Abstract
Objective
The purpose of this narrative review is to provide the vascular surgery community with updated recommendations and information regarding the use of Targeted Muscle Reinnervation (TMR) for both the prevention and treatment of chronic pain and phantom limb pain occurring in patients after undergoing lower extremity amputation for peripheral artery disease.
Methods
Current available literature discussing TMR is reviewed and included in the article in order to provide a succinct overview on the indications, clinical applications, and surgical technique for TMR. Additionally, early studies showing favorable long-term results after TMR are discussed. Patient consent for publication was obtained for this investigation.
Results
TMR has been demonstrated to be an effective means of both treating and preventing neuroma-related symptoms including chronic pain and phantom limb pain. It has been proven to be technically feasible, and can help patients to have improved utilization of prostheses for ambulation, which can conceivably lead to a reduction in mortality.
Conclusions
TMR is an important tool to consider for any patient undergoing lower extremity amputation for a vascular-related indication. A vascular-plastic surgeon dual team approach is an effective means to prevent and reduce neuromas and associated chronic pain in this patient population.
Keywords
Introduction
There are approximately 185,000 amputations each year in the United States. 1 The majority of amputations are attributed to peripheral vascular disease, with over two-thirds of these patients also having a comorbid diagnosis of diabetes. 2 Complications of diabetic foot wounds, including wound infection, osteomyelitis, and sepsis are leading causes of major amputations.3,4
Approximately 70–80% of below knee amputations in the United States, particularly those secondary to peripheral vascular disease and diabetic complications, are performed by vascular surgeons. 5 Below knee amputations performed by vascular surgeons have been associated with fewer postoperative complications, lower revision rates, and decreased length of stay for patients in comparison to those performed by other specialties.5,6 Previous literature has not explored the differences between vascular surgery and other specialties in neuroma formation and phantom limb pain after lower extremity amputation. That being said, most amputations today are still performed in largely the same manner as those performed during the Civil War.7,8
Despite improved outcomes in the immediate postoperative period, the salvaged limb continues to be a source of poor function and chronic pain for vascular surgery patients undergoing amputation. 3 With standard techniques, it is estimated that 75–86% of patients who have undergone amputation will develop neuromas and associated chronic limb or phantom limb pain.9,10 Neuromas develop in amputated limbs at the cut ends of nerves. This is due to disorganized axonal regeneration beyond the neural sheath and epineurium. This is a particularly significant problem in large sensory nerves as the bundled sensory axons become encased in scar tissue that forms a disorganized collagen cap. Patients may experience pain and sensitivity in the area in response to a variety of triggers including light touch, temperature, pressure, or at rest.11,12 Lack of afferent feedback in severed sensory and motor nerves also leads to phantom limb sensations. 13 These sensations can range from a benign feeling that the limb is still intact, to severe and disabling pain sensation perceived as somewhere in the previously amputated limb.
Current techniques for management of neuroma pain include both non-operative and operative modalities, both of which have been limited in their efficacy. Conservative measures have included alpha agonists, anticonvulsants, antidepressants, local anesthetics, electrical stimulation, and massage. 12 Surgical options have focused on dividing the nerve as proximally as possible, or wrapping/burying the severed nerve into adjacent structures including muscle, bone, or vein.14,15 However, these methods have shown limited efficacy. 12 Current non-surgical treatments for phantom limb sensations/pain include mirror therapy, virtual reality therapy, anticonvulsants, opioids, and memantine. 13 Surgical management of phantom limb sensations/pain includes stump revision, nerve block, and neurectomy. 16 The efficacy of these interventions is highly variable as well.
