Abstract
Background
Phantom limb pain (PLP) and symptomatic neuroma can be debilitating and significantly impact the quality of life of amputees. However, the prevalence of PLP and symptomatic neuromas in patients following dysvascular lower limb amputation (LLA) has not been reliably established. This systematic review and meta-analysis evaluates the prevalence and incidence of phantom limb pain and symptomatic neuroma after dysvascular LLA.
Methods
Four databases (Embase, MEDLINE, Cochrane Central, and Web of Science) were searched on October 5th, 2022. Prospective or retrospective observational cohort studies or cross-sectional studies reporting either the prevalence or incidence of phantom limb pain and/or symptomatic neuroma following dysvascular LLA were identified. Two reviewers independently conducted the screening, data extraction, and the risk of bias assessment according to the PRISMA guidelines. To estimate the prevalence of phantom limb pain, a meta-analysis using a random effects model was performed.
Results
Twelve articles were included in the quantitative analysis, including 1924 amputees. A meta-analysis demonstrated that 69% of patients after dysvascular LLA experience phantom limb pain (95% CI 53-86%). The reported pain intensity on a scale from 0-10 in LLA patients ranged between 2.3 ± 1.4 and 5.5 ± .7. A single study reported an incidence of symptomatic neuroma following dysvascular LLA of 5%.
Conclusions
This meta-analysis demonstrates the high prevalence of phantom limb pain after dysvascular LLA. Given the often prolonged and disabling nature of neuropathic pain and the difficulties managing it, more consideration needs to be given to strategies to prevent it at the time of amputation.
Introduction
Amputation of the lower extremity is one of the most frequently performed surgical interventions worldwide, 1 with national incidences ranging from 5.8 to 31 per 100,000. 2 With the rapid rise in the prevalence of diabetes mellitus, particularly in low- and middle-income countries, the rates of associated peripheral arterial occlusive disease (PAD), diabetic neuropathy, and soft tissue sepsis have increased as well.3,4 Together, PAD and complications from diabetes mellitus have become the leading cause of lower extremity amputation (LLA), with dysvascularity responsible for 82% of limb loss in the United States. 5
Over 70% of amputees experience pain in the residual limb. 6 Residual limb pain can have many causes, including bone spurs, pressure points, or deep tissue infections, but can also be the consequence of neuroma formation. Furthermore, many patients experience phantom limb pain (PLP) following lower extremity amputation. PLP is the sensation of burning, tingling, or electrical shooting pain originating from an area of body tissue that is not physically present. 7 PLP and neuropathic pain from a symptomatic neuroma can be debilitating and significantly impact the quality of life of amputees and be difficult to manage. The pain decreases the patient’s ability and desire to wear prosthetics, impairing mobility status and function of the limb. 8
The prevalence of PLP and symptomatic neuromas in patients following dysvascular LLA has not been reliably established. The traditional rationale was that dysvascular amputees were thought to experience less neuropathic pain and PLP due to longstanding peripheral neuropathy as a consequence of diabetes mellitus, leading to loss of sensation and pain. 7 However, research by Clark et al 7 reported no significant difference in PLP between dysvascular and non-dysvascular amputees. The impact of PLP and symptomatic neuromas on the patient’s mobility status and quality of life make it essential to gain an understanding of the magnitude of the problem in this patient population that already experiences higher complication and mortality rates than non-vascular amputees.9,10 This systematic review examines the prevalence and incidence of phantom limb pain and symptomatic neuroma after dysvascular LLA.
Methods
Literature Search
A systematic review was conducted to study the prevalence and/or incidence of phantom limb pain and symptomatic neuroma following dysvascular LLA. The study was performed in accordance with the Preferred Reporting Items for Systematic Reviews and Meta-analyses statement (PRISMA guidelines, Appendix, Supplemental Digital Content 1). 11 Databases Embase, MEDLINE, Web of Science Core Collection, and Cochrane Central were searched from inception to 5th October 2022 (Appendix, Supplemental Digital Content 2).
Study Selection
Baseline Characteristics of Included Studies.
