Abstract
Background:
Obesity is common among patients with HIV. The objective of this study was to characterize response to antiretroviral therapy (ART) in a cohort of obese incarcerated adults compared to a nonobese cohort.
Methods:
A retrospective matched cohort study was conducted in an HIV telemedicine clinic. Patients with body mass index (BMI) >30 kg/m2 who received the same ART with >95% adherence for at least 6 months were matched to nonobese patients by age, gender, ART, CD4 count, and viral load at baseline.
Results:
Twenty pairs were included, with an average BMI of 24 kg/m2 in the nonobese cohort and 35 kg/m2 in the obese cohort. No difference was observed in the proportion of patients who achieved virologic suppression or the change in CD4 count from baseline to 6 to 12 months.
Conclusion:
This study revealed no differences in immunologic recovery or virologic suppression between obese and nonobese patients in an adult correctional population.
Introduction
The body composition of patients with HIV infection has shifted over time, with obesity now more common than the wasting syndrome described in the early years of the disease. 1 Cohort studies in the United States and Canada report that among patients newly diagnosed with HIV, 24% to 31% are overweight and 17% to 20% are obese. 2,3 Within HIV ambulatory practices, 31% to 37% of patients on treatment are reported to be overweight and 25% to 28% obese, with patients often gaining weight after initiating antiretroviral therapy (ART). 2,4 This changing demographic does not appear to result in poor disease control; in fact, overweight or obese patients with a body mass index (BMI) of approximately 30 kg/m2 have improved CD4 T-cell counts after ART initiation compared to normal-weight patients. 5 -7 However, previously published studies of HIV-infected adults with obesity have typically not investigated the potential impact of specific antiretroviral regimens on clinical outcomes, despite the fact that obesity-related pharmacokinetic alterations may affect individual antiretroviral regimens differently. The objective of this study was to characterize clinical response to ART in a cohort of incarcerated patients with HIV infection and obesity compared to a nonobese matched cohort receiving the same ART regimen. The incarcerated population is unique for retrospective HIV studies as adherence can be explicitly monitored and documented.
Methods
We conducted a retrospective matched cohort study of incarcerated adult patients receiving care at an HIV telemedicine clinic at the University of Illinois at Chicago Hospital and Health Sciences System. Inmates in the Illinois Department of Corrections with known or newly diagnosed HIV or AIDS are referred to the clinic, where they elect to receive remotely provided clinical consultation with a physician, pharmacist, and social worker. 8 The study was approved by the institutional review board at the University of Illinois at Chicago, and appropriate safeguards were taken to maintain the confidentiality of collected protected health information in this vulnerable population throughout the study. Patients who received care in the telemedicine clinic between November 1, 2010, and November 30, 2015, were identified by an internal patient database. Patients classified as obese according to the Centers for Disease Control and Prevention definition, with a BMI ≥30 kg/m2, were eligible for inclusion in the study if they had at least 2 sets of immunologic (CD4 count) and virologic (HIV RNA viral load) laboratory markers. 9 A 6-month data collection period was identified for each patient, defined as the first period of at least 6 consecutive months that the patient was on a stable ART regimen with >95% adherence documented in clinic notes. Both treatment-naive and treatment-experienced patients were included in the cohort. Eligible obese patients were then matched 1:1 to patients with a BMI <30 kg/m2 by gender, age within 20 years, antiretroviral regimen, and laboratory values at the beginning of the observation period. Matched patients had to have a CD4 count within the same category of <200 cells/mm3, 200 to 500 cells/mm3, and >500 cells/mm3 and a viral load within the same category of ≤75 copies/mL, 76 to 20 000 copies/mL, and >20 000 copies/mL. In the case of multiple matching controls, the patient with the closest CD4 count was selected. Patients were excluded in the case of incomplete records or if they could not be matched based on the study criteria. The study end points included absolute CD4 count at 6 and 12 months, change in CD4 count from observation baseline, and achievement of virologic suppression (defined as HIV RNA viral load less than 75 copies/mL) at 6 and 12 months.
