Abstract
Background. The current diagnostic criteria of urinary tract infection (UTI) in male patients with spinal cord injury (SCI) are not clear. Methods. The authors studied 381 episodes of “symptomatic” UTI (209 participants) and 277 episodes of “asymptomatic” UTI (205 participants) in male SCI patients using intermittent catheterization. UTI was defined as a bacterial count ≥102 colony-forming units (cfu)/mL (American Paraplegia Society criterion). Univariate analysis and receiver operating characteristic (ROC) curve analysis were used to determine optimal cfu and white blood cell (WBC) thresholds. Results. The most prevalent clinical signs, alone or in combination, were cloudy and/or malodorous urine (51.4%), onset of urinary incontinence (51.2%), fatigue (41.7%), fever (30.7%), and increased spasticity (30.2%). Urine cfu and WBC levels in patients with only one sign, including fever, were not significantly higher than those in asymptomatic controls. WBC, but not cfu, levels increased significantly with the number of signs (P = .026). Univariate analysis and ROC curve analysis failed to identify cfu, WBC, or a combination of cfu and WBC count thresholds, allowing discrimination between the symptomatic and asymptomatic UTI groups. Conclusions. Clinical signs of UTI correlate poorly with the urine cfu and WBC levels in SCI patients, except for a positive relationship between WBC counts and the number of signs. Fever alone has no higher diagnostic value. There are no satisfactory cfu and WBC thresholds: thresholds more restrictive than the current American Paraplegia Society criteria provide higher specificity values but with equivalent loss of sensitivity.
Keywords
Introduction
Urinary tract infection (UTI) is the most common medical complication in patients with spinal cord injury (SCI). 1-3 Neurological deficits following SCI result in incomplete bladder emptying such that bladder drainage with catheters is required; both these factors predispose to UTI. 1,3 Although the introduction of intermittent catheterization (IC) in the mid-1970s dramatically reduced the incidence of UTI in SCI patients, 4,5 about two thirds of SCI patients under IC develop one or more episodes of bacteriuria. 1,6
There is a general consensus that only symptomatic UTI in SCI patients should be treated. Asymptomatic episodes of bacteriuria are common in this population and do not appear to increase the risk of infectious complications and long-term sequelae. 1,7 In addition, repeated administrations of antimicrobial agents may facilitate antimicrobial resistance, which is already a serious problem in SCI patients. 8,9 However, there is currently no consensus about the clinical criteria for UTI in such patients. 2 Whereas a number of authors consider any suggestive sign or symptom, 10 others attribute a greater diagnostic value to fever 5,11 or only consider as significant the presence of combinations of signs or symptoms. 5 Moreover, clinical manifestations of UTI are often atypical and nonspecific in SCI patients and may involve only aggravation of preexisting neurological symptoms, whereas some very suggestive symptoms such as fever may have a noninfectious origin. 12
Bacteriological criteria of UTI in SCI patients are also a matter of debate. The American Paraplegia Society (APS) recommends basing the diagnosis of UTI solely on bacteriuria, with a threshold of 102 colony-forming units (cfu)/mL for SCI patients under IC. 5 However, up to 75% of samples taken from totally asymptomatic SCI male patients under IC contain ≥102 cfu/mL. 2,13,14 Thus, although very sensitive, a threshold of 102 cfu/mL is associated with low specificity, leading to the treatment of an excess of patients, with the risk of the emergence of resistant bacteria, unwanted side effects, and unnecessary expense. 8,9 This is why some groups propose that the diagnosis of UTI should use more specific cfu thresholds, generally 105 cfu/mL. 9,11,14 It has also been proposed to take pyuria into account because pyuria reflects the inflammatory state of the bladder and may help distinguish true infection from colonization 15,16 and predict subsequent morbidity. 17 However, the optimal method and quantitative criteria for assessing pyuria in persons with SCI have not been determined. 2
The current APS recommendations are primarily based on clinical consensus. Some previous studies have looked for correlations between cfu and white blood cell (WBC) counts in urine samples collected in parallel either via the urethra (catheter or clean-voided specimens) or via suprapubic aspiration with the aim of identifying positivity criteria applicable to simple catheter samples in daily practice. 13,18 However, these studies included a small number of patients and no control group. We conducted a large, prospective, cohort-based study to determine the diagnostic criteria of UTI in male SCI patients under IC. In this study, we tried to answer the following specific questions: (1) Is there any evidence that some clinical signs are better than others for the diagnosis of UTI in SCI patients, and what is the pertinence of fever and “neurological signs” (ie, increased spasticity and autonomic hyperreflexia)? (2) Is there any benefit in considering combinations of signs and symptoms? and (3) What cfu and WBC threshold values should be used to provide optimal sensitivity to specificity ratios?
