Abstract
Fenestrated endovascular repair (FEVAR) has become a safe and effective option for the treatment of complex aortic aneurysms (CAA). Complications can occur, including endoleaks like type IIIc endoleak, characterized by flow between a fenestration and its bridging stent. Although some endoleaks may resolve spontaneously, most require secondary interventions to prevent further expansion of the aneurysmal sac and other complications. We describe a percutaneous “stent-and-plug” rescue maneuver performed in 5 consecutive patients with refractory type IIIc endoleaks after company-manufactured FEVAR grafts. Under moderate sedation and bilateral femoral access, the target-vessel stent was balloon-protected while the free space of the fenestration causing the endoleak was catheterized contralaterally; a 5 × 16 mm iCast™ stent was deployed within the fenestration, and an Amplatzer vascular plug was positioned inside the new stent. Then, the renal-stent balloon was re-inflated to crush the stent-plug complex and achieve a seal in the fenestration. Endoleaks emerged at a mean of 2.47 months (range 1.10-7.1) after the index repair, persisting despite one (n = 3) or 2 (n = 2) prior angioplasty or re-stenting attempts. The stent-and-plug technique achieved technical success in 4 of the 5 cases; the remaining case required one additional procedure to achieve complete sealing. No renal branch occlusion, dialysis-requiring renal injury, or procedure-related mortality occurred. The mean follow-up after rescue was 8.96 months (range 1.4-15.3), with no sac growth observed. Stenting and plugging the fenestration-free space offers a feasible option for treating persistent type IIIc endoleaks when conventional secondary interventions fail; extended surveillance is necessary to confirm long-term durability.
Objective
To report 5 cases of persistent Type IIIc Endoleak treated with a new technique using a stent and a plug outside of the bridging stent after Fenestrated/Branched endovascular aortic repair (F/B-EVAR) using company-manufactured patient-specific devices (CMD).
Introduction
Fenestrated/branched endovascular aortic repair (F/B-EVAR) has shown favorable outcomes, with low morbidity and mortality rates, when compared to historical results of open surgical repair, when treating thoracoabdominal and complex abdominal aortic aneurysms.1,2
Including target vessels in the repair process can lead to instability issues with these vessels, such as endoleaks. Therefore, a limitation of the FEVAR technique is the potential need for secondary interventions (SI) to maintain target vessel patency and ensure continued exclusion of the aneurysmal sac.3-5 Target vessels can be incorporated using either branches or fenestrations. Fenestrations can vary in size; the commonly used configurations are 8*6 mm and 6*6 mm. The use of the former was found to be associated with a higher incidence of type IIIc endoleak and the need for SI. 6 Target vessels may develop either a type Ic or type IIIC endoleak. A type III endoleak is defined as leakage due to detachment between or defects in stent components.
We are reporting 5 cases of Type IIIC endoleak that failed to resolve after at least one conventional secondary intervention.
