Abstract
This study aimed to determine the effect of antiretroviral therapy (ART) on glycemia and transaminase levels in HIV-infected patients in Limbe, Cameroon. A total of 200 ART-experienced patients and 30 ART-naive HIV-infected controls were recruited after submitting signed informed consent forms. The blood samples collected were screened for hepatitis B and C, and liver transaminases and random blood sugar (RBS) levels were determined spectrophotometrically. No significant correlation existed between the RBS and transaminase levels with either the duration of ART or the duration since HIV diagnosis. Although both groups of patients appeared to have similar transaminase and RBS levels, after controlling for confounders, patients on ART had significantly higher aspartate aminotransferase levels, significantly lower alanine aminotransferase levels, and slightly (but not significantly) lower glycemia levels (respective mean differences of 14.92 IU/L, P = .00; 11.95 IU/L, P = .00; and 7.60 mg/dL, P = .19). These changes need to be considered in monitoring patients on ART in this setting and other similar settings.
Introduction
The number of people on antiretroviral therapy (ART) keeps on rising worldwide and in resource-limited settings. The use of ART has been associated with metabolic abnormalities and drug-related toxicities. 1 –4 These drug-related toxicities—hepatotoxicity and abnormalities of glucose metabolism—often lead to treatment interruptions and could be a significant impediment to continued successful clinical outcomes. 5
Patients on ART thus need to be monitored for potential metabolic adverse effects. However, this monitoring needs to take into account the baseline data on the effects of HIV as well as expected clinical levels of transaminases and glycemia in each setting. The latter could be affected by the epidemiology of other causes of hepatotoxicity or hepatitis such as alcohol consumption and viral hepatitis.
There are limited data from resource-constrained settings for an evidence-based approach to monitor the occurrence and evolution of liver transaminase and glycemia abnormalities in HIV/AIDS individuals on ART. 6 This study was therefore aimed at describing the effect of ART (including the duration of therapy) on glycemia and liver transaminase levels in HIV-infected patients in Limbe, Cameroon.
Methods
Study Site and Design
This was a cross-sectional study carried out at the Limbe Regional Hospital Antiretroviral Treatment Center, in the South West Region of Cameroon. The population included HIV-infected ART-experienced and ART-naive adults receiving care at this center.
Participant Recruitment and Study Procedures
Patients attending the Limbe Regional Hospital Antiretroviral Treatment center were approached, informed about the study, and recruited after obtaining written informed consent. They were assisted to complete a structured questionnaire in a face-to-face interview. The questionnaire included assessments of demographic characteristics, medical history, and HIV care (including type of ART, duration since HIV diagnosis, and duration of ART).
Venous blood (4 mL) was collected into 2 tubes (dry and fluoride oxalate) per participant using the vacutainer needles. The fluoride oxalate samples and samples in the dry tubes were taken to the Limbe Regional Hospital laboratory for separation, aliquoting, and hepatitis screening. Plasma from anticoagulated tubes and serum from the dry tubes were obtained by centrifugation at 3000 rpm for 5 minutes. These were then transferred into the labeled Eppendorf tubes using Pasteur pipettes and stored at −20°C for batch analysis on a later day, at the University of Buea, Faculty of Health Sciences laboratory. After aliquoting, 2 to 3 drops of the sera were used to screen for hepatitis B surface antigen (HBsAg) and anti-hepatitis C virus (HCV) antibodies using Acumen Diagnostics HBsAg test strips and Acumen Diagnostics HCV antibody test strips (Acumen Diagnostics Incorporated, China). The frozen sera and plasma were then de-frozen and used to measure, spectrophotometrically, the levels of transaminases (International federation of Clinical Chemistry, Inmesco GmbH, Neustadt, Germany) and glucose (phenol + 4-aminoantipyrine, Inmesco GmbH). All analyses were conducted according to manufacturer’s guidelines.
Statistical Analysis
Data collected were entered into the MS Access interface in EPI-INFO version 3.5.1 software (CDC/World Health Organization [WHO], Atlanta, Georgia) and analyzed using STATA version 10 (StataCorp, College Station, Texas). The mean (± standard deviation) for continuous characteristics (such as age) and the frequency of categorical characteristics (such as gender) were described for the 2 groups of participants and compared using Student t tests and chi-square tests for proportions (or Fisher exact tests when appropriate), respectively. The mean glycemia and transaminase levels in participants on ART were compared with those in ART-naive participants using Student t tests. To control for potential confounding by characteristics such as age, gender, hepatitis, duration on ART, and duration with HIV, multivariate analysis was conducted using multiple linear regression models. Linear regression models were also used to ascertain the average change in the levels of glycemia and transaminase with duration since HIV diagnosis and duration on ART. For all these analyses, the level of statistical significance was set at .05.
