Abstract
Background. A loss of sensation in the lower limbs, observed in individuals with diabetes as well as elderly individuals, contributes to postural instability, altered gait patterns, increased risk of falling, and decreased quality of life. Objective. To determine if somatosensory cues delivered to sensate areas of the lower limbs above the ankle joints enhance the control of posture in individuals with peripheral neuropathy. Methods. Twelve subjects with sensory neuropathy due to diabetes participated in static and dynamic balance tests with and without auxiliary sensory cues provided to the lower limbs without stabilizing the ankle joints. During the tests the subjects were required to stand on a fixed or moving computer-controlled platform with their eyes open or closed. Equilibrium scores and response latency were obtained. Results. For all tests, equilibrium scores were significantly larger in experiments with auxiliary sensory cues in comparison with conditions without cues (P < .05). Smaller latency scores were recorded in conditions with auxiliary sensory information. The results indicate that auxiliary sensory cues improved automatic postural responses. Conclusions. The observed enhancement of automatic postural responses has clinical implications that aid in the understanding of postural control in individuals with peripheral neuropathy. Future controlled trials could examine whether devices that provide auxiliary sensory cues can improve balance, mobility, and the performance of daily activities.
Introduction
Approximately 12 million people have been diagnosed with diabetes in the United States. This number is projected to increase to around 14.5 million by 2010, thus raising the cost associated with medical expenses and lost productivity to an estimated $156 billion. 1
Individuals with diabetic neuropathy frequently experience somatosensory deficits such as loss of position, vibration, light touch sense, and sensory ataxia with the loss of ankle reflexes. 2 The loss of sensation secondary to diabetic distal sensory neuropathy has a markedly detrimental effect on postural stability. 3 Since ankle proprioception is a critical factor for postural stability, 4,5 peripheral neuropathy affects the way individuals with diabetes control their posture while standing. Thus, laboratory studies have demonstrated that individuals with peripheral neuropathy show a larger postural sway and sway velocity 6 and delayed postural responses to surface perturbations 7 than patients with non-neuropathic diabetes.
In addition, the loss of sensation associated with diabetic neuropathy contributes to altered gait patterns, feelings of being less safe while standing and walking, increased risk of falling, 8,9 and decreased quality of life. 10 Literature data show that among people aged 60 years and older, individuals with diabetes had a 1.6-fold to 35-fold increase in falls compared with individuals without diabetes. 9,11 Diabetes also appears to increase the risk for fracture in the case of a fall. 12
Recent studies indicate that impaired limb sensation, linked to a large-fiber peripheral nerve dysfunction, is quite common in otherwise healthy people aged 65 years and older and has become substantially more frequent with an increase in age. 13 In particular, age-related impairments in foot and ankle sensitivity have been shown to contribute to poor balance abilities and reduced mobility functions in the elderly. 13,14
Several approaches were developed to address these problems. Among them are therapeutic shoes, inlays, and ankle–foot orthoses (AFOs) that influence tactile and proprioceptive mechanisms resulting in improvements in balance and a reduced risk of falling. 15 Improvements in balance while using these devices are associated with increased feedback from cutaneous receptors in the foot and ankle. 15,16 Subsensory mechanical noise applied to the soles of the feet via vibrating insoles may also lead to reduced postural sway in healthy young and elderly individuals 17 and those with diabetic neuropathy. 18
A number of studies reported that the provision of additional somatosensory cues could attenuate postural sway in standing healthy subjects. 19,20 For example, contact of the index finger with a stationary surface was associated with the reduction of postural sway. 20,21 Fingertip signals may specify the direction and velocity of the body motion and guide the execution of compensatory innervations to minimize body sway well before vestibular thresholds are exceeded. 22 Moreover, sensory input to the hand and arm through a cane can reduce postural sway in individuals without a functioning vestibular system. 23 Provision of an index finger touch on a stable surface also improves postural stability in individuals with peripheral neuropathy. 24 However, if this approach is implemented outside of the laboratory, it would require using either a cane 23 or touching walls. For example, using a cane makes one feel old. 25 and nearly half of seniors who could benefit from using a cane or walker refuse to use one. 26 Conversely, providing sensory information to the lower extremities could potentially be an alternative approach helping individuals with peripheral neuropathy to better control their posture while walking. However, no literature data exist on whether the improvement of postural stability in individuals with peripheral neuropathy could be accomplished by providing additional sensory cues to the lower extremities.
