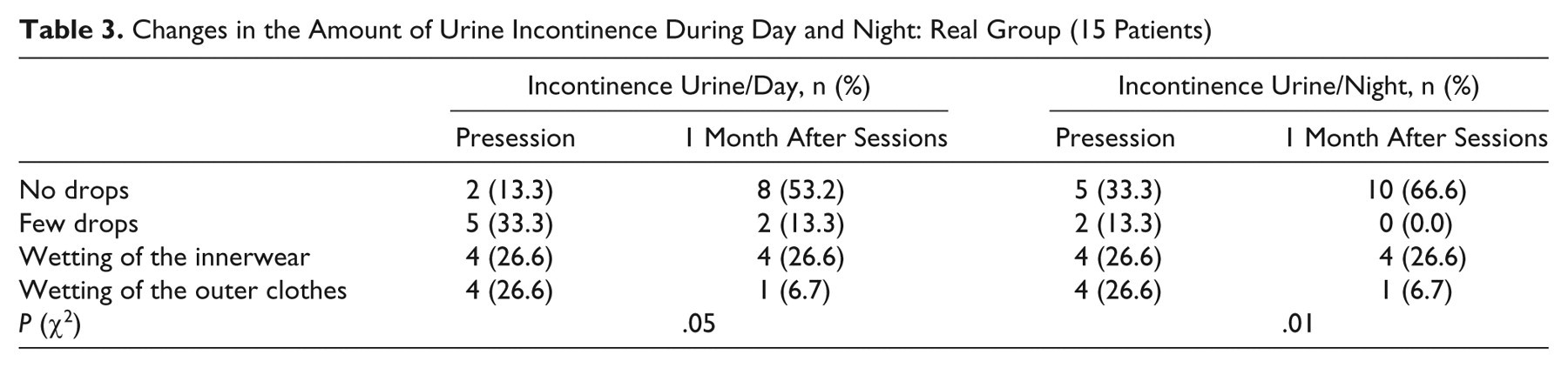
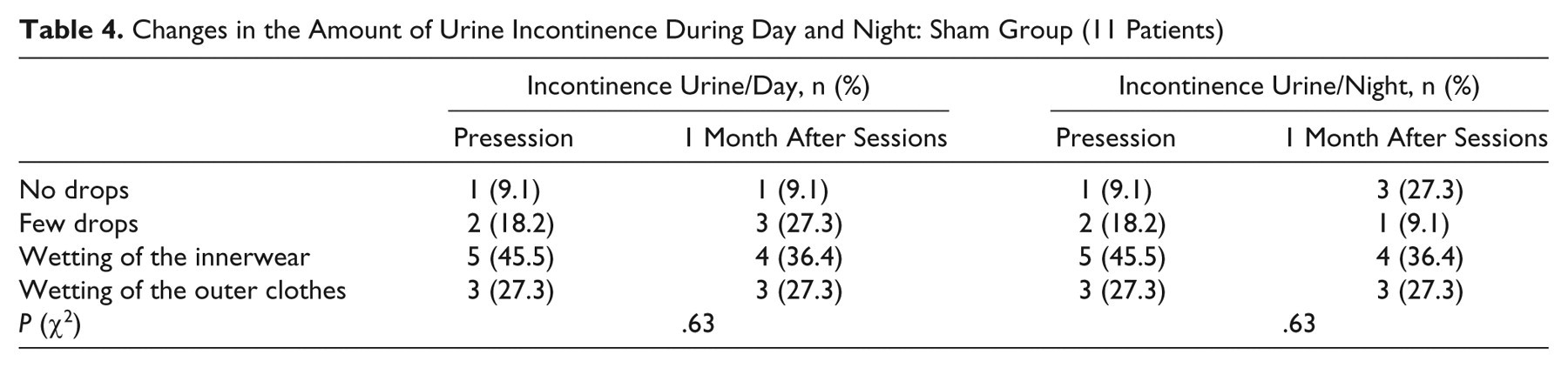
The mean duration of timed voiding of patients who received real-rLMS increased significantly (F = 55.05, df = 1.58 (17.4), P = .001) compared with sham (F = 3.55, df = 1.1 (9.7), P = .08), but no significant differences were found between groups (F = 2.1, df = 1.18 (23.7), P = .15; Figure 3). Tables 3 and 4 show the changes in the amount of incontinent urine during day and night, respectively, in the patients who had received real-rLMS. Two-way ANOVA showed a significant “time × group” interaction (F = 11.15, df = 1.2 (33.3), P = .001) for the VAS in favor of the real-rLMS.