Statistical Analysis
There was no formal power analysis for this retrospective study, as all eligible patients from the study period were included. Normally distributed data are presented as mean (standard deviation) and compared to the independent samples t test. Nonnormally distributed data are presented as median and interquartile ranges and compared to the Mann-Whitney U test. Categorical data were compared to the Fisher exact test. Change in CD4 count from baseline to 6 and 12 months was analyzed with a paired Student t test. All analyses were conducted in SPSS (IBM Corporation).
Results
Twenty matched pairs were identified from clinic records, with an average BMI in the nonobese cohort of 24 kg/m2 (range: 21.4-27.5 kg/m2) and in the obese cohort of 35 kg/m2 (range: 30.8-47.7 kg/m2). Demographic characteristics are presented in Table 1. The majority of patients were males on a single tablet regimen of emtricitabine–tenofovir (TDF)–efavirenz (EFV; n = 12 pairs). Five pairs were on ritonavir (RTV)-boosted protease inhibitor regimens, with 4 receiving darunavir and 1 atazanavir (ATV). Three additional pairs received emtricitabine–TDF and raltegravir, emtricitabine–TDF and rilpivirine, and emtricitabine–TDF–elvitegravir–cobicistat with ATV. At the observation baseline, 75% of patients were virologically suppressed on treatment and few had a CD4 count that would categorize them as having AIDS at observation baseline. There were no statistically significant differences between groups in terms of CD4 count at observation baseline.
Demographic Information of Incarcerated Patients in the Obese and Nonobese Cohort.a
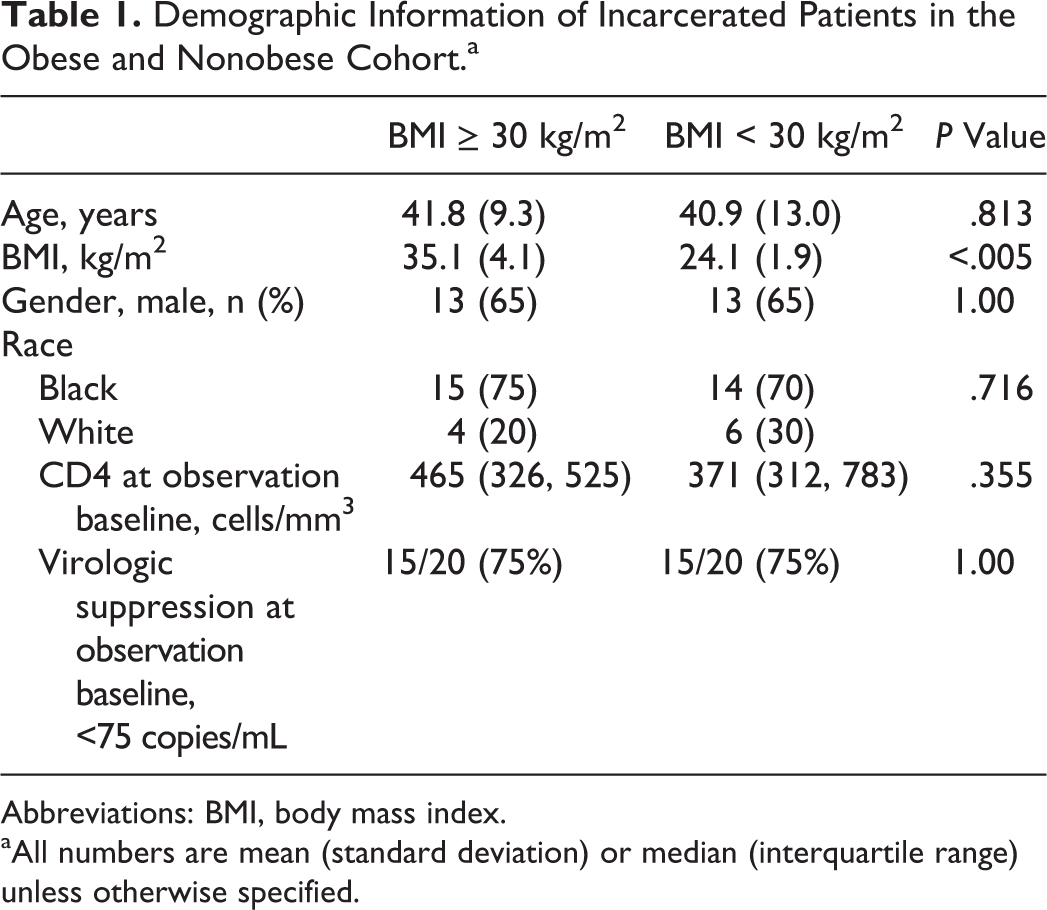
Abbreviations: BMI, body mass index.
