Abstract
Objective:
To evaluate the clinical application and outcomes of physician-modified endografts (PMEG) made from a prefenestrated custom-made device (CMD) from COOK Medical with one reinforced fenestration and prefabricated diameter-reducing ties for the treatment of complex abdominal aortic aneurysms.
Methods:
All patients treated with this adapted PMEG technique were included in the study. The single prefabricated fenestration was located 57 mm from the proximal edge. Two proximal graft diameters (34 mm and 36 mm) tapered to 22 mm distally, with tapering beginning 67 mm from the top and extending 40 mm were used. All grafts had a fixed length of 160 mm.
Results:
Ten patients, of which 8 male (80%) and with a mean age of 74 (IQR: 70–78 y) were included. Indications included degenerative abdominal aortic aneurysms (n=6 [60%]), penetrating aortic ulcers (n=2 [20%]), and mycotic aneurysms (n=2 (20%). Three (30%) presented with contained rupture, six (60%) were symptomatic without rupture, and one (10%) was asymptomatic. Median diameter of the aneurysm was 53mm (IQR 48.3–73.2 mm). Physician modification involved creation of at least three additional fenestrations, all reinforced with Goose Snare wires and secured with sutures. All target vessels were successfully bridged. The median operating time was 208.5 min (IQR: 161.3–290 min), while the median modification time was 71.5 min (IQR 55–96 min). The in-hospital mortality rate was 20 %, both were patients with ruptured mycotic aneurysms. The remaining eight patients have all regained their baseline functional status and are currently undergoing follow-up (median follow-up time: 4.5 months; IQR: 1–13 months), with maintained patency of all target vessels to date. Postoperative CT revealed one type III endoleak, one type Ib, and two type II endoleaks. The type III endoleak between the TEVAR and the fenestrated aortic graft was treated with a giant Palmaz stent (one endovascular reintervention), while the others remain under follow-up.
Conclusions:
Use of a simplified PMEG with one prefabricated fenestration and diameter-reducing ties is feasible and offers favorable early outcomes with technical advantages in modification and deployment.
Clinical Impact
Using an endograft with a single prefabricated fenestration as the outset graft for physician-modified endografts enhances procedural efficiency by reducing customisation time and simplifying the modification process. This approach lowers technical complexity, improves procedural reproducibility, and broadens the applicability of PMEG techniques. Consequently, PMEGs can be more readily used across a wider range of complex aortic anatomies, expanding endovascular treatment options for patients who might otherwise have limited or higher-risk surgical alternatives.
Introduction
The use of fenestrated endoprostheses has enabled treatment of complex aortic aneurysms with low periprocedural mortality of 3.4% and freedom of aneurysm-related mortality of 97% at 5 years.1-4 Because of the time required to fabricate custom-made fenestrated endoprostheses (CMD), they are not suitable for urgent cases. Similarly, in patients with large aneurysms, the risk of rupture during the manufacturing process of a CMD device is also increased. 5 Nowadays, endoprostheses with branched configurations are available off-the-shelf. In numerous cases, these off-the-shelf options can facilitate the treatment of juxtarenal and pararenal aneurysms. However, a major disadvantage of these prostheses is that they require much longer coverage of healthy aorta compared to fenestrated endografts. 6 Alternative endovascular techniques, such as chimney endovascular aortic repair (ChEVAR), in situ fenestration, and physician-modified endografts (PMEG) offer additional options for treating complex aortic aneurysms in urgent setting. ChEVAR was not only associated with a higher 30-day mortality compared to PMEG (6.1% vs. 2.7%), but also with an increased perioperative stroke rate. 7 As for in situ laser fenestration to the visceral arteries, a recent systematic review found that the available research, albeit limited, showed high technical success rates up to 95.6% and acceptable mortality rates. 8
The use of PMEGs on the other hand, even in urgent situations, are associated with a technical success of as high as 100% and perioperative mortality of 5%.9,10 Long-term results after PMEG procedures have shown high primary target vessel patency of up to 91% and low aortic related mortality of 2% at 5 years. 11 While PMEGs have shown promising outcomes in urgent cases, the limitations lie in the time needed for back-table modifications and the expertise required in performing the constraining ties. 9 This restricts their application to patients who are hemodynamically stable. In an effort to minimize the required modification time, previous research presented the approach of modifying a 4x fenestrated CMD. 12 While these existing fenestrations do contribute to a significant reduction in modification time, they also limit the applicability of this technique to a broader range of patient anatomies.
