Abstract
Nevirapine (NVP) was the first nonnucleoside reverse transcriptase inhibitor (NNRTI) approved by the US Food and Drug Administration (FDA) in 1996, for the treatment of HIV infection. Current treatment guidelines include NVP as a component of a recommended alternative NNRTI regimen, which may be the preferred regimen for patients with established cardiovascular risk factors since NVP has minimal untoward effects on serum lipids. Two randomized and controlled clinical trials established the noninferior virologic efficacy of twice-daily NVP versus ritonavir-boosted atazanavir (ATV/r), a protease inhibitor with limited effects on serum lipids, each drug on a background regimen of once-daily (QD) tenofovir (TDF)/emtricitabine (FTC). An extended-release (XR) formulation of NVP was developed since QD dosing and reduced pill burdens have been shown to improve regimen adherence. This formulation (Viramune XR 400 mg) was recently FDA approved based on the results of 2 randomized, controlled clinical trials. The XR formulation will provide additional treatment options for patients who may benefit from NVP-based regimens.
Introduction
Nevirapine (NVP) is one of the oldest antiretroviral (ARV) drugs for the treatment of HIV infection and is still in widespread use. With over 1 million patient-years of accumulated experience worldwide, a substantial data set has been generated, providing an increased understanding of this nonnucleoside reverse transcriptase inhibitor (NNRTI). 1 Nevirapine is among the most extensively used ARV drugs in Africa 2 and Asia, 3 giving it a prominent role in the global effort to control HIV infection.
As the first NNRTI approved by the US Food and Drug Administration (FDA), NVP has a well-understood and extensively characterized safety profile. 4 Although it may have some disadvantages, it may also be an acceptable regimen in some populations according to the latest Department of Health and Human Services guidelines. 5 With the institution of new CD4 count treatment initiation recommendations (<400 cells/mm3 for men, <250 cells/mm3 for women), the overall incidences of hepatotoxicity, rash, and Stevens-Johnson syndrome now reported with NVP treatment are comparable with the levels seen with several other ARV drugs. 1,6,7
Due in large part to the success of modern ARV regimens, HIV-infected patients are now living longer. If the current trends continue, by the year 2015 at least 50% of HIV-infected patients will be aged 50 years or older. 8 As a result, the effects of chronic ARV treatment on a patient’s metabolic profile become increasingly important and can influence the choice of treatment regimens. Numerous studies have shown that NVP-based regimens are associated with minimal untoward effects on serum lipid profiles in comparison with other ARV regimens. 4,6 In addition, switching from a protease inhibitor (PI) to a NVP-based regimen has been shown to improve markers of glucose metabolism, including insulin levels and Homeostasis Model of Assessment-Insulin Resistance. 9 These characteristics have important implications for the overall management of cardiovascular disease risk factors in HIV-infected patients.
Although PI-based regimens are generally considered more effective than NNRTI regimens, results from 2 recent randomized clinical studies showed NVP has noninferior efficacy versus ritonavir-boosted atazanavir (ATV/r) when each was administered on a common backbone of once-daily (QD) tenofovir/emtricitabine (TDF/FTC). 6,10
In trial 2 of the OCTANE study, 500 women without prior exposure to NVP were randomized to receive either NVP or ritonavir-boosted lopinavir (LPV/r), both on a background regimen of TDF/FTC. Both arms demonstrated equivalent virologic efficacy as measured by the primary end point of virologic failure or death, which was 14% in each arm. 11
Treatment simplification strategies (eg, reducing dosing frequency or pill burden) have been shown to improve ARV regimen adherence rates, especially in difficult-to-treat populations. 12 Nevirapine has a long half-life, achieving steady-state concentrations exceeding the half maximal inhibitory concentration of NVP, suggesting that a full dose (400 mg) of the original, immediate-release (IR) formulation of this drug could be administered QD. Formal studies comparing key pharmacokinetic parameters between twice-daily (BID) and QD dosing regimens demonstrated equivalent AUC0-24 h (h/μg per mL), half-lives, and clearance. 13 However, mean peak and trough plasma concentrations of NVP were higher and lower with the QD (400 mg) regimen, leading to fears of increased liver toxicity and emergent virologic resistance. 13
Larger clinical trials that directly compared QD NVP (400 mg) to BID dosing (200 mg) generally found no significant difference in virologic efficacy or safety between the 2 dosing regimens. The 2NN trial, with a total of 1216 patients, reported 43.6% and 43.7% of patients as treatment failures at 48 weeks (QD and BID, respectively). 14 Also, no significant differences were seen in virologic efficacy or increases in CD4 counts from baseline. 14 The DAUFIN study reported virologic failure in 8 (22%) of 36 patients in the NVP QD arm. 15 However, the small number of patients and early termination of this trial (with no preset criteria) makes interpretation of these results difficult. Benzie et al reported 89% virologic efficacy (VL <50 copies/mL) at 48 weeks in a group of 173 participants treated with NVP, with 28% of the patients on a QD regimen of NVP. 16 The NODy Study Group also reported that QD NVP was noninferior to BID dosing, with no significant differences in the proportions of patients with virologic failure, nor differences in changes in CD4 counts from baseline. 17 In the recent ArTEN trial, with 569 randomized and treated patients, 67% versus 66.5% of patients on NVP (QD and BID, respectively) achieved the primary end point (VL <50 copies/mL) at 2 consecutive visits prior to week 48. 18 It should be noted that all these results were obtained with the original, IR formulation of NVP.