aAll numbers are mean (standard deviation) or median (interquartile range) unless otherwise specified.
There was no statistically significant difference in the absolute 12-month CD4 count, proportion of patients who were virologically suppressed at 12 months, or changes in immunologic counts from baseline, as described in Table 2.
Immunologic and Virologic Response to Antiretroviral Therapy in Obese and Nonobese Cohort of Incarcerated Adults.a
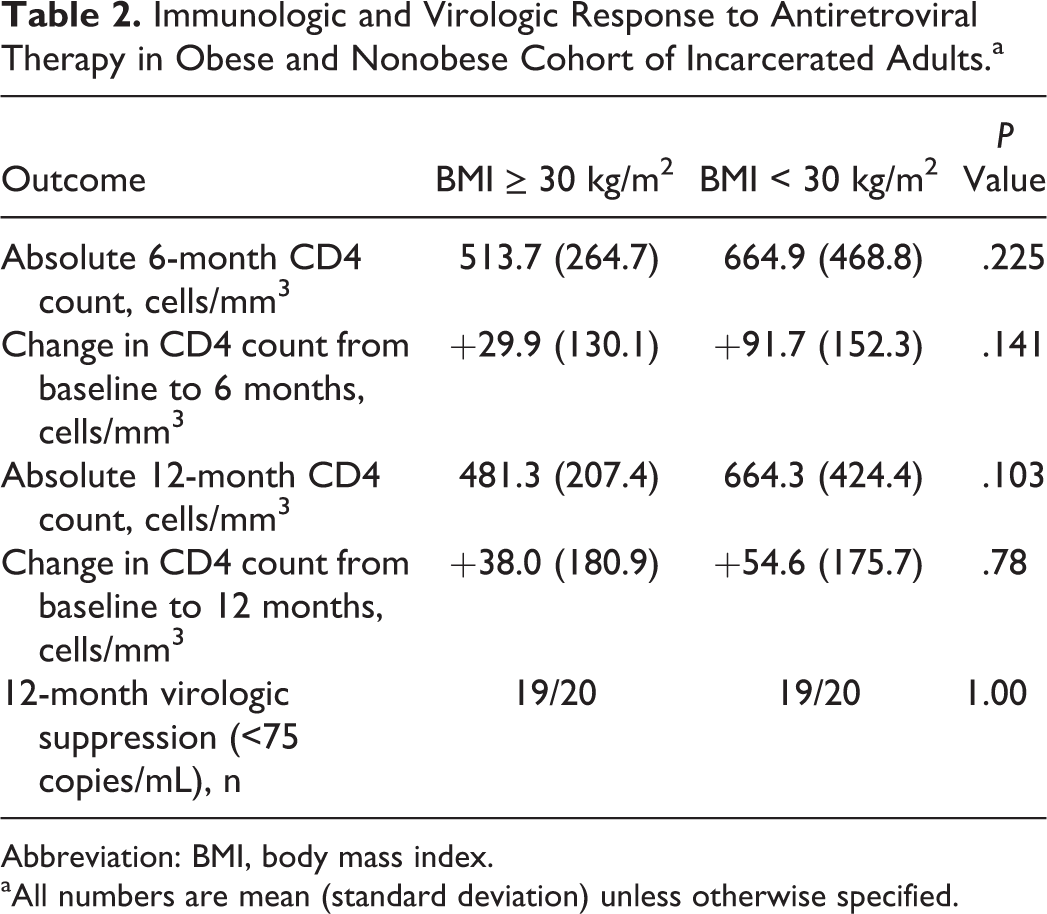
Abbreviation: BMI, body mass index.
aAll numbers are mean (standard deviation) unless otherwise specified.
