Abstract
Inflammatory pain is a key component of acute traumatic pain and chronic rheumatic disease, which significantly reduces the quality of life of those who suffer from it and is often refractory to treatment. One contributor to the failure of current treatments is that the majority of pain testing has historically been performed in male subjects while the majority of pain patients are women. To better manage inflammatory pain, first the baseline sex differences in its experience must be assessed. Therefore, we evaluated C57BL/6J male and female mice for baseline sex differences in the formalin model of inflammatory pain, further investigating the observed significant sex differences through both assessing female mice at each phase of the estrous cycle and through examining the effects of gonadectomy (ovariectomy or castration) within the formalin model of inflammatory pain. Female mice in the metestrus or diestrus phase had decreased inflammatory pain relative to both male mice and female mice in the proestrus or estrus phase. Ovariectomy resulted in decreased pain, which was restored through treatment with estradiol (E2). Castration similarly reduced pain in male mice. Injection of the G-protein coupled estrogen receptor (GPER) agonist G1 resulted in significant antinociception in both female and male mice, in both mice that had received sham surgery or gonadectomy. These results establish baseline sex differences in the formalin model of inflammatory pain and support the need for further investigation into the interaction between estrogen, its receptors, and testosterone in the regulation of nociception.
Introduction
Inflammatory pain is caused by the combination of peripheral and central tissue hypersensitization and is associated with both acute traumatic pain and chronic rheumatic disease.1–3 Chronic rheumatic disease impacts approximately 20% of US adults and causes a significant reduction in patient quality of life.4,5 Despite its prevalence and severity, there is currently a paucity of adequate treatment options and as many as 21% of patients have refractory pain while others cease treatment due to severe side effects.6,7 Due to treatment inefficacy and adverse effects, approximately 52% of all diagnosed adult rheumatic disease patients do not have a current treatment plan. 8 Female sex has repeatedly been found as a risk factor for chronic pain, with this effect frequently attributed to differences in sex hormone functioning and estrogen having been identified as playing a critical role.9–12 Despite this known effect, most pain research has historically been predominantly male focused. 13
Murine models have similarly revealed significant sex differences in both acute and chronic models of pain. 14 Mice have a four-stage estrous cycle, during which estrogen levels fluctuate significantly and predictably, and these estrogen fluctuations are believed to contribute to the observed sex and estrous cycle phase differences in pain sensitivity.15–18 There are three natural estrogens, with 17β-estradiol (E2) being most common in pre-menopausal women and primarily produced in ovarian tissue, estrone (E1) being most common in post-menopausal women and produced by ovarian, adrenal, and adipose tissue, and estriol (E3) being most common in pregnant women and largely produced by placental tissue.19,20 In women, menopause results in an approximately 67% decline in circulating E2 within 2 years of the final menstrual period. 21 This decline in E2 is associated with the primary symptoms of menopause, for example, hot flashes and mood swings, and with the onset of menopause-associated musculoskeletal, migraine, and vulvovaginal pain conditions. 22 In mice, bilateral ovariectomies are commonly performed to produce a murine model of menopause and result in a similar decrease in E2 within 4 weeks post-ovariectomy.23,24
The most potent form of endogenous estrogen is E2, which acts at both traditional nuclear estrogen receptors to induce transcriptional changes and at the G-protein coupled estrogen receptor (GPER; formerly known as GPR30) to produce comparatively rapid non-genomic effects. 25 Estrogen has been found to play a complex role in the regulation of pain. In murine models of inflammatory pain, estrogen has previously been shown to be either pro-nociceptive26,27 or anti-nociceptive,28–30 or to produce no significant effect. 17 GPER has previously been shown to couple in neurons to either inhibitory Gi/o or excitatory Gs G-proteins, depending on the neuron population, while in astrocytes and glia it has been linked to modulatory Gq G-proteins.31–33 GPER has been found to be widely distributed throughout the central nervous system, peripheral nervous system, various organs, musculature, bones, and reproductive systems.34,35 Although GPER is widely distributed in both sexes, there are notable sex differences in its expression, and it has been shown to fluctuate significantly throughout the estrous cycle.36,37 GPER has previously been shown to play a role in nociception, with GPER activation in the dorsal horn of the spinal cord producing spontaneous pain responses and systemic activation of GPER, through selective agonist G1, serving to sensitize ongoing pain.38,39 Activation of GPER in the rostral ventromedial medulla has been shown to enhance in the severity of chronic pain and to contribute to its persistence, with both of these effects attributed in part to downstream μ-opioid receptor phosphorylation.40,41 Conversely, activation of GPER has been shown to be anti-inflammatory and anti-nociceptive in murine models of chronic inflammatory and chronic visceral pain.42,43 From the diversity of the pain findings and associated G-proteins, it is apparent that GPER plays a complex role in the regulation of pain and may contribute to the often-conflicting results observed when assessing the effects of estrogen on pain in varying models.
Testosterone is primarily produced in the testes and has been found to be generally anti-inflammatory and anti-nociceptive in animal models, though these beneficial effects are inconsistently seen in humans.44,45 Testosterone may protect against the development of chronic pain46,47 though elevated testosterone has also been shown to increase the perception of pain in men. 48 While there is not a male equivalent of menopause, testosterone levels do decrease in men between 1% and 2% per year each year after 30 years old.49,50 In contrast to murine ovariectomies, which broadly mimic age-related loss of E2 due to menopause, bilateral castration reduces circulating testosterone to below 1% of its initial levels, a far greater decline than that observed with natural aging.51,52 Despite this, castration remains the accepted means of assessing the effect of testosterone depletion on murine behavior.
