Abstract
Bjørk M-H, Zoega H, Leinonen MK, Cohen JM, Dreier JW, Furu K, Gilhus NE, Gissler M, Hálfdánarson Ó, Igland J, Sun Y, Tomson T, Alvestad S, Christensen J. JAMA Neurol. 2022;79(7):1-10. doi:10.1001/jamaneurol.2022.1269 Women with epilepsy frequently need antiseizure medication (ASM) to prevent seizures in pregnancy. Risk of neurodevelopmental disorders after prenatal exposure to AMSs is uncertain. To determine whether children exposed prenatally to ASMs in monotherapy and duotherapy have increased risk of neurodevelopmental disorders. The Nordic register-based study of antiepileptic drugs in pregnancy (SCAN-AED) is a population-based cohort study using health register and social register data from Denmark, Finland, Iceland, Norway, and Sweden (1996-2017; analysis performed February 2022). From 4 702 774 alive-born children with available mother-child identities and maternal prescription data, this study included 4 494 926 participants. Children from a multiple pregnancy or with chromosomal disorders or uncertain pregnancy length were excluded (n = 207 848). Prenatal exposure to ASM determined from maternal prescription fills between last menstrual period and birth. We estimated cumulative incidence at age 8 years in exposed and unexposed children. Cox regression adjusted for potential confounders yielded adjusted hazard ratios (aHRs) with 95% CIs for autism spectrum disorder (ASD), intellectual disability (ID), or any neurodevelopmental disorder (ASD and/or ID). A total of 4 494 926 children were included; 2 306 993 (51.3%) were male, and the median (IQR) age at end of follow-up was 8 (4.0-12.1) years. Among 21 634 unexposed children of mothers with epilepsy, 1.5% had a diagnosis of ASD and 0.8% (numerators were not available because of personal data regulations in Denmark) of ID by age 8 years. In same-aged children of mothers with epilepsy exposed to topiramate and valproate monotherapy, 4.3% and 2.7%, respectively, had ASD, and 3.1% and 2.4% had ID. The aHRs for ASD and ID after topiramate exposure were 2.8 (95% CI, 1.4-5.7) and 3.5 (95% CI, 1.4-8.6), respectively, and after valproate exposure were 2.4 (95% CI, 1.7-3.3) and 2.5 (95% CI, 1.7-3.7). The aHRs were elevated with higher ASM doses compared with children from the general population. The duotherapies levetiracetam with carbamazepine and lamotrigine with topiramate were associated with increased risks of neurodevelopmental disorders in children of women with epilepsy: levetiracetam with carbamazepine: 8-year cumulative incidence, 5.7%; aHR, 3.5; 95% CI, 1.5-8.2; lamotrigine with topiramate: 8-year cumulative incidence, 7.5%; aHR, 2.4; 95% CI, 1.1-4.9. No increased risk was associated with levetiracetam with lamotrigine (8-year cumulative incidence, 1.6%; aHR, 0.9; 95% CI, 0.3-2.5). No consistently increased risks were observed for neurodevelopmental disorders after prenatal exposure to monotherapy with lamotrigine, levetiracetam, carbamazepin, oxcarbazepine, gapapentin, pregabalin, clonazepam, or phenobarbital. Conclusions and relevance: In this cohort study, prenatal exposure to topiramate, valproate, and several duotherapies were associated with increased risks of neurodevelopmental disorders.Importance:
Objective:
Design, setting, and participants:
Exposures:
Main outcomes and measures:
Results:
Commentary
In the past 2 decades, following the increase in awareness of valproate-associated adverse fetal and neurodevelopmental outcomes, clinicians have been searching for anti-seizure medications (ASMs) that are “not valproic acid” for patients with epilepsy who may become pregnant. Topiramate is a broad-spectrum ASM that was “not valproic acid.” However, recent studies have shed light on the fact that in utero topiramate exposure is associated an increased risk of oral clefts. 1 In addition, infants exposed to topiramate have a higher risk of having low birth weight and being small for gestational age. 2 Our understanding of the effect of in utero topiramate exposure on cognitive outcomes, however, has been limited to a few studies with variable methodologies, mostly small sample sizes and contradictory findings (Table 1). 3 -7 In a recent neurodevelopmental outcomes study that is the focus of this review, Bjørk and colleagues raise concern that topiramate is, in fact, a significant behavioral and cognitive teratogen. 8
Prior Studies on Neurodevelopmental Outcomes With In Utero Topiramate Exposure.
