Abstract
The double-barrel stenting (DBS) is a technique in which 2 parallel stents are simultaneously deployed through the same reinforced fenestration, into 2 adjacent target vessels. Prior reports describe the application of this technique for the treatment of superior mesenteric artery dissection with aneurysmal degeneration, coronary artery bifurcations, aortic arch branches, and intracranial aneurysms. The DBS technique is particularly useful in the context of fenestrated repair (FEVAR) of complex anatomy aortic aneurysms when the origin of visceral arteries branch off the aorta very close to each other or present early branches. We herein describe a case series including 7 patients who underwent a FEVAR procedure for thoracoabdominal and juxtarenal aortic aneurysms in which the presence of accessory renal arteries (ARA) or early renal branches was the reason for the application of this technique. Technical success was 100% and all stents were patent in the last follow-up CT scan (follow-up range: 1.8-62.8 months). There was only 1 small endoleak from indetermined source potentially related to the DBS, but the aneurysm sac decreased in size during follow-up and no secondary intervention was needed. Therefore, the DBS technique is a viable option for the incorporation of ARA or early renal branches to a fenestrated repair of aortic aneurysms with complex anatomy.
Keywords
Introduction
The treatment of complex anatomy aortic aneurysms with a fenestrated endovascular repair (FEVAR) has largely increased worldwide in the last 2 decades, which allowed the management of thousands of high-risk patients who were not candidates for an open or hybrid repair.1,2 In addition to the extension of the aneurysm being a