Abstract
Purpose:
Fenestrated stent-grafts with proximal bare-metal fixation stent (BMFS) may provide better rotational movement to facilitate vessel catheterization, but novel designs with covered fixation stents (CFSs) have been increasingly utilized with lower profile fabric. This study aimed to evaluate procedural metrics and outcomes of fenestrated endovascular repair (FEVAR) using BMFS and CFS devices.
Methods:
Clinical data of 502 consecutive patients enrolled in a prospective, non-randomized investigational device exemption (IDE) study were reviewed. Patients with 4-vessel fenestrated stent-grafts with BMFS or CFS for treatment of complex abdominal aortic aneurysms (CAAAs) and Extent IV thoracoabdominal aortic aneurysms (TAAAs) were included. Patients with Extent I-III TAAAs and stent-grafts designed with <4 fenestrations or scallops were excluded. Endpoints were technical success, radiation exposure, major adverse events (MAEs), patient survival, freedom from secondary interventions, and target artery instability (TAI).
Results:
There were 147 patients (29%; 85% male, median 75 years old) treated by FEVAR using 4-vessel fenestrated stent-grafts with BMFS in 79 patients or CFS in 68 patients. Patients treated with BMFS designs had significantly (p<0.05) higher prevalence of cigarette smoking and peripheral artery disease and less often had Extent IV TAAAs. Use of BMFS designs decreased from 88% (64/73) to 20% (15/74) after 2018 (p<0.001). Technical success was similar in patients treated with or without BMFS designs (99% vs 100%, p=0.47), but total operating time and cumulative air karma were significantly lower (p<0.05) in patients with CFS designs. Overall 30-day mortality was 1%, with no difference in mortality and MAEs among patients treated with or without BMFS (1% vs 1%, p=1.0 and 15% vs 12%, p=0.59), respectively. Patient survival at 1 and 3 years was 95%±2%, and 85%±4% for all patients with no difference between groups (p=0.51). Freedom from secondary interventions was 88%±3.6% for patients with BMFS and 86%±4.6% for those with CFS at 1 year (p=0.71), with also no difference at 3 years (p=0.55).
Conclusion:
FEVAR for CAAAs and Extent IV TAAAs was performed with high technical success, low mortality, and MAEs, independent of the use of BMFS. There is no difference in patient survival and freedom from secondary interventions in patients treated with or without BMFS.
Clinical Impact
One hundred forty-seven patients were treated with bare-metal fixation-stent (79) or covered fixation stents (68). Technical success was similar (p=.47). Overall 30-day mortality was 1%, with no difference in major adverse events (MAEs; p=.59). At 1- and 3-year, there was no difference in patient survival (p=.51), freedom from secondary intervention (p=.71). In conclusion, FEVAR for CAAAs and Extent IV TAAAs was performed with high technical success, low mortality and MAEs, independent of the use of bare-metal fixation stent.
Keywords
Introduction
Fenestrated stent-graft devices were traditionally designed with a proximal bare-metal fixation stent (BMFS) for active aortic fixation and to facilitate rotational movement of the device during deployment. The fixation mechanism is intended to decrease migration risk and is specially used for infrarenal aortic aneurysms, pursuing suprarenal fixation, which is less likely to dilate overtime. The role of active fixation for fenestrated stent-grafts to treat complex abdominal aortic aneurysms (CAAAs) and thoracoabdominal aortic aneurysms (TAAAs) is not as well established, but it is assumed that the same principles are needed. In addition to fixation barbs, securing the device into the delivery system may provide better rotational movement to facilitate vessel catheterization. Novel fenestrated stent-graft designs with CFSs have lower-profile fabric and apply the same principles of thoracic stent-grafts. These have been increasingly utilized due to concerns that uncovered stents may induce trauma to the thoracic aorta and are not necessarily needed. The aim of this study was to evaluate procedural metrics and differences in outcomes of fenestrated endovascular repair (FEVAR) using devices with BMFS or devices with CFSs.
Materials and Methods
The study was approved by the Institutional Review Board of the 2 participating institutions, and all patients consented for participation in a prospective, non-randomized investigational device exemption (IDE G130266 and G130030). A total of 502 consecutive patients enrolled in the IDE were reviewed, including the prospectively collected clinical and imaging data. Patients with 4-vessel fenestrated stent-grafts with BMFS or CFS indicated for treatment of CAAAs and Extent IV TAAAs from 2013 to 2022 were included in the analysis. Patients with Extent I-III TAAAs and stent-grafts designed with less than 4 fenestrations or with scallops were excluded.