Results
Study Participant Characteristics
Study participant characteristics are detailed in Table 1. Patients on ART were generally similar to ART-naive controls except for the distribution in age, gender, and use of medications. The group on ART had a significantly higher proportion of women (86% vs 57%), were significantly older (25% vs 3% aged 50 years or more), and were significantly less likely to have recently received a drug known to have an effect on the liver (4.8% vs 7.6%).
Characteristics of Study Participants.a

Abbreviations: ART, antiretroviral therapy; HBV, hepatitis B virus; HCV, hepatitis C virus; WHO, World Health Organization.
a Some totals may not sum up to 200 or 30 because of missing data.
b Recent refers to within the past month; antiretrovirals were excluded in this tally.
c Medications with known effect on the liver.
d Medications with known effect on glycemia levels.
e Medications with no known effect on the liver or glycemia levels.
Impact of ART on Transaminases and Glycemia
The transaminase and glycemia levels in each group of patients are shown in Table 2. Only 1 patient on ART had alanine aminotransferase (ALT) and aspartate aminotransferase (AST) levels >200 IU/L. Among the patients on ART, the proportion with AST and ALT levels <40 IU/L was 76% and 75%, respectively, as compared to 43% and 57% in ART-naive patients. As many as 4% of patients on ART had random hyperglycemia (>140 mg/dL).
Distribution of Transaminase and Glycemia Levels in ART-Experienced and ART-Naive Patients in the Limbe Regional Hospital, Cameroon.

Abbreviations: ART, antiretroviral therapy; ALT, alanine aminotransferase; AST, aspartate aminotransferase.
aPercentage may not sum to 100 because of rounding.
The impact of ART on the levels of transaminases and glycemia is described in Table 3. Although the ART-naive and ART-experienced patients appeared to have similar mean transaminase and glycemia levels in bivariate analysis, after controlling for confounders, patients on ART had significantly higher AST levels, significantly lower ALT levels, and slightly lower (but not significant) glycemia levels (respective mean differences of 14.92 IU/L, P < .001; 11.95 IU/L, P < .001; and 7.60 mg/dL, P = .19).
Comparison of Mean Transaminase and Glycemic Levels in ART-Experienced and ART-Naive Patients in the Limbe Regional Hospital, Cameroon.a
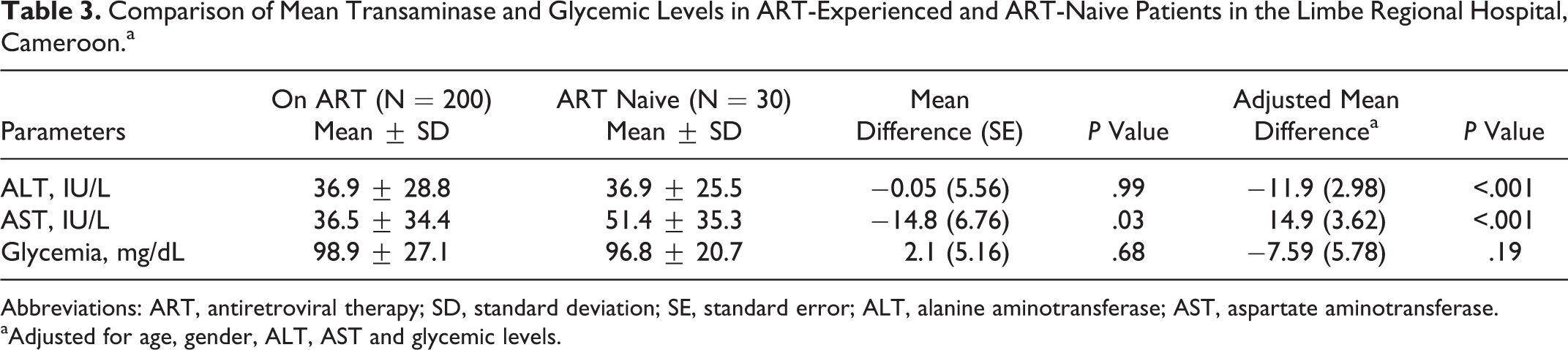
Abbreviations: ART, antiretroviral therapy; SD, standard deviation; SE, standard error; ALT, alanine aminotransferase; AST, aspartate aminotransferase.
aAdjusted for age, gender, ALT, AST and glycemic levels.
Correlation between Time since HIV Diagnosis and Duration on ART with Each of Transaminases and Glycemia
The time since HIV diagnosis ranged from 0 to 96 months (median of 36 months; interquartile range [IQR]: 11-56 months). For those on ART, the duration on ART ranged from 2 to 90 months (median of 34 months; IQR: 14-55 months). No significant correlation existed between the glycemia and transaminase levels with either the duration of therapy or the duration since HIV diagnosis among the patients on ART (Table 4).
Average Change in the Levels of Transaminases and Glycemia over the Duration since HIV Diagnosis and Duration on Antiretroviral Therapy in 200 Participants on ART in the Limbe Regional Hospital, Cameroon.