Case Series Presentation
5 patients were treated using fenestrated endovascular graft under Investigational device exemption, supplied by Cook Medical (Bloomington, IN). These patients subsequently developed type IIIc endoleak (example Figure 1), 3 from the right renal and 2 from the left renal arteries (Table 1). Shows a type IIIc endoleak from the right renal artery Information Regarding Cases and Attempted Secondary Interventions and Outcomes Abbreviations: AVP: Amplatzer Vascular Plug, Fen: Fenestration, F/u: Follow-up, LRA: Left Renal Artery, RRA: Right Renal Artery, SI: Secondary Intervention.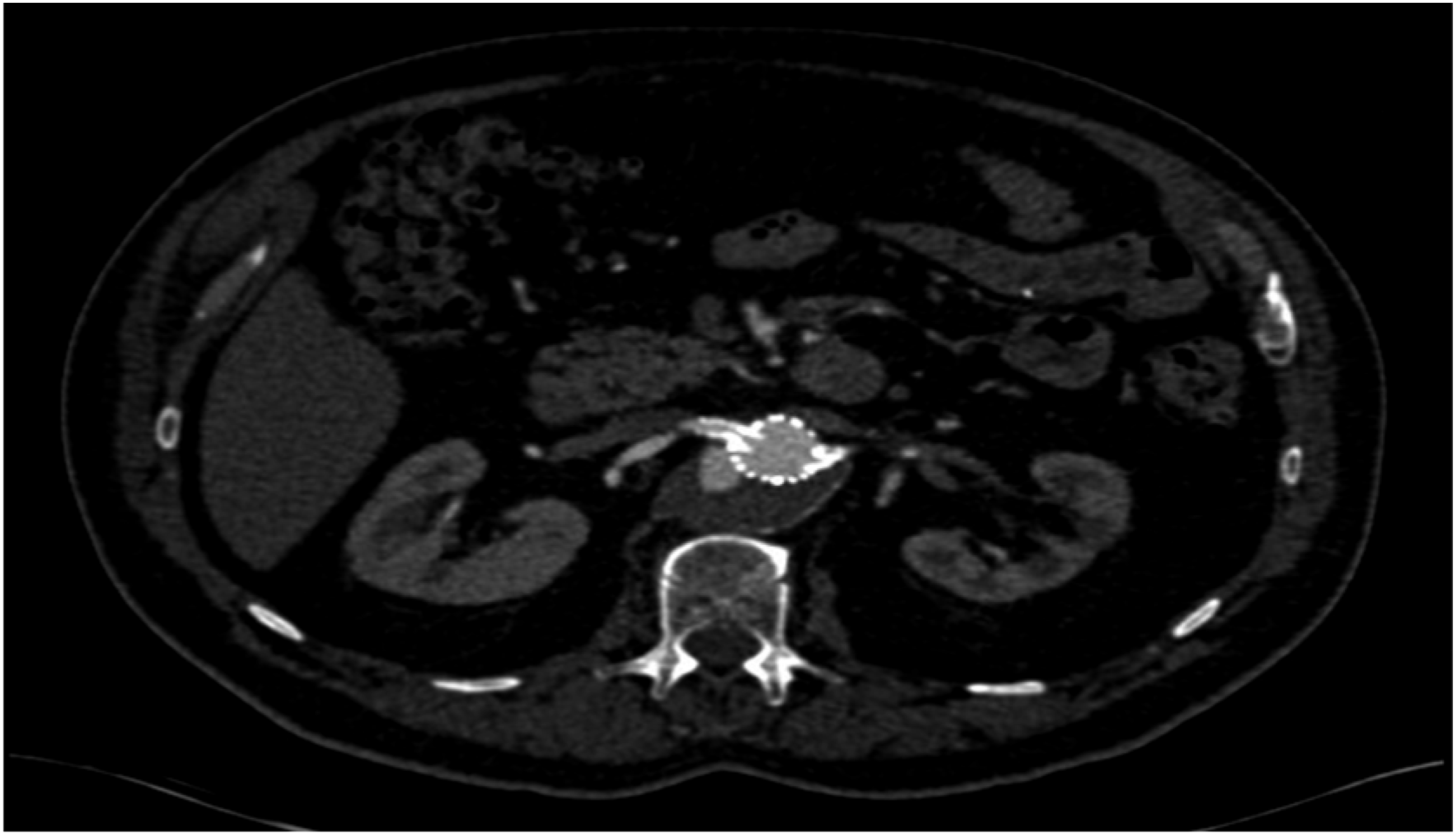
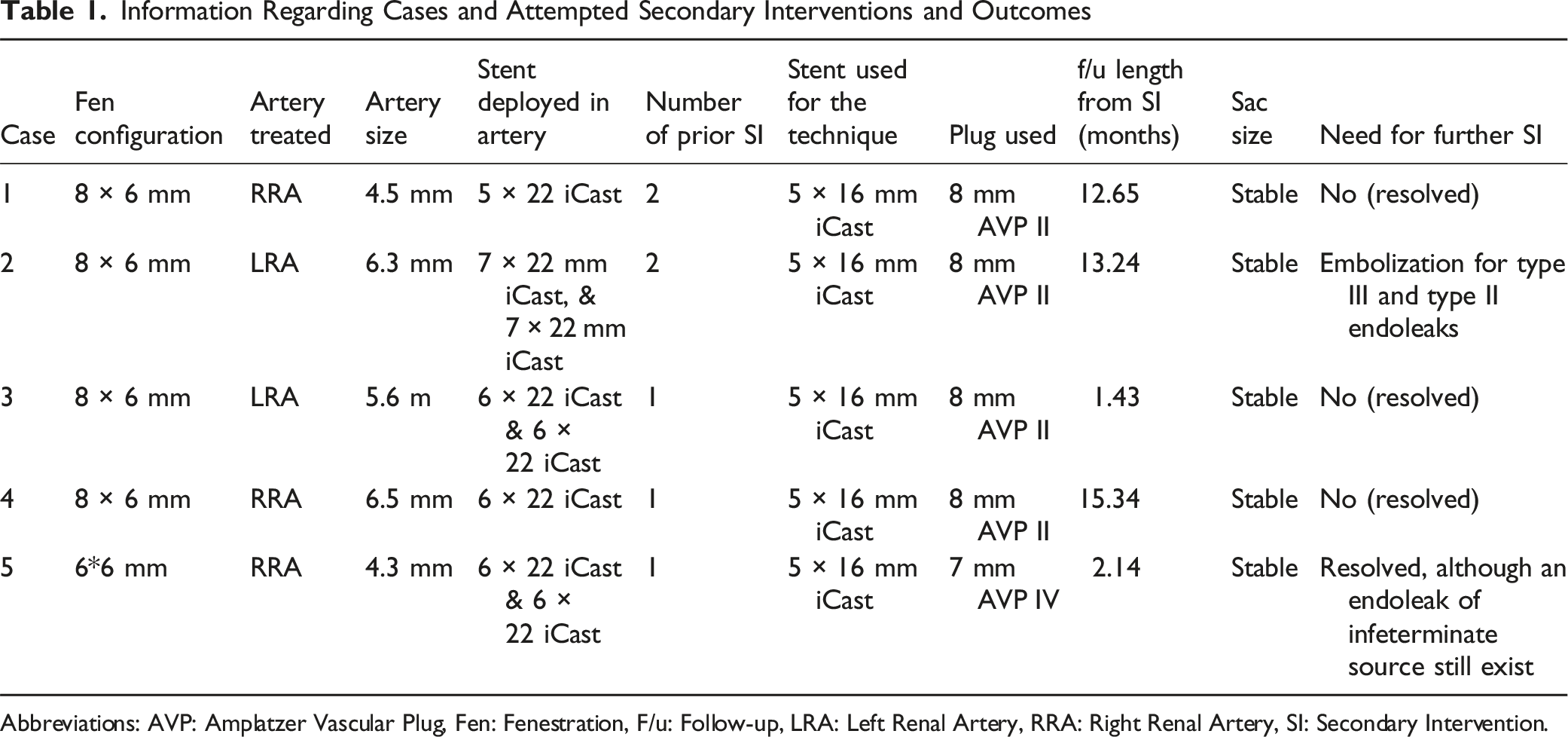
Prior to attempting this technique, all patients underwent conventional type IIIc endoleak reintervention through either angioplasty or restenting of the target artery. Two patients had undergone 2 previous interventions for their type IIIc endoleak, while 3 other patients had only undergone one intervention prior to implementing this technique. Three patients underwent relining using iCast (Getinge, Wayne, NJ), while 2 patients underwent angioplasty.
In all 5 cases, the secondary intervention was performed percutaneously via femoral access, and all were under moderate sedation. Four patients had 8*6 fenestrations, and only one had 6*6 fenestration.