Thus, the goal of the current study was to find out whether proprioceptive information delivered to the proximal parts of the lower legs could be used for the control of posture. We hypothesized that auxiliary sensory cues, which bypass the disrupted pathways in the lower legs, enhance the balance in individuals with diabetic neuropathy.
Methods
Subjects
Twenty-three individuals with diabetic neuropathy referred by primary care physicians and neurologists to participate in the study were evaluated by a physician specialized in neurological rehabilitation. Twelve subjects were selected to participate in the study based on satisfying the inclusion criteria. Inclusion criteria were: (a) clinically confirmed peripheral neuropathy with significant sensory loss and no other neurological or musculoskeletal disorders that could have affected their balance, such as ataxia due to central nervous system disorder or osteoarthritis of the knee; (b) ankle, knee, and hip range of motion within normal limits; (c) strength at the toes, ankles, knees, and hips as determined by manual muscle tests of at least 3/5; (d) difficulty in balance maintenance and ambulation not associated with vestibular abnormalities; (e) ability to walk independently with or without any assistive devices; and (f) ability to stand unassisted for 5 minutes. There were 9 men and 3 women; their mean age was 69.5 ± 14.1 years, and the mean time from the disease onset was 14.4 ± 6.2 years. All subjects had clinically confirmed diabetes and sensory neuropathy based on testing for bilateral foot sensation using a Semmes–Weinstein aesthesiometry set (Research Designs Inc, Houston, TX) and vibration using 128-Hz tuning fork; they also had loss of muscle stretch reflexes. The Semmes–Weinstein monofilaments were applied to 3 dorsal and 2 plantar foot zones, 5 times at each site. Correct detection of touch provided by a filament in 3 of 5 trials was considered to be that subject’s sensory threshold.27 The average score for the group of subjects was 4.71 ± 0.9; a score of more than 3.84 is associated with a diminished protective sensation. Sensitivity to vibration was also graded as normal, diminished, or absent by detection of the cessation of vibration applied with a 128-Hz tuning fork placed at the lateral malleolus and hallux.28 Furthermore, we were able to corroborate the diagnosis in 5 subjects participating in the sensory nerve conduction study and found that all of them had an absence of sural sensory nerve action potentials. All subjects had numbness in their feet, and all but 1 patient reported loss of balance while walking. The subjects’ demographics are in presented in Table 1. The Semmes–Weinstein monofilaments and 128-Hz tuning fork technique were also used to assess the sensation above the ankle joint by applying monofilaments to the middle calf area and the tuning fork to the middle tibia. The tests revealed that all of the subjects had normal sensation above the ankle joint (Table 1).
The study protocol was approved by the Marianjoy Rehabilitation Hospital Institutional Review Board. Prior to obtaining all participants’ written consent, the objectives and method of the study were explained.