This case series proposes a novel approach for modifying an outset graft from the ‘CMD’-COOK series with a single prefabricated fenestration tailored to PMEG procedures, which significantly reduces both the time and complexity of the modification process, as well as providing prefabricated diameter-reducing ties.
Methods
Patient Selection and Data Collection
This was a single center, retrospective case series on the outcomes of endovascular repair of complex abdominal aortic pathologies with a PMEG based on an outset graft with a single premade fenestration and prefabricated diameter-reducing ties. All patients treated with this technique between July 2023 and March 2025 were included. Ethical approval was obtained (No. 20-148), and all patients were informed that the use of the PMEG procedure is an off-label procedure.
Electronic patient database records were scrutinized regarding preoperative demographic patient characteristics (age, sex), comorbidities, as well as clinical and anatomical characteristics. These included details about the target vessel orientation and diameter, perioperative details, as well as postoperative outcomes in the first 30 days and during follow-up. Aortic outcomes were assessed using follow-up computed tomography angiography (CTA) at 30 days, 6 months, and then annually. Mortality was determined through in-office visits or follow-up phone calls.
Technical success was the primary endpoint of the study. Technical success was defined as successful completion of the planned procedure with patency of the target vessels and no type I or III endoleaks on the final angiogram. Early death was defined as death within the first 30 days after the procedure or during the hospital stay.
Secondary endpoints were postoperative morbidity (including stroke, vascular and surgical complications) and reintervention rate in the early postoperative period (30 days) and during follow-up.
Endograft Design
A straight graft with the reinforced fenestration and diameter-reducing ties was planned and was on stock when the patients arrived to the hospital. The graft is based on the foundations of the fenestrated CMD prostheses from COOK Medical (Cook Medical, Bloomington, Indiana) and does not differ from the other fenestrated CMD prostheses (Figure 1). All grafts had a uniform proximal diameter of 34 mm or 36 and a distal taper to 22 mm. The existing reinforced fenestration was located at 57 mm from the top of the graft. The taper of the endograft started 67 mm below the upper end of the graft and extended over 40 mm, which allowed wall apposition of the endoprosthesis at the level of the renal arteries.