In this study, we investigated male and female C57BL/6J mice for baseline sex differences in the formalin murine model of acute and inflammatory pain. Following our initial significant findings, that female mice had decreased inflammatory pain, we further investigated this finding by comparing male mice to female mice currently in each of the four estrus phases (proestrus, estrus, metestrus, and diestrus), revealing that the metestrus and diestrus phase female mice had significantly reduced pain relative to both male and proestrus or estrus phase female mice. We then assessed the effects of gonadectomy (ovariectomy or castration) in our C57BL/6J mice, observing that gonadectomies decreased pain in the formalin test. A separate cohort of mice was used to assess the efficacy of ovariectomy through its effects on plasma 17β-estradiol levels. Assessment of E2 in female mice revealed a pro-nociceptive effect while investigation of G1, a selective GPER agonist, in male and female mice revealed an anti-nociceptive effect. These findings emphasize the importance of sex differences in pain research and the need for further investigation into the interplay between estrogen, its receptors, and testosterone in establishing these well-observed sex differences.
Materials and methods
Animals
Experiments were conducted using 10- to 12-week-old female and male C57BL/6J mice obtained from Jackson Laboratories (Bar Harbor, ME, USA). Mice were group housed (four mice per cage) with a 12:12 h light-dark schedule (lights on 07:00, lights off 19:00) and provided standard mouse chow containing 19.8% protein ad libitum (Purina Lab Diet, St. Louis, MO, USA). The Institutional Animal Care and Use Committee (IACUC) of Texas Tech University Health Sciences Center approved all animal care and experimental procedures, which conformed with all National Institute of Health guidelines found in the Guide for the Care and Use of Laboratory Animals. 53
Drugs
Formalin was obtained from Thermo Fisher Scientific (Waltham, MA, USA). G1 was obtained from Cayman Chemical (Ann Arbor, MI, USA) and administered at a dose of 0.2 mg/kg i.p., with the dose of G1 being determined based on demonstrated in vivo efficacy in previous studies.54–56 E2 was obtained from Cayman Chemical (Ann Arbor, MI, USA) and was administered at a dose of 0.2 mg/kg i.p., with this dose being determined based on previously demonstrated in vivo efficacy.57,58 G1 and E2 were administered in a vehicle solution consisting of 5% dimethylsulfoxide, 5% ethanol, 5% Tween-80, and 85% physiological saline at a volume of 10 μL/gram of body weight (i.p.) 30 min prior to behavioral testing.
Gonadectomy
Mice randomly assigned to receive either ovariectomy or castration had these surgical procedures completed at 6- to 8-weeks old. Anesthesia was induced using 4% isoflurane and maintained for the duration of the procedures at 2% isoflurane. A small (15 mm) longitudinal incision of the abdominal wall, followed by an incision of the fascia, to expose the peritoneal cavity.59,60 In female mice, the uterine horn was identified and ligatured bilaterally using absorbable silk (5.0) suture inferior to each ovary and oviduct and the ovaries were then excised using forceps. 59 In male mice, the testicular fat pad was identified and pulled using forceps, with blood vessels proximal to the testis (avoiding the vas deferens) were tied bilaterally using absorbable silk (5.0) suture and the testes were then excised using scissors and forceps. 60 Incisions in the fascia were closed using absorbable silk (4.0) sutures and the skin was closed using surgical staples, at which point mice were allowed to recover in a heated recovery chamber. Mice were monitored daily until removal of the staples 7 days after surgery and allowed to recover for 30 days prior to experimental testing to allow for stabilization of hormone levels and healing of surgical wounds.
Estrous cycle tracking
The estrous cycle was tracked per the previously described protocol.61,62 Mice had their vaginal area exposed and a 1–200 μL pipette was used to repeatedly aspirate 50 μL of distilled water at the entrance of their vaginal canal. The liquid was applied to a glass slide, allowed to dry at room temperature, and stained using crystal violet prior to observation through a light microscope. The vaginal lavage occurred prior to any injections or behavioral testing on that day. Estrous cycle stage was determined by assessing the proportions of cell type present: proestrus (predominance of nucleated epithelial cells), estrus (predominance of cornified epithelial cells), metestrus (mix of cornified epithelial cells and leukocytes), and diestrus (predominance of leukocytes).
Formalin test
The formalin test was performed by injecting 10 µL of dilute 2.5% formalin solution into the right hind paw of each mouse, with pain behavior being observed and scored over the following hour.63,64 Mice are observed while in a 10 cm × 10 cm × 10 cm clear plexiglass house on a glass observation table, with a mirror situated underneath at a 45-degree angle to aid in viewing. The formalin test consists of both an acute phase (phase 1, 0–15 min) that is mediated by TRPA1 peripheral nociceptors 65 and an inflammatory phase (phase 2, 15–60 min) that is mediated by peripheral and central sensitization that is mediated through inflammatory processes.66,67 Pain behavior is scored in four categories: no pain behavior, favoring (injected paw has limited weight bearing), lifting (injected paw is not in contact with any surface), and rapid licking/shaking/biting. Pain behavior is divided into 12 5-min intervals and the composite pain score is computed using the composite pain score-weighted scores technique (CPS-WST 0,1,2). 68 Area under the curve (AUC) is calculated, via the trapezoidal rule, for both the acute and inflammatory phases of formalin pain. 69 Mice are humanely euthanized after the completion of the formalin test.