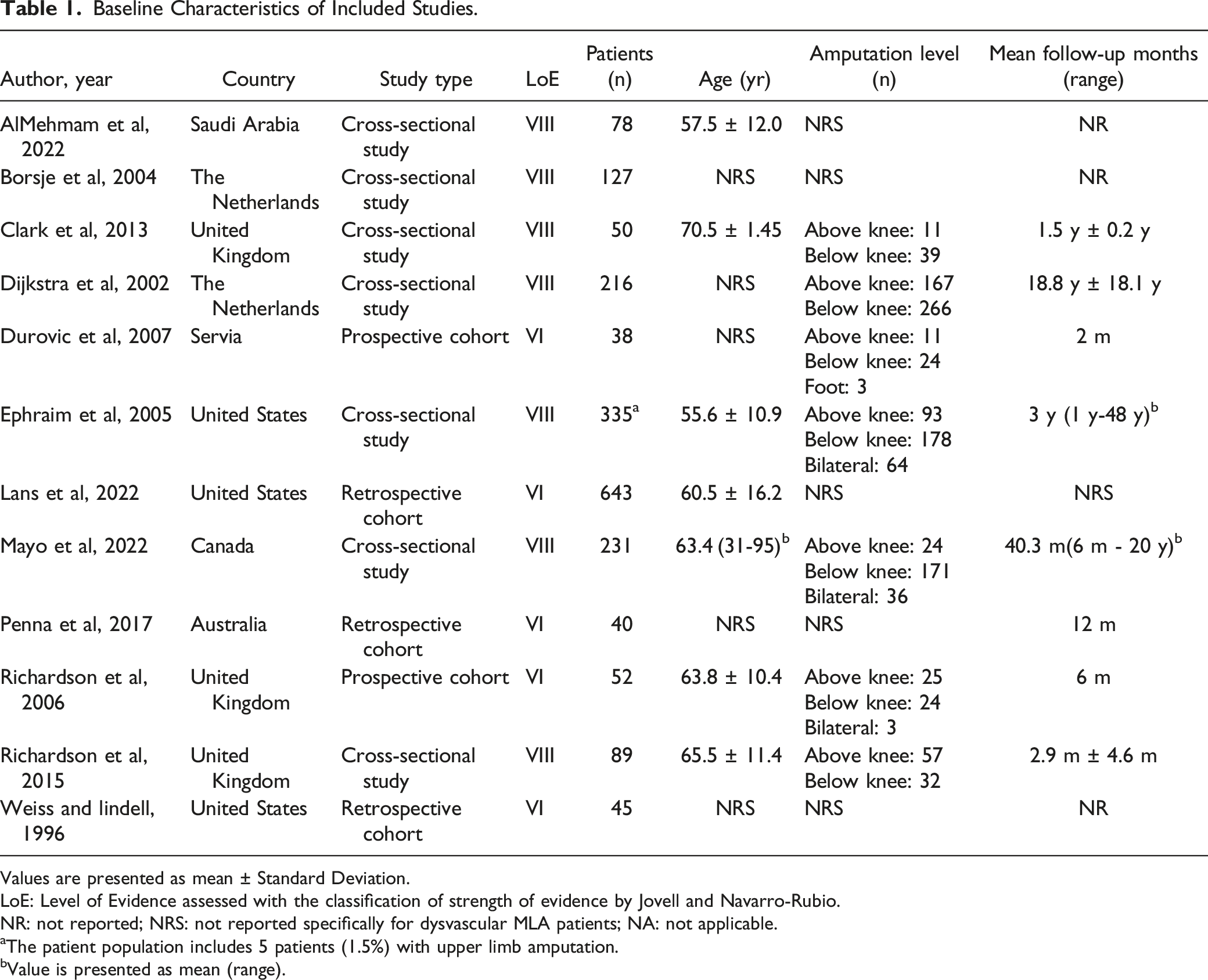
Values are presented as mean ± Standard Deviation.
LoE: Level of Evidence assessed with the classification of strength of evidence by Jovell and Navarro-Rubio.