Type IIIc endoleaks are usually identified during follow-up visits. In our institution, the follow-up protocol includes obtaining CT scans of the chest, abdomen, and pelvis at specific intervals after the initial procedure. These intervals are scheduled at 1, 6, 12, 18, and 24 months, then annually for 5 years. A renal-mesenteric duplex is performed every 6 months for the first 2 years and annually afterward. Additionally, after a secondary intervention, we typically schedule a CT scan one month later to confirm the intervention’s success.
The follow-up duration ranged from 1.43 to 15.34 months after the procedure. In one case (Case #2), an additional intervention was required to address the endoleak, despite the sac size remaining stable, due to a type II endoleak. The fifth patient may require reintervention; however, the sac size has remained unchanged, and the source of the endoleak remains uncertain, as the right renal artery—initially suspected—showed no evidence of endoleak at the end of this technique.
Description of the Technique
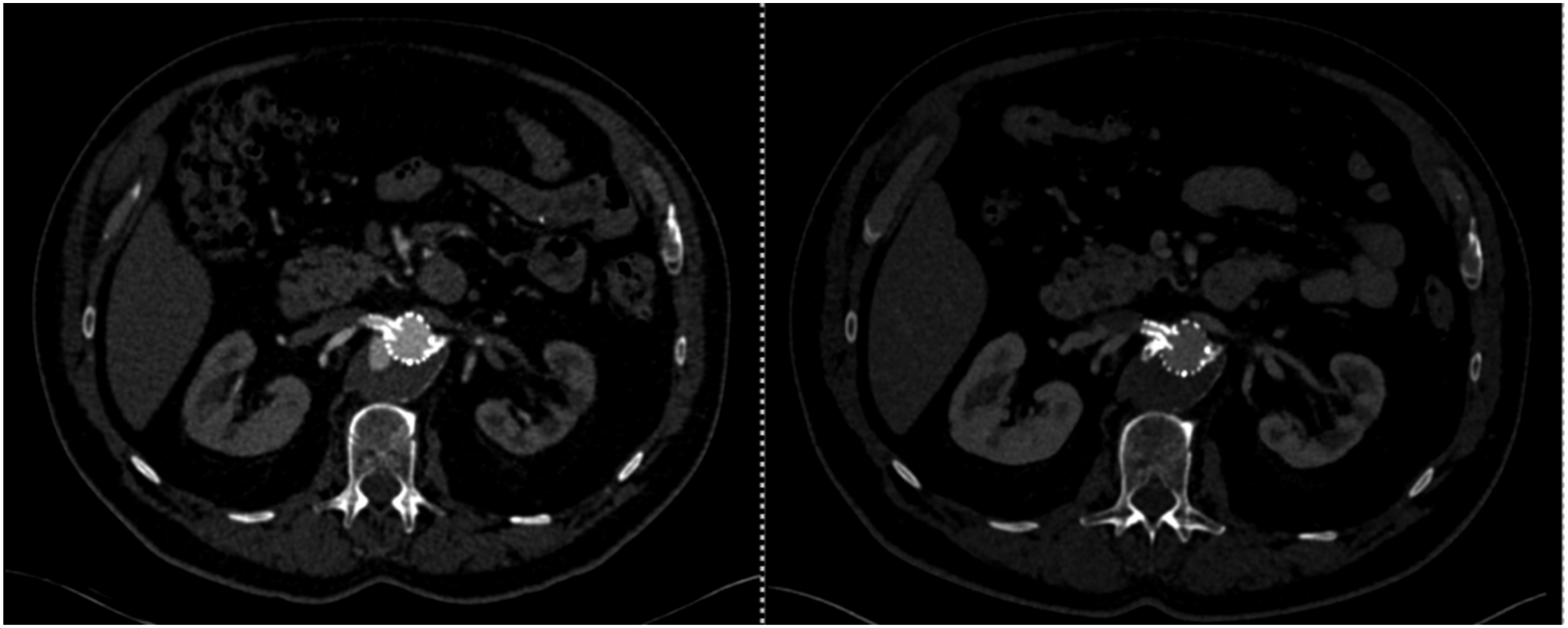
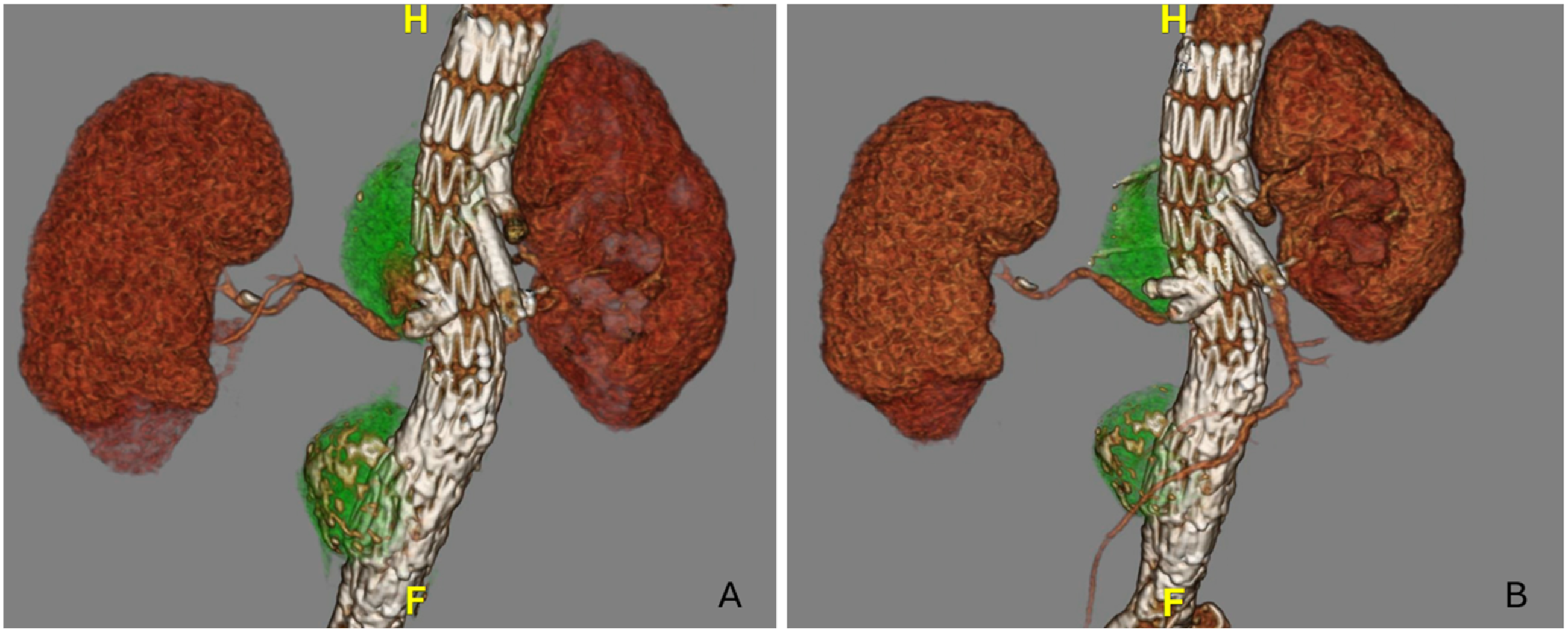