ELISA quantification of estradiol plasma levels
Competitive enzyme-linked immunosorbent assay (ELISA) techniques were used to analyze estradiol concentration in a separate cohort of estrous cycle identified or ovariectomized female mice (Abcam Estrogen Kit, product ID: ab108667, Cambridge, MA).62,70 Plasma was isolated via 15,000 RPM at 4°C blood centrifugation for 5 min and the plasma was stored at −80°C until ELISA was performed. ELISA was performed using undiluted plasma and conducted in triplicate, in accordance with all manufacturer’s instructions. In short, µL of plasma sample (or supplied standards or control samples) were added to the microplate wells with 200 µL 17-β Estradiol-HRP conjugate. The plates were covered with foil, incubated at 37°C for 2 h, and the well solutions were then aspirated and washed three times with provided washing solutions. 100 µL of TMB Substrate Solution was pipetted to each well and the plate was allowed to incubate for 30 min in the dark at room temperature. 100 µL of Stop Solution was then added to all wells and the plate was gently shaken. A microplate reader at 450 nm was then used to measure absorbance and mean background subtracted absorbance was calculated for the standards and plasma standard, with the mean value of the absorbance plotted against this value. A four-parametric logistic curve was calculated, and absorbance values were interpolated using this curve to determine estradiol concentrations in pg/mL (GraphPad Prism 10.6.0).
Data analysis and statistics
Mice were randomly assigned to experimental conditions and conducted in a blinded manner. Data is presented as mean ± SEM. Data analysis was performed via analysis of variance (ANOVA) for repeated measures with Greenhouse-Geisser correction (for composite pain score data), one-way ANOVA (for AUC data) with Bonferroni post-hoc multiple comparisons, independent samples T-test (for AUC data if only two groups), or two-way ANOVA (for comparison of sex differences in drug response) with Bonferroni post-hoc multiple comparisons. Degrees of freedom for significant interactions are reported as the uncorrected values. Components of total variation were settled via a priori use of multiple regression analysis. 71 Analyses were performed using GraphPad Prism (version 10.6.0, GraphPad Software, San Diego, CA, USA). p < 0.05 was considered significant.
Results
Baseline sex differences in the formalin test
In the formalin test, significant differences were noted between male and female mice (F1,22 = 8.473, p = 0.009) but these differences were found to be non-significant when evaluated in a time-group (F11, 242 = 1.605, p = 0.19) dependent manner (Figure 1(a)). Assessment of potential differences in the area under the curve (AUC) revealed no significant difference during the acute phase (t22 = 0.8627, p = 0.40; Figure 1(b)). Assessment of the AUC during the inflammatory phase revealed significant differences (t22 = 3.248, p = 0.004), with female mice showing a decreased pain response during this phase (Figure 1(c)).

C57BL/6J mice show significant sex differences in the formalin test of inflammatory pain. Female mice had significantly decreased Composite Pain Scores (a). Assessment of area under the curve (AUC) revealed no significant differences in the Acute Phase (b) while female mice had significantly reduced pain behavior in the Inflammatory Phase (c). Data is expressed as mean ± SEM, with individual data points (male mice represented by triangles; female mice represented by circles) included on all bar graphs to better represent variability (male: n = 12; female: n = 12, with female mice evenly distributed between proestrus, estrus, metestrus, and diestrus phases). Repeated measures one-way ANOVA with Greenhouse-Geisser Correction used to calculate significance for (a). Independent samples T-test used to calculate significance for (b) and (c). Relative to male mice: *p < 0.05, **p < 0.01, ***p < 0.001.
Estrous cycle phase differences in the formalin test
When comparing male mice versus female mice in known estrous cycle phases in the formalin test, significant differences were noted between the groups (F4,35 = 4.636, p = 0.004) in a time-group (F44,385 = 1.736, p = 0.03) dependent manner (Figure 2(a)). Multiple comparison analysis revealed that female mice in the diestrus phase (female – diestrus) had significantly reduced CPSs in the inflammatory phase relative to female mice in the proestrus phase (female – proestrus; at 35 min, p = 0.01), relative to female mice in the estrus phase (female – estrus; at 35 min, p = 0.002) and relative to male mice (at 35 min, p = 0.02; at 40 min, p = 0.01; Figure 2(a)). Further analysis revealed that female mice in the metestrus phase (female – metestrus) had significantly reduced CPSs in the inflammatory phase relative to female – proestrus (at 35 min, p = 0.04) and relative to female – estrus (at 35 min, p = 0.006). Assessment of potential AUC differences revealed no significant differences during the acute phase (F4,35 = 0.5100, p = 0.73; Figure 2(b)). Assessment of AUC during the inflammatory phase revealed significant differences (F4,35 = 6.348, p < 0.001; Figure 2(c)). Multiple comparison analysis revealed that female – diestrus had significantly decreased pain relative to male mice (p = 0.01), female – proestrus (p = 0.02), and relative to female – estrus (p = 0.01; Figure 2(c)). Female – metestrus had significantly decreased pain relative to male mice (p = 0.03) and relative to female – estrus (p = 0.03; Figure 2(c)).

C57BL/6J female mice in the metestrus and diestrus phases showed significantly decreased pain in the formalin test of inflammatory pain. Composite Pain Scores (a) were significantly decreased in female mice in the diestrus phase (relative to male, female – proestrus, and female – estrus) and in female mice in the metestrus phase (relative to female – proestrus and female – estrus). Area under the curve (AUC) analysis revealed no significant differences in the Acute Phase (b) while female mice in the metestrus phase and female mice in the diestrus phase had significantly reduced pain behavior in the Inflammatory Phase (c). Data is expressed as mean ± SEM, with individual data points (male mice represented by triangles; female mice represented by circles) included on all bar graphs to better represent variability (male: n = 12; female – proestrus: n = 7; female – estrus: n = 8; female – metestrus: n = 7; female – diestrus: n = 6). Repeated measures one-way ANOVA with Greenhouse-Geisser Correction used to calculate significance for (a). One-way ANOVA with Bonferroni post-hoc used to calculate significance for (b) and (c). Relative to male mice: *p < 0.05, **p < 0.01, ***p < 0.001; relative to female – proestrus mice: #p < 0.05, ##p < 0.01, ###p < 0.001; relative to female – estrus mice: @p < 0.05, @@p < 0.01, @@@p < 0.001.
