Abstract
Seizures and epilepsy affect people of all sexes and genders. In the last several years, funding agency initiatives such as the U.S. National Institutes of Health policy on sex as a biological variable (SABV) have intended to encourage researchers to study both males and females from cell to tissue to organism and analyze and report the resulting data with sex as a factor. Preclinical epilepsy research, however, continues to be plagued by confusion regarding both the SABV policy and its implementation, reflecting similar beliefs in the larger neuroscience research community. This article aims to address some common misconceptions and provide practical tools and suggestions for preclinical epilepsy researchers in implementing SABV and analysis of the female ovarian cycle (estrous cycle in rodents) in their research programs, with a focus on studies using rodent models. Examples of recent publications in preclinical epilepsy research highlighting the value of incorporating SABV and information on the estrous cycle are included. The specifics of how best to address SABV and the estrous cycle can vary depending on the needs and goals of a particular research program, but an embrace of these physiological factors by the preclinical epilepsy research community promises to yield more rigorous research and improved treatment strategies for all people with epilepsy.
Introduction
Seizures and epilepsy affect people of all sexes and genders.1‐3 Much like neuroscience research more broadly,4‐6 however, the inclusion of female animals in preclinical epilepsy studies has lagged far behind that of males. National funding agencies, most prominently the U.S. National Institutes of Health (NIH), now require the inclusion of female animals, cells, and tissues in basic and preclinical research. Adhering to the NIH policy entails factoring sex as a biological variable (SABV) into the research design, analysis, and reporting of collected data. 7 This policy has been active since 2016, so nearly all current NIH grants were initiated with this policy in place. Despite these initiatives, however, one commonly finds newly published papers in preclinical epilepsy and neuroscience research either not including females, not providing the data disaggregated by sex, and/or not including any statistical analysis addressing SABV.
Why are researchers hesitant to incorporate females in experimental cohorts? Common explanations include a perception that addressing SABV requires doubling experimental group numbers, thus increasing time and money; alleged lower variability in males; the “complication” of the estrous cycle in females; an assumption that a lack of phenotypic sex differences suggests results from one sex will translate to the other; and a belief that females can be examined after a main study of males is complete.
Here, I aim to address these misconceptions and provide practical strategies to better incorporate SABV analysis and design into preclinical epilepsy studies, together with suggestions for when and how to integrate evaluation of the female ovarian cycle (Figure 1). At the outset, however, it is important to state that just as epilepsy is highly heterogeneous, there is no “one size fits all” approach to incorporating SABV and the ovarian cycle. Therefore, my goal here is to help the members of the preclinical epilepsy research community to better understand the value of addressing these factors in their own work and provide practical guidance in doing so. For an in-depth analysis of studies examining SABV and ovarian cycle effects in epilepsy, please see our comprehensive recent review, 1 with the addition of more recent works in the preclinical epilepsy literature as cited below.