Materials and Methods
Studied Groups
Between December 1, 1999, and November 30, 2004, we prospectively recorded all symptomatic UTI episodes diagnosed in male SCI patients under clean IC, managed after the acute injury phase at the Department of Physical Medicine and Rehabilitation of the Raymond Poincaré hospital (“symptomatic UTI” group). Symptomatic UTI was defined according to the criteria of the APS for catheter specimens collected from SCI patients on IC 3 : (1) at least 1 sign or symptom among fever (temperature ≥38.5°C—T), cloudy and/or malodorous urine (U), onset of urinary incontinence or modification of bladder behavior (I), fatigue or sense of unease (G), increased spasticity (S), and autonomic hyperreflexia (H; signs and symptoms will hereafter be collectively referred to as “clinical signs” for simplicity) and (2) a bacterial count ≥102 cfu/mL. Criteria for controls (asymptomatic UTI group) included all consecutive episodes of asymptomatic UTI (bacterial count ≥102 cfu, without any clinical manifestation suggestive of UTI) identified during the study period in SCI male patients before urodynamic evaluation or cystoscopy. UTI episodes, whether or not symptomatic, were excluded if the patient had received antibiotics in the 7 days prior to sample collection. Repeated inclusions were allowed for individual patients provided that the period between UTI episodes, whether or not symptomatic, was ≥30 days. No ethical approval was required for this study (no blood sampling).
Laboratory Methods
Urine samples were collected using standard procedures and were immediately sent to the microbiology laboratory. The samples were processed within 2 hours of collection. WBC counts in unspun fresh urine were determined using a hemocytometer chamber. 16 Gram stain examination was also performed on unspun urine. Cfu were counted by the surface streak method using CPS agar (BioMérieux, Marcy l’Etoile, France). The minimal detection limit was 20 cfu/mL. Other culture plates were used as appropriate according to the results of Gram stain smear examination. Bacterial species were identified according to standard methods.
Statistical Methods
Statistical analyses were performed with Stata software, Version 9.2 (2006 Stata Corporation, College Station, Texas). Characteristics of the studied groups were compared using the Mann-Whitney test for quantitative variables and with the χ2 test (or Fisher test if necessary) for qualitative variables. Urine cfu and WBC counts are reported as medians and interquartile ranges and clinical parameters as frequencies and percentages. Distributions of cfu and WBC levels were compared between groups with nonparametric tests: the Kruskal-Wallis (global test) or the Mann-Whitney test (2 by 2 test). A nonparametric trend test was used if there was a significant trend in cfu and WBC levels according to the number of clinical signs. A P value of <.05 was considered as statistically significant. Univariate odds ratios (ORs) with their 95% confidence intervals (95% CI) were calculated for different WBC and cfu thresholds. ORs > 2 were considered as significant. The optimal cfu and WBC thresholds were determined by receiver operating characteristic (ROC) analysis. 19
Results
Characteristics of the Studied Groups
The characteristics of patients included in the symptomatic (n = 209) and asymptomatic (n = 205) UTI groups were similar with respect to age and proportion of patients with spinal cord lesion ≥T6 (ie, patients at risk of autonomic dysreflexia; Table 1). The mean number of urine samples per patient was slightly, but significantly, higher in the symptomatic group (P < .001). The ranges of cfu and WBC counts in the 2 groups were similar. The number of bacterial species per sample was higher in the asymptomatic group than in the symptomatic group (number of samples with ≥2 species: 82/277 [29.6%] vs 69/381 [18.1%]; P = .001; Table 1). The bacterial species recovered also differed slightly between the 2 groups, with a Gram-negative to Gram-positive ratio of about 3:1 in the symptomatic group and 2:1 in the asymptomatic group (Table 1). Bacterial changes in urine (number and/or nature of species recovered) more than 30 days after a UTI were observed in 73% of participants.