Discussion
This small observational study of incarcerated adult patients with HIV infection did not demonstrate a significant difference in the proportion of patients achieving virologic suppression among obese and nonobese patients treated with the same antiretroviral regimen. There was also not a significant difference in absolute CD4 count at 6 and 12 months or in the change from baseline.
The proportion of overweight and obese individuals with HIV has increased dramatically since the beginning of the epidemic. Several epidemiologic studies suggest that obesity is problematic not only in developed countries but also in developing nations, where as many as 27.9% to 46% of patients in certain African communities were classified as overweight. 10 -12 The effect of obesity on infectious diseases-related outcomes is variable, with some reports describing the “obesity paradox” in reference to the inverse relationship between BMI and infection-related mortality observed in pneumonia. 13,14 This phenomenon is difficult to explain in the context of dose–response relationships with antimicrobials and bacterial killing and altered pharmacokinetics in patients with a higher body mass. Although antiretroviral regimens have less clearly defined pharmacodynamic targets than antibiotics, studies have linked treatment efficacy and emergence of resistance with minimum plasma concentrations of nonnucleoside reverse transcriptase inhibitors and protease inhibitors. 15 To date, no studies have clearly linked obesity-related pharmacokinetic changes among antiretroviral regimens to poor clinical outcomes. A single case report has described suboptimal plasma levels of EFV achieved with standard dosing in a morbidly obese patient (BMI 66 kg/m2) whose viral load was not fully suppressed until the EFV dose was increased. 16 In the report, EFV 600 mg daily produced an 18-hour level of 806 ng/mL and 1800 mg daily produced an 11-hour level of 2030 ng/mL; the minimum effective concentration for EFV is generally considered to be 1000 ng/mL. 17 In contrast, a recent case–control study of obese and nonobese patients on maintenance ART conducted in a health system with routine therapeutic drug monitoring for antiretroviral regimens demonstrated that although ART concentrations below the typical efficacy thresholds were more common in patients with obesity (>15% for EFV, nevirapine [NVP], etravirine, and raltegravir and up to 24.4% for lopinavir [LPV]), virologic failure was not associated with obesity or subtherapeutic ART concentrations after adjustment for other variables in a multivariate regression. 18 It was not reported whether ART doses were increased in response to subtherapeutic concentrations. Clinical data have supported adequate virological control in cohorts of EFV-treated patients with obesity as well. 19,20 HIV-infected adults starting an EFV-containing regimen from the Collaboration of Observational HIV Epidemiologic Research Europe network reported that body weight >95 kg was not associated with increased time to viral load suppression or a lower absolute CD4 count after treatment initiation. It was important to note that a limited number of patients with a BMI >40 kg/m2 have been included in these studies. 18,20
Our study supports previous literature showing that obese patients have comparable immunologic and virologic outcomes compared to normal-weight patients. There are several limitations to the study that must be addressed. Most (75%) of the study cohort was virologically suppressed at baseline, indicating that they were not treatment naive. We were unable to determine whether these patients had been switched from a previously ineffective antiretroviral regimen prior to the data collection period, which may have biased our observations toward more effective antiretroviral regimens in obese patients. In addition, the majority of patients received an EFV-based regimen, which limited our data about the role of integrase- or protease-based regimens in obese patients. Furthermore, we did not collect information regarding concomitant disease states or medications, which may affect attainment of clinical goals related to HIV infection. Future studies should focus on integrase and protease inhibitors as therapeutic options in obese patients as well as determination of a BMI threshold at which point the pharmacokinetics of antiretroviral agents may be pronounced enough to cause clinically relevant outcome differences.
Conclusion
In a small cohort of obese and nonobese incarcerated HIV-infected adults, obesity did not play a role in the attainment of virologic and immunologic response to standard dosing of antiretroviral regimens.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