major concern during the planning and execution of the repair, anatomical features such as the presence of accessory renal arteries (ARA) and early bifurcation of visceral vessels can make the incorporation of these arteries to the repair a challenging task to be accomplished. The double-barrel stenting (DBS) is a technique in which 2 parallel stents are simultaneously deployed from the same fenestration, into 2 target vessels. Prior reports describe the application of this technique for the treatment of superior mesenteric artery dissection with aneurysmal degeneration, 3 coronary artery bifurcations, 4 aortic arch branches, 5 and intracranial aneurysms. 6
This technique can be particularly useful in the context of FEVAR for complex anatomy aortic aneurysms, especially in cases in which the visceral arteries branch off the aorta from origins very close to each other or in the presence of early branches. To our knowledge, there are no case series in the literature of the use and mid-term follow-up of the DBS technique for the renal arteries associated with FEVAR devices. We present a case series including 7 patients submitted to a FEVAR procedure for thoracoabdominal and juxtarenal aortic aneurysms in which the presence of ARA or early renal artery bifurcation was the reason for the application of the DBS technique.
Case Series Presentation
Information Regarding Demographics, Aneurysm and Renal Arteries Anatomy, Stents Deployed, and Follow-up.
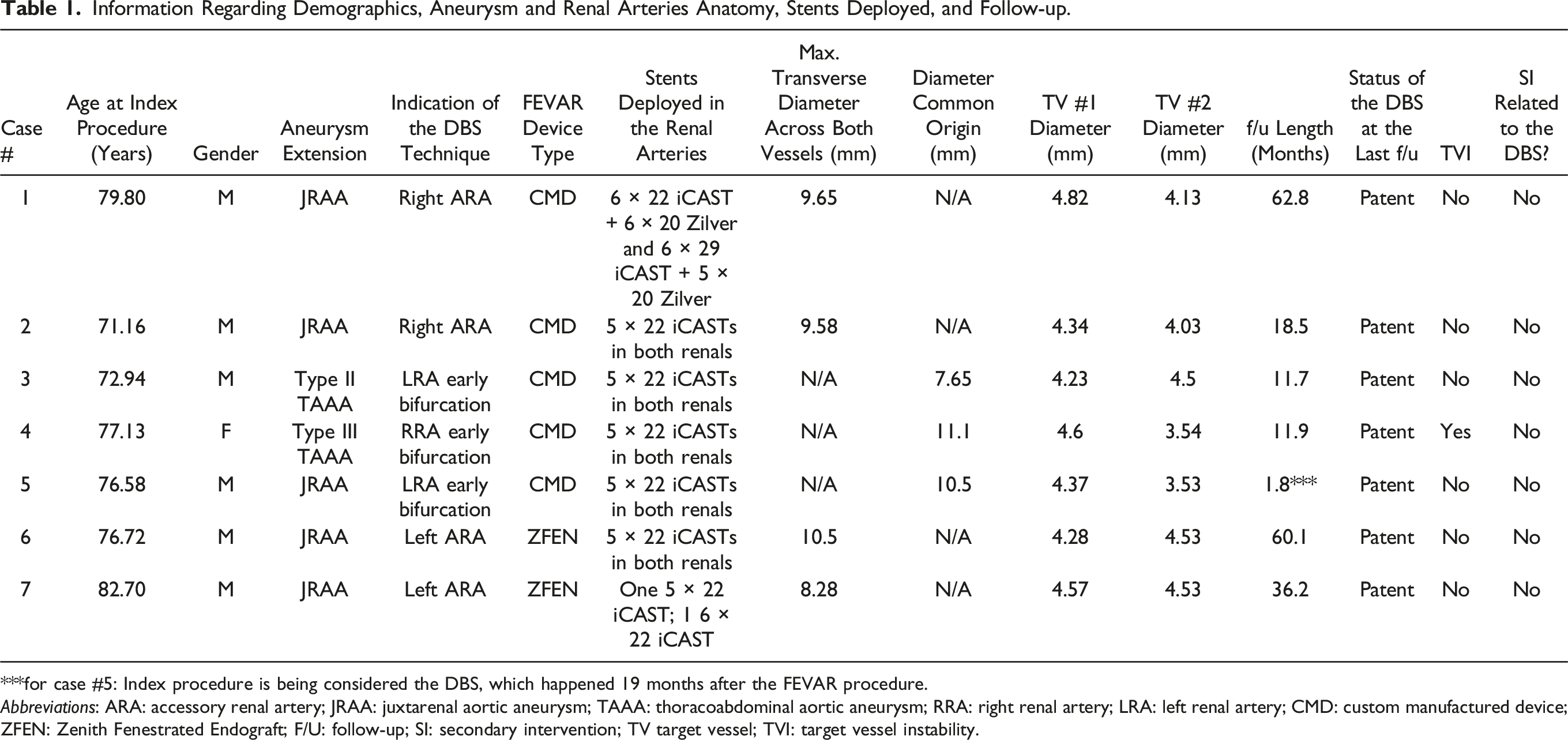
***for case #5: Index procedure is being considered the DBS, which happened 19 months after the FEVAR procedure.
Abbreviations: ARA: accessory renal artery; JRAA: juxtarenal aortic aneurysm; TAAA: thoracoabdominal aortic aneurysm; RRA: right renal artery; LRA: left renal artery; CMD: custom manufactured device; ZFEN: Zenith Fenestrated Endograft; F/U: follow-up; SI: secondary intervention; TV target vessel; TVI: target vessel instability.