Ovariectomy effects and functional reversal via E2 in the formalin test
When comparing sham surgery male mice (male – sham), sham surgery female mice (female – sham), ovariectomized female mice (female – OVX), and ovariectomized female mice injected with E2 (female – OVX – E2; 0.2 mg/kg), significant differences were noted between the groups (F3,22 = 11.35, p < 0.001) in a time-group (F33,242 = 3.099, p < 0.001) dependent manner (Figure 3(a)). Multiple comparison analysis revealed that female – OVX had significantly reduced CPSs in the inflammatory phase relative to male – sham (at 25 min, p = 0.01; at 30 min, p = 0.03; at 35 min, p = 0.04) and relative to female – sham (at 25 min, p = 0.02; Figure 3(a)). Treatment with E2 reversed this reduction, resulting in female – OVX – E2 having significantly increased inflammatory phase nociception relative to female – OVX (at 25 min, p = 0.002; at 30 min, p = 0.002; at 35 min, p = 0.02; Figure 3(a)). Assessment of potential AUC differences revealed no significant differences during the acute phase (F3,22 = 0.9728; Figure 3(b)). In the inflammatory phase, AUC assessment revealed significant differences (F3,22 = 11.33, p < 0.001; Figure 3(c)). Multiple comparison analysis revealed that female – sham had significantly decreased pain relative to male – sham (p = 0.02), that female – OVX had significantly decreased pain relative to male – sham (p < 0.001) and female – sham (p = 0.050), and that female – OVX – E2 had significantly increased pain relative to female – OVX (p = 0.010; Figure 3(c)).
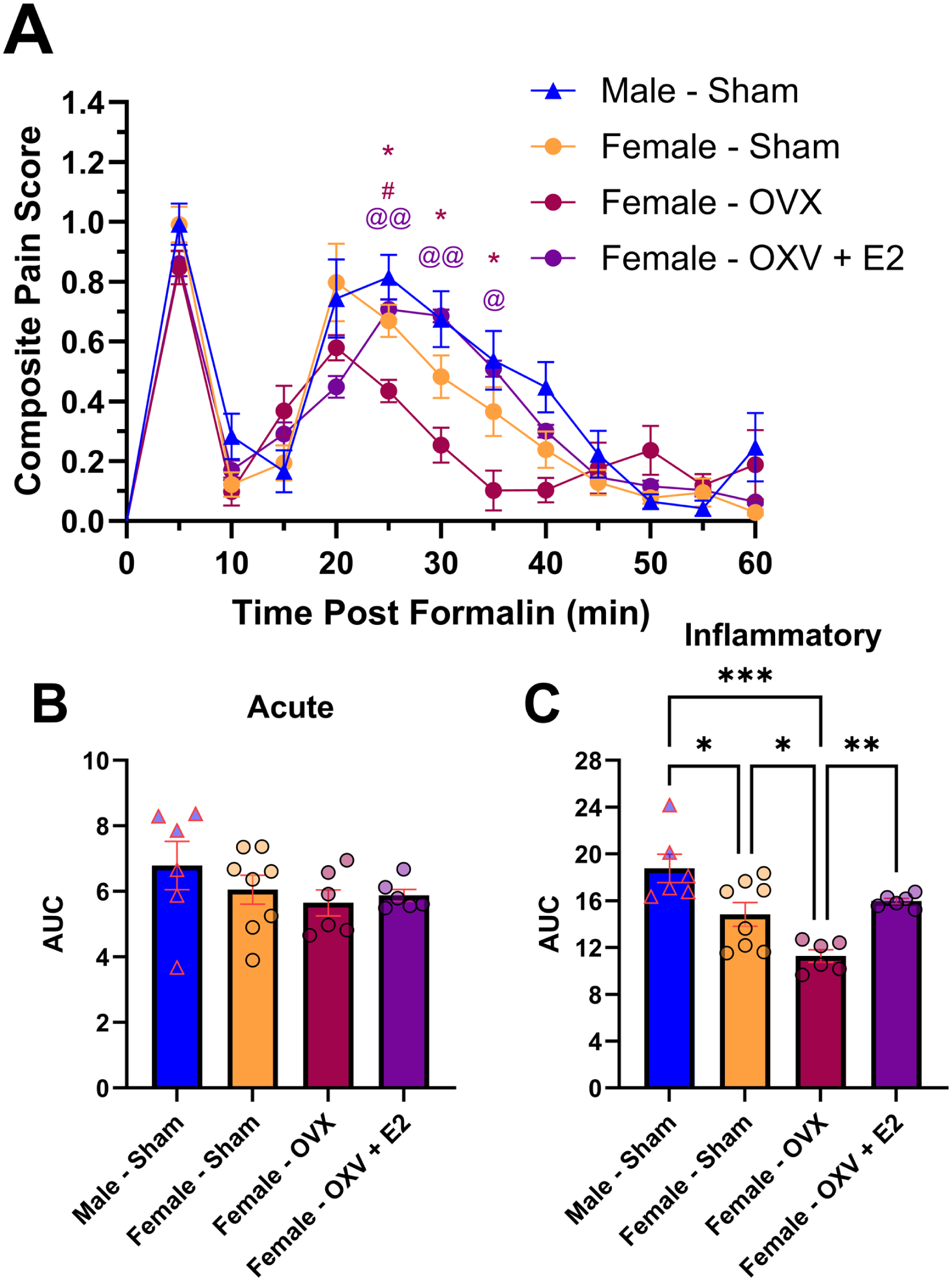
Ovariectomy of C57BL/6J female mice had significantly decreased pain that was restored by administration of E2 (0.2 mg/kg, i.p.) in the formalin test of inflammatory pain. Composite Pain Scores (a) were significantly decreased in ovariectomized female mice (relative to male and female mice that had undergone sham surgeries) and were significantly increased in ovariectomized female mice that received E2 (relative to ovariectomized female mice). Area under the curve (AUC) analysis revealed no significant differences in the Acute Phase (b). AUC analysis in the Inflammatory Phase (c) revealed significantly reduced pain in female mice that underwent sham surgery (relative to male mice with sham surgeries) and in ovariectomized female mice (relative to male and female mice that underwent sham surgeries) while injection with E2 in ovariectomized mice significantly increased their pain (relative to ovariectomized female mice). Data is expressed as mean ± SEM, with individual data points (male mice represented by triangles; female mice represented by circles) included on all bar graphs to better