Characteristics of the Groups Studied
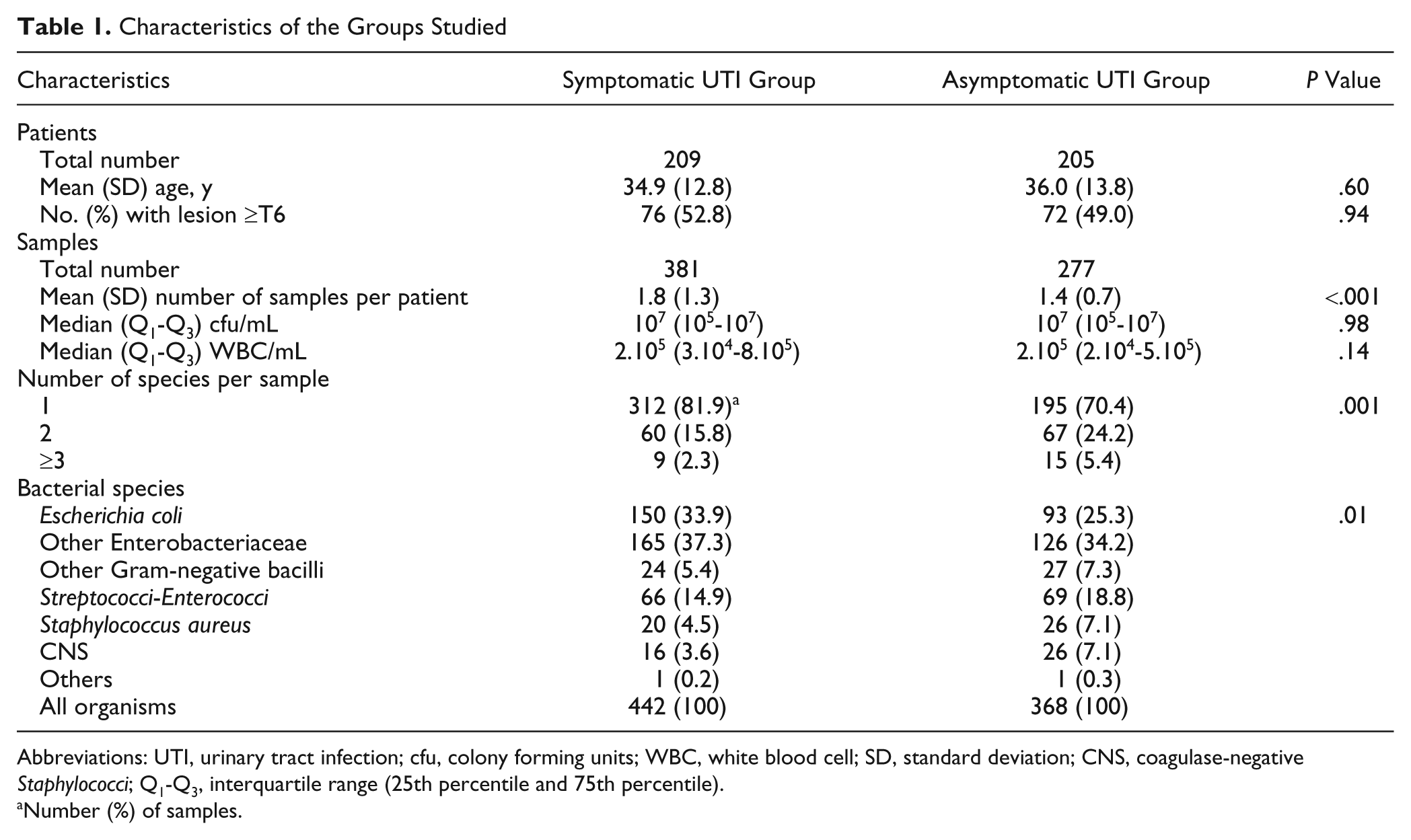
Abbreviations: UTI, urinary tract infection; cfu, colony forming units; WBC, white blood cell; SD, standard deviation; CNS, coagulase-negative Staphylococci; Q1-Q3, interquartile range (25th percentile and 75th percentile).
Number (%) of samples.