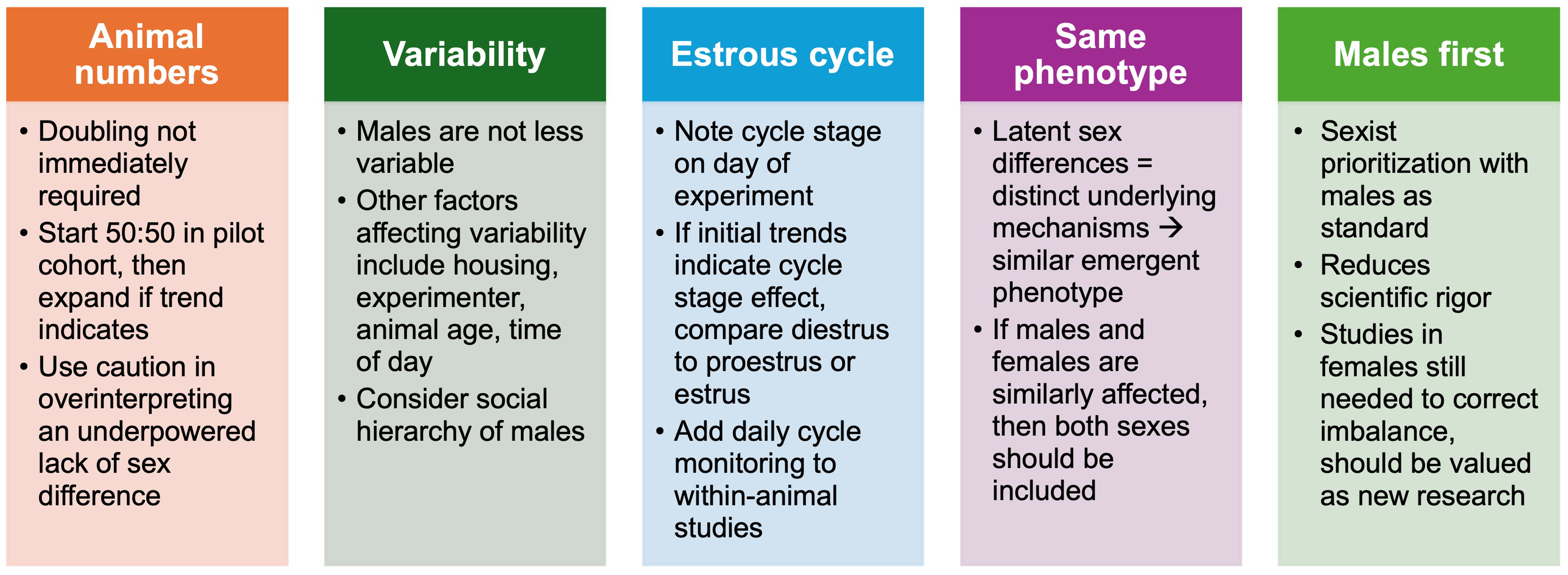
Summary of common misconceptions and strategies in addressing sex as a biological variable and the ovarian cycle in preclinical studies.
The focus here will be on rodent models (rats and mice). Therefore, I will use the term “sex” as the terminology to distinguish between the biologic and physical characteristics of males and females. 8 In humans, “gender” refers to a nonbinary spectrum of psychosocial norms, roles, and interactions that may not align as conventionally expected with the biological sex. These aspects cannot be meaningfully assessed or determined in preclinical models, so sex is the correct term to use in studies using nonhuman animals. It is important to note that sex is not a perfect binary, 9 but for simplicity and relevance to the vast majority of preclinical work, I will refer to males and females. In addition, although the human ovarian cycle is a menstrual cycle, female rodents have an estrous cycle, so I will refer to the estrous cycle throughout.
Addressing Common Myths About SABV and the Estrous Cycle
Including females will cost more time and money since we have to use twice as many animals.
At the outset of a study, it is appropriate to begin with experimental groups at an approximately 50:50 ratio of males to females. After completing a pilot experiment, analyze and examine the data with the variables disaggregated by sex (e.g., by using different symbols or colors in the data visualization plots). If the data points strongly overlap, then it is unlikely that increasing the animal numbers further will yield a biologically meaningful sex difference. If there appears to be little or no overlap between the data points from males and females, thus suggesting the potential for a sex difference, a secondary power analysis should be conducted incorporating the magnitude of this difference to account for sex as a statistical factor. Further details regarding this experimental strategy are available elsewhere.10‐12 A recent preclinical epilepsy paper provides a nice example of the value of expanding a study to examine sex differences based on initial trends indicating a sex effect. 13
Even if expanding a study does indeed require more time and animals, it is an investment in rigor and reproducibility and avoids overinterpretation of a lack of difference in a small cohort. For example, although early studies of spike-and-wave discharge (SWD) activity in six male and six female WAG/Rij rats did not indicate a sex difference, 14 a recent study using longitudinal monitoring in a group over three times as large showed age-dependent increases in male-dominant SWDs. 15
An important step, no matter the sample size, is to report the data in a manner that allows identification of which data came from which sex.
Males are easier to use because they are less variable.