represent variability (male – sham: n = 6; female – sham: n = 8; female – OVX: n = 6; female – OVX – E2: n = 6). Repeated measures one-way ANOVA with Greenhouse-Geisser Correction used to calculate significance for (a). One-way ANOVA with Bonferroni post-hoc used to calculate significance for (b) and (c). Relative to male – sham: *p < 0.05, **p < 0.01, ***p < 0.001; relative to female – sham: #p < 0.05, ##p < 0.01, ###p < 0.001; relative to female – OVX: @p < 0.05, @@p < 0.01, @@@p < 0.001.
Antinociceptive effects of GPER agonism (G1) in female mice in the formalin test
When comparing female mice that received either sham surgery or OVX and either vehicle or G1 (0.2 mg/kg i.p.), significant differences were noted between the groups (F3,26 = 22.50, p < 0.001) in a time-group (F33,286 = 5.099, p < 0.001) dependent manner (Figure 4(a)). Multiple comparison analysis revealed that G1 significantly reduced CPSs in both sham (female – sham – vehicle vs female – sham – G1; at 5 min, p = 0.02; at 30 min, p = 0.02; at 35 min, p = 0.009) and OVX mice (female – OVX – vehicle vs female – OVX – G1; at 25 min, p = 0.02; Figure 4(a)). Further, the mice that received both OVX and G1 had significantly reduced CPSs relative to both sham – G1 mice (at 5 min, p = 0.008; at 25 min, p = 0.009) and relative to sham – vehicle mice (at 5 min, p < 0.001; at 25 min, p < 0.001; at 30 min, p = 0.02; at 35 min, p = 0.007; Figure 4(a)). Assessment of potential AUC differences revealed significant differences during the acute phase (F3,26 = 16.04, p < 0.001; Figure 4(b)). Multiple comparison analysis revealed that OVX – G1 mice had significantly reduced pain relative to sham – vehicle (p < 0.001) and OVX – vehicle (p = 0.003) mice while sham – G1 mice had significantly reduced pain relative to sham – vehicle (p < 0.001) mice (Figure 4(b)). In the inflammatory phase, AUC assessment revealed significant differences (F3,26 = 19.61, p < 0.001; Figure 4(c)). Multiple comparison analysis revealed that OVX – vehicle mice had decreased pain relative to sham – vehicle mice (p = 0.02), that OVX – G1 mice had decreased pain relative to sham – vehicle (p < 0.001) and OVX – vehicle (p = 0.006) mice, and that sham – G1 mice had decreased pain relative to sham – vehicle mice (p < 0.001; Figure 4(c)).
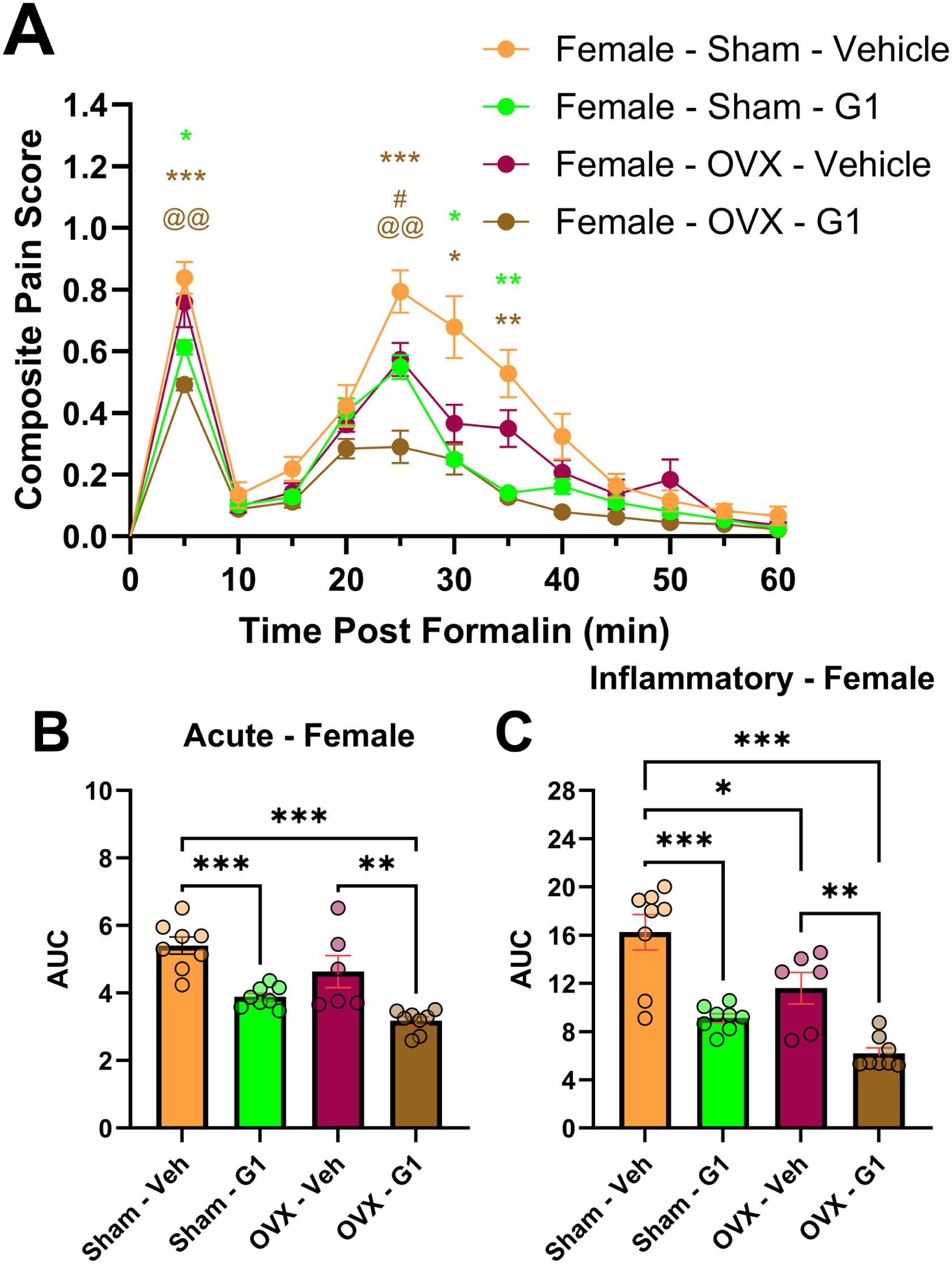