A new extended-release (XR) QD formulation of NVP (NVP XR), designed to provide more stable 24-hour plasma concentrations, was recently approved by the FDA based on the results of 2 randomized clinical trials demonstrating that the NVP XR has noninferior efficacy and safety in both treatment-naive 19 and treatment-experienced patients 20 compared with the original BID formulation of NVP IR.
New Clinical Trials Addressing Safety and Efficacy of NVP versus a First-Line PI
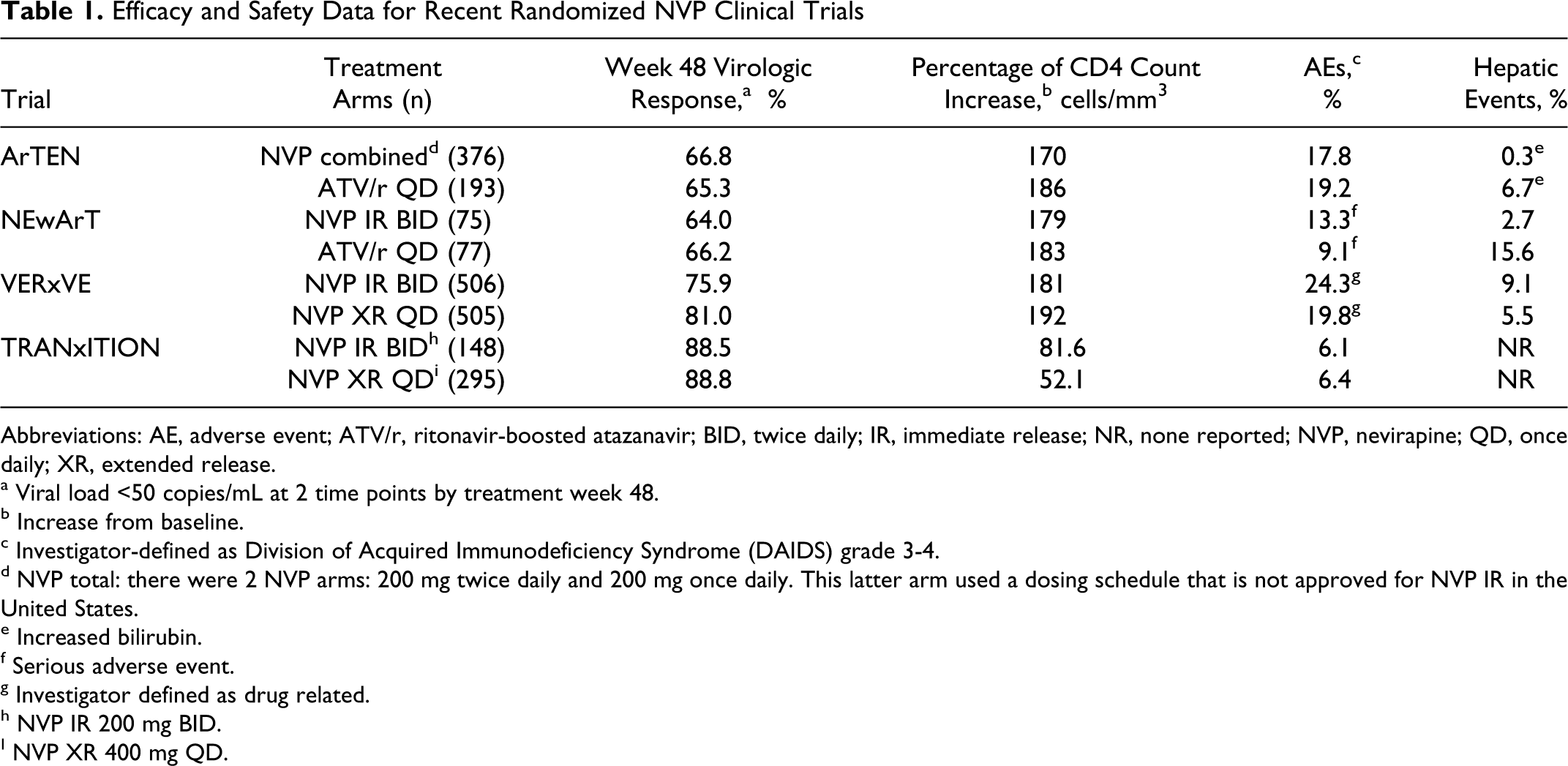
The recent international trial ATV/r on a background of TDF and FTC (Truvada, Gilead Sciences, Foster City, California) versus NVP (ArTEN) was a randomized, noninferiority study comparing NVP (either 400 mg QD [not approved in the United States] or 200 mg BID [US-approved dosing]) versus the PI ATV/r 300 mg/100 mg QD in treatment-naive HIV-infected patients (Table 1). The NVP treatment initiation proceeded by adhering to current CD4 count treatment initiation guidelines; NVP IR at 200 mg QD was administered for a 14-day (lead-in) period in combination with the background regimen of fixed-dose TDF/FTC 300 mg/200 mg QD.
6
If no rashes or laboratory findings consistent with hepatotoxicity were seen, then the lead-in period was followed by a full-dose of NVP IR BID.
6
Of the 569 patients who were randomized and treated in this study, 66.8% of NVP-treated (both 200-mg QD and BID arms) and 65.3% of ATV/r-treated patients reached the primary study end point of HIV RNA<50 copies/mL at 2 consecutive visits prior to week 48. Serious adverse events occurred in 9.6% of NVP-treated patients versus 8.8% of ATV/r-treated patients.
6
Also, NVP-treated patients had a significantly greater increase in high-density lipoprotein cholesterol (HDL-C) and apolipoprotein A1 from baseline than was seen in ATV/r-treated patients, whereas serum triglycerides increased significantly more with ATV/r treatment than with NVP. The mean change from baseline to week 48 in total cholesterol (TC):HDL-C ratio was −0.24 for NVP IR and 0.13 for ATV/r (