Targeted Muscle Reinnervation (TMR) offers an alternate effective means toward neuroma and phantom limb control. TMR is a microsurgical technique that was first described in 2002 17 and originally designed not as a pain management modality, but to facilitate myoelectric interfaces for enhancement of volitional control of complex activities with upper extremity prostheses. 18 TMR refers to the coaptation of a large donor nerve end terminus to a smaller recipient motor nerve, innervating a muscle belly within the adjacent field. This requires microsurgical technique, tissue handling, dissection, and use of a nerve stimulator to identify suitable target motor nerves. The movement in the reinnervated motor units is detected by a specialized prosthesis and translated into predetermined motions. 18 In early studies the patients that had these procedures performed noted an incidental reduction in neuroma pain and phantom limb sensations. The technique has since been trialed specifically for this purpose in traumatic amputees and has been found to reduce rates of symptomatic neuroma formation after amputation.11,14,19,20 Consequently, TMR has also been employed in lower extremity amputation patients as either treatment for, or surgical prophylaxis against chronic limb pain and phantom limb pain.14,21
The mechanism by which TMR works to prevent phantom limb sensations/pain and neuroma pain is not completely clear. Theoretically, by establishing a pathway for more directed and purposeful axonal regeneration, TMR prevents the nerve from overgrowing in a disorganized way and forming a neuroma. In addition, the reestablishment of afferent and efferent signals from the cortex to the muscle that has been reinnervated likely prevents the phantom limb symptoms as well. 15 More succinctly, by “giving the nerve somewhere to go and something to do,” these conditions are avoided. 20 While TMR has been described in the plastic surgery, surgical oncology and orthopedic surgery literature10,22 recognition of this technique by vascular surgeons and has been limited. 23,24 The purpose of this paper is therefore to outline for the vascular surgery community the practical technical aspects of TMR and to illustrate the significance of collaboration with plastic surgeons on these cases to improve outcomes in vascular surgery patients undergoing amputation. Patient consent for publication was obtained for this investigation.
Indications
TMR should be considered in any patient undergoing above knee amputation or below knee amputation. At present TMR is indicated primarily for traumatic or oncologic amputees and secondarily for patients experiencing symptoms related to neuroma formation following amputation. At the authors’ institutions, TMR is considered for any patient undergoing major lower extremity amputation that meet the following criteria: ambulatory prior to presentation, no known severe neuropathy, younger than 65, and no medical contraindications to adding approximately 1 hour of operative time to a primary amputation procedure or performing a secondary procedure lasting 1–2 h. The inclusion criteria utilized by our institution coincides with the recommendations of previously published investigations on TMR.10,17,18,19,20,22 All patients who underwent TMR were ambulatory patients at age of 65 or younger. Patient characteristics included comorbidities common to patients with peripheral arterial disease, including diabetes, hypertension, hyperlipidemia, coronary artery disease, and chronic kidney disease. Most patients undergoing amputation with TMR had distal ulcerations with no revascularization option to the distal extremity, but suitable perfusion to ensure healing after BKA or AKA.
For patients undergoing delayed TMR, they undergo a trial of conservative management, where they are followed closely in the early postoperative period. During this time, patients are evaluated for neuropathic pain and phantom limb pain. If recovery from neuropathic pain is delayed or symptoms are persistent and debilitating, we proceed with the delayed TMR procedure. Approximately 50% of the patients in this investigation underwent delayed TMR. There were many different factors that play a role in the decision for immediate versus delayed TMR. This includes other medial comorbidities, surgeon availability, and operating room availability.
While all age groups should be considered candidates for TMR, studies have indicated that there is a higher likelihood of success in younger patients likely owing to improved neuronal regeneration in younger age groups.25,26 Additionally, younger working-age patients are more likely to benefit from prophylactic management of neuroma and phantom limb pain, as these can cause significant impact on performance at work.26. The percentages vary, but approximately 20–40% of patients who undergo amputation by the providers in this investigation qualify for TMR. This is both practice and patient population dependent. Typically, younger diabetic patients undergoing amputation more frequently qualify for TMR. In contrast, practices caring for older nursing home practices are less likely to have patients who qualify for TMR.
While there are no absolute contraindications to TMR described in the literature, the authors generally consider as highest priority those patients that are likely to pursue rehabilitation and ambulation with a prosthetic. As such, TMR is generally deferred in patients who are bedbound, demented, or of very advanced age. Exceptions can be made for patients who are particularly concerned about phantom limb symptoms. The presence of existing neuropathy pre-operatively may limit the utility of TMR, but has not been extensively explored and further research is warranted on the impact this factor has on patient outcomes. Physical exam and review of systems was utilized to assess neuropathy in this investigation. Review of systems involved inquiring if patients had symptoms that included numbness, burning, stabbing pain, “pins and needles” sensation, sharp pain, and weakness.