This common myth is dispelled by metaanalyses demonstrating that, across a variety of traits commonly measured in preclinical neuroscientific and biomedical studies, the coefficients of variation were virtually identical in male and female rodents.16‐18 Moreover, in some traits, males were more variable than females.
16
With respect to epilepsy, our recent work in the intrahippocampal kainic acid (IHKA) mouse model of temporal lobe epilepsy demonstrated similar variances in seizure parameters in both sexes.
19
Of note, other factors such as housing conditions,
20
differences in experimenters,21,22 time of day of experimentation,
23
and age and body weight
24
can also shape the variability of experimental outcomes. In group-housed conditions, males develop hierarchies dominated by one “alpha” individual, and circulating testosterone and corticosterone levels can differ between dominant and subordinate males.25,26 Male social status is entirely unexamined and unreported in preclinical epilepsy research.
Studying females is complicated because of the estrous cycle.
The rodent estrous cycle consists of four main stages (in order): proestrus; estrus; metestrus; and diestrus. For clarification on the spelling of these terms, see Figure 2. Proestrus is akin to the late follicular phase of the human menstrual cycle and is characterized by high circulating estrogen levels. Rodents do not have a true luteal phase as in the menstrual cycle, but progesterone levels are elevated on diestrus compared to the other stages. Most rats and mice spend 1 to 2 days in diestrus, resulting in an average cycle length of 4 to 5 days. Changes in vaginal cytology reflect these changes in circulating levels of ovarian steroids. With a bit of practice, assessment of estrous cycle stage is relatively straightforward and noninvasive, and our group and others have produced protocols and tools to assist in this regard.27‐29 Although visual inspection of the vagina is easier technically and can also provide an indication of estrous cycle stage, 28 the features vary by strain, are mostly visible only on proestrus and estrus, and are more prone to observer interpretation. Similarly, the accuracy of commercially available vaginal impedance monitors is highly questionable. 30

Clarification on the spelling of terms related to the estrous cycle.
Multiple studies have indicated an impact of estrous cycle stage on various aspects of seizure parameters, with estrous rodents often observed to display increased susceptibility to seizure induction. 1 However, these findings do not automatically mean that all experiments must be performed on all cycle stages, or that a female must be carefully monitored for many days prior to testing. In the vast majority of studies, an appropriate approach would be to note the estrous cycle stage on the day of examination (e.g., the day of seizure induction testing), without specifically timing examination in all females to the same cycle stage. An argument that the magnitude of cycle-associated changes is typically not large enough to warrant this extra effort relies on such an assumption being true. That is, if no cycle stage information is collected at the time of experimentation, then it is guaranteed that any trend indicating otherwise will be missed. By contrast, incorporation and reporting of estrous cycle stage information, without specifically timing the experiments to one particular stage, allows one to match the initial data collected to the cycle stage to identify trends toward stage-dependent shifts and indicate whether further studies comparing different stages are warranted. However, a study designed to specifically examine estrous cycle influences does not necessarily require quadrupling animal numbers to account for all four cycle stages. An appropriate strategy starting with just two groups of females is to use diestrus (low estrogen:progesterone ratio) as a baseline stage and compare it to proestrus or estrus (high estrogen:progesterone).31‐33
For studies in which the same animals are continuously monitored across several days, such as chronic electroencephalography recordings, the relatively simple addition of daily estrous cycle staging provides the benefit of enabling within-animal longitudinal statistics that are more statistically powerful and can yield novel insights into interactions between estrous cycle stage and seizure phenotypes. For example, our group recently found that cycle stage-dependent shifts in seizure burden can be resolved in IHKA female mice when the same animals are monitored across multiple days of the cycle, with diestrus used as an internal control for each mouse. 34 Similar shifts with estrous cycle stage in SWDs and interictal spikes have also been observed in rat epilepsy models.35,36 It is important to note that rodent epilepsy models are often characterized by estrous cycle disruption,37‐40 and tethered EEG recording in and of itself can be a stressor that temporarily disrupts the cycle in control animals. 34 Therefore, it is important to perform estrous cycle staging each day during recording and not rely on extrapolation from more infrequent monitoring.