NR: not reported; NRS: not reported specifically for dysvascular MLA patients; NA: not applicable.
aThe patient population includes 5 patients (1.5%) with upper limb amputation.
bValue is presented as mean (range).
Data Extraction and Quality Scoring
Two authors (R.B. and M.L.) extracted data from the selected articles. Collected variables included the year of publication, country, number of patients, age of patients, level of amputation, time to follow-up, and reported outcome measure.
The primary outcome was the proportion of patients that experienced PLP after LLA. PLP was defined as pain that is felt in the portion of the limb that has been amputated. Secondary outcomes were the proportion of patients with a symptomatic neuroma in the amputation stump of the affected lower limb following LLA, and pain score measured with the Visual Analog Scale (VAS)/Numerical Rating Scale (NRS).
The Jovell and Narvarro-Rubio classification was used to classify eligible articles by strength of evidence (Appendix, Supplemental Digital Content 3). A quality assessment was performed using the Study Quality Assessment Tools of the National Institutes of Health (NIH) (Appendix, Supplemental Digital Content 4) (2).
Statistical Analysis
Descriptive data was summarized in tables. For each study, the absolute number and proportion of patients reporting PLP was determined. Those numbers were statistically combined in a meta-analysis to generate an overall pooled proportion with a 95% confidence interval. This proportional meta-analysis was performed in R (version 4.0.5.) using a random-effects model with logit transformation. The random-effects model was selected because of the expected heterogeneity between the included studies. In this model, studies are equally weighted. A back-transformation from logit was performed to provide the pooled proportion of PLP. Heterogeneity testing was performed with a generalized/weighted least-squares extension of Cochran’s Q-test. The results of the analysis are graphically presented in a forest plot. A funnel plot was used to analyze publication bias.
Results
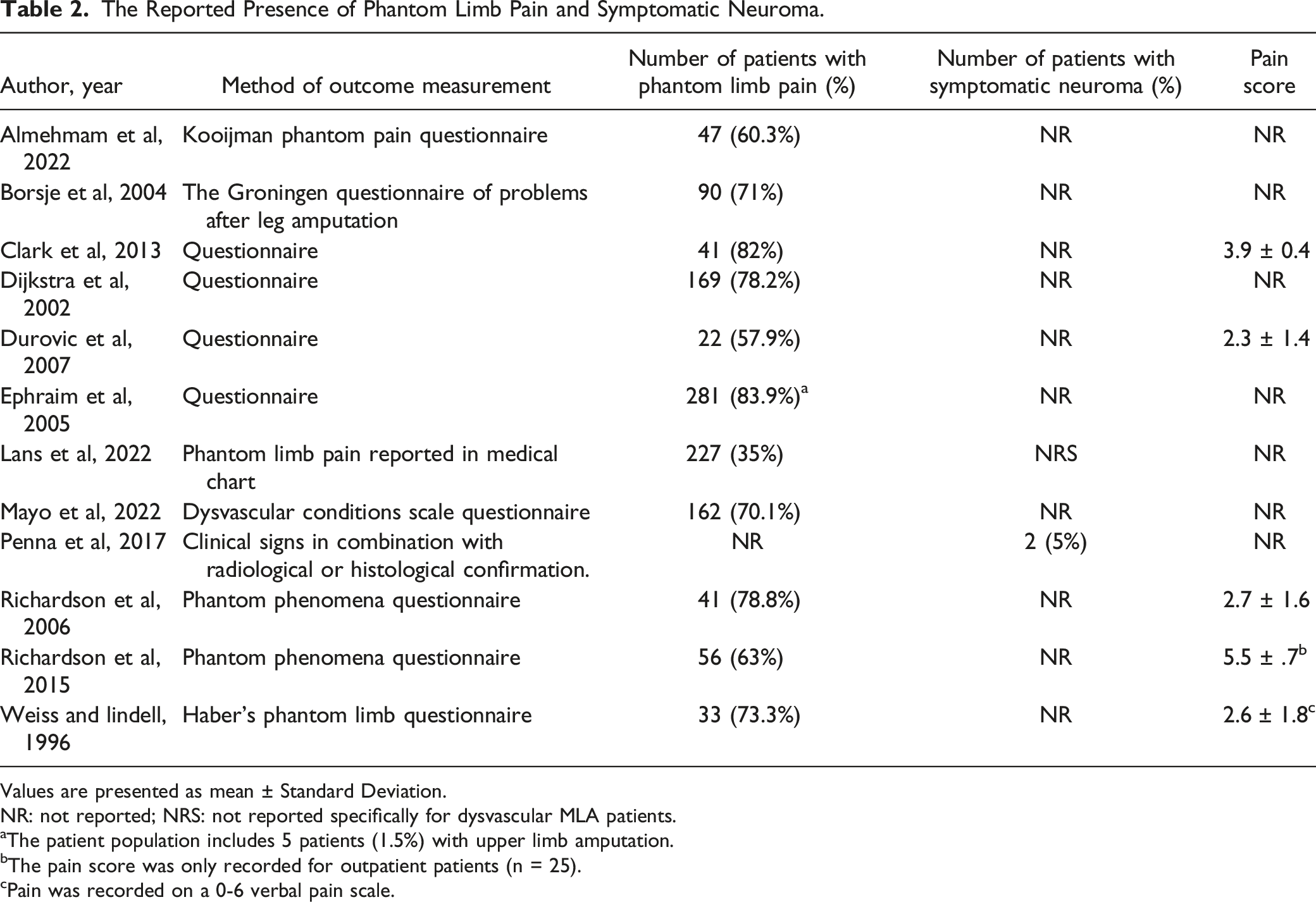