Efficacy and Safety Data for Recent Randomized NVP Clinical Trials
Abbreviations: AE, adverse event; ATV/r, ritonavir-boosted atazanavir; BID, twice daily; IR, immediate release; NR, none reported; NVP, nevirapine; QD, once daily; XR, extended release.
a Viral load <50 copies/mL at 2 time points by treatment week 48.
b Increase from baseline.
c Investigator-defined as Division of Acquired Immunodeficiency Syndrome (DAIDS) grade 3-4.
d NVP total: there were 2 NVP arms: 200 mg twice daily and 200 mg once daily. This latter arm used a dosing schedule that is not approved for NVP IR in the United States.
e Increased bilirubin.
f Serious adverse event.
g Investigator defined as drug related.
h NVP IR 200 mg BID.
I NVP XR 400 mg QD.
The NEvirapine compared with Atazanavir, boosted with ritonavir, on a backbone of Truvada (TDF and FTC; NEwArT) trial (ClincialTrials.gov, NCT00552240) was a US-based clinical study designed to support results from the international ArTEN trial, which had similar design and study end points, in a different clinical population (Table 1). Again, all study participants met treatment initiation guidelines for CD4 counts at baseline. In NEwArT, 152 patients were randomized 1:1 to open-label NVP IR 200 mg BID after lead-in dosing or ATV/r (300/100 mg) QD, each on a backbone of TDF/FTC (300/200 mg QD). 10
The primary end point (HIV RNA <50 copies/mL at 2 consecutive visits prior to week 48 without rebound) was achieved in 61.3% of NVP IR-treated and 64.9% of ATV/r-treated patients.
10
Overall grade 3 adverse event (AE) frequencies were similar between treatment arms: 10.7% in NVP-treated patients and 9.1% in ATV/r-treated patients, although grade 4 AEs were higher with NVP IR (9.3%) compared with ATV/r treatment (3.9%). Equal proportions of patients in each arm experienced AEs leading to discontinuation (12.0% vs 11.7%, NVP IR vs ATV/r, respectively).
