Abstract
Background
The average age of patients starting antiretroviral therapy (ART) in resource-limited settings is below 40 years 1,2 ; however, as the current HIV-positive population ages and as access to treatment increases in resource-limited settings, the absolute number of older patients on treatment is increasing. Few studies have explored the outcomes of older patients on ART specifically in resource-limited settings. Research in developed countries has shown that older HIV-positive individuals progress faster from AIDS diagnosis to death or morbidity compared to younger populations. 3 However, there is a mixed evidence as to whether older HIV-infected patients have a higher risk of mortality or different immunological or virologic responses to ART. 4 -20 In resource-limited settings like South Africa, few sites have large numbers of patients over 50 years. This may limit the power of analyses of data from these sites to determine if outcomes differ among higher age groups.
The Themba Lethu Clinic in Johannesburg, South Africa, is one of the largest HIV clinics in the country, with 968 patients ≥50 years initiated onto ART between April 2004 and December 2008. We explored whether the treatment outcomes, including failure to achieve immunological response and virologic suppression on ART, differ with increasing age among HIV-positive patients.
Methods
Cohort Description
The study was conducted at Themba Lethu Clinic in Johannesburg , South Africa, which enrolled over 18 800 patients in care between 2004 and 2008; over 12 000 of those have initiated ART. Care at Themba Lethu Clinic is provided according to South African National Department of Health guidelines. 21 All patient data at Themba Lethu Clinic are collected and stored in a standardized way using an electronic patient management system (TherapyEdge-HIV, Advanced Biological Laboratories SA, Luxembourg). Demographic, clinical history, and examination data as well as laboratory results (including CD4 counts, full blood counts, and liver function tests) are captured at initiation of first-line ART. At each subsequent medical visit, information on regimen changes, tuberculosis symptom screen, weight, other vital signs, and any new clinical conditions diagnosed including new opportunistic infections are recorded. Treatment monitoring is done with CD4 counts and viral loads (VLs) between 4 and 7 months after initiation of a new regimen (median 3.9 months; interquartile range [IQR] 3.7-4.7) and then approximately 6 monthly thereafter unless clinically indicated. CD4 counts are done using pan-leucogated CD4 flow cytometry (FlowCount Fluorospheres, Beckman Coulter-Immunotech, France), while HIV-1 RNA VL tests are conducted using NucliSENS EasyQ HIV-1 assay (bioMérieux Clinical Diagnostics, France). At each medical visit, patients are seen by a nurse, a doctor, and when appropriate, a counselor. All visit information is collected in real time in the clinic by the clinical staff attending to the patients.
Early on, the treatment patients were scheduled for medical visits every month and 6 monthly thereafter once stable; in between these visits, patients return every 1 to 2 months to collect antiretroviral (ARV) drugs. Visit scheduling is tracked electronically and allows for tracing of patients who have missed visits and for categorization of patients as loss to follow-up (defined as having missed a scheduled medical or antiretroviral pickup for >3 months). Active tracing of those who miss scheduled clinic visits is attempted by telephonic contact and home-based tracing within a month of the missed visit.
Use of Themba Lethu Clinic data was approved by the Human Research Ethics Committee of the University of the Witwatersrand. Approval for analysis of deidentified data was granted by the Boston University Institutional Review Board.
Eligibility Criteria
Our analysis included nonpregnant HIV-infected treatment-naive patients, ≥18 years of age, who initiated ART at Themba Lethu Clinic between April 2004 and December 2008. We limited the analysis to patients initiated onto standard government first-line ART regimens (stavudine [d4T] or zidovudine [ZDV] with lamivudine [3TC] and either efavirenz [EFV] or nevirapine [NVP]). 21
Study Variables
This cohort study compared ART outcomes (all-cause mortality—hereafter referred to as mortality—and loss to follow-up at 12 and 24 months) by age at ART initiation (categorized as 18-29.9, 30-39.9, 40-49.9, 50-59.9, and ≥60 years). We limited the outcomes to the first 24 months on treatment, as long-term mortality would be expected to be higher among older patients. We explored the relation between age and failure to achieve a CD4 response and HIV VL suppression by 6 and 12 months on ART. Mortality is ascertained through the South African National Vital Registration System. 22 -24
Statistical Analysis
Log-binomial regression was used to estimate the relative risk of age on failure to achieve CD4 response (≥50 cells/mm3) and failure to suppress VL (<400 vs ≥400 copies/mL) by 6 and 12 months on treatment. Cox proportional hazard models were used to estimate the hazard of death and loss to follow-up by age category and to identify predictors of mortality and loss to follow-up. For death and loss to follow-up analyses (the only 2 times to event analyses), person–time accrued from ART initiation until the earliest (1) date of death; (2) date of loss to follow-up; (3) date of transfer; or (4) completion of 24 months of follow-up. The proportionality assumption was tested with log (−log(survival probability)) versus time plots for each covariates in the final models and was not violated. Potential confounding factors such as gender, baseline hemoglobin, baseline CD4 count, body mass index (BMI), and World Health Organization (WHO) clinical stage 25 were included in the models where appropriate.