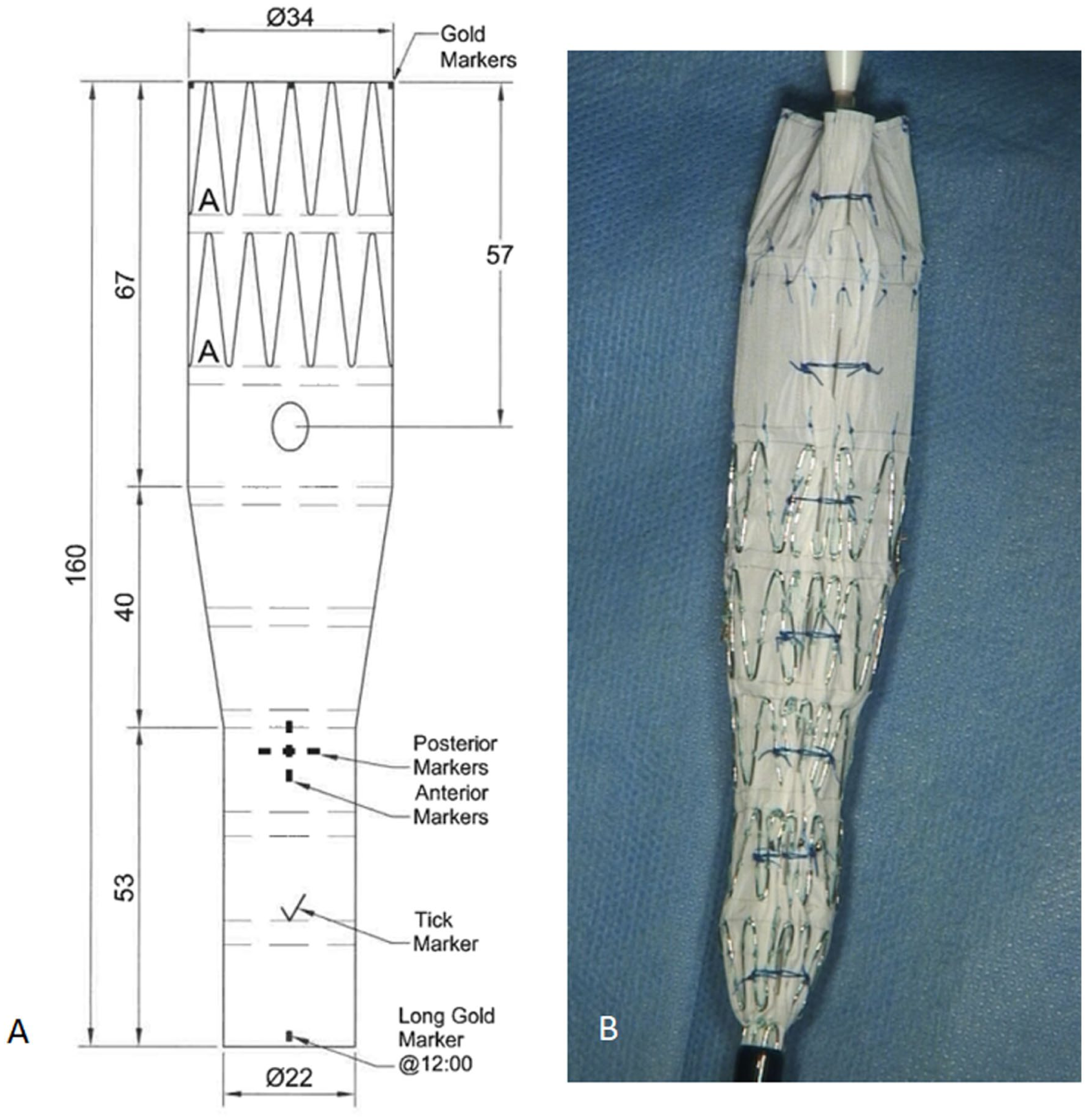
(A) Technical droving of the CMD device; (B) diameter-reducing ties.
Technique Description and Procedure
Preoperative planning was performed using a dedicated 3D workstation (TeraRecon, San Mateo, California). The flat distances of the fenestrations was calculated using a mathematical formula based on the inner diameter of the aorta and the clock-face positions of the target vessels. These distances were then transferred to the graft on the back table. Following measurement completion, graft modification was carried out on the back table in the hybrid operating room. In all cases, the same graft design with a single fenestration was employed, with the only variation being the proximal diameter: 34 or 36mm.
For the modification, the graft was released on the back table. The positions for the fenestration were transferred to the graft. Ophthalmic cautery was used to create the fenestrations for the target vessels. The created fenestrations were reinforced by using a double layer Goose Snare wire (ev3 Endovascular, Plymouth, MN, USA) and fixed with a running 4.0 polyfilament suture. The prefabricated fenestration was used either for the superior mesenteric artery or the truncus coeliacus. The main body was then resheated using a Mersilene band and 2.0 polyfilament sutures. The graft was irrigated with rimfapicin solution.
All procedures took place urgently in a hybrid operating room equipped with Artis Zeego (Siemens Healthineers, Erlangen, Germany) under general anesthesia. The patients underwent full heparinization to achieve an activated clotting time within the range of 250–300 seconds.
The endograft was then advanced through the femoral access. Once positioned, the endograft was deployed with careful attention to graft orientation. Catheterization of the endograft was subsequently carried out via the contralateral side. If needed the main body was extended with a bifurcated graft.
Results
Study Population
During the study period, 111 fEVAR or bEVAR procedures were performed, including 13 physician-modified endografts (PMEG). Ten cases used the described single-prefenestration PMEG.outset graft and were included in the study. Three cases were not included because they were treated with Zenith TX2 Dissection Endovascular Graft (ZDEG Cook Medical, Bloomington, Indiana) outset-grafts, two of which due to unavailability of the prefenestrated graft and one because the diameter of the renovisceral segment was too small to accommodate the available fenestrated CMD graft. The demographic data of the 10 included patients is summarized in Table 1.