The selective GPER agonist G1 (0.2 mg/kg, i.p.) significantly reduced pain in both sham surgery and ovariectomized (OVX) female mice. Composite Pain Scores (a) were significantly decreased in both sham surgery and OVX female mice that received injections of G1 (relative to those receiving vehicle injections). Area under the curve (AUC) analysis revealed significant differences in the Acute Phase (b), with G1 reducing pain in both sham surgery and OVX female mice. AUC analysis in the Inflammatory Phase (c) revealed that OVX mice had decreased pain relative to sham surgery female mice and that injections of G1 significantly reduced inflammatory pain in both sham surgery and OVX female mice. Data is expressed as mean ± SEM, with individual data points (male mice represented by triangles; female mice represented by circles) included on all bar graphs to better represent variability (sham – vehicle, n = 8; sham – G1, n = 8; OVX – vehicle, n = 6; OVX – G1, n = 8). Repeated measures one-way ANOVA with Greenhouse-Geisser Correction used to calculate significance for (a). One-way ANOVA with Bonferroni post-hoc used to calculate significance for (b) and (c). Relative to sham – vehicle: *p < 0.05, **p < 0.01, ***p < 0.001; relative to OVX – vehicle: #p < 0.05, ##p < 0.01, ###p < 0.001; relative to sham – G1: @p < 0.05, @@p < 0.01, @@@p < 0.001.
Antinociceptive effects of GPER agonism (G1) in male mice in the formalin test
When comparing male mice that received either sham surgery or castration (CAS) and either vehicle or G1 (0.2 mg/kg i.p.), significant differences were noted between the groups (F3,28 = 57.48, p < 0.001) in a time-group (F33,308 = 6.783, p < 0.001) dependent manner (Figure 5(a)). Multiple comparison analysis revealed that CAS significantly reduced CPSs relative to sham (at 5 min, p = 0.02; at 25 min, p = 0.004; at 30 min, p = 0.01; at 35 min, p = 0.01; Figure 5(a)). Both sham and CAS mice receiving G1 had significantly reduced CPSs relative to sham – vehicle mice (sham – G1: at 5 min, p = 0.01; at 25 min, p = 0.004; at 30 min, p = 0.005; at 35 min, p = 0.001; CAS – G1: at 5 min, p = 0.003; at 25 min, p = 0.001; at 30 min, p = 0.002; at 35 min, p < 0.001; at 40 min, p = 0.02) and CAS – G1 had a reduced CPS relative to CAS – vehicle mice (at 40 min, p = 0.050; Figure 5(a)). Assessment of potential AUC differences revealed significant differences during the acute phase (F3,28 = 20.87, p < 0.001; Figure 5(b)). Multiple comparison analysis revealed significantly recued pain relative to sham – vehicle for sham – G1 (p < 0.001), CAS – vehicle (p < 0.001), and CAS – G1 (p < 0.001) conditions (Figure 5(b)). In the inflammatory phase, AUC assessment revealed significant differences (F3,28 = 53.83, p < 0.001; Figure 5(c)). Multiple comparison analysis revealed that CAS reduced pain (sham – vehicle vs CAS – vehicle, p < 0.001) and that G1 reduced pain relative to vehicle in all relevant comparisons (sham – vehicle vs sham – G1: p < 0.001; sham – vehicle vs CAS – G1: p < 0.001; CAS – vehicle vs CAS – G1: p = 0.009; Figure 5(c)).