Changes in the Amount of Urine Incontinence During Day and Night: Real Group (15 Patients)
Changes in the Amount of Urine Incontinence During Day and Night: Sham Group (11 Patients)
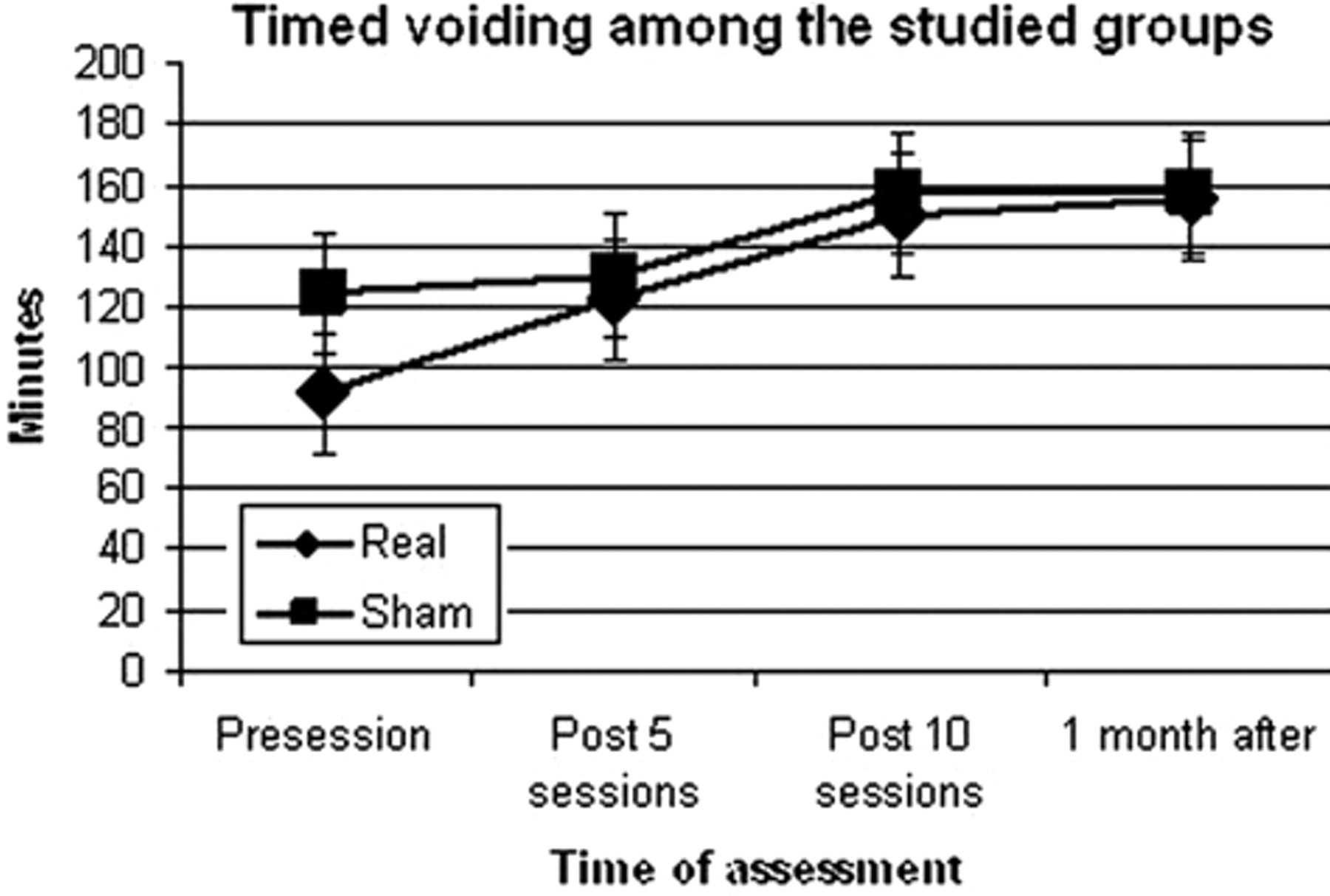
Changes in mean duration of the timed voiding among the real and sham groups along the course of stimulation. The data show that although the mean duration of the timed voiding of the patients who received real-rLMS (15 Hz) was increased significantly more over the course of the treatment than those who received sham-rLMS, the difference was not significant.