Clinical data including demographics, cardiovascular risk factors, procedural metrics, and early and late outcomes were prospectively collected in case report forms and stored in the MediRave database. Clinical events were adjudicated by a clinical event committee and data safety monitoring board, internally monitored, and reported annually to the Food and Drug Administration. Preoperative computed tomography angiography (CTA) was analyzed with Aquarius iNtuition software (TeraRecon, Foster City, California) to assess the anatomical extent of the aneurysm and for device planning. Technical success was defined as successful implantation of the investigational fenestrated stent-graft and all intended target vessel stents, patency of all aortic modular stent-graft components and intended side branch components, and absence of type I or III endoleak. The Society for Vascular Surgery (SVS) reporting standards for endovascular repair of complex aortic aneurysms were used for the definition of endpoints. 1
Endpoints were technical success, radiation exposure, MAEs, patient survival, and freedom from secondary interventions, and target artery instability (TAI). The MAEs were defined per SVS guidelines and included any 30-day mortality, myocardial infarction, respiratory failure requiring prolonged mechanical ventilation or reintubation, renal function decline resulting in >50% decline in estimated glomerular filtration rate or new-onset dialysis, bowel ischemia not resolving with medical therapy, major stroke, and paraplegia (grade 3 spinal cord injury). Our follow-up protocol includes clinical examination, laboratory studies, and imaging at baseline, 6 to 8 weeks, 6 months, 12 months, and annually thereafter for 5 years. Imaging included CTA or CT without contrast and duplex ultrasound of the renal-mesenteric arteries at each follow-up interval. Before 2018, a CTA was part of our protocol before discharge, but this changed after 2018 when we replaced it with a cone-beam computed tomography scan intra-operatively. All procedures were performed in hybrid operating rooms using imaging platforms that changed over time: Artis Zeego (Siemens Medical Solutions, Erlangen, Germany) from 2013 to 2015; Discovery IGS 740 (GE HealthCare, Chicago, Illinois) from January 2016 to June 2020; Artis Pheno (Siemens Healthineers, Malvern, Pennsylvania) from July 2020 to June 2021; and Allia IGS 740 (GE HealthCare, Chicago, Illinois) from July 2021 to 2022.
For statistical analysis, the Pearson χ2 test or Fischer’s exact test was used for categorical variables, and they were reported as percentages and numbers. Continuous variables were reported as mean ± standard deviation and were analyzed with the 2-sample t-test or Mann-Whitney U-test. A p-value less than 0.05 was considered statistically significant. For time-dependent results, Kaplan-Meier curves were utilized. To compare time-dependent outcomes, log-rank tests were applied. IBM SPSS Statistics 25 (IBM, Armonk, New York) was the software employed for statistical analysis.
Results
Patients
A total of 502 patients were treated by fenestrated-branched endovascular aortic repair (FB-EVAR) for pararenal and TAAAs in the prospective IDE during the study period. Of these, 147 patients treated by FEVAR using 4-vessel fenestrated stent-grafts with BMFS in 79 patients or CFS in 68 patients were included in the analysis. There were 125 male (85%) and 22 female (15%) patients with a median age of 75 years old (Interquartile Range [IQR]=71-79 years old). Patients treated with BMFS designs had significantly greater prevalence of any history of cigarette smoking, peripheral artery disease and pararenal aneurysms and lower proportion of Extent IV TAAAs (Table 1). The remaining variables had similar distribution in both groups. The use of BMFS designs decreased overtime from 88% (64/73) to 20% (15/74) after 2018 (p<0.001).
Characteristics of 147 Patients Treated With 4-Vessel Fenestrated Endovascular Aortic Repair With Proximal Bare-Metal Fixation Stent (BMFS) or Covered Fixation Stents (CFSs).