In the preoperative CT scan, ARA were observed in 4 cases and early renal branching/bifurcation was found in 3 patients (example in Figure 1). In the cases with ARA, the distance between the main vessel and the accessory one was too short to enable the incorporation of the 2 vessels through separated fenestrations (Table 1). In addition, in all cases there was no space to adequately accommodate directional branches by the renal arteries, therefore justifying the choice for dual renal fenestration with double barrel stenting (Figure 1). 3D reconstruction of preoperative CT scan. This patient had a type B aortic dissection with aneurysmal degeneration, initially treated with a TEVAR landing just proximal to the celiac artery. Early bifurcation of the RRA is observed.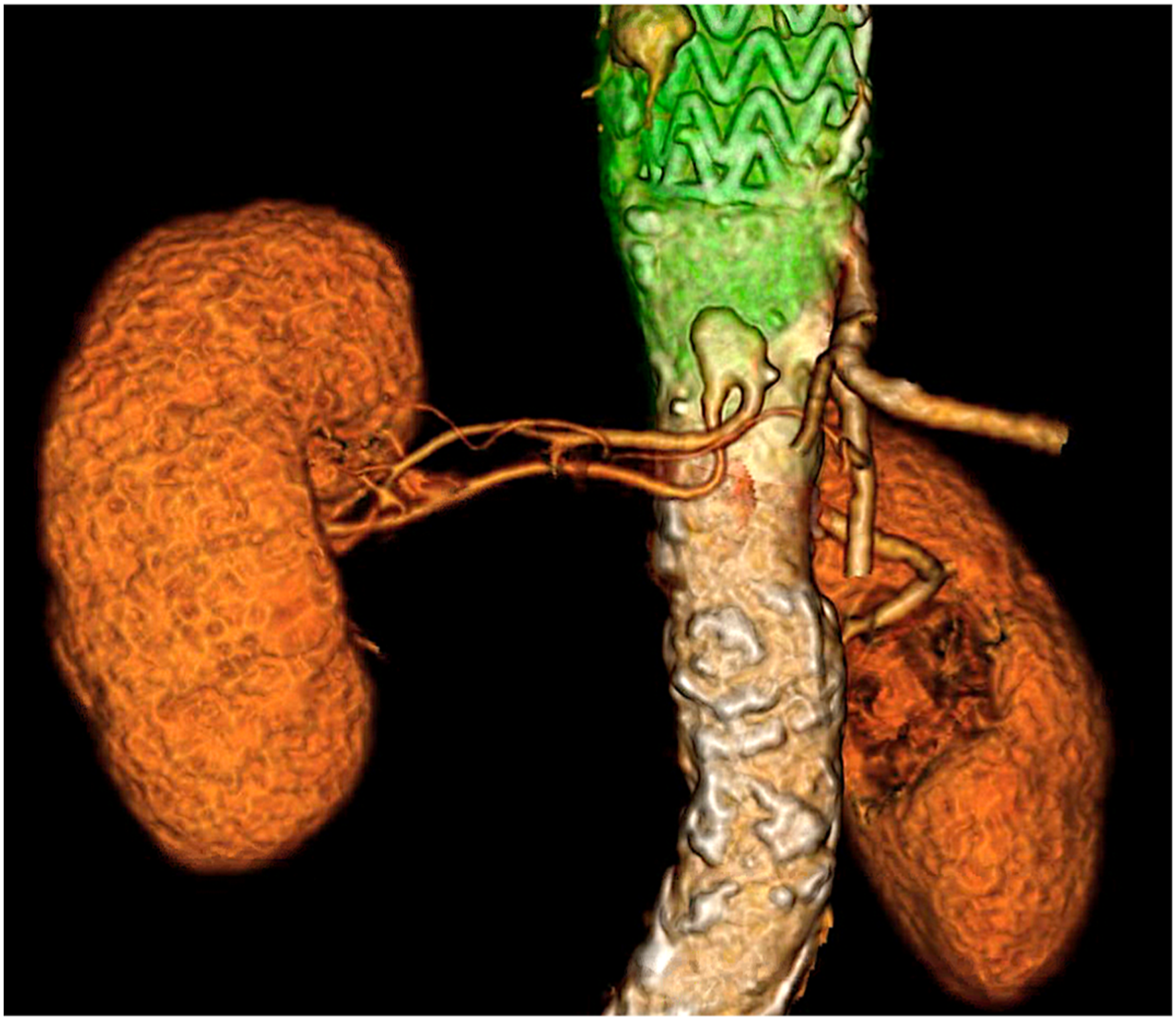
In all cases, the double barrel renal stents were performed during the FEVAR procedure under general anesthesia, with exception of 1 patient that had this technique applied as a secondary intervention for the treatment of an endoleak associated with the left renal fenestration. The LRA fenestration was not primarily stented during the FEVAR procedure because no connection was observed intraoperatively between this artery and the aneurysm sac. However, in the follow-up CT scans, an endoleak from this fenestration was demonstrated, and the patient was submitted to a secondary intervention under moderate sedation and local anesthesia, with application of the DBS technique due to the early branching of the LRA.