Clinical Signs of UTI
We analyzed the prevalence of clinical signs recorded in the 381 episodes of symptomatic UTI. The most prevalent clinical signs, alone or in combination, were U (n = 196, 51.4%), I (n = 195, 51.2%), and G (n = 159, 41.7%), then T (n = 117, 30.7%) and S (n = 115, 30.2%); H was much less frequent (n = 21, 5.5%). A variety of clinical presentations were associated with the 381 episodes of UTI (Table 2). One isolated sign—mostly I, U, or T—was recorded in about one third of episodes; a combination of 2 signs—mostly IU, GT, GU, or GI—in about one third; and a combination of 3 signs or more—mostly GIU, ISU, GTU, or GSTU—in the final third. The “neurological” signs —S and H—were found as isolated signs in only 3.1% and 0.3% of all episodes, respectively; G was also rarely found as an isolated sign (2.1%).
Clinical Presentation of the 381 Symptomatic UTI Episodes

Abbreviations: UTI, urinary tract infection; T, fever; U, cloudy and/or malodorous urine; I, onset of urinary incontinence or modification of bladder behavior; G, fatigue or sense of unease; S, increased spasticity; H, autonomic hyperreflexia.
Evaluation of Clinical Signs
In a first attempt to identify the most “informative” signs, we studied the levels of cfu and WBC counts in the various monosymptomatic subgroups (U, I, T, G, S, and H alone). However, S, G, and H alone were excluded from the analysis because too few episodes involved these signs in isolation (number of episodes = 12, 8, and 1, respectively). Thus, only I, U, and T were evaluated (Table 3). Urine cfu and WBC levels in patients with either I, U, or T alone were not significantly higher than those in asymptomatic controls. Unexpectedly, T tended to be associated with lower cfu levels than controls, but the difference did not reach significance (Table 3).
We then tested whether cfu and/or WBC counts correlated positively with the number of clinical signs (episodes with 1, 2, or ≥3 signs): correlation was demonstrated for WBC (P = .026) but not for cfu, with significantly higher WBC levels in patients presenting with 3 signs or more than in those with 1 or 2 signs (P = .012); WBC levels, however, did not differ significantly between patients having 1 sign and those with 2 signs or more (P = .18; Figure 1).

Urine colony forming units (cfu) and white blood cell (WBC) counts. Cfu counts trend test, P = .375. Wbc counts: trend test, P = .026; Mann-Whitney test, ≤ 2 versus ≥ 3 signs, P = .012).
WBC and cfu Counts in Monosymptomatic UTI a
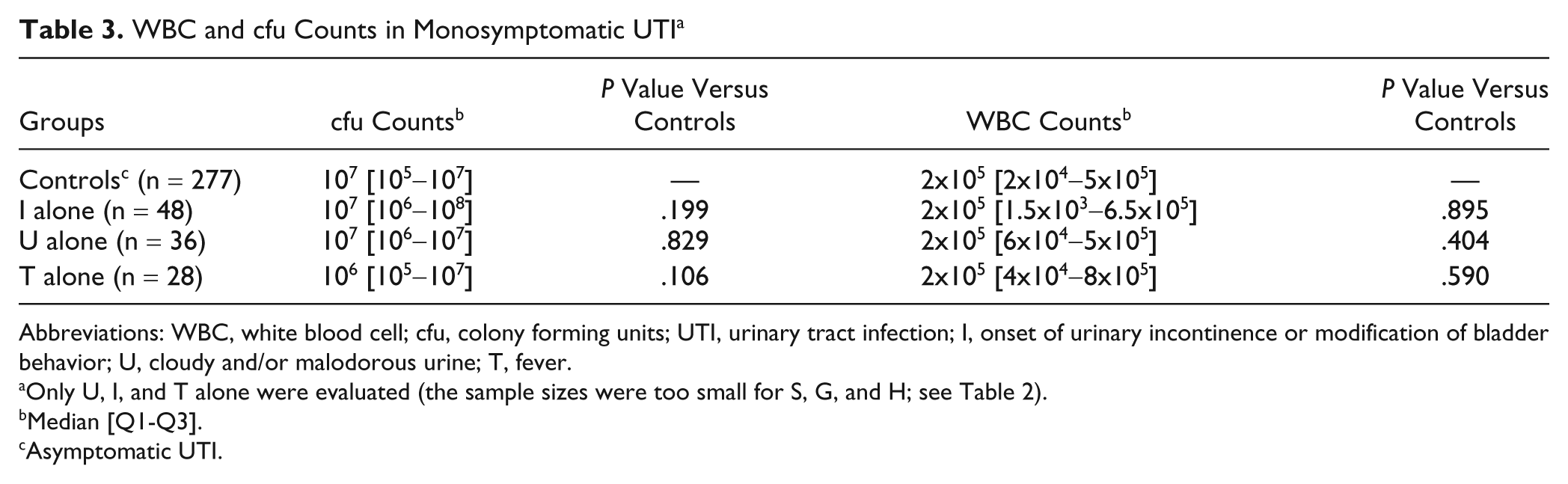
Abbreviations: WBC, white blood cell; cfu, colony forming units; UTI, urinary tract infection; I, onset of urinary incontinence or modification of bladder behavior; U, cloudy and/or malodorous urine; T, fever.
