Abstract
Final outcome after surgical repair of peripheral nerve transections varies. Here, we present the first longitudinal functional magnetic resonance imaging (fMRI) obserof cortical somatosensory reorganization patterns after surgery. A 43-year-old man presented with isolated complete transecof the right median nerve and underwent immediate epineural end-to-end coaptation. Applying standardized vibrotactile median nerve stimulation, 3 T brain activation maps were evaluated at 1, 7, 15 weeks and 1 year after surgery. Initially, the affected hemisphere showed no primary activation but increased frontoparietal activity. After 1 year, primary activation had recovered, and frontoparietal activity was decreased relative to the nonaffected hemisphere. Based on these longitudinal fMRI patterns, we propose a new marker for restoration of somatosensory function, which may not be provided by electrophysiological methods.
Keywords
Introduction
Final outcome after surgical repair of peripheral nerve transections varies. Cross-sectional cortical neuroimaging data indicate that neuroplastic changes occur throughout the regeneration process. 1 However, the time course of such changes is not known, although it may be expected that the time course of brain activation changes is associated with the final outcome. In this article, we present the first longitudinal functional magnetic resonance imaging (fMRI) observations of cortical somatosensory reorganization patterns after isolated peripheral nerve transection acquired with special emphasis on standardized and replicable methodology.
Case Study
A 43-year-old man presented with isolated complete transection of the right median nerve at the volar side of the wrist and underwent immediate epineural end-to-end coaptation. Initial clinical assessment revealed sensory and motor deficits, which corresponded to a typical complete median nerve lesion at the wrist, including distal loss of sensation of digits II and III, loss of thumb opposition, and weakening of thumb abduction. During the period of loss of sensation in the territory of the median nerve, the patient was treated by an enhanced sensory relearning program using transmodal sensory input via 3-dimensional audiovisual signals and kinesthetic training. 2
Clinical evaluation was performed with serial somatosensory and motor function testing, performed at weeks 8 and 12 and months 6, 9, and 12, using a protocol designed for quantification of peripheral nerve repair. 3 Somatosensory evaluation of the distal phalanx of digit II was done with Semmes–Weinstein monofilaments 4 —synthetic filaments that are pressed against the skin, thereby bowed to a C-shape for 1.5 seconds. Filaments vary in their rigidity and are numbered according to the weight required for bowing (logarithmic force on a base 10, ie, filament 6.65 requires log 10 × 6.65 g for bowing). Clinical testing showed a trend of improvement over time: numbers of monofilaments for the respective time points of 0, 0, 6.65, 4.56, and 4.31, corresponding to a sensitivity level of 66% relative to the healthy hand by 12 months after injury. Motor function was tested by thumb abduction and a Jamar dynamometer; strength progressed from 32% (week 8) to 66% (month 12) relative to the healthy hand.
Applying a standardized vibrotactile stimulus 5 and an individually constructed head fixation helmet for exact head repositioning with repeated measurements, 6 we evaluated fMRI brain activation maps at 3 T at 1, 7, and 15 weeks and 1 year after surgery. We stimulated digits II and III, left versus right hand, 10 runs each, in a block design with alternating rest and activation periods (20 seconds each, 3 rest, 4 activation periods). Data analysis was performed using SPM8 (Wellcome Department of Cognitive Neurology, London, UK; http://www.fil.ucl.ac.uk/spm): All volumes were realigned to the first volume of the first session, spatially smoothed with a kernel twice the voxel size, and blood-oxygen-level-dependent responses were modeled by a canonical hemodynamic response function function.
Corresponding to previous literature 7 and to recent suggestions to use peak of activation analysis as a marker of reorganization, 8 we identified the peak t value of activation clusters as an indicator for brain activation changes over time. Cross-sectional data from Taylor et al 1 indicated that primary somatosensory cortex and frontoparietal areas of the task associated network 9 are major candidates for functional changes. Therefore, functional regions of interest were defined for these areas (comprising all significant voxels at P < .01 uncorrected) and their peak t values monitored during functional recovery. The peak t values were used to calculate ratios comparing injured and healthy hand stimulation (Figure 1A and B). Within primary somatosensory cortex, peak t values at week 1 were 0.0 (injured hand)/6.6 (healthy hand) but changed to 6.3/8.8 after 1 year. Corresponding peak t values for the frontoparietal cortex were 4.3/3.2 and 4.5/5.2. To illustrate reorganization of primary activation in relation to surrounding secondary frontoparietal activation, a “regeneration index” was calculated from these injured/healthy ratios (ie, a new ratio of primary vs frontoparietal cortex; Figure 1C). Furthermore, linear regression of the postoperative time points was performed and showed a highly linear decline for the frontoparietal activation ratios (r = .993; P = .007) and a nearly linear increase for primary activation ratios (r = .904; P = .096; see Figure 1).