Demographics and Comorbidities.
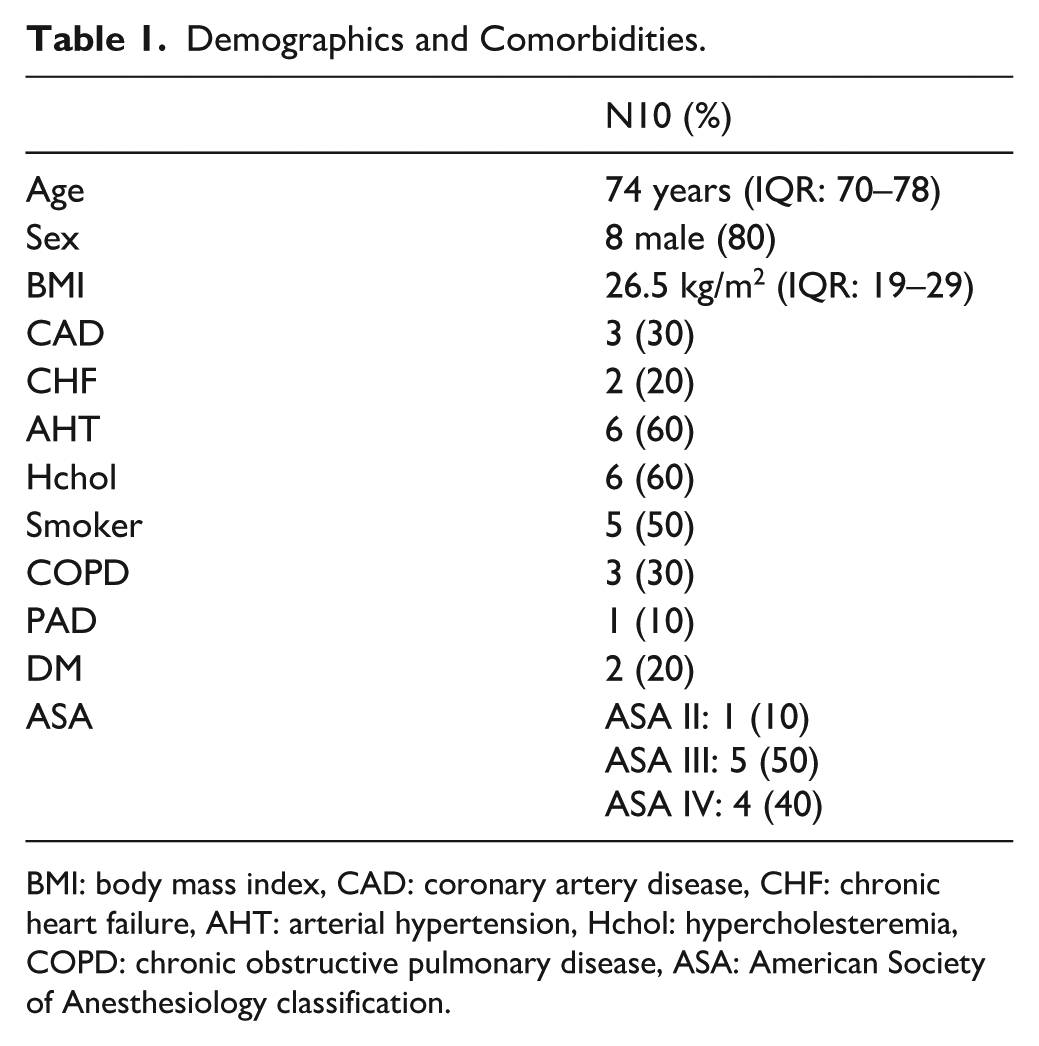
BMI: body mass index, CAD: coronary artery disease, CHF: chronic heart failure, AHT: arterial hypertension, Hchol: hypercholesteremia, COPD: chronic obstructive pulmonary disease, ASA: American Society of Anesthesiology classification.