Only U, I, and T alone were evaluated (the sample sizes were too small for S, G, and H; see Table 2).
Median [Q1-Q3].
Asymptomatic UTI.
Determination of Optimal cfu and WBC Thresholds With Univariate Analysis
We evaluated the capacity of different cfu and WBC thresholds, alone or in association, to distinguish between the symptomatic and asymptomatic UTI groups (Figure 2). Whatever the threshold used, the rate of positivity was not significantly higher in the symptomatic group than in the asymptomatic group. An OR value greater than 2 was found with only 1 threshold—103 cfu/mL (OR = 2.8; Figure 2A)—the difference being nonsignificant (95% CI = 0.7-11.3).
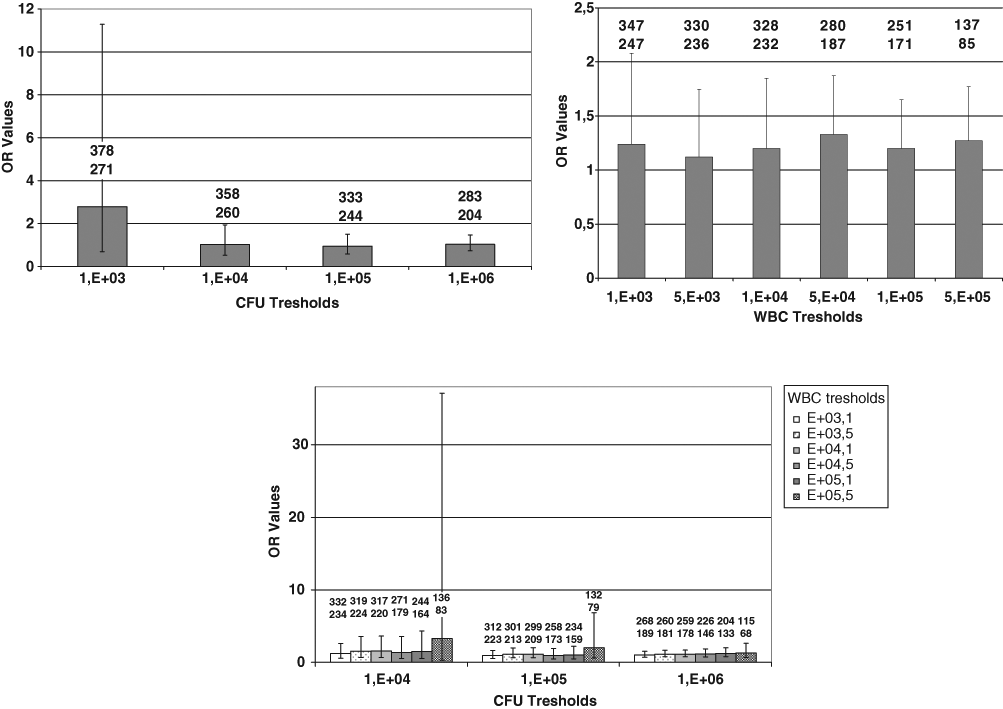
Univariate comparison of the symptomatic and asymptomatic UTI groups: A. cfu thresholds. B. WBC thresholds; C. Combinations of cfu and WBC thresholds. The number of symptomatic samples (upper) and asymptomatic UTI samples (lower) are indicated for each threshold or combination of thresholds. Error bars indicate 95% confidence interval values.