The steps of the technique can be summarized as follows: After bilateral groin access is achieved, we first select the target vessel. Then, we insert a balloon and leave it in the stent to protect it while working in the free space of the fenestrations (Figure 2A). From the contralateral femoral artery, we gain access to reach the free space of the fenestration and place a Rosen Wire (Figure 2B). Subsequently, we advance a 5*16 mm iCast stent into the free space, leaving 8 mm of the stent in the aorta (Figure 2C). After deployment, a plug is advanced into the new stent. Next, we inflate the balloon in the main stent to crush the new stent with the plug inside (Figure 2D). A completion angiogram is performed to ensure good flow through the target vessel and to confirm the resolution of the endoleak Figure 3 shows the differences before and after using this technique on CT axial images with the resolution of the endoleak, and Figure 4 shows the differences on 3D reconstruction. Securing the original stent with a balloon (A), gaining access to fenestration outside the stent (B), Inserting a stent in the free space of the fenestration (C), inserting a plug into the new stent (D) The left image shows an axial view of the endoleak, the right image shows the resolution of the endoleak after stenting the free space Shows the 3D reconstruction of the aorta before (A) and after the reintervention (B)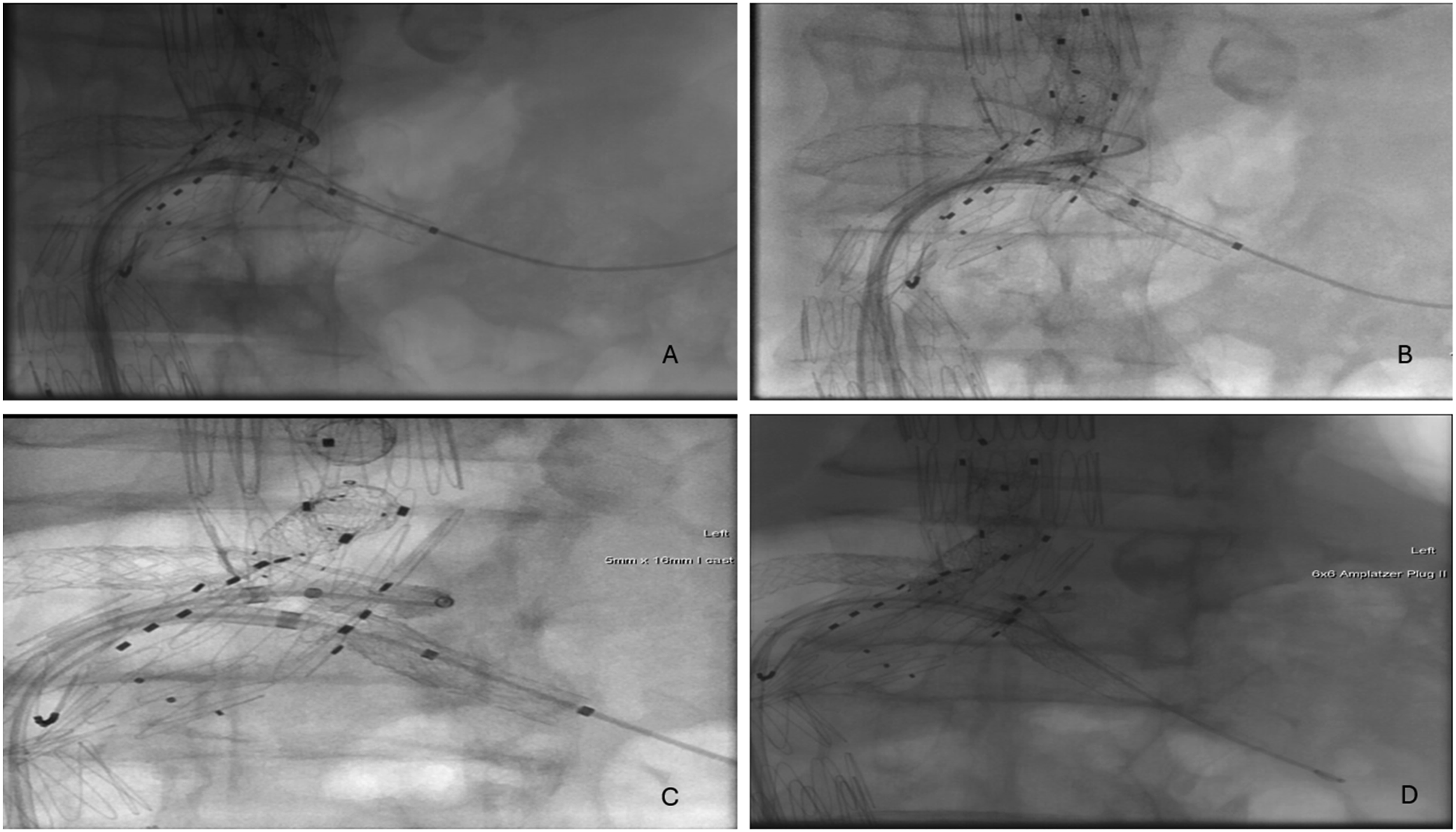
Discussion
Type IIIc endoleak is a concerning adverse event that can occur in any target artery involved in a fenestrated aortic repair, as it can lead to an increase in aneurysmal sac size. It is the most common target vessel complication at 5 years.5The reported incidence of this adverse event in literature ranges from 0 to 5.6% at one month follow-up, 3.4 to 7.1% on long-term follow-up7,8 and up to 9.2% at 5 years. 5 While many of these endoleaks resolve on their own, intervention is necessary when they increase sac size. 9
Gomes et al. 6 conducted a comparison between fenestration configurations of 8 × 6 mm and 6 × 6 mm. The incidence of Type IIIc endoleak was 4.9% compared to 2% in the respective groups, with a significant P-value of 0.005. This resulted in a higher rate of secondary intervention. The choice between the 2 options depends on the surgeon’s preference and the diameter of the target vessel.