10
Safety data for plasma lipids indicated a significantly greater increase at week 48 in mean plasma HDL-C in the NVP arm versus the ATV/r arm (9.6 vs 3.5 mg/dL, respectively;
In summary, these 2 recent studies support the observation that the virologic efficacy of NVP IR was noninferior to that of ATV/r when each drug was taken in combination with a TDF/FTC QD backbone. 6,10 The reported rates of AEs were similar between the 2 comparator arms in both studies 6,10 and no cases of Stevens-Johnson syndrome, toxic epidermal necrolysis, or skin- or liver-toxicity-related deaths were observed in either study. 6,10
Durability of Virologic Suppression
Established durability is an important consideration when choosing among potential ARV regimens. Among European patients in clinical care who initially tolerated and experienced virologic suppression with their ARV regimen, a NVP-based regimen demonstrated comparable long-term durability to other ARV regimens (containing efavirenz [EFV] or LPV/r) when followed over 5 years, based on all-cause discontinuations and long-term AEs. 21
In a long-term follow-up of a cohort of French patients infected with HIV, researchers found that 592 of 745 patients receiving NVP were still being followed at the end of the observational period (1996-2008) and that 61% (361 of 592) of patients tracked were still taking NVP.
22
The median observational period for all patients in this cohort was 176 weeks (range 0.3-600 weeks). Efficacy as measured by median CD4 counts increased from 377 (range, 8-1449) to 549 cells/mm3 (range, 144-1621).
22
Of the patients in the cohort followed for 5 years, ARV regimen durability rates were 60.9% for NVP, 41.4% for EFV, and 23% for LPV/r (
Recent Clinical Trials Testing the New QD Formulation of NVP
Treatment simplification strategies (eg, reducing dosing frequency or pill burden) have been shown to improve ARV regimen adherence rates, especially in difficult-to-treat populations. 12 Two recent randomized clinical trials have demonstrated that a recently approved NVP XR QD formulation has noninferior efficacy and safety in both treatment-naive 19 and treatment-experienced 20 patients compared with the original BID IR formulation.
The NVP XR QD formulation was approved by the FDA on March 11, 2011, for use in HIV-infected adults. 23 This approval was based, in part, on the results of VERxVE (A Randomised, Double Blind, Double Dummy, Parallel Group, Active Controlled Trial to Evaluate the Antiviral Efficacy of 400 mg QD neVirapine Extended-Release Formulation in Comparison to 200 mg BID neVirapinE Immediate Release in Combination With Truvada in Antiretroviral Therapy naive HIV-1 Infected Patients; ClinicalTrials.gov, NCT00561925; Table 1). The VERxVE was a recent multinational phase III trial, with a double-blind, double-dummy, actively controlled study design that compared the safety and efficacy of NVP XR (400 mg QD) to the original formulation of NVP IR (200 mg BID). All patients received a nucleoside reverse transcriptase inhibitor (NRTI) backbone regimen of fixed-dose TDF/FTC QD. 19 Treatment-naive, HIV-infected adult patients were randomized and treated with either NVP XR (n = 505) or NVP IR (n = 506) after a 14-day lead-in period with NVP IR (in accordance with the product label for NVP treatment initiation) in all eligible patients meeting the CD4 count guidelines. 19
The primary end point of VERxVE was sustained virologic response through week 48 (defined as 2 consecutive viral load measurements <50 copies/mL, taken 2 or more weeks apart) with no subsequent virologic rebound or change in ARV therapy, according to the FDA’s time to loss of virologic response algorithm. 19 Extended release formulation of NVP was shown to be noninferior to NVP IR, based on the proportion of patients meeting the week 48 primary end point. At this key milestone, 81.0% (409 of 505) of patients in the XR arm versus 75.9% (384 of 506) in the IR arm had undetectable viral loads. 19
No significant differences were seen in the tolerability and safety profiles for patients in the 2 study arms, including the incidence of hepatic events (6% with XR and 9% with IR). 19 However, 2 cases of Stevens-Johnson syndrome were reported—1 grade 3 and 1 grade 4—during the 14-day lead-in phase, when all patients were receiving the IR formulation 19 ; therefore, neither patient was randomized. An additional 3 cases of Stevens-Johnson syndrome were reported, all within the first 30 days of treatment. 19,24