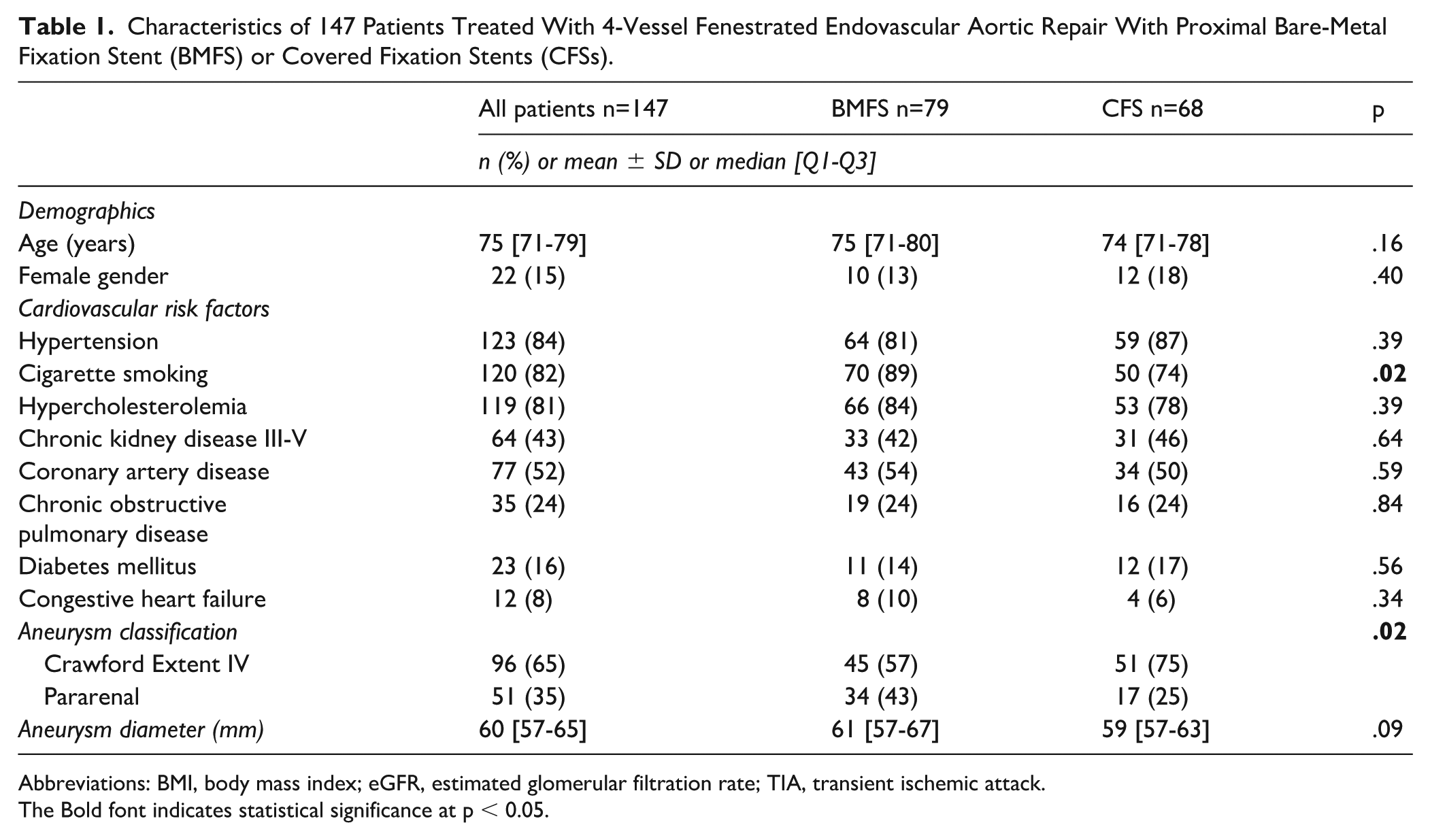
Abbreviations: BMI, body mass index; eGFR, estimated glomerular filtration rate; TIA, transient ischemic attack.
The Bold font indicates statistical significance at p < 0.05.
Procedural Metrics
Technical success was similar in patients treated with BMFS or CFS designs (99% vs 100%, p=0.47), but total operating time and cumulative air karma were significantly lower in patients with CFS designs (Table 2). Technical failure was due to inability to catheterize 1 target vessel, resulting in a residual endoleak. The success rate of intended target vessel incorporation with bridging stent was 99.8%. Median volume of contrast use was 137 mL (IQR=108-170 mL) in patients with BMFS and 130 (IQR=90-168 mL) in patients with CFS (p=0.39). There were no perceived technical difficulties during device deployment among patients with CFS designs.
Procedure Details of 147 Patients Treated With 4-Vessel Fenestrated Endovascular Aortic Repair With Proximal Bare-Metal Fixation Stent (BMFS) or Covered Fixation Stents (CFSs).