Demographic Data
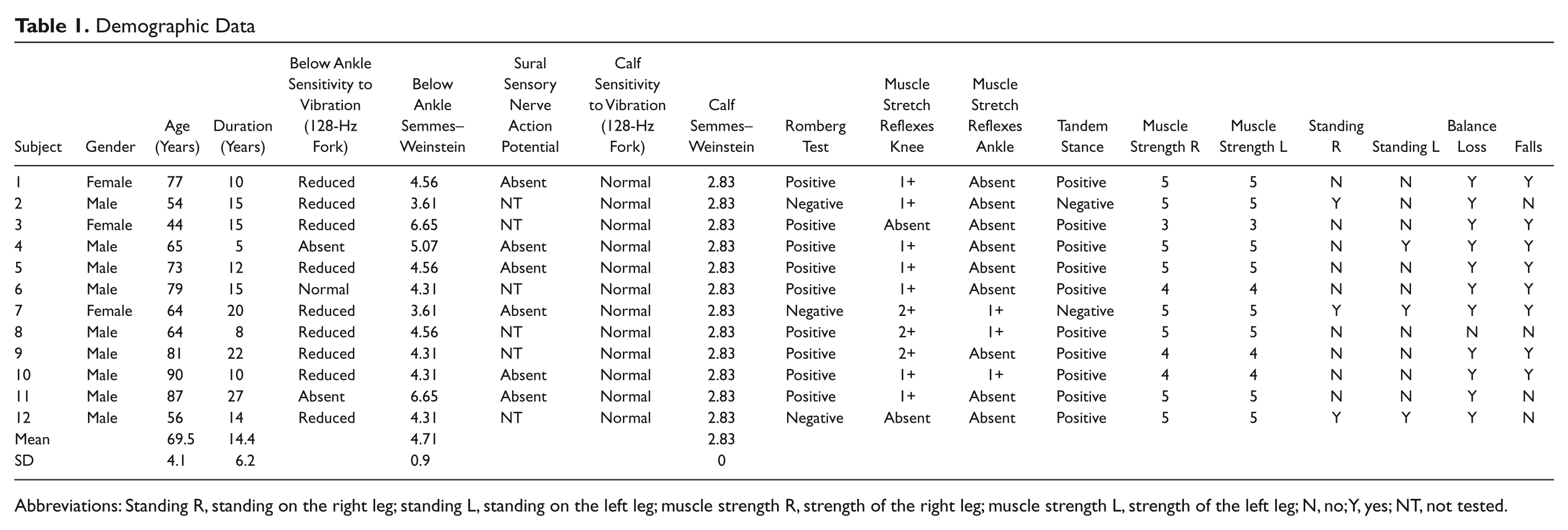
Abbreviations: Standing R, standing on the right leg; standing L, standing on the left leg; muscle strength R, strength of the right leg; muscle strength L, strength of the left leg; N, no; Y, yes; NT, not tested.
Instrumentation
A Computerized Dynamic Posturography (CDP) technique (NeuroCom International Inc, Clackamas, OR) was used. The CDP includes the Sensory Organization (SOT) and Motor Control (MCT) tests. SOT evaluates the efficiency of the subject’s proprioception in balance control using an equilibrium score (ES). ES is a measure of postural stability based on degrees of offset of the subject from the centered position in the anterior–posterior plane. The MCT assesses the efficacy of the patient’s postural responses to platform translations of different magnitudes and directions. A measure of postural response to translations is based on calculating the time (latency) between the onset of the force platform translation and the start of a mechanical response initiated by the subject. 29
The use of CDP in the current study is justified by the following: First, the CDP quantifies both the balance-stabilizing responses (during standing in an unperturbed stance with or without misleading visuals and/or lower leg proprioceptive information, SOT test) and balance-correcting responses (that involve support-surface translations, MCT test). Second, the CDP examination is an established test of postural stability 30 that is widely used for clinical and research purposes in studies of stroke, 31,32 Parkinson disease, 32 vestibular impairment, 33 peripheral neuropathy, 34,35 elderly individuals, 29,36 children, 37,38 and post–space shuttle flights for astronauts. 39 Third, the analysis of the literature suggests that CDP is a reliable and valid test of postural stability. 30 Healthy subjects retested on different days show a minimal learning effect with repeated CDP tests. 40 Indeed, SOT scores that decreased during galvanic stimulation returned to baseline thereafter, despite the retest experience of the subjects. 41
Protocol
During the test session, subjects were asked to stand erect on the force platform with their hands at their sides. Their feet were shoulder width apart, with the medial malleoli over the placement strip imprinted on the platform. The instructions were to stand upright and relaxed throughout the test, with knees slightly bent and arms hanging loosely at the sides of the body. Prior to each testing condition subjects were instructed that during testing the force platform would move.
Sensory organization test
All subjects were exposed to 4 conditions that challenged their ability to integrate somatosensory, visual, and vestibular inputs in maintaining standing balance. Accordingly, in conditions 1 and 2, subjects stood on a fixed support surface with eyes open (SOT1) and closed (SOT2). Test conditions 4 and 5 involved standing on a sway-referenced support surface with eyes open (SOT4) and closed (SOT5).