Abbreviations: ALT, alanine aminotransferase; AST, aspartate aminotransferase.
Discussion
In this study, we compare transaminase levels and glycemia in patients on ART with that in ART-naive patients in Limbe, Cameroon. The mean ALT levels for the ART group were reduced by 11.95 IU/L compared with that for the ART-naive group, and the mean AST levels for the ART group was increased by 14.92 IU/L. The mean glycemia in the ART group was reduced by 7.60 mg/dL compared with the ART-naive group, but the difference was not statistically significant.
It is worth noting that elevated liver enzymes may not be directly caused by the medication in question as patients often take alternative and complementary medicines in association with ART, and several of these have been associated with clear-cut drug-induced liver injury. 7 In addition, an elevation in transaminases may only be a sensitive indicator for liver injury which may not be specific or even clinically relevant as most instances of elevations in transaminases improve despite drug continuation. 8 While statistically significant, the magnitude of the differences in transaminases (all <20 IU/L) probably has little or no clinical significance. Furthermore, ALT levels are more specific for hepatic injury than AST levels. Based on our a priori hypothesis of increased transaminases because of ART-induced hepatotoxicity, the finding of lower ALT levels in patients on ART was unexpected. However, this could potentially be explained by ART being able to reduce the viral load and thus reduce mild viral hepatitis due to HIV. Unfortunately, we cannot unequivocally verify this as the HIV viral loads of participants were not ascertained. Nevertheless, this hypothesis would be consistent with the trends toward a reduction in ALT levels with longer duration of ART.
Several earlier retrospective studies and case reports have associated liver enzyme elevations with specific ART regimens. Sulkowski noted the highest percentages of WHO grade 3 enzyme elevations with ritonavir (RTV), in up to 30% of patients. 9 Nevirapine (NVP) was associated with such elevations in up to 13% of cases; however, only 2.6% were symptomatic. 10 Hepatitis C virus was associated with a 2- to 3-fold chance of “hepatotoxicity” in any ART regimen. 11
The observed similarity in glycemia between the 2 groups is comparable to the findings of Ramezani et al, 12 who reported no significant difference by ART group (11.6% treatment group vs 8.9% in untreated patients).
There was no correlation between the duration of therapy and either transaminase or glycemia levels. This is in contrast to prior findings such as those observed in cohort of patients on ART in south-western France (wherein it was noted that 8% of patients had transaminases >200 IU/L at 1 year after initiating ART and that regimens including 2 nucleoside analogs led to enzyme elevations an average of 3 months after initiating protease inhibitor–based regimens). 11 This is in contrast to prior findings such as those of Kamga et al 3 where a positive correlation was found between duration of therapy and each of AST (r = .81) and ALT (r = .99). The difference may be due to the different study design, period considered, and duration of ART. Kamga et al 3 carried out a 3-year prospective study, where participants at the start of the study were not on ART but about to be initiated on ART, while this study was cross-sectional, participants were already on ART and more than 50% had been on ART for more than 34 months.
Overall, these findings suggest that from a population (rather than individual) perspective, ART seems to have little impact on the levels of transaminases and glycemia. This suggests that monitoring of patients need not be systematic for all patients but may rather need to be based on individual characteristics such as individual risk of viral hepatitis, drug and nondrug toxicities, and alcohol consumption. Furthermore, the transaminase and glycemia levels do not seem to be influenced by the duration on ART.
Some limitations need to be considered in interpreting these data. First, the study used a cross-sectional design with a single measure of transaminases and glycemia being done for each participant. In patients with chronic liver disease, there is a fluctuation in transaminases and a single measurement can underestimate disease burden. 13 A prospective study with multiple samples per participants is thus recommended to better elucidate the time trends in individual patients. The absence of information on participants’ viral load, adherence to ART, information on body mass index and blood pressure (potential indicators of the metabolic syndrome), and other assays of hepatic function such as alkaline phosphatase and γ-glutamyl transferase are also noteworthy limitations. Nevertheless, the strength of this study includes its conduct in a real-life resource-limited setting (noncontrolled as would be data from clinical trials), the inclusion of patients with as long as 7 years on ART, and the assessment and control of other sources of hepatotoxity such as alcohol intake and viral hepatitis B and C.
In this population of patients in Cameroon, those on ART appear to have higher AST levels and slightly lower ALT levels than ART-naive patients. Within the first 7 years, transaminase and glycemia levels also do not appear to be affected by the duration of therapy. These changes need to be considered in monitoring patients on ART in this and other similar settings.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: JA and PMN were partially supported by a grant from the European and Developing Countries Trial Partnership to the Central African Network against Tuberculosis AIDS and Malaria. The Limbe Regional Hospital and the Laboratory of the Faculty of Health Sciences are supported by the Government of Cameroon.