Technique
TMR can be performed for above knee or below knee amputations. In addition, TMR can be performed in an immediate fashion, at the time of amputation, or in a delayed fashion separate from the amputation procedure. Unique surgical approaches are required in each distinct scenario. In all of these scenarios, however, large (named) nerve ends are transferred to small (unnamed) segmental motor nerves within the wound. These segmental motor nerves are readily identified using the Checkpoint Nerve Stimulator Device (Checkpoint Surgical Inc, Independence, OH) (Figure 1). The nerve stimulator is only used to identify target motor nerves. The five major nerve bundles encountered during a BKA are identified by their anatomic location and caliber during dissection. The neurorrhaphy is performed using 8–0 or 9–0 nylon and standard microsurgical techniques. These nerve transfers can be performed with either loupe magnification or the operating microscope. Checkpoint Nerve Stimulator used to identify motor nerves for neurorrhaphy in Targeted Muscle Reinnervation procedure.
The quality of the muscle bed is essential for successful outcomes in patients undergoing any lower extremity amputation and furthermore those utilizing TMR. The muscle bed that is innervated by the target nerve is assessed when stimulating the target with the nerve stimulator. If there is good contraction of the muscle, it is viable for reinnervation by our donor nerve. Additionally, all muscle beds should have no evidence of active infection or ischemia.
Immediate Targeted Muscle Reinnervation in Below Knee Amputation