Results
Of the 12 146 patients initiating ART at Themba Lethu Clinic between April 2004 and December 2008, 968 patients were of age ≥50 years. We excluded 47 patients <18 years of age, 1857 nonnaive patients, 974 patients on nonstandard first-line regimens, and 129 pregnant women from the original 12 150 individuals, leaving a sample of 9139 patients eligible for analysis. This included 831 individuals aged ≥50 years who were eligible for further analysis. Patients in this study had a median age of 36.3 years (IQR 31.3-42.5) and a baseline CD4 count of 74 cells/mm3 (IQR 27-144). They were predominantly female (61.8%) and primarily initiated on d4T/3TC/EFV (89.4%). Patients were followed for a median of 24.0 months (IQR 13.5-24.0). We identified 721 (7.9%) patients 50 to 59.9 years and 110 (1.2%) patients ≥60 years old (Table 1). Although the cohort is predominately female, the proportion of males increases steadily with age, with those ≥60 years comprising more males (57.3%) than females. Additionally, older patients, particularly those in the ≥60 category, presented with higher median baseline CD4 counts, BMI, and had fewer WHO stage III/IV conditions compared to younger patients. Greater proportions of patients were initiated on EFV-based regimens with each increasing age category.
Baseline Characteristics and Outcomes by 24 Months on ART Stratified by Age for Patients at the Themba Lethu Clinic in Johannesburg, South Africa (n = 9139)

Abbreviations: ART, antiretroviral therapy; Hb, hemoglobin; LTFU, lost to follow-up; BMI, body mass index; IQR, interquartile range; d4T, stavudine; 3TC, lamivudine; EFV, efavirenz.
Death and Loss to Follow-Up
Among the 9139 patients, 992 (10.9%) died and 1353 (14.8%) were lost to follow-up during the 24-month follow-up period (Table 1). Median follow-up time for patients that died or were lost to follow-up was 4.5 months (IQR 1.6-10.1) and 7.2 months (IQR 4.1-14.3), respectively. The proportion of patients who died increased with age. Over 14% of patients ≥60 years old died during follow-up, compared to 9.0% of patients <30 years. The mortality rate increased from 8.6/100 person years in the 18 to 29 group to 13.5/100 person years among those >60 years. We observed a U-shaped relationship between age and proportion of those lost to follow-up, with patients <30 and ≥60 years having the highest proportion of those lost to follow-up (19.0% and 21.0%, respectively); while only 14.6% of patients aged 50 to 59.9 were lost to follow-up.
Adjusted mortality rates at 12 and 24 months increased with increasing age (Table 2). The largest estimate of this association was in the first 12 months of follow-up among patients aged 50 to 59.9 years (HR 1.67; 95% CI: 1.24-2.23) and, although imprecise, patients ≥60 years (HR 1.54; 95% CI: 0.81-2.95) versus those <30 years had a higher risk of death. Relative increases in risk of death were similar when the follow-up was extended to 24 months. Other predictors of mortality within the first 12 months included male gender (HR 1.23; 95% CI: 1.06-1.42), initiating CD4 count <50 cells/mm3 (HR 2.11; 95% CI: 1.60-2.79), anemia (Hb <10.0 ug/dL) at ART initiation (HR 1.71; 95% CI: 1.47-1.99), and baseline WHO stage III/IV condition (HR 1.21; 95% CI: 1.04-1.42). Predictors of mortality were similar at 24 months.
The Relation between Age and Mortality and Lost to Follow-Up within 12 and 24 Months on Antiretroviral Therapy at Themba Lethu Clinic, Johannesburg, South Africa (n = 9139)


Abbreviations: ART, antiretroviral therapy; BMI, body mass index; WHO, World Health Organization; CI, confidence interval.
a Death obtained from South African National Vital Registration System.
b Hazard ratio (HR) estimated from a Cox proportional hazard model.
c Models also adjusted for baseline ART regimen.
d Lost to follow-up is defined as having missed a clinic appointment by at least 3 months after the scheduled visit date.