In all cases, the DBS was performed through femoral access, percutaneously in 4 cases and with open exposure of the arteries in 3 patients. Three different sizes of fenestrations were utilized: 8 × 6, 8 × 8 and 10 × 10 mm. In 3 cases treated with a CMD endograft, the dual renal fenestration was preloaded. The FEVAR procedure demands the catheterization of each fenestration so the bridging stents can be deployed into the visceral arteries. When the fenestrated CMD endografts (Cook Medical - Bloomington, IN) are designed with preloaded guidewires or catheters, the fenestrations are already catheterized with the aforementioned component, which potentially save time during the procedure as fewer catheter manipulation to access the fenestrations is needed. 7
After all the visceral stents were in place, the fenestrated device was fully deployed. The renal stents were simultaneously deployed (kissing stents, Figures 2(A), 2(B) and 2(C)) and then post-dilated and flared into the fenestrated device with balloons to avoid type IIIc endoleaks. In some cases, a secondary stent distal to the iCAST (Getinge, Wayne, NJ) was deployed in the renal artery to ease its tortuosity. A renal angiogram (Figure 2(D)) was performed in all cases. A, Both RRAs stents (iCASTs) already in position (arrows). B, Kissing stenting of the RRAs (arrows). C, Both RRA stents already deployed (arrows). D, Final renal angiogram demonstrating patency of both RRAs. Yellow dashed ellipses highlight the position of the dual renal fenestration.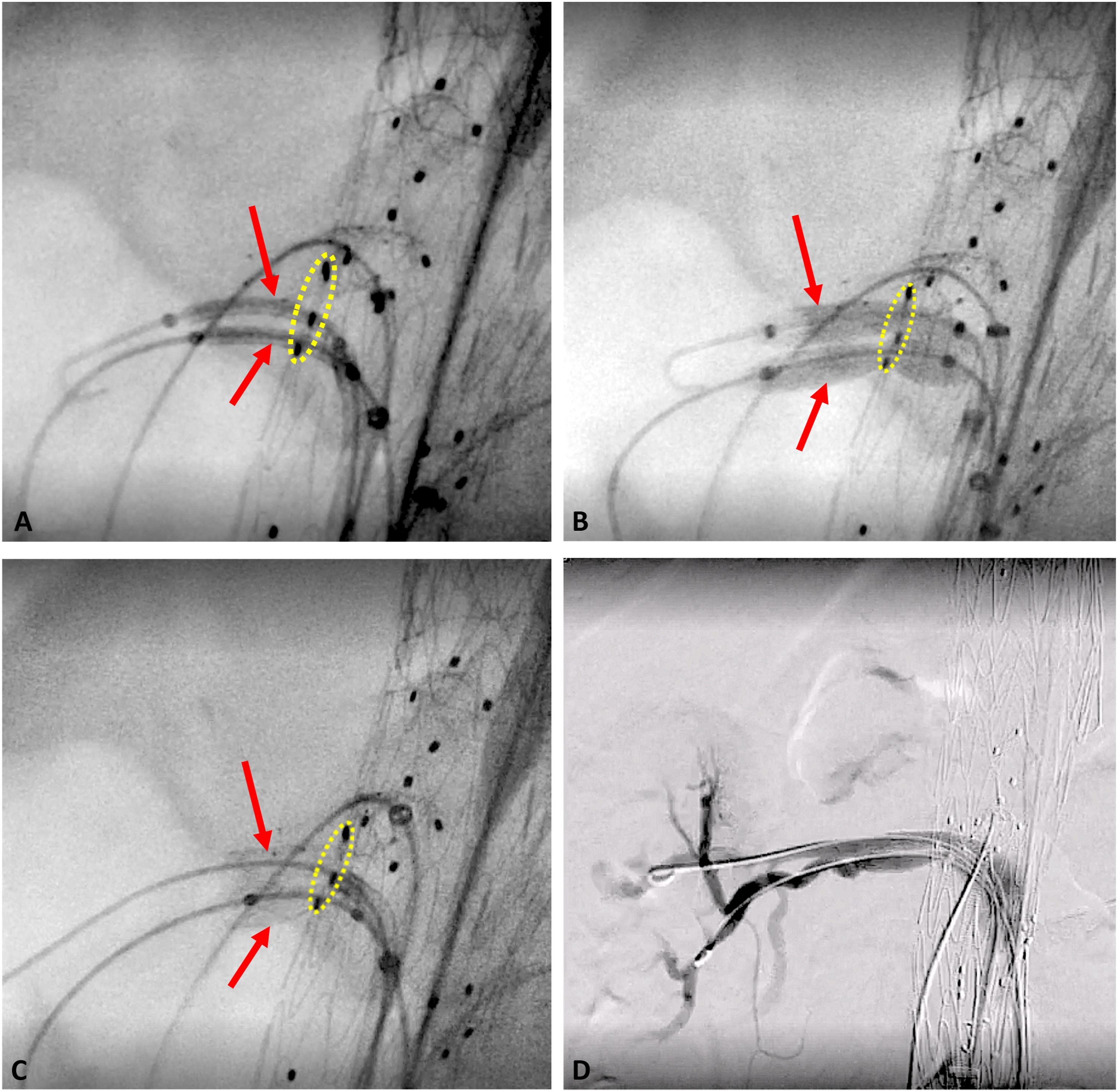
At discharge, all patients were prescribed dual antiplatelet therapy (daily aspirin 81 mg + clopidogrel 75 mg) for a minimum duration of 3 months and continued ASA thereafter. As for the FEVAR follow-up protocol in our institution, a renal-mesenteric duplex is performed every 3 months, and thorax, abdomen, and pelvis CT scans are obtained at 1, 6, 12, 18, and 24 months after the index procedure, and yearly thereafter until 5 years (Figures 3(A) and (B) and 4). After this, follow-up with imaging can be less frequent, usually scheduled in 2-year intervals in the absence of complications. A, 