When coapting an oftentimes large nerve terminus to a smaller motor target there is often a size-mismatch upwards of 10:1. 15 However, size-mismatch has not been shown to provoke neuroma formation. Transfer of the larger motor nerve can enhance EMG signal over 10x and as such increasing fascicular input to a motor nerve at an amputation stump can actually maintain and even enhance stump muscular bulk over time. 15 As such, there is no concern about denervating the muscle from which our target motor nerve is selected.
In a BKA, the five major nerves of the lower extremity are cut. These include the tibial nerve, deep and superficial peroneal nerves, the saphenous nerve, and the sural nerve. In our center, the plastic surgery and vascular surgery teams work in conjunction through a standard posterior flap incision for immediate TMR during a BKA. Both teams will start the procedure working together to identify the superficial peroneal, deep peroneal, tibial, and saphenous nerves at the ankle and dissect them proximally to the level of the tibial osteotomy so that they can be isolated and protected for later transfer. The sural nerve is identified in the posterior flap and dissected free following the completion of the amputation. Once the osteotomies have been performed and the specimen has been removed by the vascular surgery team, the five nerves are prepared for transfer into small segmental motor nerves of the residual stump musculature by the plastic surgery team (Figure 2). Once these nerve transfers are complete the amputation can be closed in a standard fashion. Immediate approach to Targeted Muscle Reinnervation in a below-the-knee amputation in the supine position with the five major sensory nerves of the lower extremity including (from left to right) the saphenous nerve, sural nerve, tibial nerve, deep peroneal nerve, and superficial peroneal nerve are identified and dissected (left photo) and sutured to adjacent motor nerves (right photo).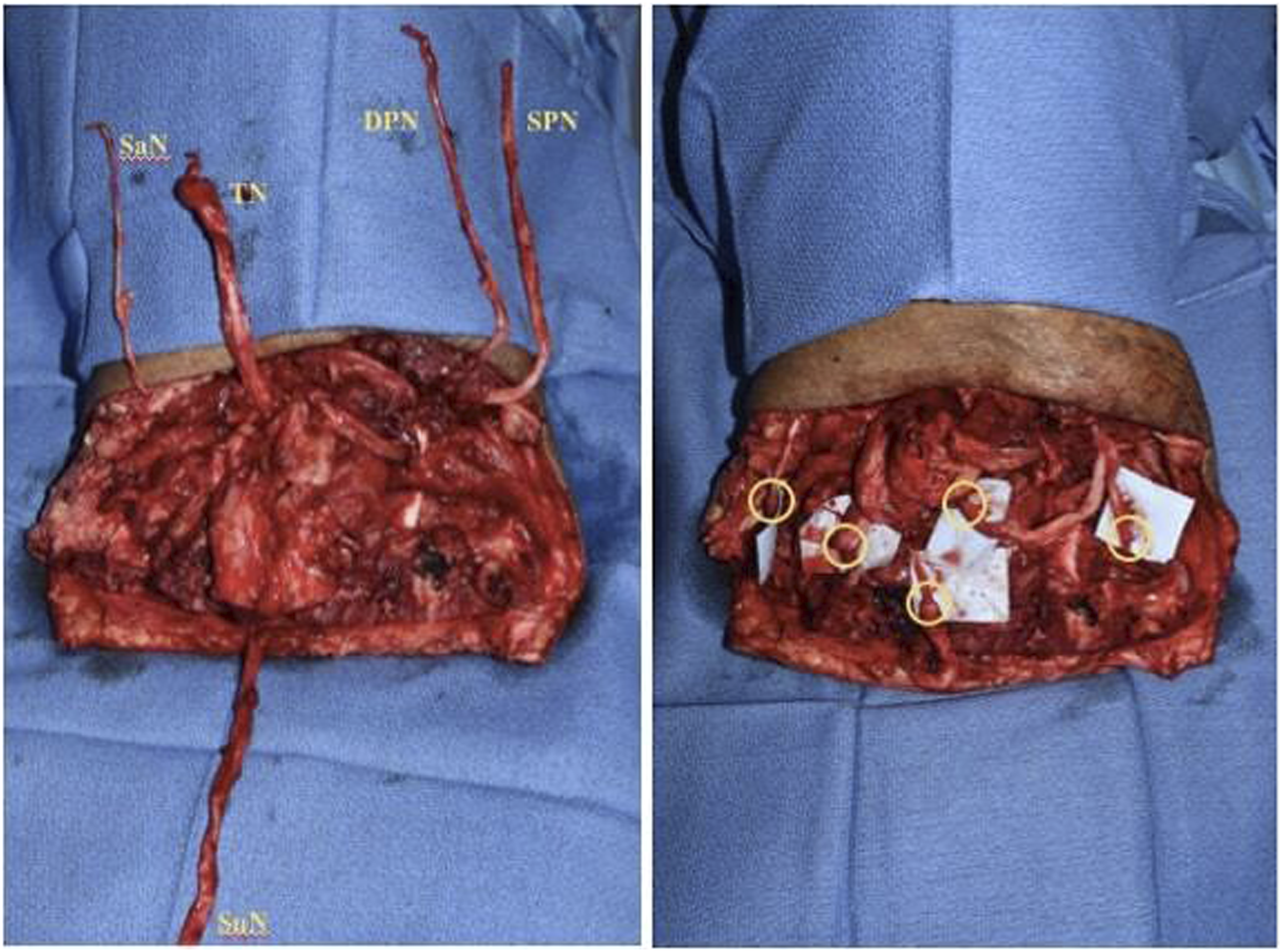