Of note, although changes with estrous cycle stage are thought to largely reflect actions of ovarian hormones, neural actions of gonadal hormones are not a uniquely female concern. Blood testosterone levels in male rodents fluctuate in a diurnal pattern,41‐43 and testosterone and testis-derived factors can shape acute seizure susceptibility in males.44‐46 The male brain also expresses aromatase, the enzyme that converts testosterone to estradiol, 47 and aromatase blockade modulates seizure susceptibility in both sexes.48,49
If estrous cycle stage influences a particular phenotype, this result provides an opportunity for a better understanding of underlying mechanisms.
50
Given that half of the human population experiences an ovarian cycle for at least some portion of the lifespan, an understanding of the impacts of this cycle (and its absence) on brain function, seizures, and epilepsy is warranted. The estrous cycle should not be used as a cudgel to prevent the inclusion of females in preclinical research.
This form of epilepsy doesn’t show a sex difference in humans or in the animal model.
A lack of visible sex difference does not necessarily confirm that the underlying mechanisms are the same in both sexes. “Latent” sex differences are distinct underlying mechanisms at the genetic, cellular, and/or circuit levels in males and females that give rise to similar emergent phenotypes.51,52 This paradigm has not yet received widespread attention in the context of epilepsy, but an understanding of its relevance to seizure mechanisms, antiseizure medication efficacy, and other aspects of epilepsy research and care can only be achieved by detailed studies of both sexes. Furthermore, if a type of seizure/epilepsy indeed appears similar in human males and females, then it stands to reason that it affects both males and females and thus both sexes must be included in preclinical experiments.
We can test males first to see if there's an effect, then follow-up to determine if females are the same.
This strategy is flawed on several fronts. First, it unjustifiably prioritizes the study of males. Unless one is studying a clearly sex-specific system or process, there is no scientific basis to rank the study of one sex as more important. Second, it treats the male as the archetypal standard, thereby inferring the female is the “other.” This reasoning is sexist and must not be tolerated and perpetuated. Third, it diminishes scientific rigor. For example, even if there is a willingness to perform subsequent studies in females, the gap in time between the two studies may be substantial, raising the risk of other factors (e.g., environmental and/or experimenter changes, genetic drift, reagent quality) either inducing an apparent sex difference where there is none, or masking a true sex-influenced effect. Lastly, the strategy relies on a promise that can easily be broken due to circumstances such as the ending of a grant funding period, the departure of certain laboratory personnel, and/or typical changes in project priority over time.
That said, it is better late than never to right the historical wrong of the exclusion of females by revisiting prior work done in males and performing near-equivalent studies in females. As a recent example, an initial study in males demonstrated that a specific microRNA inhibitor could robustly reduce spontaneous seizures in a mouse model of chronic epilepsy. 53 In a subsequent study of females from the same laboratory, however, this antiseizure effect was reduced, 33 providing important mechanistic insight into the prospects for using this particular microRNA inhibition to achieve effective seizure suppression. There is also value in female-only studies confirming similarity to previous findings in males, as in a recent study of antiseizure medications and drug resistance in IHKA mice. 54 It is vital that journal editors, grant funding agencies, and university hiring and promotion committees recognize the importance of this work by considering it as novel research and not simply replication studies. 55
Concluding Remarks
Implementation of the NIH SABV policy still falls short of the stated goals, particularly with respect to publications and data reporting. One remedy is for manuscript reviewers and editors to require the inclusion of females and disaggregation of data by sex as criteria for publication unless a science-based justification for studying only one sex is provided (Figure 3).