Preoperative Characteristics
Of the 10 cases analyzed, 3 patients (30%) presented with contained ruptures, 6 patients (60%) with symptomatic non-ruptured aneurysms, and 1 patient (10%) with an asymptomatic non-ruptured aneurysm. The underlying pathologies included degenerative abdominal aortic aneurysms in 6 cases (60%), penetrating aortic ulcers in 2 cases (20%), and mycotic aneurysms in 2 cases (20%). Median diameter of the aneurysms was 53 mm (IQR 48.3–73.2 mm). The median diameter of the proximal landing zones was 30.5 mm (IQR 27.3–32.3 mm). Detailed preoperative measurements are summarized in Table 2.
Anatomic Parameters of the Aorta and Target Vessels.
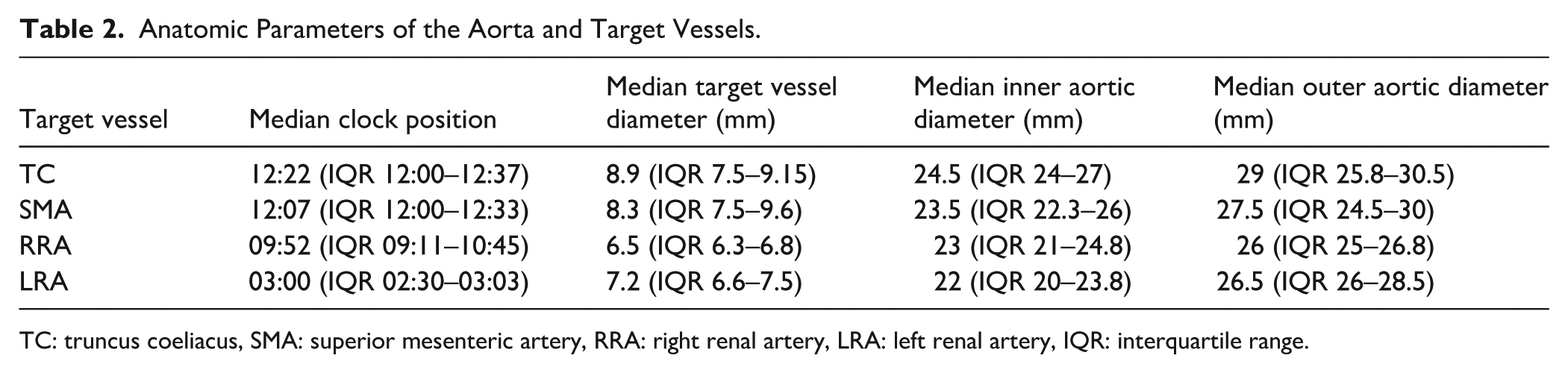
TC: truncus coeliacus, SMA: superior mesenteric artery, RRA: right renal artery, LRA: left renal artery, IQR: interquartile range.
Intraoperative Data
Supplementary Table 1 presents the intraoperative data. The physician’s modifications included the creation of 3 additional fenestrations in all but one case in which 4 were made. The median operating time was 208.5 min (161.3–290 min), while the median modification time was 71.5 min (55–96 min)
Outcomes
Technical success was achieved in 9 out of 10 cases (90%). In one patient, postoperative (CT) imaging revealed a type III endoleak at the junction between the thoracic endograft and the fenestrated device. Despite the overall technical success, early postoperative mortality occurred in two patients within 30 days of the procedure.
One patient was treated for a contained rupture of a 110-mm mycotic aneurysm. The procedure included a thoracic endovascular aortic repair (TEVAR) in combination with a physician-modified endograft (PMEG) featuring four fenestrations. Postoperative CT imaging revealed a type III endoleak, as described earlier this leak between the TEVAR and the PMEG was successfully relined. Despite adequate sealing and maintained stent patency on subsequent imaging, the patient developed an aortoesophageal fistula, confirmed by gastroscopy, necessitating esophagectomy. This was further complicated by bowel ischemia, requiring total colectomy and partial ileal resection. The patient developed a refractory infection unresponsive to broad-spectrum antimicrobial therapy (vancomycin, meropenem, fluconazole) and succumbed to multi-organ failure on postoperative day 22.