ROC Curve Analysis
We performed ROC curve analysis to identify the laboratory criteria giving the best sensitivity to specificity ratio. The ROC curves obtained using cfu thresholds alone (Figure 3A), WBC thresholds alone (Figure 3B), and combinations of cfu and WBC thresholds (Figure 3C) were all straight. Thus, there were no optimal thresholds: all increases in specificity were accompanied by an equivalent loss in sensitivity and vice versa (Table 4).
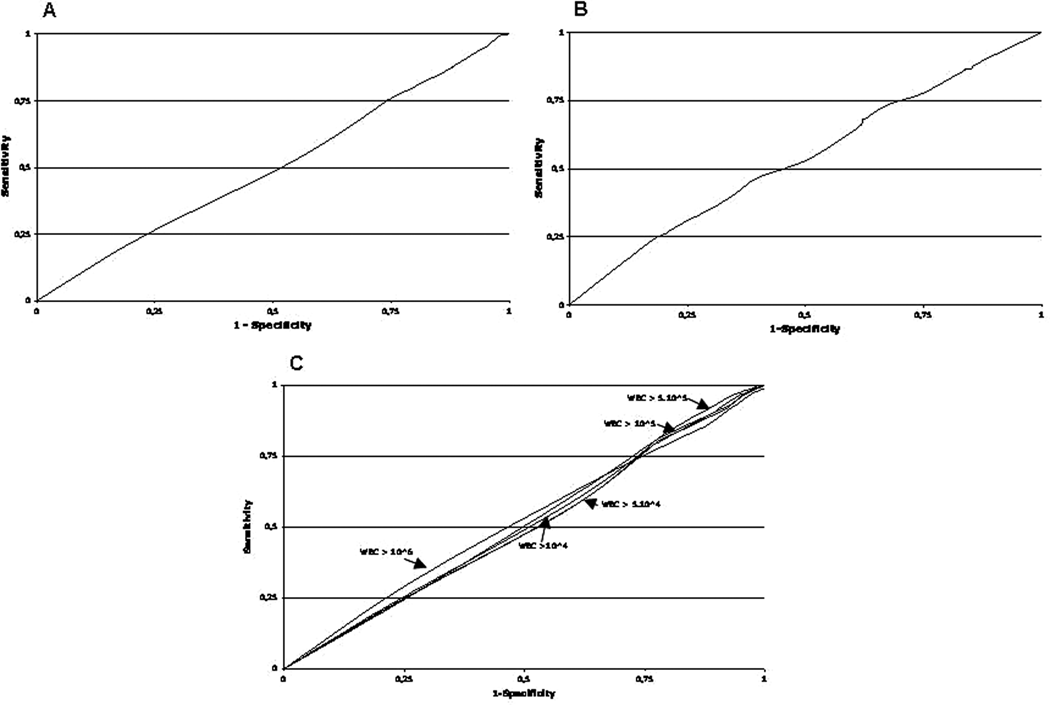
ROC curve analysis: A. cfu thresholds. B. WBC thresholds. C. Combinations of cfu and WBC thresholds.