The literature search yielded 5668 publications (Figure 1). After duplicates were removed and abstracts were screened, 5610 articles were excluded. Fifty-eight studies were read in full text and assessed for eligibility. In twenty-six studies the outcome of interest in dysvascular patients was non-distinguishable from other causes for major limb amputation. Other reasons for exclusion were no outcome of interest (n = 2), sample size below 30 patients (n = 1), article not available in the English language (n = 3) and previously unidentified duplicative articles (n = 3). Eleven articles were not available in full text. A total of 12 articles were included in qualitative analysis and 11 in quantitative analysis (Table 2).1,7,12-21 Flowchart regarding the selection of included articles according to the PRISMA standards. The Reported Presence of Phantom Limb Pain and Symptomatic Neuroma. Values are presented as mean ± Standard Deviation. NR: not reported; NRS: not reported specifically for dysvascular MLA patients. aThe patient population includes 5 patients (1.5%) with upper limb amputation. bThe pain score was only recorded for outpatient patients (n = 25). cPain was recorded on a 0-6 verbal pain scale.
Seven of the 12 included articles were cross-sectional studies (Table 2),7,12-14,16,18,20 two prospective cohort studies,15,19 and three retrospective cohort studies.1,15,17,19,21 The studies were conducted in seven different countries and published between 1996 and 2022. The number of patients in each study ranged between 38 and 643, with a median of 70.5 (Table 2). Seven studies included both above and below-knee amputations,7,14-16,18-20,22 while five studies did not report amputation levels specifically for dysvascular MLA patients (Table 2).1,12,13,17,21 Three studies included bilateral amputations.16,18,19 The mean age of included patients in each study ranged between 57.7 and 70.5 years (Table 2).
Articles used various outcome measures to assess the presence of PLP and symptomatic neuroma. Ten studies used questionnaires including the Kooijman Phantom Pain Questionnaire, the Groningen Questionnaire of Problems after Leg Amputation, the Dysvascular Conditions Scale Questionnaire, the Phantom phenomena questionnaire and Haber’s Phantom Limb Questionnaire (Table 2).7,12-16,18-21 One study by Lans et al 17 retrospectively based the prevalence of PLP on reporting in medical charts. A single study by Penna et al 1 evaluated symptomatic neuroma’s presence by combining clinical signs with radiological or histological confirmation. Five studies used numerical pain scores to assess the severity of the PLP. Four studies used the 0-10 VAS scale or 0-10 NRS scale.7,15,19,20 Weiss and Lindell 21 used a 0-6 verbal pain scale.