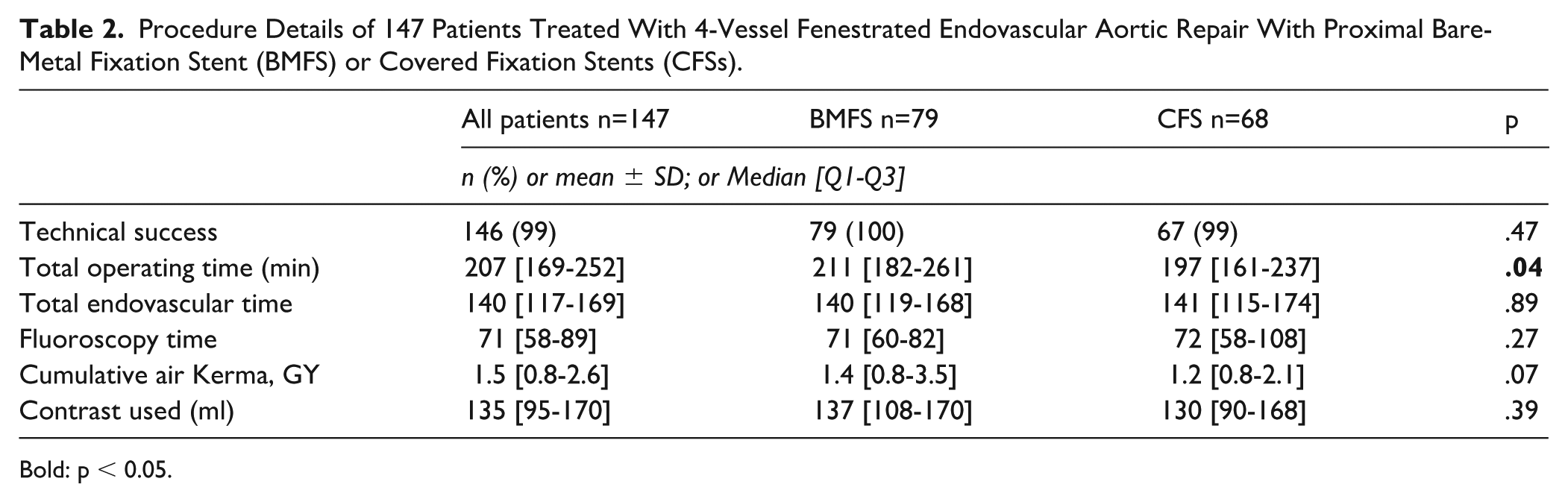
Bold: p < 0.05.
Early and Mid-term Outcomes
There were 2 patients (1%) who died within the first 30 days or hospital stay, with no difference between groups (Table 3). Causes of death included myocardial infarction and mesenteric ischemia in 1 patient each. Major adverse events occurred in 20 patients (14%), with no significant difference in patients with BMFS or with CFS (15% vs 12%, p=0.59), respectively. None of the early mortality or MAEs could be directly attributed to the fixation stent. The median follow-up was 25±21 months. Patient survival at 1 and 3 years was 95%±2%, and 85%±4% for all patients with no difference between groups (Figure 1). Freedom from secondary interventions was 88%±3.6% for patients with BMFS and 86%±4.6% for those with CFS at 1 year (p=0.71), with no difference at 3 years (Figure 2). Table 4 specifies all reinterventions in this cohort. Similarly, there was no difference in freedom from TAI at 1 year for patients