The selective GPER agonist G1 (0.2 mg/kg, i.p.) significantly reduced pain in both sham surgery and castration (CAS) male mice. Composite Pain Scores (a) were significantly decreased in CAS versus sham surgery male mice and in mice that received injections of G1, relative to sham male mice receiving vehicle injections. Area under the curve (AUC) analysis revealed significant differences in the Acute Phase (b), with CAS reducing pain relative to sham surgery conditions and G1 reducing pain relative to sham vehicle male mice. AUC analysis in the Inflammatory Phase (c) revealed that CAS mice had decreased pain relative to sham surgery male mice and that injections of G1 significantly reduced inflammatory pain in both sham surgery and CAS male mice, relative to sham – vehicle and CAS – vehicle conditions, respectively. Data is expressed as mean ± SEM, with individual data points (male mice represented by triangles; female mice represented by circles) included on all bar graphs to better represent variability (n = 8 per group). Repeated measures one-way ANOVA with Greenhouse-Geisser Correction used to calculate significance for (a). One-way ANOVA with Bonferroni post-hoc used to calculate significance for (b) and (c). Relative to sham – vehicle: *p < 0.05, **p < 0.01, ***p < 0.001; relative to OVX – vehicle: #p < 0.05, ##p < 0.01, ###p < 0.001; relative to sham – G1: @p < 0.05, @@p < 0.01, @@@p < 0.001; relative to CAS – vehicle: #p < 0.05, ##p < 0.01, ###p < 0.001.
Assessment of 17β estradiol (E2) throughout the estrous cycle and after ovariectomy
When comparing the concentration of E2 in female during different phases of the estrous cycle or after ovariectomy, significant differences were noted between the groups (F4,35 = 19.91, p < 0.001; Figure 6). Multiple comparison analysis revealed that the metestrus and diestrus phases had significantly decreased estradiol relative to the proestrus and estrus phases (metestrus vs proestrus, p < 0.001; metestrus vs estrus, p = 0.002; diestrus vs proestrus, p < 0.001; diestrus vs estrus, p = 0.01) and that ovariectomy resulted in circulating E2 that was significantly lower than proestrus (p < 0.001) and estrus (p = 0.001) phase circulations (Figure 6). Injection of E2 at a concentration of 0.2 mg/kg i.p. in ovariectomized mice resulted in an approximately 60-fold increase in circulating E2 (Figure 6).

Assessment of circulating 17β-estradiol (E2) concentration at different estrous stage cycles and following ovariectomy, with or without subsequent E2 (0.2 mg/kg i.p.) administration. E2 was significantly elevated in the proestrus and estrus phases, significantly decreased following ovariectomy, and experienced an approximately 60-fold increase following E2 administration. Data is expressed as mean ± SEM, with individual data points (male mice represented by triangles; female mice represented by circles) included on all bar graphs to better represent variability (n = 8 per group). One-way ANOVA with Bonferroni post-hoc used to calculate significance. *p < 0.05, **p < 0.01, ***p < 0.001.
Discussion
Despite the severity and prevalence of diseases associated with inflammatory pain, there remains a lack of adequate treatment options and a significant number of patients suffer from a reduced quality of life due to their refractory pain.4–7 One possible explanation for the refractory nature of this type of pain due to the maturity of drug development and the fact that pain research has been male focused 13 while women are more likely to suffer from chronic pain. 12 These sex differences are even observed in preclinical models of acute and chronic pain, 14 with fluctuations of estradiol during the estrous cycle being suggested as a potential cause.15–18 Our results demonstrated that there are significant sex differences in the formalin murine model of inflammatory pain, with female mice having lower inflammatory pain than male mice. Further investigation revealed that these significant sex differences were driven by female mice having lower inflammatory pain in the metestrus and diestrus phases, while their proestrus and estrus phase results were comparable to male mice. In comparison to humans, the proestrus phase roughly corresponds with the follicular phase, the estrus phase with ovulation, and the metestrus and diestrus phases with the early and late luteal phase. 72 Both women and female mice have variations in circulating estradiol that occur and contribute to the aforementioned phases, with mice having relatively low circulating estradiol during the metestrus and diestrus phases and relatively high circulating estradiol during the proestrus and estrus phases. 73 These previous findings were mirrored in our study, as E2 was significantly lower in the metestrus and diestrus phases than in the proestrus and estrus phases. While estrogen has previously been shown to be either pro-nociceptive26,27 or anti-nociceptive,28–30 it is possible that other circulating sex hormones are contributing to the observed effect. Future studies should investigate progesterone, which is elevated in the late luteal/diestrus phase and has been shown previously to be anti-inflammatory 74 and to decrease the experience of pain, possible through its influence on emotional regulatory networks.75,76 While this study demonstrates that both circulating E2 and inflammatory pain are decreased in the metestrus and diestrus phases, suggesting a pro-nociceptive role of E2, it is also possible that the relative decline in progesterone during the diestrus phase is responsible for the change in pain sensitivity. Further studies investigating the relative pro- or anti-nociceptive contributions of estradiol, progesterone, and other circulating sex hormones would allow for clarification in this matter.