Motor control test
The subjects were exposed to medium and large forward and backward perturbations of the platform. Each perturbation was applied 3 times within each trial. The duration of platform translations was 300 and 400 milliseconds for the medium and large perturbations accordingly. The amplitude of translation was adjusted by computer software for differences in the subject’s height. The sequence of testing was a medium and large backward translation of the platform followed by medium and large forward translation of the platform. The motor control test was administered with the eyes open. The response latency to perturbations that is defined as the time, expressed in milliseconds, between the onset of translation and the onset of the subject’s active response to the support surface movement was calculated.
Each subject participated in the SOT and MC testing sessions twice, first while standing in a regular erect posture (Figure 1A) that was not associated with any auxiliary sensory information and then with the device providing auxiliary sensory information without stabilizing the ankle joints (Figure 1B). The device was designed as 2 ankle–foot orthoses, each of them consisting of the shank of the brace (1) connected with the foot bed (2) via a semirigid element (3). Velcro calf straps (4) were used to secure the shank of the brace to the leg. The device provided sensory information to the calf via the shank of the brace and to middle tibia via calf straps but at the same time did not provide any ankle stabilization as the connection of the shank of the brace with the foot bed was flexible. A licensed orthotic practitioner was involved in designing of the sensory cues–only device.
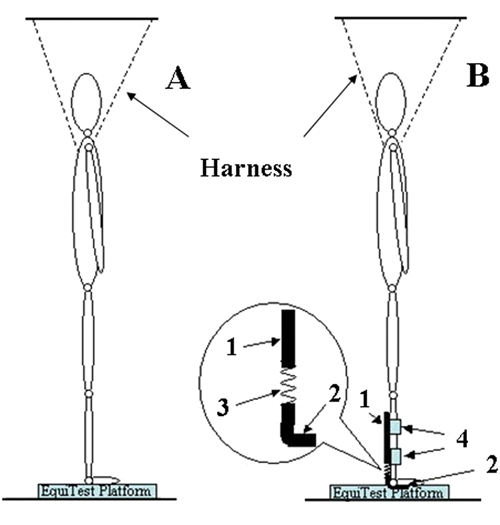
Schematic representation of the experimental set up with a subject positioning on the platform of the Computerized Dynamic Posturography system. (A,) Regular stance without sensory cues; (B) Stance with the device providing sensory cues to the calf area. 1, Shank of the brace; 2, Foot bed; 3, Semirigid element; 4, Velcro calf straps
To be sure that the shank of the brace connected with the foot bed via a semirigid element did not provide any ankle joint stabilization, we measured the horizontal force applied to the calf by that device in a pilot experiment. A miniature load cell, ELFS-B3-100N (Entran Devices, Fairfield, NJ), was taped to the inside of the shank of the brace at the level of the Velcro strap. The signal from the load cell was amplified (Coulbourn Instruments, Whitehall, PA) and collected at a sampling frequency of 100 Hz with 16-bit resolution using customized LabView software. The subject was standing vertical and then swayed backwards for several seconds, simulating sway-referenced CDP conditions. The force applied by the sensory cue only apparatus was about 0.9 N. This value is below the force required to provide mechanical support but is associated with the provision of sensory cues. 20,42 Thus, the results of the pilot experiment confirmed that the device could provide sensory information while not restricting movements in the ankle joint. The order of tests was randomized.
The subjects wore the same standard shoes during all the tests and a safety harness that is required by the manufacturer of the CDP system. No feedback was given to subjects about their performance before the whole assessment was completed. One of the authors stood behind the subject throughout the duration of the tests to assist the subject in regaining balance if needed.
Data Analysis
The SOT scores were obtained for each experimental condition to describe the efficiency of the subject’s proprioception in balance control. The response latencies were calculated to quantify the subject’s active response to the support surface movements.
The independent variable was the availability of sensory information (regular stance and standing with the device providing sensory cues). The dependent variables were SOT scores and latency of the postural responses. Separate repeated-measures analyses of variance were performed on each of the 2 dependent variables. The least significant differences (LSD) post hoc test was performed when needed. Significance levels for multiple comparisons were adjusted with the Bonferroni correction.