The second patient who died had initially presented with a ruptured mycotic aneurysm of the renovisceral aorta and a retroperitoneal hematomaA PMEG with four fenestrations was successfully deployed, with distal sealing in the infrarenal aorta. Postoperative blood cultures revealed Staphylococcus aureus, and targeted antibiotic therapy (flucloxacillin and fosfomycin) was initiated. Although early CT confirmed technical success—patent target vessels and no significant endoleaks—the patient developed acute respiratory failure and died on postoperative day 16.
Postoperative imaging in the entire cohort revealed four endoleaks in total: one type III endoleak, which was successfully treated with a Palmaz stents (Cordis, Johnson & Johnson, Miami Lakes, FL, USA), one type Ib endoleak related to a short, ectatic left common iliac artery (No iliac branch device was used to reduce operative time, a staged treatment was planned) and two type II endoelaks. The type Ib endoleak was no longer observed at the 6-month follow-up examination. All target vessels were patent on postoperative CT.
The remaining eight patients are under active follow-up and have returned to their preoperative baseline functional status. The median follow-up duration was 4.5 months (IQR: 1–13 months). No deaths or target vessel instability were observed during this period. One patient underwent a lobectomy for newly diagnosed lung cancer, which was unrelated to the endovascular procedure.
Discussion
To our knowledge, this is the first report of the use of a pre-manufactured single fenestrated endograft as an outset graft for PMEG procedures.
The treatment of ruptured and symptomatic juxtarenal or suprarenal aneurysms is a major challenge.
Although open repair is a viable treatment, it is associated with an increased incidence of renal insufficiency and proved particularly challenging in ruptured cases. 13 The introduction of endovascular techniques has revolutionized the treatment landscape for complex aneurysms. Today, these endoprostheses can be tailored to the specific anatomy of the patient to achieve optimal results. However, the common manufacturing time of 6 to 8 weeks poses a significant risk in urgent cases or extensive aneurysms. 5 The CMD devices are therefore not suitable for a ruptured aortic aneurysm or in symptomatic cases.
Off-the-shelf devices, on the other hand, offer immediate availability. However, they often necessitate covering larger extents of non-aneurysmal aortic tissue compared to fenestrated devices, potentially increasing the likelihood of spinal cord ischemia, which is exacerbated in cases of substantial blood loss.
Emergent endovascular procedures, including Chimney grafts (ChEVRA), in situ fenestration, and physician-modified endografts (PMEG), have shown promising results for the treatment of urgent aortic pathologies. The literature shows favorable perioperative and long-term outcomes associated with PMEG, albeit with the limitations of requiring technical expertise for graft modification and time constraints. 14
In urgent cases, we sometimes use CMD grafts that are in stock, but we avoid those that are reserved for patients who are still waiting for treatment to avoid delays. For fenestrated devices, reuse is often limited by the graft design and the patient’s anatomy; therefore, a branched graft is usually preferred. Our group already introduced a modification technique for a four-fold fEVAR that significantly reduce modification time. However, its applicability to a broad patient population is limited due to preexisting fenestrations. To overcome these limitations while maintaining the benefits of existing fenestrations and diameter-reducing ties, we further refined the technique. A pre-manufactured endograft with a fenestration and diameter-reducing ties was developed, which served as the basis for subsequent modifications. This leads to simplification of the modification and leads to a shortening of the modification time.