The decline of estrogen in menopause has previously been associated with the onset of a number of inflammatory pain-associated rheumatic diseases, 77 though the exact mechanism is as of yet unknown and attempts to prevent or treat these conditions using hormone replacement therapy have provided inconsistent results.78–80 In our study, we found that ovariectomy, which mimics menopause in its decrease of estradiol, resulted in the reduction of pain and that this pain was restored through estradiol injection.23,24 Assessment of estradiol levels revealed a significant decrease in circulating estradiol after ovariectomy and injection of E2 at a dose of 0.2 mg/kg resulted in significantly elevated levels of circulating estradiol. While the dose of E2 was chosen on the basis of previous in vivo research demonstrating its efficacy, it resulted in a supraphysiologic level of circulating estradiol, which has previously been shown to produce a pro-neuroinflammatory phenotype, downregulation of cannabinoid receptor 1, and depressive-like symptoms. 81 Further research should be conducted using reduced doses of E2 to ensure replication of the observed pro-nociceptive effects at physiological levels. Estrogen primarily exerts its effects through its actions as its two traditional nuclear estrogen receptors (ERα and ERβ) and through GPER. 25 Similar to the effects of estrogen, agonism of ERα and ERβ have been shown to produce either pro-nociceptive or anti-nociceptive effects, depending on the pain model, and may be responsible for the sex differences observed in pain.82–84 In a model of inflammatory pain, ERβ was found to fulfill a pro-nociceptive regulatory role while ERα was distinctly anti-nociceptive, with both also producing an anti-inflammatory effect. 85 When we assessed GPER agonism in both sham surgery and ovariectomized mice, we found that it produced a distinct anti-nociceptive effect, in both surgical conditions, in the acute and inflammatory phases of pain. Our results support the concept that the diversity of estrogen’s pro- and anti-nociceptive effects are due to the varying effects of its receptors and their potential differential activation in different models and types of pain.
While estrogen produces a variety of pro- or anti-nociceptive effects depending on the model used, testosterone has generally been found to be anti-inflammatory and anti-nociceptive in animal models, even if this effect is not consistently observed in humans.44,45 Testosterone has generally been found to protect against a pro-inflammatory state, with this protection providing beneficial effects for the experience of pain in animal models.44,86 In the carrageenan rat model of inflammatory pain, it was found that testosterone provided an anti-inflammatory and anti-nociceptive benefit, with neonatal castration resulting in a hyperalgesic state. 87 It has also been shown that testosterone inhibits the upregulation of transient receptor potential vanilloid 1 (TRPV1) within sensory ganglia in a rat model of chronic inflammatory pain. 88 However, testosterone injection in intact male mice produced no significant effect in the formalin model of inflammatory pain 89 and recent research has identified a pro-nociceptive role of testosterone in murine models of pain hypersensitivity. 90 In our study, we found that castration resulted in decreased pain in both the acute and inflammatory phases of the formalin pain model. One possible explanation for this finding lies in the definition of pain, which, according to the International Association for the Study of Pain, is “an unpleasant sensory and emotional experience associated with actual or potential tissue damage.” 91 In men, elevated testosterone has been shown to increase the perception of pain. 48 In male mice, it has been demonstrated that castration results in the elimination of conspecific aggression and social aggressive behaviors, while also promoting depressive-like symptoms.92–94 The emotional changes in castrated mice have further been associated with decreased pain behavior and stress been repeatedly found to similarly alter pain perception.95,96 One limitation of our castration model is that it fails to address the effects of castration on the emotional state and stress levels, along with their impact on pain perception, in our castrated male mice. Future studies need to be conducted to evaluate the effects of castration on stress, anxiety, and depression within our C57BL/6J castration murine model. In these male mice, G1 provided significant acute phase antinociception in sham male mice and significant inflammatory phase antinociception in both sham and castrated male mice, further supporting our findings that GPER activation in antinociceptive within the formalin model of inflammatory pain. Future studies should further investigate possible interactions between testosterone and GPER, as it has previously been shown that GPER activation can regulate testicular development, testosterone production, and spermatogenesis in male mice.97–99 Our findings demonstrate an anti-nociceptive role for G1 within the formalin model of acute inflammatory pain. Previous studies have demonstrated that GPER activation can promote pro-nociceptive effects in chronic models of pain, with regulation of downstream calcium signaling and the μ-opioid receptor having been suggested as possible mechanisms.38–41 Conversely, GPER activation has been shown to produce anti-nociceptive effects in models of visceral and inflammatory pain.42,43 Further research is needed into the mechanisms of GPER-dependent nociceptive regulation to reconcile these findings.
These results demonstrate the existence of baseline sex differences within the formalin murine model of inflammatory pain and their dependence on the estrous cycle. The significant differences between the different estrous phases necessitate further investigation to determine the hormonal mechanisms at play and their potential clinical significance in pain management for women. Further studies investigating the interplay of estrogen, its various receptors, and testosterone within inflammatory pain should be conducted to better understand the mechanisms behind the well demonstrated sex differences in pain.
Footnotes
Acknowledgements
The authors thank the Laboratory Animal Resources Center at Texas Tech University Health Sciences Center for their assistance in the care of the animals used in this study.
Author contributions
R. Barnes, J. Guindon: Participated in research design.
R. Barnes, J. Guindon: Conducted experiments.
R. Barnes, A. Alanis, H. Quick, J. Guindon: Performed data analysis.
R. Barnes, A. Alanis, H. Quick, J. Guindon: Wrote or contributed to the writing of the manuscript.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work has been supported by the National Institute on Drug Abuse DA044999-01A1 (JG), Texas Tech University Health Sciences Center School of Medicine 121035 (JG), and Texas Tech University Health Sciences Center Office of Research (JG), The CH Foundation grant (JG). To be noted CH needs to be underline but not able to do it as per Foundation request!!!
Ethical considerations
The Institutional Animal Care and Use Committee (IACUC) of Texas Tech University Health Sciences Center approved all animal care and experimental procedures, which conformed with all National Institute of Health guidelines found in the Guide for the Care and Use of Laboratory Animals.