with BMFS and CFS (97±0.9 vs 94%±2.0%, p=0.07), respectively. Branch instability occurred in 4% of all incorporated target vessels and was secondary to occlusion or stenosis in 46% of the cases and to endoleak in 54% with equal distribution in both groups. There was no type Ia endoleak or main stent-graft migration during follow-up in both groups.
Mortality and Major Adverse Events (MAE) <30 Days of 147 Patients Treated With 4-Vessel Fenestrated Endovascular Aortic Repair With Proximal Bare-Metal Fixation Stent (BMFS) or Covered Fixation Stents (CFSs).
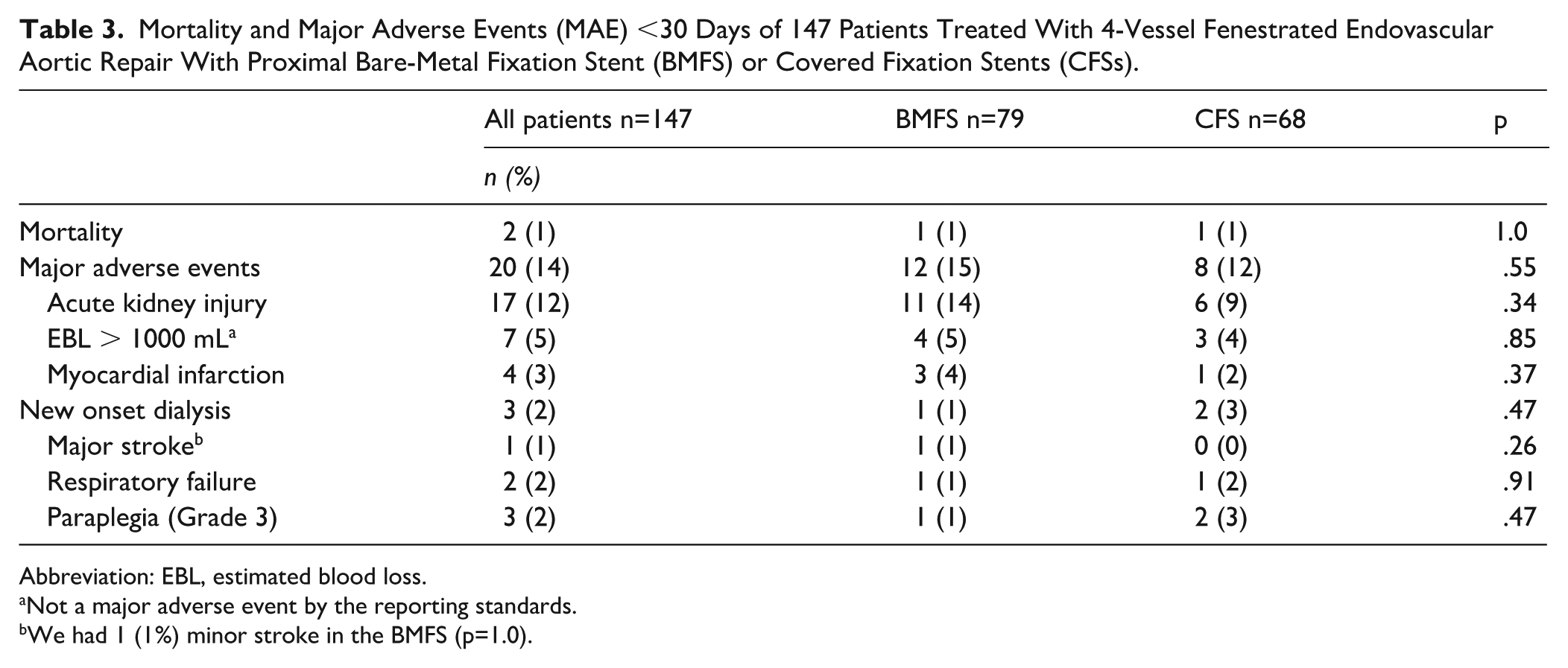
Abbreviation: EBL, estimated blood loss.
Not a major adverse event by the reporting standards.
We had 1 (1%) minor stroke in the BMFS (p=1.0).
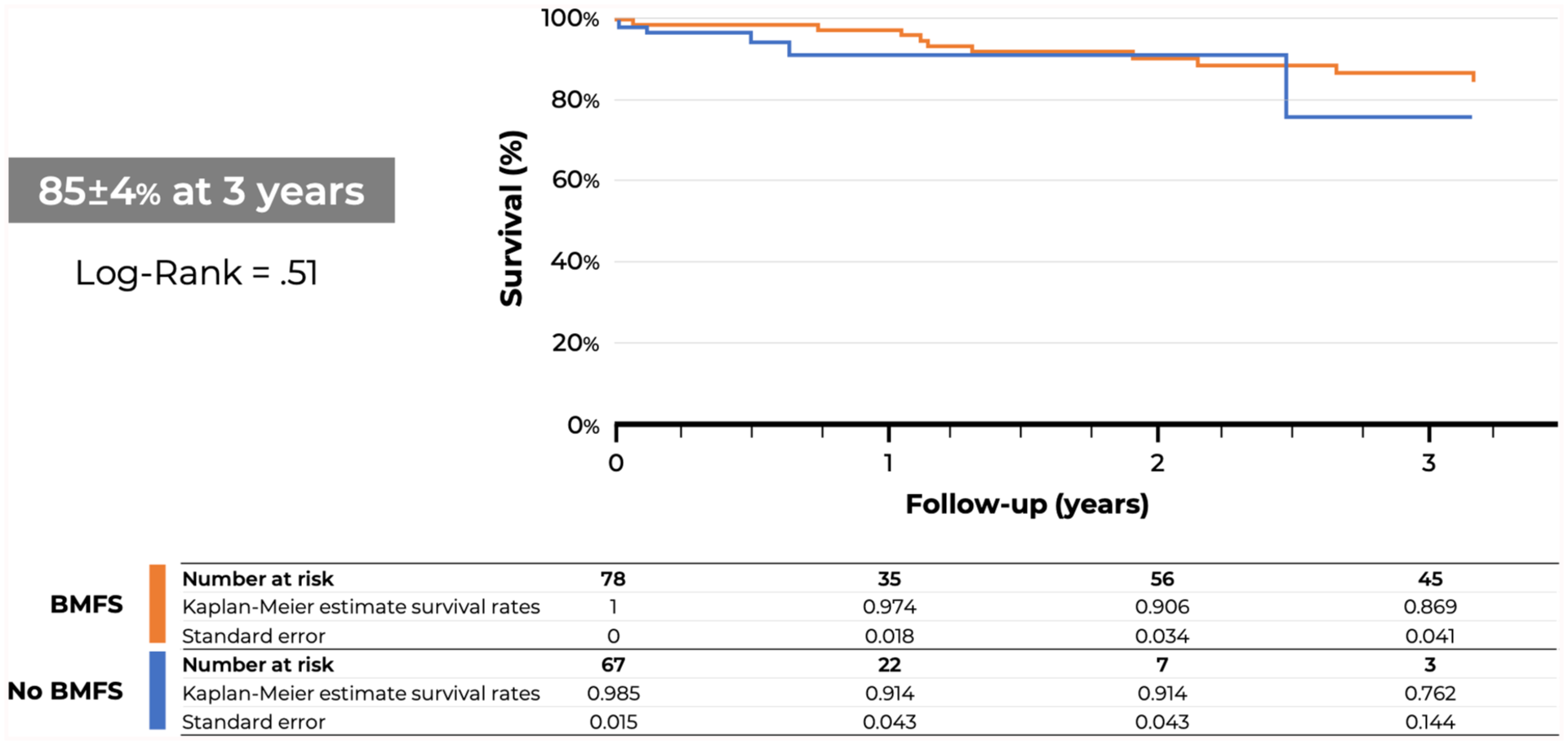
Kaplan-Meier estimates of patient survival (%) in 147 patients treated with 4-vessel fenestrated endovascular aortic repair with proximal bare-metal fixation stent (BMFS) or covered fixation stents (CFSs).