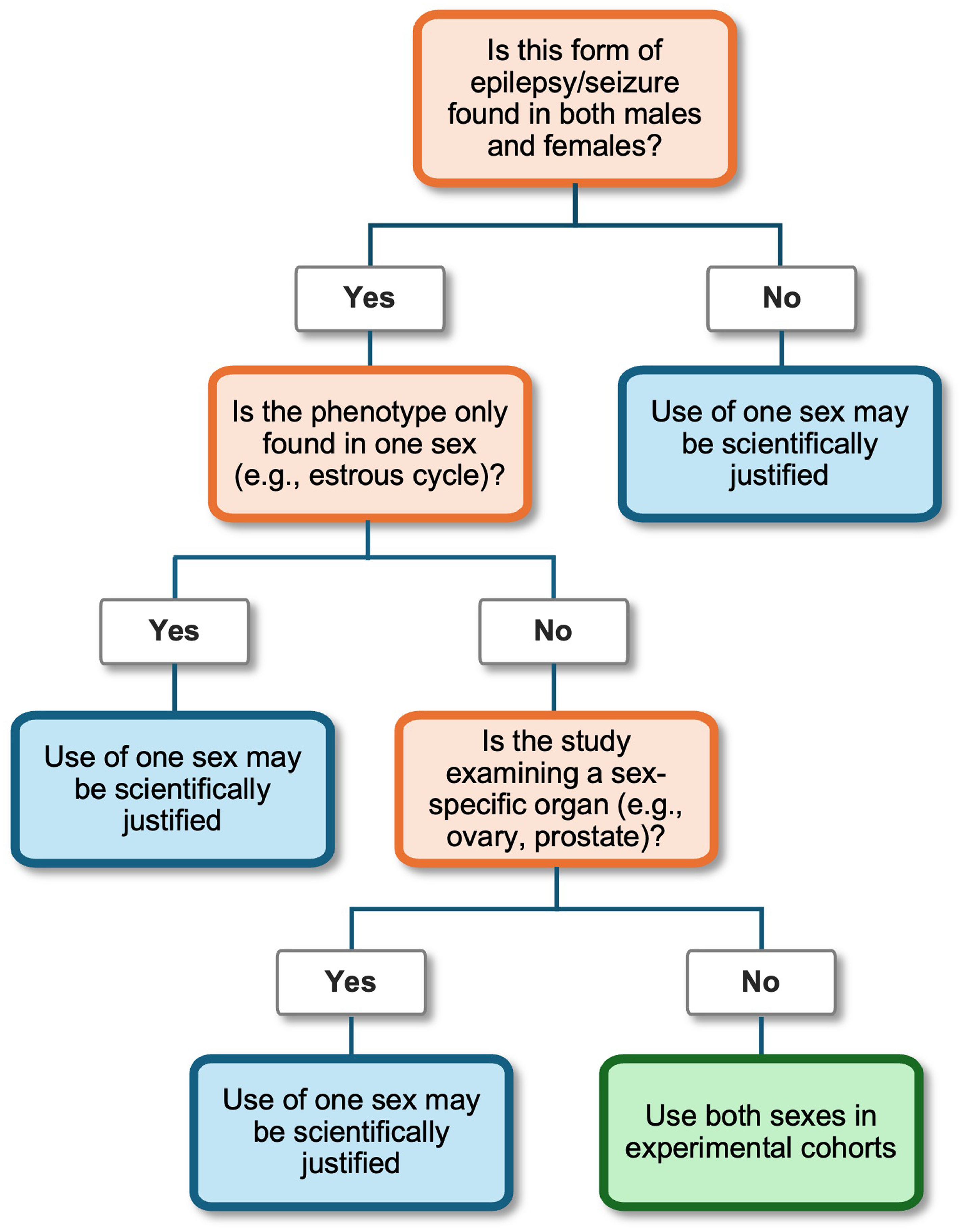
Decision tree for determination of scientific justification for use of only one sex in a preclinical epilepsy study.
If SABV and the estrous cycle are viewed by preclinical epilepsy and neuroscience community as aspects of physiology to be embraced rather than feared, the prospects for more rigorous, sex-inclusive research promise to yield improved care for all people with epilepsy and other neurological disorders.
Footnotes
Declaration of Conflicting Interests
The author declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Institute on Aging and the National Institute of Neurological Disorders and Stroke (Grant Nos. R21 AG077694, R01 NS105825, R03 NS103029, and R21 NS124980).