6-month follow-up CT scan (3D reconstruction) demonstrating the patency of both RRAs. B, Detailed image showing both RRA stents through the dual fenestration. Right lateral view of the fenestration with double-barrel stents to the right renal arteries. The red arrows point to the two renal stents and the blue asterisks indicate three of the four fenestration markers positioned at 3, 6, and 9 o’clock around the edge of the fenestration.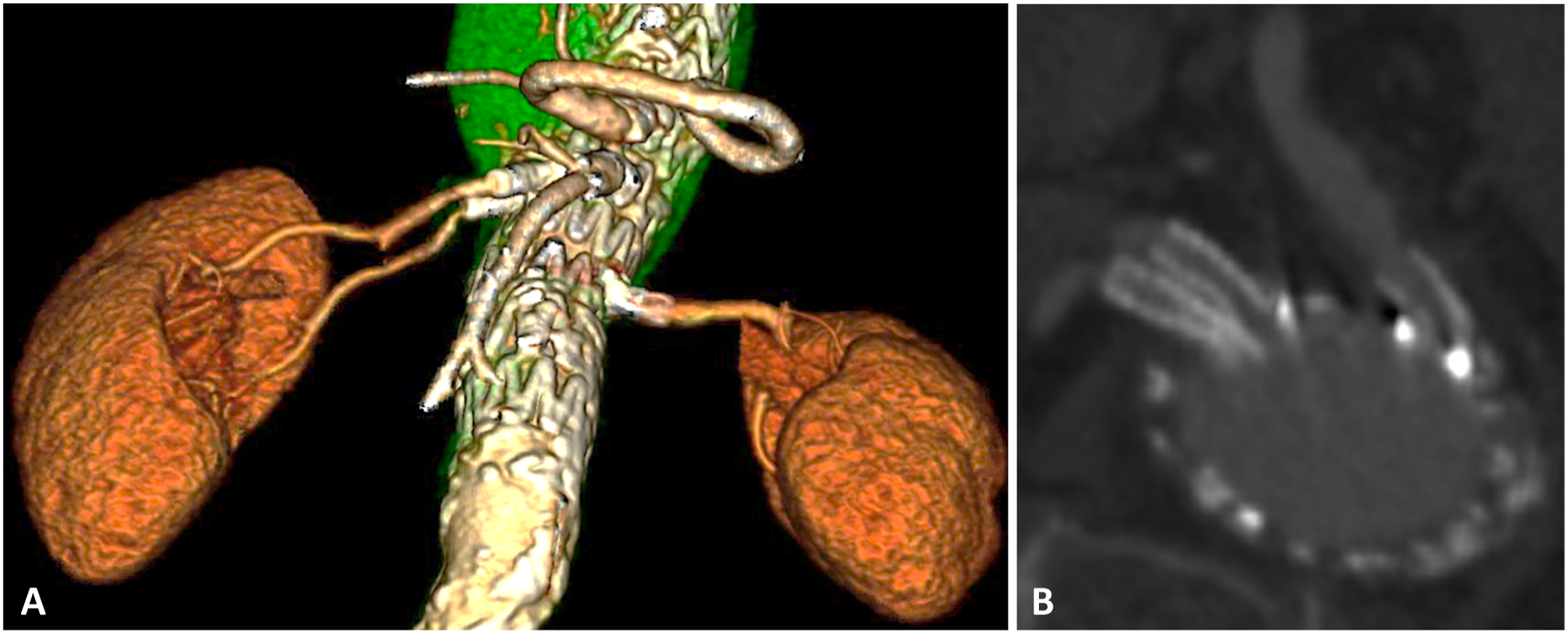
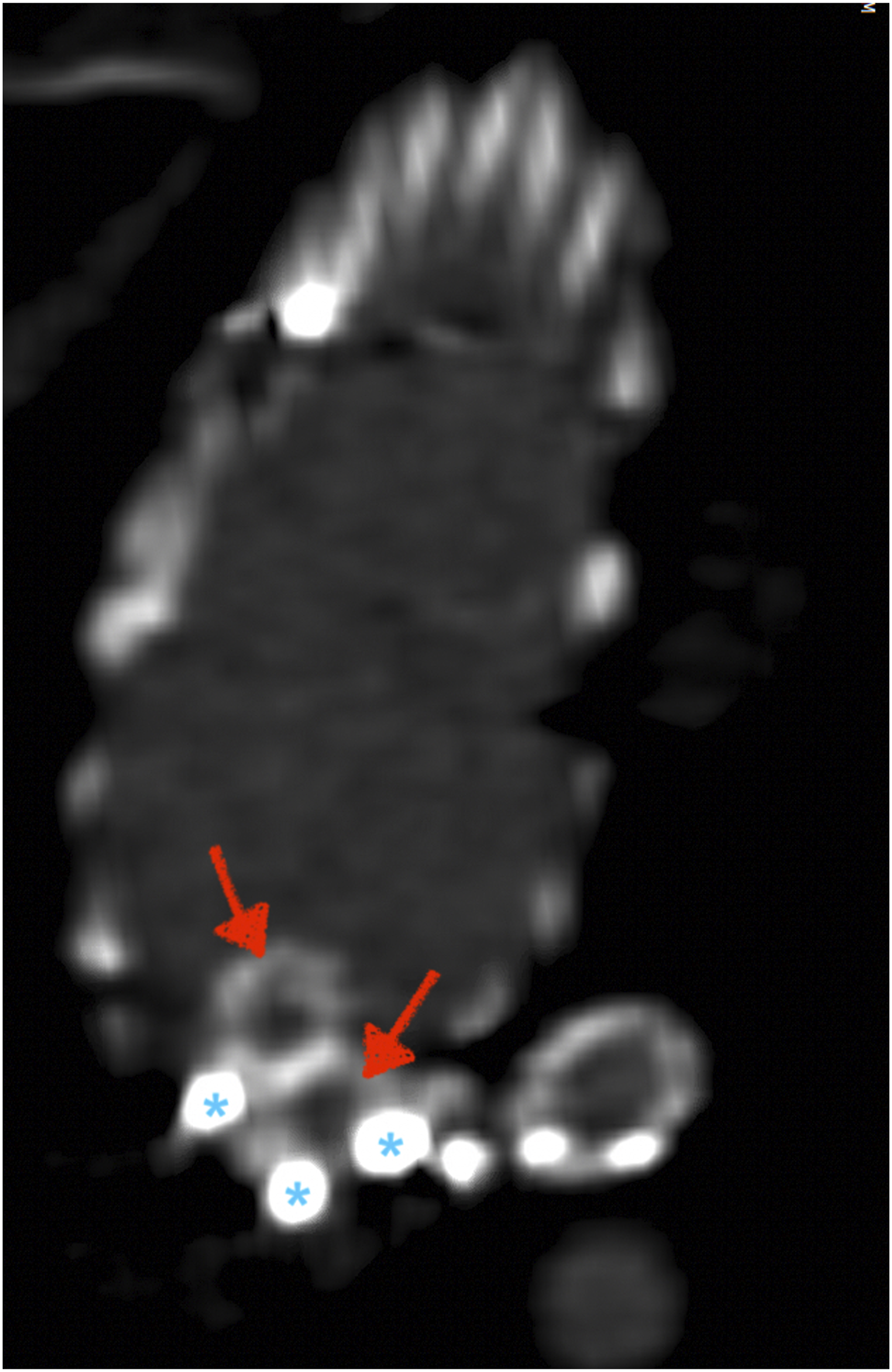
The follow-up duration ranged between 1.8 and 62.8 months, and all stents were patent at the last follow-up CT scan. Only 1 small endoleak from indetermined source was potentially related to the DBS in 1 case, but the aneurysm sac decreased in size during follow-up and no secondary interventions were required to date. One patient died 2 months after the index procedure due to causes unrelated to the aneurysm repair (congestive heart failure). Another patient died 7 years after the FEVAR procedure without presenting any complications related to this operation.