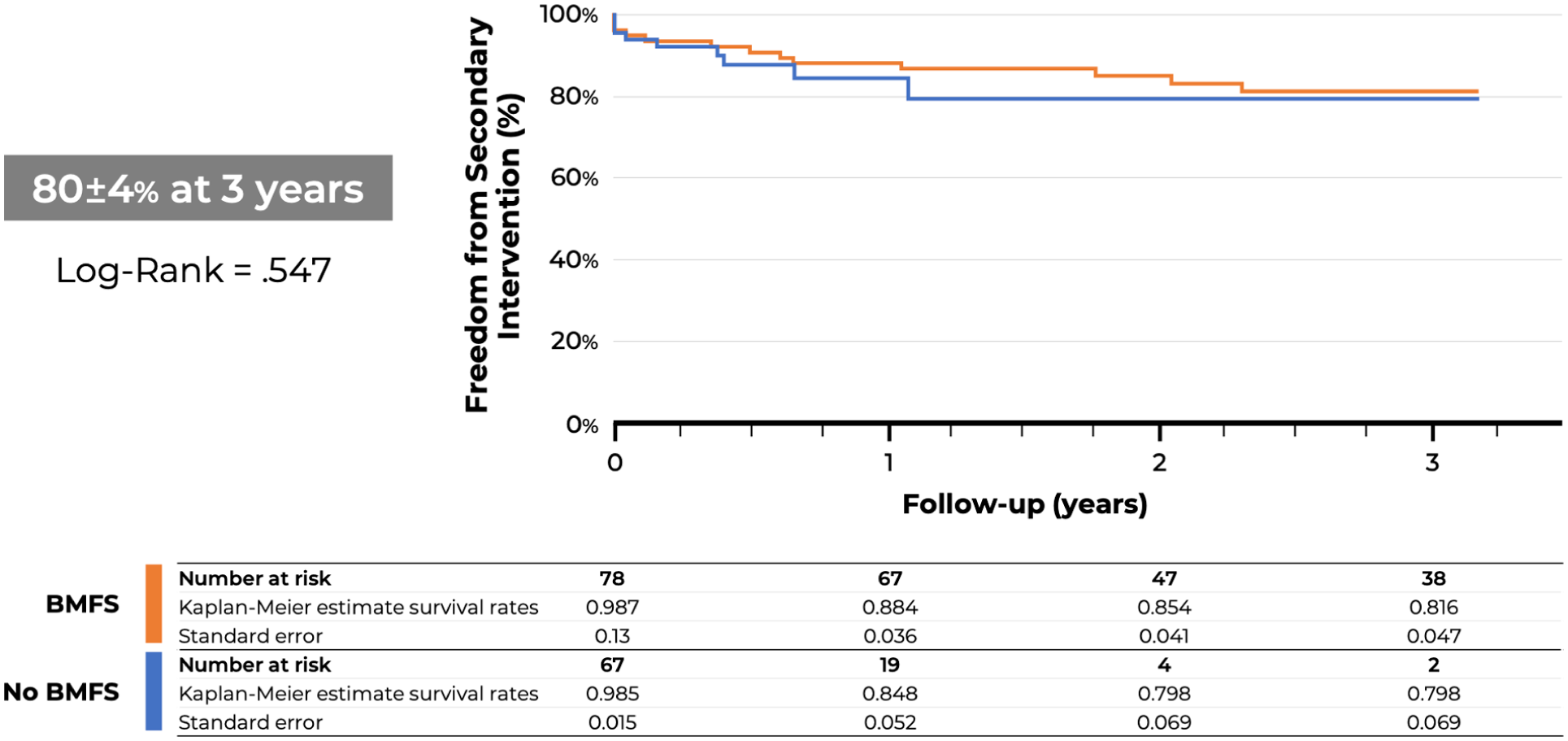
Kaplan-Meier estimates of freedom from secondary intervention (%) in 147 patients treated with 4-vessel fenestrated endovascular aortic repair with proximal bare-metal fixation stent (BMFS) or covered fixation stents (CFSs).