Typically, when a persistent type IIIc endoleak is identified and it causes an increasing aneurysmal sac size, relining the original stent or, in some cases, performing angioplasty alone would suffice. It’s rare for a type IIIc endoleak to persist after these interventions. In such situations, stenting the free space within the fenestration and plugging it, while protecting the main stent with a balloon, then crushing the outer stent and the plug might be a viable option. We used iCast stents (Getinge, Wayne, NJ) because of their proven ability to deliver the needed radial force for sealing and their fatigue resistance.10,11
Ueyama et al. 12 outlined a comparable method for addressing paravalvular leak (PVL) following Transcatheter aortic valve replacement (TAVR). They used a variety of combinations involving a covered self-expandable stent (Viabahn) and Amplatzer vascular plug II (AVP II) in a total of 8 patients. The technique is called the Tootsie roll technique, and it was applied to all 8 patients, all of whom experienced successful treatment of their leak.
Following the 1-month follow-up to assess the effectiveness of the secondary intervention, patients resume their regular long-term monitoring schedule as outlined in the FEVAR protocol. This helps detect any new adverse events and confirm that the aneurysmal sac remains excluded, indicated by either a reduction or stable size of the sac and the absence of contrast within the sac. In one patient, a persistent endoleak was observed during follow-up despite a stable sac size. Although the right renal artery was treated using this technique and showed no endoleak on follow-up angiography, it remains unclear whether this vessel is still the source or if the leak originates elsewhere. This diagnostic uncertainty, even after a technically successful intervention, highlights the challenges of endoleak surveillance and the limitations of imaging when sac behavior does not clearly indicate the need for reintervention.
Conclusion
Stenting and plugging the free space within the fenestration after Fenestrated aortic repair is a promising technique for managing persistent Type IIIc endoleaks when used by experienced vascular surgeons, especially in cases where traditional secondary interventions have failed. Long-term follow-up is essential to ensure continued sac exclusion and monitor for potential complications.
Footnotes
Author Note
This study was presented as an oral presentation section at The Southern Association of Vascular Surgery 2025, that was held in St Thomas, USVI, USA, in January 2025.
Ethical Consideration
This study was approved by the University of North Carolina Institutional Review Board (IRB; Study #25-1285). The IRB determined that the research involves no more than minimal risk and granted a waiver of informed consent in accordance with 45 CFR 46.116(f) as well as a waiver of HIPAA authorization in accordance with 45 CFR 164.512(i)(2)(ii).
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Mohammad Alsarayreh: no disclosures. Mark A Farber: Received clinical trial support and is a Consultant for Cook, W. L. Gore & Associates, Getinge, and ViTAA. He has received research support from Cook and has stock options in Centerline Biomedical. Federico Ezequiel Parodi: Received research support from Cook and has stock options in Centerline Biomedical.