Quality Assessment of Included Studies
Five cohort studies scored a level of evidence of six according to the classification of strength of evidence according to the Jovell and Navarro-Rubio classification.1,15,17,19,21 The remaining seven cross-sectional studies scored a level of evidence of eight.7,12-14,16,18,20 When scored with the NIH Risk of Bias tool, all studies had an average risk of bias (Appendix, Supplemental Digital Content 4). No studies were excluded based on the quality assessment or risk of bias tool.
Proportion of Patients with PLP Following Dysvascular LLA
Eleven studies reported phantom limb pain for a total of 1924 LLA (Table 3).7,12-21 A meta-analysis of these studies demonstrated that 69% of the patients experience phantom limb pain (95% CI 53-86%) (Figure 2). Meta-analysis of proportions of PLP in LLA.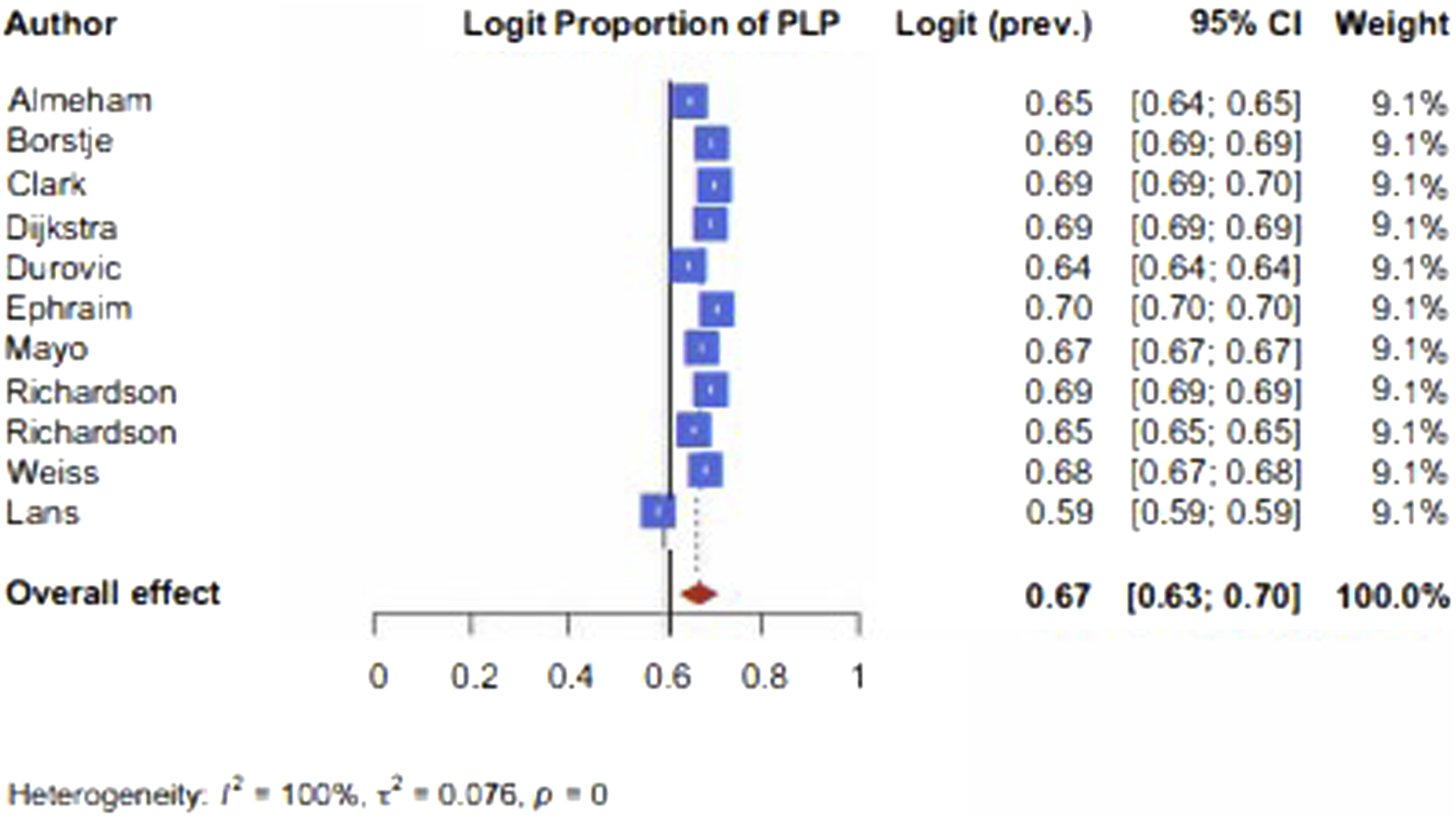
Pain Intensity of PLP
Reported pain intensity scores of PLP on the 0-10 VAS/NRS scale ranged between 2.3 ± 1.4 and 5.5 ± .7 (Table 3). Weis and Lindell evaluated pain intensity on a 0-6 pain scale and reported a score of 2.6 ± 1.8. 21
Proportion of Patients with Symptomatic Neuroma Following Dysvascular LLA
A single study reported the number of patients that developed a symptomatic neuroma following dysvascular LLA. 1 In a study population of 40, Penna et al 1 reported that two patients (5%) developed a symptomatic neuroma (Table 3).
Discussion
PLP and symptomatic neuromas can significantly impact an amputee's quality of life and mobility status, and insight into the magnitude of the problem is necessary. This systematic review aimed to examine the prevalence of PLP and symptomatic neuroma after dysvascular LLA. Overall, 69% of patients report experiencing PLP following LLA (95% CI 53-86%). In current literature and this review, evidence for the percentage of symptomatic neuroma following dysvascular LLA is lacking. However, a single study reported an incidence of 5%. 1
In this systematic review, we report a similar prevalence of PLP in dysvascular amputees as has been established in literature for the total population of all-cause amputees. Limakatso et al 23 systematically reviewed the prevalence of PLP in surgical and traumatic amputations and reported a pooled prevalence estimate of PLP of 64% (95% CI 60.01-68.05). Additionally, Schwingler et al 24 reported a pooled prevalence of PLP of 53% (95% CI 40%-66%) following all-cause LLA. Stankevicius et al 25 conducted a rapid systematic review without