Detailed Consecutive 26 Reinterventions of the 147 Patients Treated With 4-Vessel Fenestrated Endovascular Aortic Repair With Proximal Bare-Metal Fixation Stent (BMFS) or Covered Fixation Stents (CFSs).
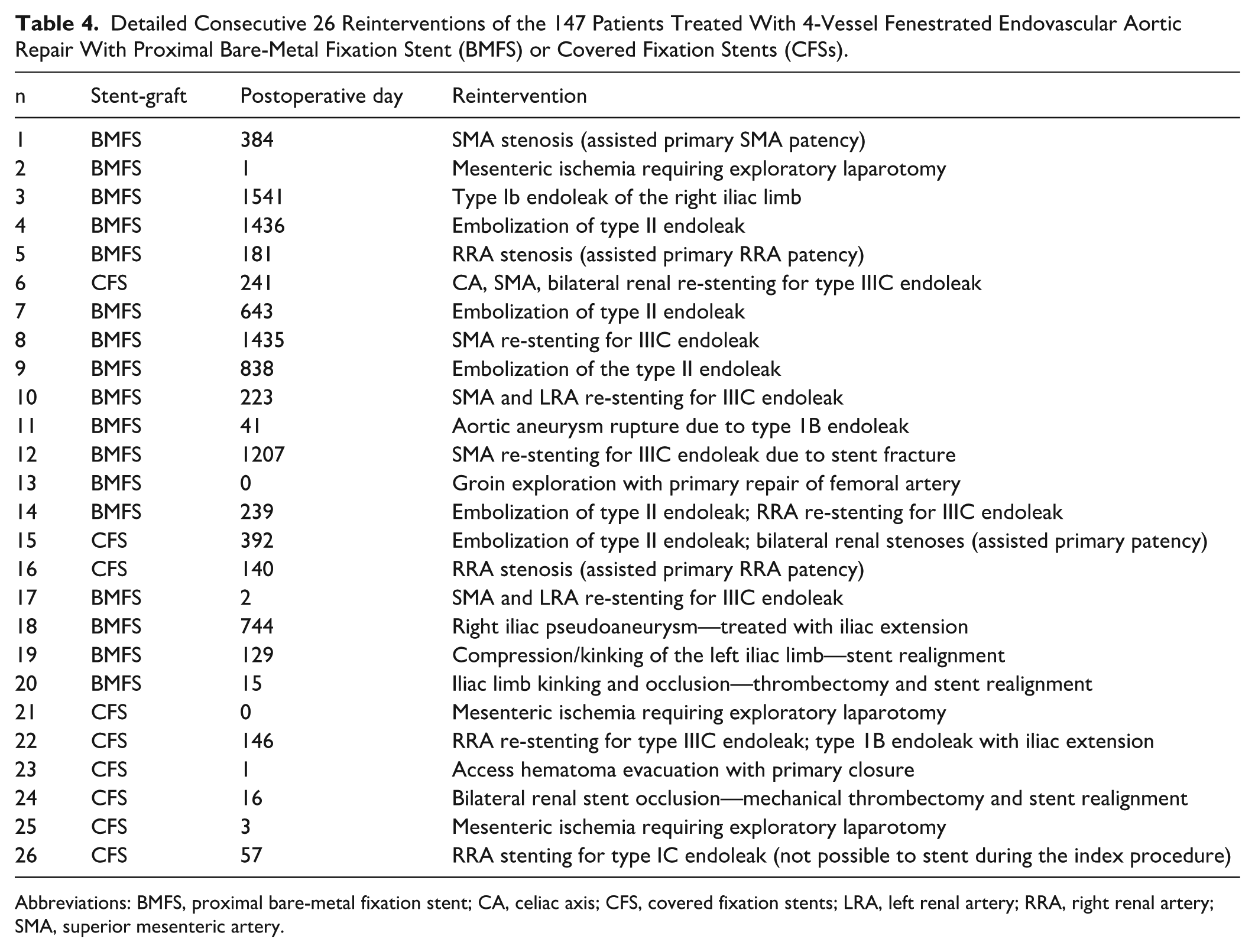
Abbreviations: BMFS, proximal bare-metal fixation stent; CA, celiac axis; CFS, covered fixation stents; LRA, left renal artery; RRA, right renal artery; SMA, superior mesenteric artery.