In adjusted models, older patients were less likely to become lost to follow-up over 12 months on ART, specifically among patients 30 to 39.9, 40 to 49.9, and 50 to 59.9 years (HR 0.84, 95% CI: 0.71-1.00; HR 0.84, 95% CI: 0.61-1.07; and HR 0.81, 95% CI: 0.61-1.07, respectively) versus <30, though these estimates were somewhat imprecise (Table 2). These hazard ratios (HRs) moved farther from the null by 24 months of follow-up for all 3 age groups. Additionally, we found that male gender (HR 1.32; 95% CI: 1.15-1.52), anemia at ART initiation (HR 1.76, 95% CI: 1.53-2.04), and low baseline BMI (HR 1.56, 95% CI: 1.35-1.80) were important predictors of becoming lost to follow-up by 12 months after ART initiation. These results were similar at 24 months of follow-up.
Immunological and Virologic Response
Of the 7652 who were alive and in care at 6 months, 1358 (17.7%) individuals had missing CD4 counts and 1760 (23%) had missing VL results. Older-age categories were equally likely to have missing values compared to the youngest group. By 6 months on treatment, the adjusted relative risk (aRR) for failure to achieve a CD4 count of ≥50 cells/mm3 was greater for older patients (50-59.9 and ≥60; aRR 1.43, 95% CI: 1.18-1.72 and aRR 1.44; 95% CI: 1.01-2.06, respectively) versus patients <30 years of age (Table 3). Older-age categories also gained, on average, fewer CD4 counts by 6 months on treatment compared to younger age groups. Those aged <30 years gained 40 cells (95% CI: 14-66), 36 cells (95% CI: 7-65), 53 cells (95% CI: 13-94), and 67 cells (95% CI: -23-156) more than the 30 to 39, 40 to 49, 50 to 59, and >60 groups, respectively.
The Relation between Age and CD4 Response and Virologic Suppression by 6 and 12 Months on Antiretroviral Therapy at Themba Lethu Clinic, Johannesburg, South Africa (n = 9139)
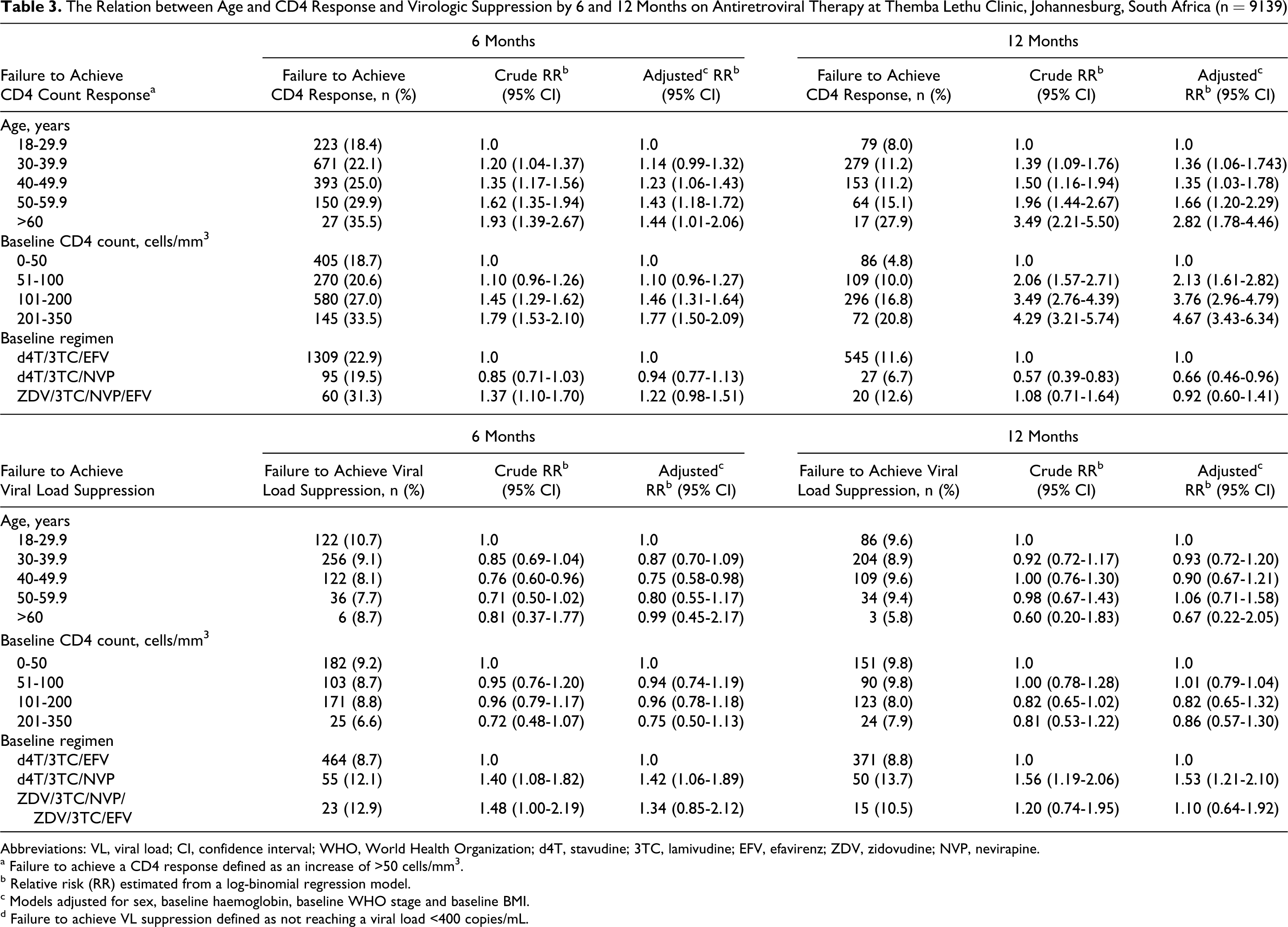
Abbreviations: VL, viral load; CI, confidence interval; WHO, World Health Organization; d4T, stavudine; 3TC, lamivudine; EFV, efavirenz; ZDV, zidovudine; NVP, nevirapine.
a Failure to achieve a CD4 response defined as an increase of >50 cells/mm3.
b Relative risk (RR) estimated from a log-binomial regression model.
c Models adjusted for sex, baseline haemoglobin, baseline WHO stage and baseline BMI.
d Failure to achieve VL suppression defined as not reaching a viral load <400 copies/mL.
Adjusted RR for failure to achieve VL suppression by 6 months on treatment of patients 40 to 49.9 and 50 to 59.9 versus those <30 years old were 0.75 (95% CI: 0.58-0.98) and 0.80 (95% CI: 0.55-1.17), respectively. The point estimates, though less precise due to smaller numbers at this time point, were further from the null after 12 months among patients ≥60 years (aRR 0.67, 95% CI: 0.22-2.05).
Discussion