The mean values of F-wave and H-reflex latencies showed a significant improvement in F-wave latencies and H-reflex latencies after real stimulation (P = .002 for F-wave and P = .036 for H-reflex), whereas no such changes were observed in the sham group. Two of 3 patients in the real group who had absent F-wave at base line regained their response and 4 patients regained their H-reflex. One patient regained H-reflexes after sham stimulation (Table 5).
Effect of rLMS on F-Wave and H-Reflex Latencies and Cauda Point of Pudendal SEP for Studied Groups a

Abbreviations: rLMS, repetitive lumbosacral magnetic stimulation; SEP, somatosensory evoked potential; NS, nonsignificant.
Numbers in parentheses indicate number of cases with no response.
Discussion
Maintenance of fecal and urinary continence depends on complex interactions between local, peripheral, and central sensorimotor processes. 9,10
Our results suggest that rLMS can improve symptoms for up to 1 month by decreasing frequency and incontinence of micturition during the day and night. Indeed, 6 patients became completely continent of urine during day time and 5 at night.
Mechanisms of Action of rLMS
The mechanism of the response to rLMS is unknown although effects may occur at both spinal and supraspinal levels. rLMS may stimulate directly the pudendal nerve efferent to both external urethral and anal sphincters and thus directly potentiate contractile mechanisms. Indeed, stimulation of the sacral nerve roots at 15 to 20 Hz has been shown to be the optimal frequency to provide contraction of the anal sphincter. 11 Stimulation will also activate directly the peripheral afferent fibers and evoke sensory input to spinal cord and additional input from the induced muscle contraction. Together these inputs could increase excitability of sensorimotor connections in the cord, perhaps accounting for the small reduction in F-wave latency as well as the increased occurrence of H-reflexes.
The same mixture of sensory inputs will also reach the supraspinal level, where it could modulate the excitability of cortico-anal pathways and drive compensatory changes within the cerebral cortex. Earlier work has demonstrated that single-pulse LMS can momentarily modulate motor cortical centers relevant to anal function. 12 Repetitive SNS can produce a longer lasting reversible change in cortico-anal excitability. 7 The combination of rLMS with pelvic floor exercise may improve relearning of the control of micturition with lasting changes in the organization of cortical motor output for micturition.
The effects of rLMS on continence were accompanied by changes in pain perception. Pujol et al 13 also noted that repetitive pulse magnetic coil stimulation can be applied successfully to reduce musculoskeletal pain for several days. Heldmann et al 14 found that prolonged peripheral magnetic stimulation could modulate the response of primary and secondary somatosensory cortices to afferent input. The effects on pain perception may also be due to the improvement of sphincteric dysfunction.
Limitations in the interpretation of our results include the small sample size of this pilot study, dropouts, and dependence on reports by participants.
Conclusion
We found that 15-Hz rLMS plus routine exercises improves urinary dysfunction in people with cauda equina and lumbosacral root lesions, which is maintained for at least 1 month beyond the period of stimulation. The mechanism of action may involve both peripheral and central effects. The procedure is noninvasive, painless, and inexpensive so it may be suitable prior to consideration of implantation of electrical sacral root stimulation in patients with lower motor neuron injuries. Further studies are required to establish the optimal profile for stimulation.
Footnotes
The author(s) declared no potential conflicts of interest with respect to the authorship and/or publication of this article.
The author(s) received no financial support for the research and/or authorship of this article.