In this study all cases received a straight prosthesis with a proximal diameter of either 34 mm or 36mm. As not both graft sizes were consistently available, diameters of 34 mm and 36 mm were chosen to maximize the proportion of patients eligible for treatment. The use of these larger sizes allowed us to accommodate the greater variability in vessel diameters, although in some cases this required a greater degree of oversizing in the renovisceral segment. To further increase adaptability of this concept, the prosthesis could be made in a variety of diameters and tapers. The fenestration, positioned 57 mm below the top of the graft, serves as the fenestration for the SMA in most cases. Its dimensions, 8 × 6 mm, correspond to the most common diameter of the visceral vessels. The distance of 57 mm from the proximal end of the graft provided sufficient space to create an additional fenestration for the celiac trunk. In cases requiring prior TEVAR implantation, the fixed fenestration could be used for the celiac trunk instead, ensuring sufficient overlap between the two grafts. The prosthesis tapers to 22 mm, starting 67 mm from the top of the graft—10 mm distal to the SMA fenestration—over a length of 40 mm, ensuring apposition to the aortic wall while preventing in-folding within the smaller renovisceral segment. A fixed fenestration may limit the flexibility of positioning additional fenestrations relative to the graft struts. To prevent misalignment, we detach and bend the struts as needed to avoid interference, then resecure them to the fenestration sutures, ensuring that no strut crosses the center of the fenestration, which could compromise target vessel patency. Alternatively, using a graft with prefabricated diameter-reducing ties but without fenestrations could provide complete flexibility in fenestration placement while still reducing the time needed for the modification.
Compared to standard thoracic endoprostheses, the graft is more tapered, which is an advantage for patients with smaller aortic diameters at the level of the renal arteries. Distally, three rows of stents ensure secure attachment to additional graft components. The selected graft length of 160 mm can be a limitation in the treatment of type Ia endoleaks after previous EVAR, as the distance between the flow divider and the renal arteries is often insufficient. In such cases, the use of a shorter graft would be advantageous. Although an access scallop was not considered in this graft design, it is essential for unilateral iliac occlusion to allow catheterization of the target vessels from above.
In our patient cohort, which included both patients with degenerative aneurysms and those with PAU, we observed more oversizing in the renovisceral segment than would normally be expected. This effect was particularly pronounced in patients with PAU, whose aortic diameter was otherwise normal. Although we did not observe any procedural complications related to this oversizing, it is an important factor to consider in preoperative planning.
The prefabricated prosthesis also offers advantages during modofication. Because of the diameter-reducing ties already in place, complete release of the prosthesis from the sheath during modification is not always required. This facilitates efficient resheathing of the graft and reduces the risk of prosthesis rotation within the sheath. In this study, we observed a wide range in graft modification times, which we attribute to the learning curve of the different surgeons performing the procedures
In our initial encounters, prosthesis modification proceeded smoothly. In all cases, successful modification of the prosthesis resulted in effective treatment of the aortic pathology. Regrettably both patients who suffered from a mycotic aneurysm and sepsis, died during the hospital stay. Despite this, postoperative CT angiography had showed a technically successful treatment of both ruptures prior to death.
The use of a single pre-manufactured fenestrated endograft as a basis for in situ laser fenestration has already been described. 15 In comparison, the PMEG method does not require the use of a special laser. In addition, the target vessels do not need to be stented in advance and the long-term results after in situ fenestration in general are not yet available.
Limitations
A major limitation of this study is the short follow-up period and its retrospective design. The other limitations of this technique are unchanged from previously described PMEG limitations; including infection risks and legal concerns related to device modification. In addition, our method is limited by the fixed size of the endograft we use. Using a custom-made device for a modification may be associated with higher costs compared to standard off-the-shelf devices.
Conclusion
In conclusion, the modification of a prefabricated single-strand endograft has improved procedural efficiency by reducing the time required for modifications and simplifying the process by eliminating the need to produce diameter-reducing ties.
Supplemental Material
sj-docx-1-jet-10.1177_15266028251397850 – Supplemental material for Optimizing PMEG Technique: Enhancing Endovascular Repair With Pre-Manufactured Fenestrated Endografts
Supplemental material, sj-docx-1-jet-10.1177_15266028251397850 for Optimizing PMEG Technique: Enhancing Endovascular Repair With Pre-Manufactured Fenestrated Endografts by Jan Stana, Laurence Bertrand, Baban Assaf, David Bijan Khangholi, Nikolaos Konstantionou and Nikolaos Tsilimparis in Journal of Endovascular Therapy
Footnotes
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Department of Vascular Surgery Ludwig-Maximilians University Hospital, Munich, Germany received travel and institutional research grants from Bentley InnoMed GmbH, Hechingen, Germany. N.T. is a consultant for Cook Medical for fenestrated/branched endovascular aneurysm repair.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