As few HIV cohorts in resource-limited settings have a large enough population >50 years of age on ART, this study is one of the first to investigate the differences in treatment outcomes among HIV-positive adults >50 years initiating ART compared to younger patients in such settings. As expected, older participants had a higher risk of mortality at 12 and 24 months. Although we estimated the all-cause mortality, HIV and treatment-related factors have been shown to exceed what is considered normal aging in the development of hypertension, hypertriglyceridemia, low bone mineral density, and lipodystrophy. 26 In addition, we found that older age groups mounted a poorer immunological response to treatment by 6 and 12 months of follow-up. However, older participants had a lower risk of becoming lost to follow-up (except in the highest age band where the estimate was imprecise) and some older age groups fared better in terms of VL suppression.
While age-related decline in immune function occurs as part of the natural physiological ageing process, there have been concerns regarding the impact that this declining immunity will have on the already immune-compromised HIV-infected older patient. 27 There is evidence that older patients show poorer CD4 count responses than their younger counterparts, 4 -7,28,29 and our results from this resource-limited setting agree with these findings. Those aged ≥60 were most at risk—this group was nearly 3 times as likely to fail to achieve a CD4 count response compared to those of 18 to 29.9 years. This was despite the fact that the ≥60 group presented for care with fewer signs of advanced disease (CD4 count and WHO stage III/IV clinical conditions) than any other age category including those aged 50 to 59.9. While our analysis of viral suppression lacked precision, our findings concur with previous works on VL suppression and age, 8,11,12,14,15,18,28,29 suggesting an advantage for older age groups. This advantage may potentially be mediated through improved adherence to ART among older populations. 13,28
In our cohort, the proportion of males increased steadily with age. This is in keeping with demographic trends of the age distribution of HIV prevalence in South Africa, 30 which likely reflect the intergenerational and age-disparate trends in sexual relations’ sex patterns (older men have sex with younger women) 31 -33 in this population. Older individuals were also more likely to be employed than the younger patients, potentially representing a more stable population than their younger counterparts. This may also explain the lower rates of loss to follow-up evident among older age groups. Patients who regularly attend their scheduled medical and pharmacy visits are likely to be adherent to treatment and as a result have better ART outcomes. 34,35 Achieving a high level of ART adherence among older patients is notable, considering this population typically have multiple comorbidities due to other age-related chronic illness and subsequently have high pill burdens.