Discussion
Variations in the renal arterial anatomy are vastly described in literature, and the incidence of ARA was described as 27.7% by Satyapal et al 8 in a morphometric study analyzing angiograms and cadaver kidneys used in renal transplantation. Early branches and additional renal arteries can be responsible for a significant percentage of the organ arterial supply, hence the importance of preserving these vessels whenever possible, especially in patients who already present a decline in renal function prior to the FEVAR procedure. Motta et al, 9 in a retrospective study including 139 F/BEVAR patients, reported intentional coverage or embolization of ARA in 87% of the 31 cases that presented this anatomical variation. The incidence of postoperative acute kidney injury among this subgroup was 7.4%. Tenorio et al, 10 in a study including 254 F/BEVAR patients, reported intentional coverage of ARA of 22%. They demonstrated that ARA coverage is a predictor for the occurrence of AKI and mid-term renal function deterioration.
In this context, the DBS technique can be an interesting option for ARA and early renal branches preservation in FEVAR cases. Although technically challenging, this strategy can produce good results if planned carefully and executed by surgeons extensively experienced in the endovascular approach of the aortic visceral branches. The DBS is an alternative tool that every endovascular surgeon used to deal with visceral arteries with challenging anatomy should be familiar with.
For these complex cases, the design of precannulated renal fenestrations can be helpful during the procedure, as the access through the renal fenestration is already secured. The simultaneous ballooning of the double-barrel renal stents is critical to guarantee a good apposition of the 2 stents against the fenestration and to avoid the crushing of one stent over the other. In addition, re-inflating both balloons inside the double-barrel stents during the insertion and deployment of the contralateral iliac limb can protect the stents against crushing. As for the bridging stent, the balloon-expandable iCAST (Getinge, Wayne, NJ) was the bridging stent of choice in all cases due to its precise deployment, rigidity and ability to flare its most proximal segment inside the main aortic device to avoid endoleaks.11,12
Close life-long follow-up with imaging is required in patients submitted to FEVAR procedures to access for adverse events, regardless if the DBS technique was applied or not. Therefore, the utilization of this technique did not increase the demand of follow-up imaging. None of the 7 patients required extra images beyond those already planned per protocol. A major concern related to this technique is the occurrence of type IIIc endoleaks. Prior publications report the incidence of this adverse event ranging between 0 and 5.6% 13 in the first 30-day period after a fenestrated repair and between 3.4 and 7.1% 14 during the long-term follow-up. In this case series, 1 case presented a small endoleak of undetermined source since the first postoperative CT scan obtained 30 days after the FEVAR procedure. Compared to a 6-month follow-up imaging, the aneurysm sac diameter decreased despite the persistence of the small endoleak in the second CT scan, demonstrating a low-pressure extravasation of contrast. No secondary intervention was indicated to treat this adverse event, considering that the aneurysm sac decreased in size. No secondary intervention related to the DBS was required for any of the 7 patients to date.
Lastly, a minimal diameter of 4 mm is usually used as a cut-off for the incorporation of accessory vessels to a fenestrated aortic repair. In this case series, arteries with a diameter of 3.5 mm were successfully included in the repair, which is the minimum value that the authors consider appropriate for the execution of the DBS. It is extremely important to note that dual stenting of a single fenestration should only be undertaken when the aortic device has significant apposition to the aortic wall to minimize the potential for a type IIIc endoleak, since the dual stenting essentially create small regions of poor apposition and seal.
Conclusion
Double-barrel renal stenting is a viable option for the incorporation of ARA or early renal branches to a fenestrated repair of aortic aneurysms with complex anatomy. This is particularly interesting for patients that already present any degree of renal function deterioration prior to the aortic procedure. Although close follow-up is imperative for life, satisfactory results in terms of patency and target vessel instability can be obtained with this technique when applied by Vascular Surgeons experienced in the endovascular management of the aortic visceral branches.
Footnotes
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Vivian Carla Gomes: no disclosures; Federico Ezequiel Parodi: Stock options from Centerline Biomedical; Elad Ohana: no disclosures; Mark A Farber: WL Gore - Consulting, Clinical Trial Support; Getinge – Consulting; Cook - Research support, Clinical Trial support; ViTTA - Consulting, Clinical Trial support; Centerline Biomedical - Stock options, Clinical Trial support.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Institutional Review Board information
Name of IRB Chairperson and Certification of Action Taken by the IRB by the time of the approval of the Research protocol #12-05644: Elizabeth Kipp Campbell, Director