Delayed Targeted Muscle Reinnervation in Below Knee Amputation
In a situation where coordination for TMR at the time of below knee amputation is not possible, or if a patient presents with symptoms following BKA, TMR can be performed in a delayed fashion. This is done via separate approaches proximal to the healed stump to avoid stump complications. The tibial nerve, deep peroneal nerve, and superficial peroneal nerve are accessed through a linear incision in the popliteal fossa and transferred to motor branches present there (Figure 3). The saphenous and sural nerves are generally not treated in these delayed cases (unless they are symptomatic) as they are considerably smaller and less likely to cause symptoms than the other three major nerves cut during BKA and would require additional surgical approaches. Delayed approach to Targeted Muscle Reinnervation in a below-the-knee amputation in the Prone position through the popliteal fossa.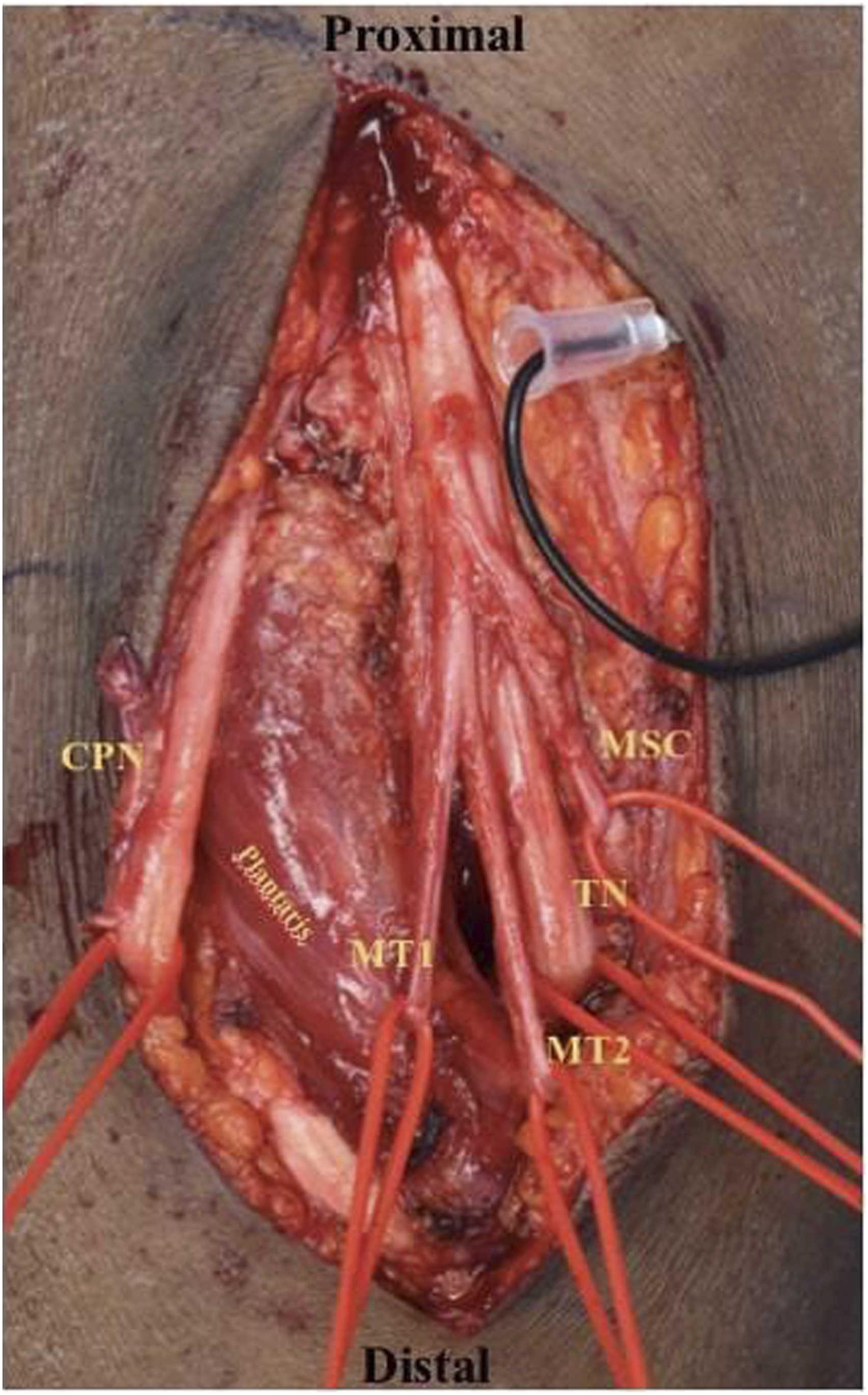
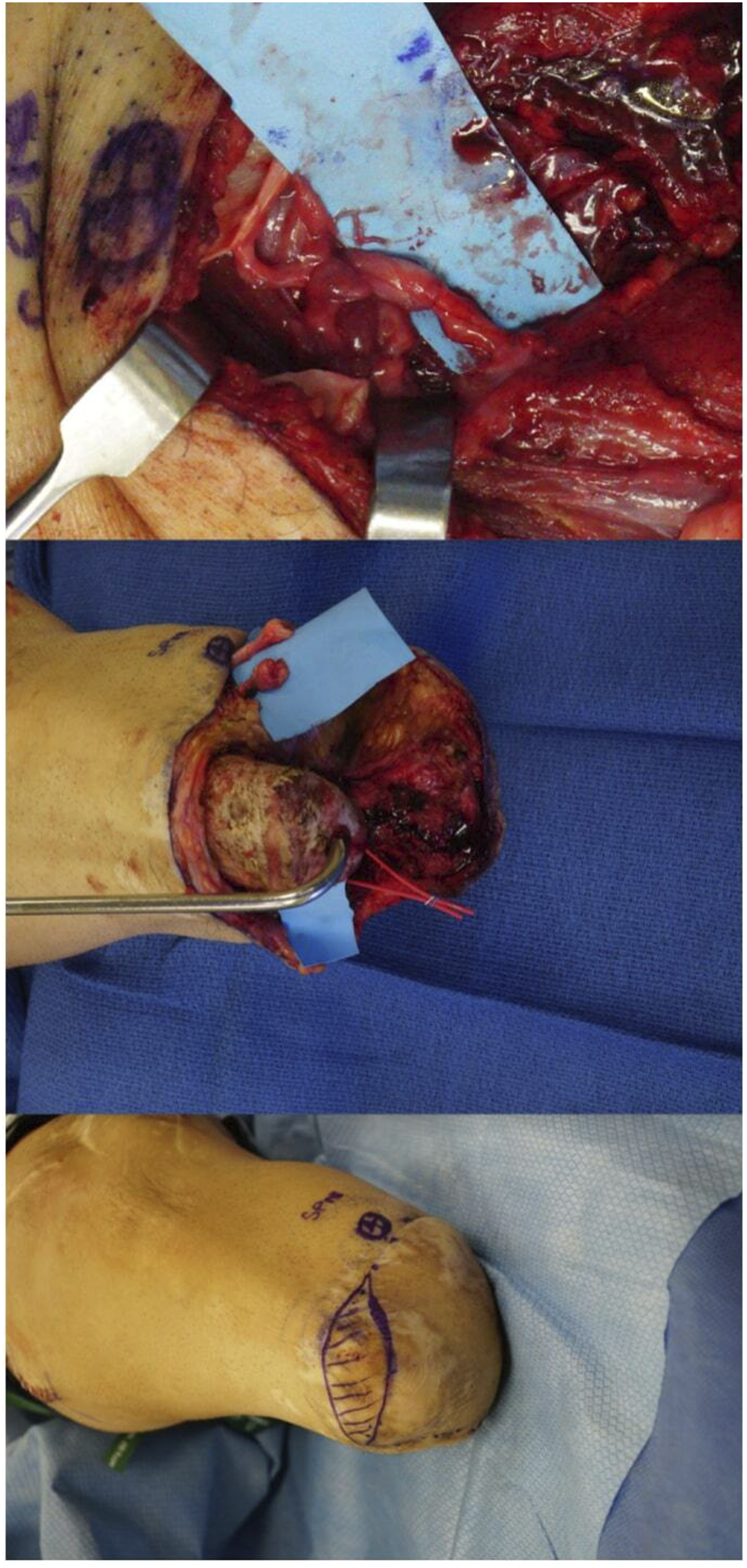
Targeted Muscle Reinnervation in the Above Knee Amputation
Targeted Muscle Reinnervation for the above knee amputation requires treatment of the tibial and peroneal components of the sciatic nerve along with the saphenous nerve. These nerve transfers can be performed either at the time of the initial amputation or in a delayed fashion, but are usually performed through separate incisions. The sciatic nerve is approached through an 8–10 cm linear incision extending proximally from the midpoint of the leg (Figure 4). Once the sciatic nerve is exposed the