Efavirenz-based regimens appeared to be favored over NVP-based regimens with each increasing age category. Nevirapine is preferred in women who are pregnant or planning to become pregnant. Efavirenz is considered potentially teratogenic and concerns have been raised regarding the potential for neural tube defects in the unborn child if used in the first trimester. 36 Older women are less likely to be pregnant and there were also fewer women in the older age categories as well. This is the likely reason for the increasing use of EFV with age. There is mixed evidence that the outcomes of those receiving EFV are different to those receiving NVP. Some show superior outcomes for those receiving EFV, 37 -39 while a Cochrane review concluded there was no difference in terms of virologic suppression and a 2-cell difference in mean change in CD4 count over 48 weeks of follow-up. 40 If differences in outcomes exist between these treatment groups, it may have confounded the estimates of the effect of age on the clinical outcomes measured. Our results did not, however, suggest any advantage for EFV-based regimens. Additionally all models were adjusted for ART regimen, so any confounding due to ART regimen is likely to be minimal.
Our findings should be considered in light of the study limitations. First, although the study site is one of the largest in South Africa, this study represents patients from only 1 government ART site and therefore may not be generalized to all patients attending national HIV clinics or those in the private sector. Similarly, the generalizability of this data to settings in the developed world may be limited, given the differences in ART regimens used in the developing world. Second, the small sample size in our oldest age group (≥60 years) may have limited our ability to accurately estimate outcomes in this group, and so we interpret these results with caution. Third, we estimate all-cause mortality and are unable to report on HIV-related cause of death versus other causes for this population. Also, the ascertainment of mortality at 24 months may be underestimated as linkage to the South African National Vital Registration System was performed prior to 2010. However, the proportion of valid national ID numbers and the likelihood of having a death ascertained in this manner were similar for all age categories and are unlikely to have strongly biased these results. Finally, we had to rely on surrogate markers of adherence which may have limited our assessment of its role.
Conclusion
The focus of HIV treatment is shifting from an acute to a more chronic approach to disease management for the increasing life expectancy of those affected. This, combined with an increasing number of older HIV-positive persons on ART, has implications for the treatment of these patients. Our findings and those of others suggest there may be benefits to having age-specific ART guidelines. 41 In light of the increased risk of mortality and poorer immune response to ART demonstrated by older patients, creating eligibility criteria allowing older patients to initiate ART at higher CD4 counts could improve outcomes in these high-risk age groups. Financial constraints are always considered in the implementation of such a recommendation particularly in resource-limited settings. Although this subset of the population accessing ART is currently small in terms of absolute numbers in resource-limited settings, these individuals often play a vital caregiver and economic role in their families. 42-43 Improvement in treatment outcomes after initiation of ART among older persons could therefore impact beyond the clinical improvement of an individual to the well-being of an entire family. Additional research to determine age-appropriate immunological criteria for initiation of ART may be a first step in improving outcomes among older HIV-infected adults.
Footnotes
Acknowledgments
We express our gratitude to Babatyi Malope-Kgokong and to the directors and staff of the Themba Lethu Clinic, in addition to Right to Care, the NGO supporting the database and study site through a partnership with US Agency for International Development (USAID). We also acknowledge the Gauteng and National Departments of Health for providing for the care of the patients at the Themba Lethu Clinic as part of the Comprehensive Care Management and Treatment plan.
MM and MF designed the study and along with AB analyzed and interpreted the data and drafted the first manuscript. PM and IS contributed to acquisition of the data and administrative support. All authors provided critical revision of the manuscript for important intellectual content and have read and approved the text as submitted.
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Funding was provided by the US Agency for International Development (USAID) under the terms of agreement 674-A-00-08-00007-00 with Right to Care. The opinions expressed herein are those of the authors and do not necessarily reflect the views of USAID, the Themba Lethu Clinic, or Right to Care. Matthew Fox was also supported by Award Number K01AI083097 from the National Institute of Allergy and Infectious Diseases (NIAID). The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institute of Allergy And Infectious Diseases or the National Institutes of Health (NIH). USAID, NIAID, and NIH had no role in the design and conduct of the study; collection, management, analysis, and interpretation of the data; and preparation, review, or approval of the manuscript.