Results
The results of the calculation of the scores for the 4 testing conditions are presented in Figure 2. In the first testing condition (SOT1), the subjects demonstrated scores of 92.37 ± 0.55 and 93.00 ± 0.41 without any devices and with the device providing auxiliary sensory information, respectively. In the second testing condition (SOT2), the scores were 85.29 ± 0.97 and 86.80 ± 0.88, respectively. The scores for the fourth testing condition (SOT4) reached 76.75 ± 4.90 and 82.79 ± 1.74 for regular stance and with the device providing auxiliary sensory information, respectively. Finally, the scores for the SOT5 condition were 21.16 ± 6.40 without any devices and 38.29 ± 7.28 with the device providing auxiliary sensory information. The improvement in the SOT scores due to the effect of the device providing auxiliary sensory information was statistically significant (F 1,23 = 9.46; P < .025). The effect of the testing conditions was also statistically significant (F 3,69 = 93.61; P < .0001). Post hoc analysis revealed that the effect of the auxiliary sensory information was statistically significant in the SOT2 (F 1,23 = 9.67; P < .005) and SOT5 (F 1,23 = 5.30; P < .030) conditions.
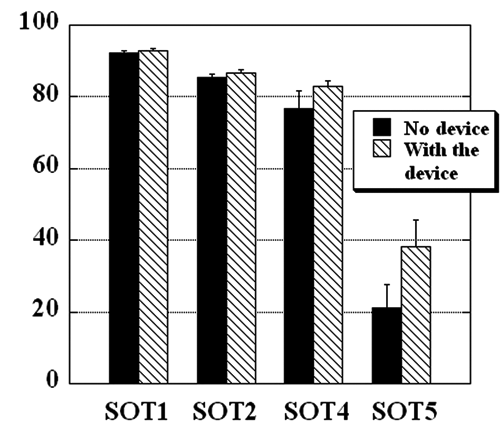
The average SOT scores for 4 balance tests performed without a device providing sensory cues and with the device. Each bar represents the group mean ± standard error
The composite latency scores, the average of the individual scores for the 2 legs calculated for each experimental series, are shown in Figure 3. The mean composite latency score while standing with no auxiliary sensory cue device was 154.11 ± 5.77 milliseconds The latency decreased reaching 147.56 ± 5.00 milliseconds in the series with the sensory cues only device. The difference between the scores was not statistically significant (P = .20).
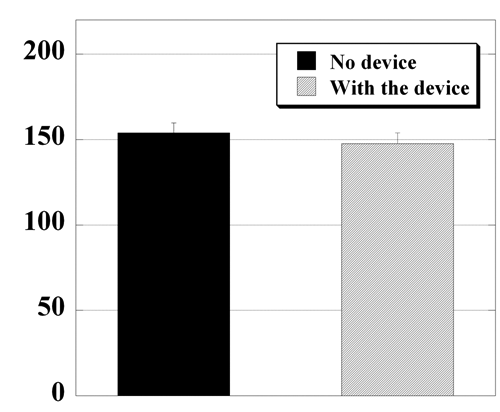
The composite latency scores, in milliseconds, for the 2 experimental conditions obtained for 9 subjects. Each bar represents the group mean ± standard error
Discussion
Studies reported the importance of proprioceptive inflow to scale the postural adjustments. 43 Moreover, several reports suggested that the ability of humans to obtain early directionally sensitive proprioceptive information is not limited to finger touch. Thus, postural sway is reduced when contact of the forehead, nose, or leg at nonsupportive force levels with a stable surface is provided. 22 Similarly, proprioceptive input from more proximal muscles, such as gluteus medius and paraspinals, could provide early directionally sensitive proprioceptive information. 44 In addition, AFOs improve automatic postural responses in individuals with peripheral neuropathy due to diabetes. 34,35 However, in our previous study, 35 improvement in automatic postural responses could have been a result of auxiliary sensory cues or additional mechanical support that became available via the AFOs. The current study was designed to distinguish between the 2 possible sources of improvement of automatic postural responses by using the device that provided sensory cues without additional stabilization of the ankle joints.