tibial and peroneal components are readily visible within the sciatic sheath and can be easily neurolysed apart from one another before being transferred to motor nerves to the long head of the biceps femoris and/or the semimembranosus (Figure 4). The saphenous nerve is approached through a separate incision on the distal portion of the medial thigh (Figure 5). It is coapted to motor nerves to the adductor longus or vastus medialis (Figure 5). Immediate approach to Targeted Muscle Reinnervation in an above-the-knee amputation demonstrating incision markings (bottom), exposed sciatic nerve (middle), and the same nerve neurolysed into the tibial and peroneal components before being transferred to motor nerves to the long head of the biceps femoris and/or the semimembranosus muscle. Immediate approach to Targeted Muscle Reinnervation in an above-the-knee amputation demonstrating the incision markings (bottom) for the medial thigh approach to the saphenous nerve on the distal portion of the medial thigh, the dissected nerve (middle), and the coapted saphenous nerve to the vastus medialis muscle (top).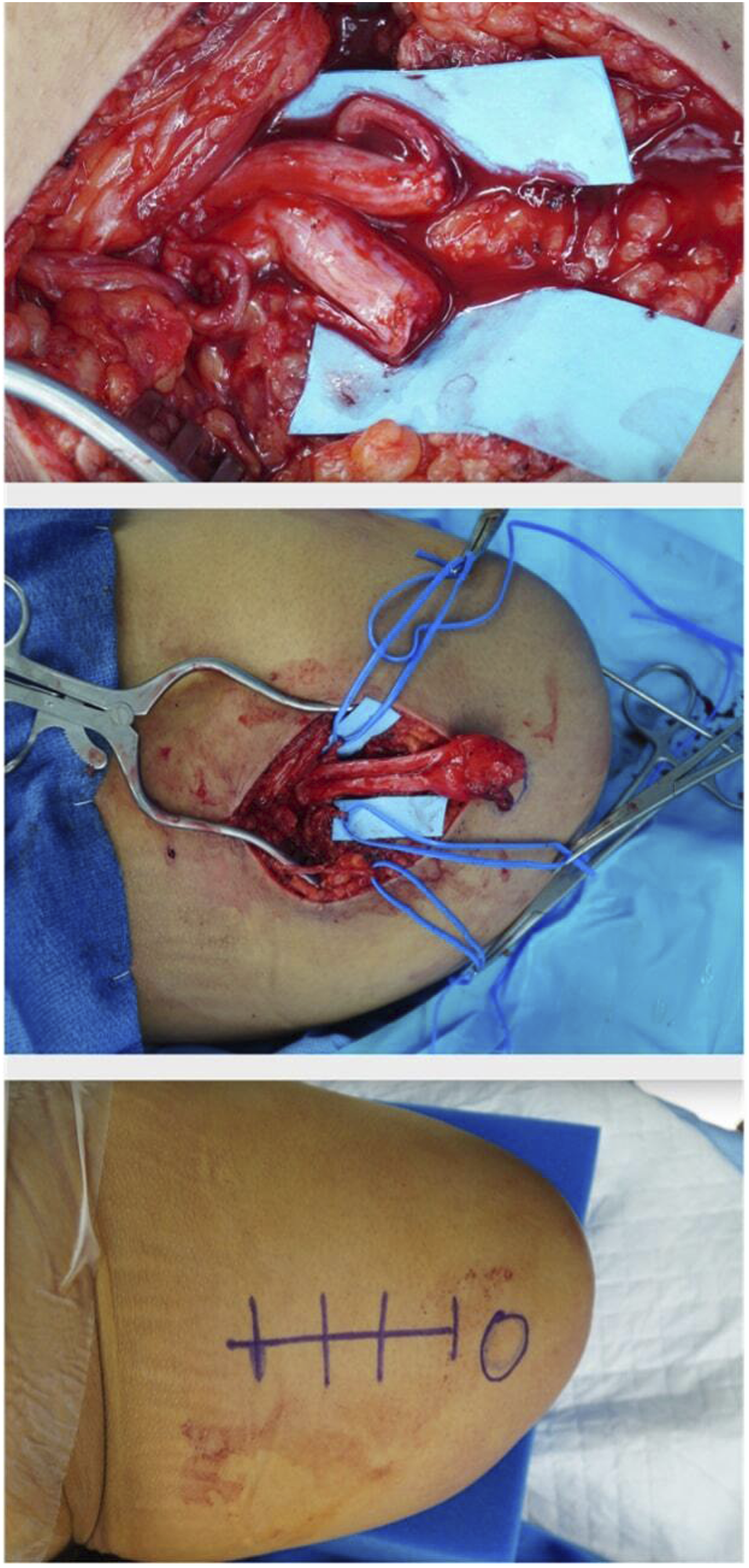
Discussion
Targeted Muscle Reinnervation is a valuable addition to the armamentarium of 21st century surgeons performing lower extremity amputations. Collaboration with plastic surgeons for TMR on these cases will yield improved long-term outcomes for our patients, as demonstrated by trials from multiple institutions implementing TMR.10,20,27 There is new data emerging demonstrating a particular benefit for the implementation of TMR in vascular amputees. 23 In general, decreased pain and phantom limb symptoms not only improve a patient’s quality of life but also allow for greater prosthesis tolerance and increased ambulation. Consequently, increased mobility has been demonstrated to reduce mortality in a patient population that is already at high risk for cardiovascular events.23,26