Discussion
Active fixation of stent-grafts is well accepted as an important design feature due to high incidence of distal migration with the first-generation infrarenal devices that lacked any fixation. 2 The evolution of infrarenal stent-grafts included proximal BMFS to achieve suprarenal fixation or CFSs placed in the infrarenal segment, both achieving similar degree of fixation and effectively minimizing device migration. Melas et al have demonstrated in experimental models using human cadaveric aortas that devices with fixation hooks also displayed higher proximal fixation and significantly increased pull-out forces. 3 Although there has been concern that suprarenal fixation across the origin of diseased renal arteries may result in deterioration of renal function over time, this has not been confirmed in several metanalysis.4 –9 Proponents of suprarenal fixation argue that the suprarenal aorta is a more stable segment for fixation, whereas those who prefer infrarenal fixation argue that this is sufficient and potentially decreases the risk of inadvertent renal artery occlusion. 10
Fenestrated stent-grafts initially evolved from the Cook Zenith infrarenal platform which uniformly applied a bare-metal fixation stent. 11 The bare-metal fixation stent also provided additional advantages for a fenestrated stent-graft. Because this stent was kept collapsed in a top cap during the initial phases of deployment, rotational movement of the device was improved, and the operator could leverage the constrained device to bounce catheters and sheaths during renal and mesenteric catheterization. In recent years, the evolution of fenestrated designs included lower-profile fabrics and the use of the same delivery system applied for thoracic stent-grafts without the BMFS and with a CFS. Initial experience raised concerns that these devices would not be as responsive to rotational movement, potentially leading to technical difficulties during deployment. However, increasing clinical experience and the potential advantages of the lower-profile platform gradually changed the paradigm from routine use of BMFS to avoidance whenever possible. In this report, the use of BMFS declined from 88% to 20% after 2018, and currently, BMFS are rarely used except when coupled with scalloped devices.
Technical success in this report was nearly identical for patients who had devices with BMFS or CFS, indicating that the perceived technical difficulties imposed by less rotational movement had no clinical consequence. There was no difference in target vessel catheterization and stenting. Although total operating time and cumulative air karma were lower in patients with CFS, this likely reflects increasing experience and other technical improvements in recent years, such as use of preloaded systems, steerable sheaths, and the avoidance of upper extremity access, all of which simplified the operation and reduced operating time. 12 There was no difference in mortality and MAEs which were low irrespective of which type of fixation stent was utilized. Finally, the cumulative incidence of secondary intervention and freedom from TAI between patients was nearly identical, indicating that the type of fixation stent has no bearing on specific causes or secondary intervention nor target vessel failure.
The study has important limitations that need to be discussed. Although the cohort was prospectively analyzed, there was no predefined algorithm for selection of the fixation stent and changes occurred due to physician preference. Patients were selected based on anatomy and clinical risk, and therefore, outcomes may not be generalized to all patients with CAAAs. There were some differences in patient characteristics, of which the higher proportion of Extent IV TAAAs in patients with CFS may negatively affect outcomes. These differences in aneurysm extent could have led to variations in endograft design, possibly resulting in a greater distance between the most proximal fenestration and the proximal edge of the device in the CFS group. Such design modifications may also have influenced endograft rotation and target vessel cannulation. Second, our midterm follow-up might be short to assess if BMFS could lead to lower stent-graft migration, reflecting lower reintervention rates. Third, the use of steerable sheaths has only been introduced in recent years, representing a significant advancement by facilitating target vessel cannulation and reducing operative time, which may confound the comparison of operative time between the 2 fixation stent configurations. Apart from these limitations, the main strengths of the study are the prospective design, independent auditing, and adjudication.
Conclusion
The FEVAR for CAAAs and Extent IV TAAAs was performed with high technical success, low mortality, and MAEs, independent of the use of BMFS or CFS. There is no difference in patient survival and freedom from secondary interventions in patients treated with or without BMFS.
Footnotes
Authors’ Note
This abstract was presented at the 2023 SCVS Annual Symposium in Miami, FL, in March 2023.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr G.S.O. has received consulting fees and grants from Cook Medical, W.L. Gore, Centerline Biomedical, and GE Healthcare (all paid to Mayo Clinic, The University of Texas Health Science at Houston, and Baylor College of Medicine with no personal income). Dr B.C.M. has received consulting fees and grants from Cook Medical and W.L. Gore (all paid to Mayo Clinic with no personal income). Other co-authors have no conflicts.