The second clinical trial leading to the approval of NVP XR was TRANxITION, an open-label, phase IIIb, randomized parallel-group study (ClinicalTrials.gov, NCT00819052; Table 1). This trial assessed the efficacy and safety of switching HIV-1-infected patients successfully treated with an NVP IR BID-based regimen to NVP XR 400 mg QD or remaining on NVP IR 200 mg BID-based regimen while maintaining their previous NRTI fixed-dose combinations. 25 For patients with virologic suppression at the time of switching, the CD4 counts could be higher than those recommended by the treatment initiation guidelines because NVP was already successfully initiated and established in these patients. The primary end point was continuation of virologic suppression (viral load <50 copies/mL) at week 24. 25 Results of the TRANxITION study showed the NVP XR QD formulation was noninferior to NVP IR, with 93.6% (276 of 295) of NVP XR-treated patients and 92.6% (137 of 148) NVP IR-treated patients maintaining virologic suppression at week 24. 25 The 48-week assessment also demonstrated virologic noninferiority of the XR formulation, with a sustained virologic response in 88.8% of patients taking NVP XR compared with 88.5% of patients remaining on NVP IR. 20 The 2 formulations also demonstrated no significant differences in mean change from baseline viral loads, mean change from baseline CD4 counts, or time to loss of virologic response. 20 Both treatments were generally well tolerated, with the rates of grade 3/4 AEs equivalent in both arms (6.4% with NVP XR and 6.1% with NVP IR), demonstrating that patients with virological suppression of NVP IR can be safely switched to the XR formulation for regimen simplification. 20
Discussion
NVP, one of the oldest ARVs, is a well-established agent and the first NNRTI to be approved for use in HIV-infected patients. As a result, there is extensive treatment experience using NVP and a very clear understanding of its use as an HIV therapeutic agent. Several years ago, the adoption of CD4 count guidelines for NVP treatment initiation, 1,24 which must be strictly followed, have been shown to reduce the frequency of rash associated with NVP treatment. 24
The management of cardiovascular risk factors is an important consideration, as HIV-infected patients are living longer or becoming infected later in life. Recent trials comparing NVP with ATV/r (another ARV with minimal untoward effects on serum lipids) demonstrated similar between-group virologic efficacy and serum lipid safety profiles when administered as part of well-considered ARV regimens.
It should also be noted that patients who are virologically suppressed on a boosted PI regimen can be safely switched to an NVP-based regimen, with no apparent adverse immunologic effects and with potential benefits to the patient’s metabolic profile. 26 Another important consideration, especially for patients with a history of depression or mental illness, is that NVP treatment was associated with the resolution of EFV-associated neuropsychiatric events in patients switched to an NVP-based regimen. 27
Clinical trials with the XR formulation in treatment-naive and treatment-experienced patients have demonstrated that the virologic efficacy and safety of the recently approved XR formulation (NVP XR) is comparable with the original IR formulation (NVP IR). 19,25 In addition, these data affirm that patients who are stable on NVP IR (BID) can be switched to NVP XR (QD), 24 allowing for regimen simplification. Better adherence and treatment outcomes with ARV regimen simplification have been reported 12 ; however, it should be noted that this has not been demonstrated in a randomized trial with a switch from the IR to the XR formulation of NVP.
Nevirapine has been shown to have comparable efficacy as other first-line NNRTIs, such as EFV. These drugs do differ in their safety and AE profiles and, therefore, individual patient parameters, including comorbidity profiles, need to be considered when making treatment choices. Worldwide use of NVP demonstrates that this NNRTI can be a component of durable, tolerable, and efficacious ARV therapy in both the United States and in diverse settings around the world.
Footnotes
Authors’ Note
The authors are fully responsible for all content and editorial decisions, were involved at all stages of manuscript development, and have approved the final version of this review that reflects the authors’ interpretation and conclusions. Boehringer Ingelheim was given the opportunity to check the data used in the manuscript for factual accuracy only.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr Bhatti has served in the following roles for Boehringer Ingelheim Pharmaceuticals, Inc (BIPI): (1) speaker for the Speakers Bureau, (2) advisor/consultant, and (3) clinical investigator for BIPI sponsored clinical trials. Dr Gladstein has served in the following roles for Boehringer Ingelheim Pharmaceuticals, Inc (BIPI): (1) speaker for the Speakers Bureau, and (2) clinical investigator for BIPI sponsored clinical trials.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Medical writing assistance, supported financially by Boehringer Ingelheim, was provided by José L. Walewski, PhD, of Envision Scientific Solutions, during the preparation of this review.