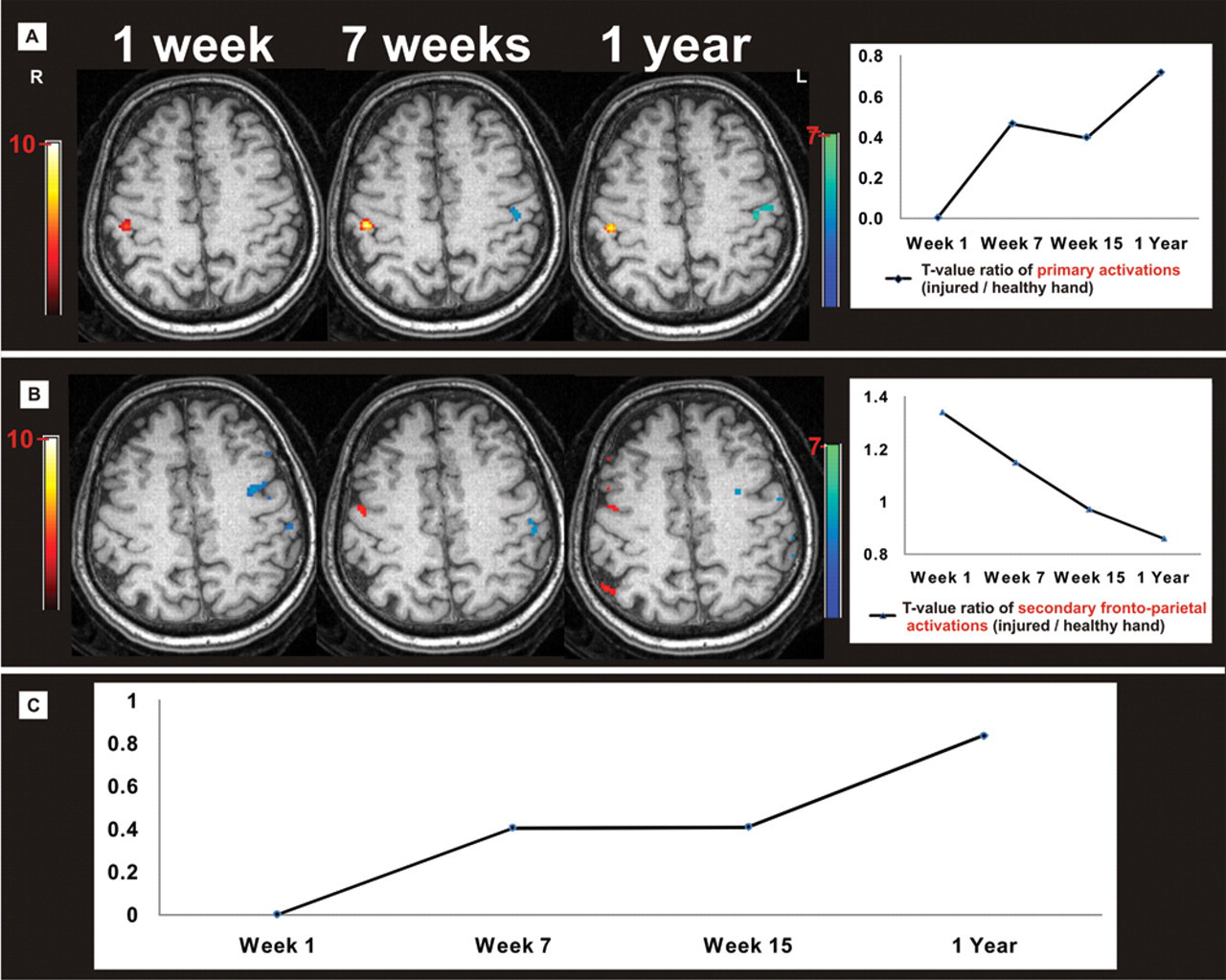
Functional magnetic resonance imaging brain activations with vibrotactile hand stimulation shown on anatomical images. The patient after complete median nerve transection of the right hand and immediate epineural end-to-end coaptation. Vibrotactile stimulation of the left and right hands, performed separately. A, Representative anatomical slice illustrating the time course of maximum t values in the primary somatosensory cortex (injured hand, blue voxels; healthy hand, red voxels). The graph illustrates the peak t value ratios of the injured/healthy hand (P < .01 uncorrected). Only top 150 t value voxels per brain volume are shown for display purposes and color bars indicate t values). B, Same illustration for frontoparietal cortex. C, Proposed regeneration index calculated from graphs A and B (Ratio primary cortex)/(Ratio frontoparietal cortex).
Discussion
Currently, only cross-sectional fMRI data about cortical somatosensory reorganization patterns after isolated peripheral nerve transection exist. 1,10 Diagnostically important information may be achieved with longitudinal observations. Relative to the hemisphere that received sensory input from the uninjured hand, the affected hand’s cortex shows a consistent change of activation patterns. Initially, primary activation was absent and activation of secondary frontoparietal areas shifted to the affected hemisphere. After 1 year, primary activation recovered, similar to frontoparietal activation in the nonaffected hemisphere. These fMRI changes corresponded to the clinical somatosensory function tests. If confirmed in larger patient cohorts, the proposed regeneration index might serve as a new comprehensive cortical marker for restoration of somatosensory function that is not available by electrophysiological methods.
Footnotes
The author(s) declared no potential conflicts of interest with respect to the authorship and/or publication of this article.
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the Austrian Science Fund (FWF P18057) and the Ludwig Boltzmann Institute for Traumatology, Vienna, Austria.