There are 2 major findings of the study that support the hypothesis that auxiliary sensory cues that bypass the disrupted pathways in the lower legs enhance balance in individuals with diabetic neuropathy. Improvement in the SOT scores was seen in all experimental conditions with the auxiliary sensory information compared with regular stance, a condition in which no additional sensory information was provided. The largest improvement of automatic postural reactions was seen in the 2 most difficult conditions in which either the somatosensory information was compromised through sway-referencing of the surface on which the subjects were standing (SOT4) or both the somatosensory and visual information was distorted through sway-referencing of the surface and the elimination of visual input (SOT5).Although the effect of auxiliary sensory information was seen in all experimental conditions, the role of additional sensory cues was the largest in the SOT5 condition in which visual information was not available. When vision is not available, the central nervous system (CNS) relies on information from the vestibular and somatosensory systems. 45 There was no change in the utilization of vestibular information between the experimental conditions, so the improvement in automatic postural responses was due to the availability of auxiliary sensory cues.
Second, overall the subjects demonstrated a 4.7% decrease in response latency when they were standing with the device delivering sensory information. This decrease in the time needed to respond to the sudden translation of the platform might reflect the fact that sensory information indeed was used in automatic postural responses. One question to consider is why the time between the onset of translation and the onset of the subject’s active response to the support surface movement was reduced only slightly. It is known that the loss of proprioception due to sensory neuropathy that usually develops over a long period of time has a markedly detrimental effect on postural stability. 3 Since all of the subjects have had a long history of diabetic distal polyneuropathy, it is possible that making available auxiliary sensory cues for a short period of time is not enough for the CNS to relearn how to integrate this information. Indeed, it is known that if the CNS has to reweigh new sensory information, it might require substantial time to establish new means of integration of information needed for efficient postural control. 46 Given the fact that the subjects were exposed only to a limited number of body perturbations while using auxiliary sensory cues, additional training might be needed to enhance their ability to integrate sensory information for postural control.
It is necessary to point out that the experimental protocol included utilization of the harness system; thus, one could suggest that it provided additional support to the body and increased body stability that might have affected the outcome. We believe that this was not the case, since the harness was attached loosely and used as a means of protection against a possible fall. Moreover, the literature data showed that the use of a loose safety harness had no effects on body sway during quiet standing. 47 Nevertheless, the effect of the harness system as a source of sensory information still needs to be studied further.
Very few studies have reported some effect of visual feedback during perturbations of a support platform 48 and exercise interventions on improving physical functioning and balance in individuals with peripheral neuropathy. 49,50 However, there is not much information available on the existence of other interventions aimed at reducing the functional consequences of peripheral neuropathy to enhance one’s mobility and quality of life. 51 In contrast, the results of the current study demonstrate that provision of auxiliary sensory information by bypassing the disrupted pathways in the lower legs could substitute for the lack of proprioceptive feedback from the lower extremities and as such to help individuals with peripheral neuropathy to generate faster and more sensitively scaled postural responses while standing. The findings also are consistent with previous studies of the effect of external vibrotactile biofeedback on stance 52 and gait. 53
Conclusion
This uncontrolled pilot study is a step toward assessing the clinical significance of using auxiliary sensory cues as a balance prosthesis to improve balance in individuals with sensory neuropathies. The next step would be to design and evaluate devices that deliver sensory cues to the calf area, such as socks that incorporate an “exoskeleton” that forms directly onto a lightweight and breathable fabric. Future studies are needed to determine whether devices providing auxiliary sensory cues are effective in enhancing performance of other dynamic balance activities, such as walking, and reducing the incidence of falls.
Footnotes
Acknowledgements
We thank the patients for their exceptional cooperation; Dan Hasso, CPO, for his help in designing ankle–foot orthoses; and Cheryl Carlson, PT, for her help in testing.
The author(s) declared no potential conflicts of interest with respect to the authorship and/or publication of this article.
The author(s) disclosed receipt of the following financial support for the research and/or authorship of this article:
This work was supported by NIH grant HD51628.