a meta-analysis and reported a wide range in point prevalences of PLP in included studies between 6.7%-88.1% and lifetime prevalences between 76%-87% in all-cause upper and lower limb amputations. Individual studies also corroborate our findings that the prevalence of PLP in dysvascular LLA patients is not lower than that in non-dysvascular amputees. Clark et al 7 compared the prevalence of PLP between dysvascular LLA and non-dysvascular amputations and found no difference between the groups.
Conflicting theories have been previously proposed for dysvascular amputees experiencing either less or more neuropathic pain than non-dysvascular amputees. Traditionally, patients with dysvascular LLA were hypothesized to experience less PLP and neuropathic pain than non-dysvascular amputees.7,26 The rationale is that the high prevalence of diabetes mellitus in this population and the associated longstanding peripheral diabetic neuropathy leads to a loss of sensation in the limb and, as a result, less pain. In their retrospective cohort study, Lans et al 17 reported diabetes mellitus as a protective factor for developing PLP and symptomatic neuroma, with the hypothesis that nerve regeneration potential is inhibited in patients with diabetes mellitus, making them less prone to develop neuropathic pain. Our meta-analysis disputes this theory, and we found a similar prevalence of PLP in dysvascular amputees as has been reported for all-cause amputees. Alternatively, dysvascularity may be associated with identified risk factors of PLP, like pre-amputation pain and depression, making dysvascular patients more likely to develop neuropathic pain. 23 While a significant portion of dysvascular amputees have a diagnosis of diabetic neuropathy that pre-dates the amputation, a pathophysiological association between diabetic neuropathy and the development of post-amputation neuropathic pain has not been sufficiently studied. 27 The mechanisms that lead to diabetic neuropathic pain are not yet fully understood either, although it is thought that the toxic effects of hyperglycemia play an important role. 28 Other theories proposed by Schreiber et al 28 include changes in blood vessels that supply the peripheral nerves, changes in sodium and calcium channels, metabolic and autoimmune disorders accompanied by glial cell activation, and central pain mechanisms. However, whether these factors also influence phantom limb pain development or symptomatic neuroma formation is not currently known.