Sensitivity to Specificity Ratios Obtained With Various cfu and WBC Thresholds, Alone or in Combination a
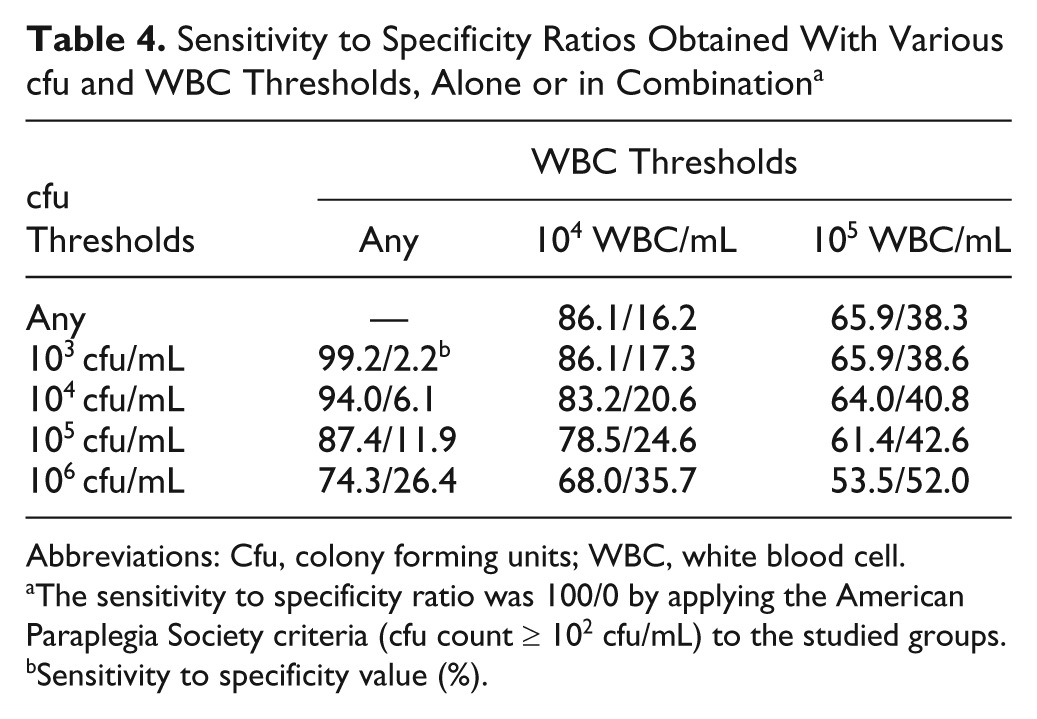
Abbreviations: Cfu, colony forming units; WBC, white blood cell.
The sensitivity to specificity ratio was 100/0 by applying the American Paraplegia Society criteria (cfu count ≥ 102 cfu/mL) to the studied groups.
Sensitivity to specificity value (%).
Discussion
There is currently a consensus that antibiotic treatment is only justified for symptomatic, and not nonsymptomatic, UTI in SCI patients. 2,3 This view is based on the notion that only symptomatic infections are associated with a significant inflammatory process and that it is this inflammatory reaction that is the main cause of the harmful effects of infection on the urinary tract. 3 Consequently, the antibiotic treatment aims to eliminate the bacteria eliciting the inflammatory reaction. In addition, bacteriuria is seen as being both frequent and trivial in SCI patients, caused by the impaired flow and repeated interventions involving the urinary tract; antibiotic therapy for such colonization increases the risks of selecting antibiotic resistance and of unwanted side effects, without having any demonstrable benefits. 20
Although a major issue, there is no clear definition of symptomatic urinary infection in SCI patients. 2 Most previous studies score an infection as symptomatic if there is fever or, in the absence of fever, combinations of signs. 5,11 The National Institute of Disability and Rehabilitation Research conference of 1992 concluded, primarily based on clinical consensus, that fever was the sole symptom that routinely warrants concern and consideration for antibiotic treatment in the setting of bacteriuria. 3 However, there is no evidence supporting this position, 2 and we are aware of no clinical study demonstrating relevant correlations.
We were able to analyze 3 signs as isolated signs: urinary incontinence, cloudy or malodorous urine, and fever. None of these 3 signs was associated with higher cfu or WBC levels than found for completely asymptomatic controls. This raises doubts about the diagnostic value of these signs in isolation; certainly, no single sign or symptom performed any better than the others. In particular, not only was fever not associated with higher WBC levels than that for controls but tended to be associated with lower cfu levels. Possibly, this was because fever can be caused by infections other than UTI (eg, viral infection) or, indeed, may have a noninfectious cause (eg, impairment of neurological body temperature control). 12 Consequently, and contradicting various recommendations, 2,3 there is no reason to attribute a greater value to isolated fever than to any other signs or symptoms for the diagnosis of UTI in male SCI patients. However, the occurrence of febrile episodes in prior years has been associated with upper-urinary-tract complications or abnormalities at follow-up several years later. 21 In this particular case of fever in SCI male patients under IC, the value of other investigations (eg, ultrasound) is being studied.
Increased spasticity and autonomic hyperreflexia are signs that are particular to SCI patients and are often suggested as being indicative of UTI in this group. Increased spasticity is thought to be a result of any nociceptive stimulation below the lesion and can be observed after any complication (including UTI, pressure sore, fecal impaction, and lithiasis). 22 Autonomic hyperreflexia can develop after acute nociceptive stimulation below the lesion in patients with spinal lesions above T6 and also by UTIs. 23 Despite the large number of UTI episodes included in our study, the number of monosymptomatic episodes involving these signs was too small for meaningful statistical analysis. Consequently, further studies are required to assess the diagnostic value of these signs, considered in isolation, relative to that of more standard signs of UTI, such as cloudy urine or fever.