In the first randomized control trial comparing TMR to the standard of care (neurectomy) in patients who had developed neuromas secondary to limb amputations in 2019, Dumanian et al. 20 demonstrated that TMR results in significantly reduced phantom limb pain and reduced residual limb pain after treatment. Additionally, Valerio et al. demonstrated that patients who were treated with TMR versus amputation alone were three-times less likely to experience phantom limb pain and nearly four-times less likely to experience residual lower limb pain after amputation. 15 TMR can also be instrumental in reducing chronic opioid use in amputation patients. 15
Additionally, in our practice we have noted a secondary educational benefit to the vascular and plastic surgical trainees, as the approaches to the nerves for neurorrhaphy closely parallel those used to expose nearby vascular pedicles. As rates of open cases continue to decrease in the field of vascular surgery with the advancement of endovascular interventions, TMR will allow for improved comfort with the individual skillsets involved with open approaches among trainees in both specialties. 28 Further research evaluating the educational benefit for residents and trainees assisting in these procedures is ongoing at our institution.
Studies have demonstrated that TMR is a very effective preventative measure for chronic post-amputation limb pain, and that it poses limited risk for additional complications. The use of TMR adds approximately 60 min to operating room time, and is therefore an acceptable addition to a simultaneous amputation case.14,29 Despite, a longer operating room time with the combined approach, this drawback is mitigated by being able to provide the patient with one surgical procedure and exposure to anesthetic with improved postoperative outcomes. TMR has been proven to be technically feasible for a below-the-knee amputations and effective donor nerve and target motor nerve branches have been identified for these cases.19,30 Outcomes have been excellent when nerve transfers are performed by trained microsurgeons who routinely perform nerve coaptations. That being said, surgeons from multiple specialties, if adequately trained in nerve transfer technique can be adept in identifying and performing the nerve coaptations required for TMR.
We are presently collecting data and compiling the results of patients undergoing TMR at our institution. Therefore, overall outcomes and statistics are not included in this current manuscript. Results are forthcoming and we plan to publish this data in a future submission.
Conclusions
Targeted Muscle Reinnervation offers promising benefits to patients undergoing amputation for peripheral vascular disease or infection specifically. In these patients, it has been demonstrated that TMR significantly reduces residual limb and phantom limb pain and postoperatively decreases opioid use while increasing ambulation rates compared to non-TMR patients. 23 Patients with peripheral vascular disease and diabetes are at a higher risk for decreased ambulation compared to other amputees 31 and thus have the potential for greater benefit from the incorporation of TMR into regular practice.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