Due to the potentially debilitating nature of PLP and symptomatic neuromas, various surgical strategies have been proposed to inhibit the development of neuropathic pain. Targeted Muscle Reinnervation (TMR) and Regenerative Peripheral Nerve Interfaces (RPNI) have been proposed as preventive procedures performed during major limb amputation. TMR is a procedure where the peripheral nerves are coapted to motor branches supplying functionally expendable portions of muscle in the vicinity of the nerve. 29 Originally developed to provide intuitive control of upper limb prostheses, Dumanian et al first noted the improvement in PLP and decrease in neuropathic pain in patients who underwent TMR at the time of amputation. 30 The rationale is that providing the transected nerves with a physiological target for reinnervation prevents hypersensitivity and the formation of symptomatic neuromas. Alternatively, during RPNI, the transected peripheral nerves are implanted into autologous free, denervated, and devascularized muscle grafts.31,32 Multiple studies have reported that performing prophylactic TMR during limb amputation significantly reduces the development of phantom limb pain, residual limb pain, and symptomatic neuroma.33-35 For example, Valerio et al 33 demonstrated an incidence of phantom limb pain in 55.8% of the patients treated with prophylactic TMR compared to 78.6% of the controls. In one study it was reported that prophylactic RPNI significantly reduced the incidence of painful neuroma and PLP, following LLA from 91% in the control group to 51% in the RPNI group. 36 Most studies have excluded dysvascular amputations focusing instead on amputation of trauma or oncological aetiology.
In dysvascular patients, there are several factors that could dissuade the surgeon from using TMR or RPNI preventively, including higher re-amputation rates, higher complication and wound infection rates, and a peri-operative mortality of 10-15%.37-39 Furthermore, TMR and RPNI lengthen the operative time and often necessitate making additional surgical wounds. However, a recent study by Chang et al 40 used prophylactic TMR in 100 patients at the time of below-knee amputation in a highly comorbid cohort and compared the outcomes with 100 patients treated with amputation with standard traction neurectomy in a non-randomized study. Of this cohort, 76% of patients underwent amputation for a diabetic wound or arterial or venous ulcer, 15% for a failed Charcot reconstruction and 10% for ischemic pain. Chang et al 40 reported significantly less pain and PLP in the TMR cohort, with 71% of TMR patients being pain-free at 9.6 ± 5.7 months follow-up compared to 36% of non-TMR patients (P < .01), and PLP occurring in 19% of the TMR patients vs 47% in the non-TMR patients (P < .01). Additionally, the TMR cohort's ambulation rate was significantly higher, while complications requiring revision or debridement occurred less than in the cohort treated with standard traction neurectomy. On average, preventive TMR added an additional 34.8 ± 75 minutes to the operative time. 40 These demonstrate that use of PLP preventive strategies at the time of dysvascular LLA in selected patients may significantly reduce pain and improve quality of life and overall health.