We found a positive relationship between the number of clinical signs and the urine WBC count. This is a strong argument in favor of the notion of including leukocyturia as a biological criterion of UTI, contrary to the current recommendations of the APS. 3 However, the number of clinical signs that should be considered as being significant remains unresolved. In our study, we only found a significant difference between 1 or 2 signs and 3 or more signs. However, this threshold would exclude about two thirds of cases (in our study) classified as UTI according to current APS criteria. Moreover, because of its design, our study did not address other major issues such as the occurrence of long-term complications. Further study is required to evaluate the prognostic value of the number of clinical signs and determine the optimal threshold with or without fever.
We expected higher urinary counts of cfu and/or WBC in symptomatic episodes than in asymptomatic episodes, thus, enabling the identification of appropriate thresholds to distinguish between the 2 groups. The results of univariate analysis showed that this was not the case. No criterion, whether based on bacteriuria, pyuria, or a combination of both, could be used to differentiate between samples from symptomatic or asymptomatic patients. This was surprising because symptomatic patients more often harbored Gram-negative bacteria (ie, Escherichia coli), which tend to cause severe inflammation associated with intense leukocyturia. This may suggest that the response mechanism of the bladder’s mucous membrane to attack by microorganisms is different in SCI patients. The high WBC in all our patients, symptomatic or not, may also be linked to bladder wall irritation caused by intermittent catheterization, which generally leads to an elevated level of leukocyturia in patients under IC, even in the absence of infection. 24
Analysis of the ROC graphs was in complete agreement with the results of the univariate analysis. Whatever the biological criterion (bacteriuria, pyuria, or a combination of the two), the graphs obtained were almost perfect straight lines: any loss of sensitivity was accompanied by an equivalent increase in specificity and vice versa. Thus, it was impossible to select a threshold that would give a better sensitivity to specificity ratio than the APS threshold. For example, replacing the APS value with a threshold of 105 cfu/mL gave a 12% increase in specificity but a loss of sensitivity of 13%. If thresholds of 105 cfu/mL and 104 WBC/mL were combined, there was a gain in specificity of almost 25%; however, the loss in sensitivity was around 22%. Our results probably explain why none of the criteria recommended to date have really proven to be more appropriate than the others. Thus, based on our findings, we were unable to suggest laboratory criteria other than those currently recommended by the APS.
In conclusion, we show that 3 elements need to be considered when establishing recommendations for the diagnosis of UTI in SCI patients under IC, after the acute phase of injury. First, there is no evidence in support of giving particular importance to fever as an isolated symptom. Second, the number of clinical signs is undoubtedly a major diagnostic factor. Third, leukocyturia correlates with the number of clinical signs and, therefore, presumably with the severity of the infection and associated inflammatory response; consequently, supporting previous opinions, 15,16 it should be included as a biological criterion of UTI. The use of these 3 elements would make the criteria more restrictive and thereby help identify those cases for which antibiotic therapy is beneficial and justified. However, our results also clearly show the limits of cfu and WBC criteria in SCI patients and the need for alternative approaches. One possibility is to identify better biological markers in blood, urine, or both, such as cytokines. 25,26 Another approach is to identify the collective criteria that allow us to decide whether to treat a patient and reconcile 2 objectives that tend to be in opposition—to avoid complications and reduce selective pressure by antibiotics. Only interventional studies accompanied by longitudinal studies for perhaps 3 years will resolve these issues. Future studies should include possibly important parameters not taken into account in our study, such as quantification of neurogenic detrusor activity and treatment.
Footnotes
Acknowledgements
In memoriam of Olivier Dizien. We thank the personnel of the department of physical medicine and rehabilitation of the Raymond Poincaré hospital for their contribution to this work. We also thank I. Sénégas for help with the manuscript.
ER and PD contributed equally to this work.
The author(s) declared no potential conflicts of interest with respect to the authorship and/or publication of this article.
The author(s) received no financial support for the research and/or authorship of this article.