There are several limitations to this study. The most significant limitation is the varying methods of outcome measurements used to assess PLP. As no universally accepted criteria exist for PLP, studies used varying definitions to evaluate its presence, such as ‘pain in the missing part of the limb’ or ‘an unpleasant sensation in the nerve distribution of the missing body part’. Furthermore, no validated method of outcome measurement exists for PLP. Most included articles used unvalidated questionnaires, while others reported PLP based on clinical signs or retrospectively assessed medical records. This may explain why the study by Lans et al reported a particularly low prevalence of PLP, as it was the only study to use retrospective chart review to evaluate the presence of phantom limb pain, which may have led to underreporting. Additionally, the varying criteria for PLP and the variability in methods of outcome measurement may have caused the reported heterogeneity between the included studies in our meta-analysis. Furthermore, while the I-squared statistic supposedly indicates significant heterogeneity between included studies in the conducted analysis, this statistic should be interpreted cautiously. Due to the nature of proportional data, the I-squared statistic tends to yield high values in proportional meta-analyses, primarily due to the fact that even small studies with limited sample sizes often exhibit small variances in their outcomes, leading to high I-squared values. 41 Consequently, the high I-square value in such cases does not automatically indicate data inconsistency, and a conservative interpretation is recommended. 41 Therefore, a pooled proportion for PLP across the included studies was presented in this analysis despite the high I-squared value. Lastly, it was impossible to perform subgroup analyses to evaluate whether the prevalence of PLP and symptomatic neuroma differ between levels of amputation and/or amputation techniques. Previous studies have reported higher rates of neuropathic pain and PLP in proximal amputations compared to distal amputations.17,42,43
There is a scarcity of literature addressing the prevalence of symptomatic neuromas resulting from dysvascular conditions. A single retrospective study by Penna et al 1 reported a prevalence of 5%. The formation of a neuroma is expected in every severed nerve without a distal nerve end, due to disorganized axial regeneration. However, it remains unclear which mechanisms determine whether a neuroma will cause symptomatic neuropathic pain in patients. Further research is necessary to investigate the influence of dysvascularity on the development of symptomatic neuromas. The diagnosis of symptomatic neuroma development is further complicated by the absence of universally accepted diagnostic criteria. To address this issue and facilitate consistent diagnosis, diagnostic criteria proposed by Arnold et al 44 have been suggested for adoption in clinical practice and future research. To receive a diagnosis of symptomatic neuroma, patients must exhibit pain with at least three “neuropathic” characteristics, symptoms in a specific neural anatomical distribution, and a history of or suspected nerve injury. Furthermore, patients must meet at least one of the following criteria: (1) a positive Tinel sign upon examination at or along the suspected nerve injury site of a cutaneous nerve, (2) a positive response to a diagnostic local anaesthetic injection, or (3) confirmation of neuroma through ultrasound or magnetic resonance imaging. Incorporating these diagnostic criteria into future research will enable the evaluation of the prevalence of symptomatic neuromas following dysvascular amputation in a universally comparable manner. 44
Conclusions
This systematic review and meta-analysis demonstrates a high prevalence of PLP after dysvascular LLA. Evidence on the prevalence of symptomatic neuromas after dysvascular amputation is still lacking, and further research is needed. There is good evidence in non-dysvascular amputations, and a single study in dysvascular amputation, that use of surgical strategies such as TMR and RNPI at the time of amputation may significantly reduce subsequent PLP. Knowledge of the prevalence of PLP and symptomatic neuroma in patients undergoing dysvascular amputation may encourage wider adoption of techniques in this patient group.
Supplemental Material
Supplemental Material - Phantom Limb Pain and Painful Neuroma After Dysvascular Lower-Extremity Amputation: A Systematic Review and Meta-Analysis
Supplemental Material for Phantom Limb Pain and Painful Neuroma After Dysvascular Lower-Extremity Amputation: A Systematic Review and Meta-Analysis by Mirte Langeveld, MD, Romy Bosman, BSc, Caroline A. Hundepool, MD, PhD, Liron S. Duraku, MD, PhD, Christopher McGhee, BNurs, J. Michiel Zuidam, MD, PhD, Tom Barker, MD, FRCS, Maciej Juszczak, PhD, FRCS, and Dominic M. Power, MA, MB BChir (Cantab), FRCS (Tr & Orth) in Vascular and Endovascular Surgery
Footnotes
Acknowledgments
The authors like to thank Elise Krabbendam, biomedical information specialist at the Erasmus Medical Center, for her assistance with the literature search.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Board and Informed Consent
This manuscript is a systematic review and meta-analysis. As such, the article is not subject to ethical approval or informed consent procedures in the Netherlands or the United Kingdom, where the authors are located.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