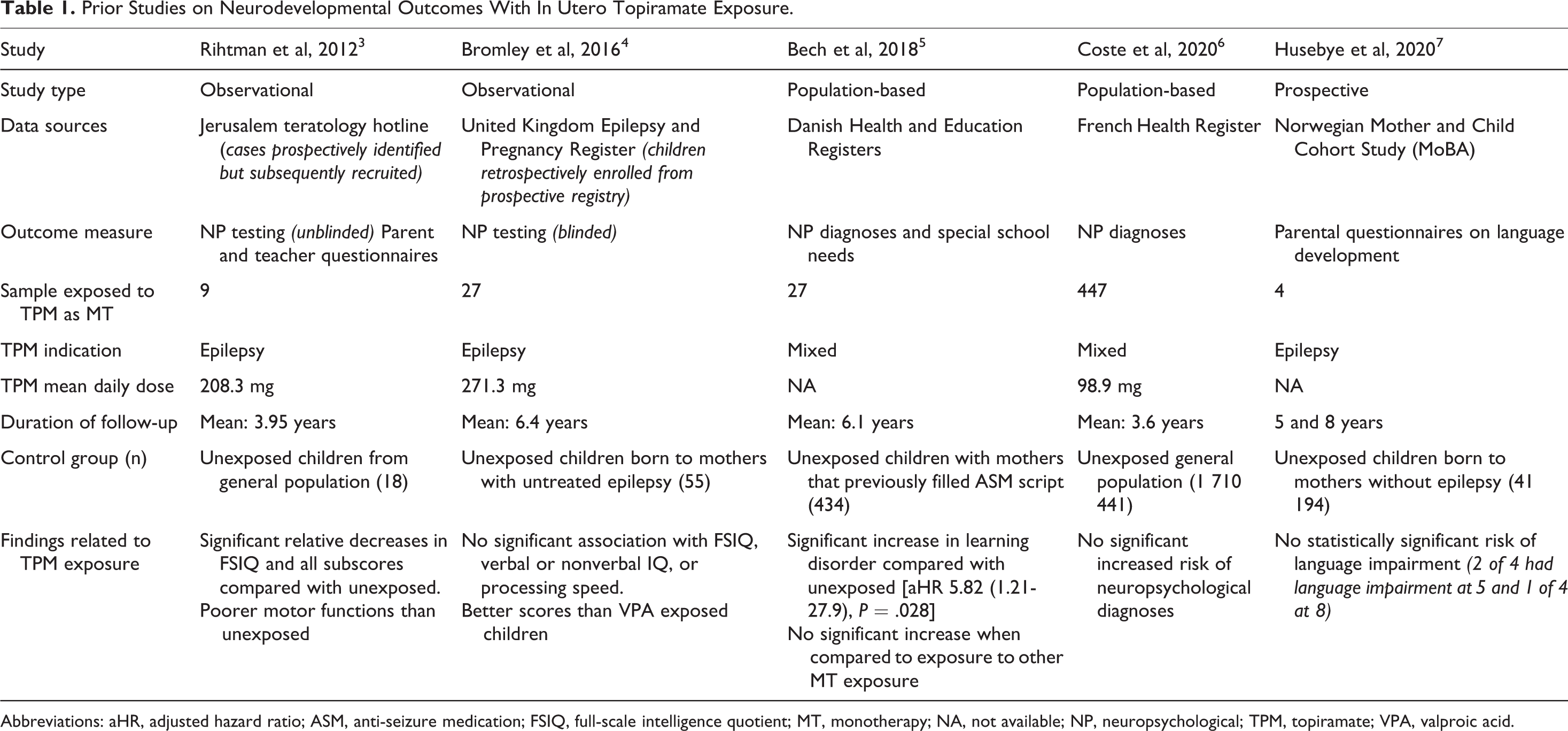
Abbreviations: aHR, adjusted hazard ratio; ASM, anti-seizure medication; FSIQ, full-scale intelligence quotient; MT, monotherapy; NA, not available; NP, neuropsychological; TPM, topiramate; VPA, valproic acid.
This is a population-based study of neurodevelopmental diagnoses among children exposed to ASMs during pregnancy. The source of the information was health registries from Denmark, Finland, Iceland, Norway, and Sweden. These Nordic countries have universal health care and each citizen is assigned an identification number which is used across registries. The authors used these identifiers to align mother-infant pairs across maternal birth registries, prescription drug registries, and the national patient register (which includes diagnoses received by mother or child). The primary outcome of the study was the risk of a formal diagnosis of autism spectrum disorder (ASD) or intellectual disability (ID) in children exposed to individual ASMs in pregnancy. Exposure during pregnancy was determined by prescription data indicating that a mother had filled a prescription for a given ASM between the time of her last menstrual period and before giving birth. There were 2 main groups studied. The first was children born to mothers with a diagnosis of epilepsy. Within this group, the children born to mothers with epilepsy taking ASMs were compared to children born to mothers with epilepsy who were not taking ASMs. It is notable that 57% of the mothers with a diagnosis of epilepsy were not taking seizure medications, suggesting that many may have had inactive or childhood epilepsy. The other group studied was the total population. The authors compared the outcomes of children born to mothers who took ASMs in the total population to those born to mothers who did not take ASMs. This total population cohort included mothers with epilepsy diagnoses. In the subgroup exposed to ASM during pregnancy, 65% of the mothers had an epilepsy diagnosis. Other common indications were anxiety (11.6%), bipolar disorder (10.4%), and depression (9.5%).
The main finding in both groups was that there was an increased risk of an ASD and ID in children born to mothers taking valproate monotherapy and in mothers taking topiramate monotherapy when compared to an unexposed control group (Table 2). The adjusted hazard ratios (aHR) were significantly elevated and similar for both the comparisons within the epilepsy group and in the total population group. Thus, although the cumulative risk and hazard ratio at first appear higher in the topiramate group compared to the valproate group, it is important to note the wide confidence intervals due to the small sample size.
Summary of Key Findings From Bjørk et al “Association of Prenatal Exposure to Antiseizure Medication With Risk of Autism and Intellectual Disability.” 8
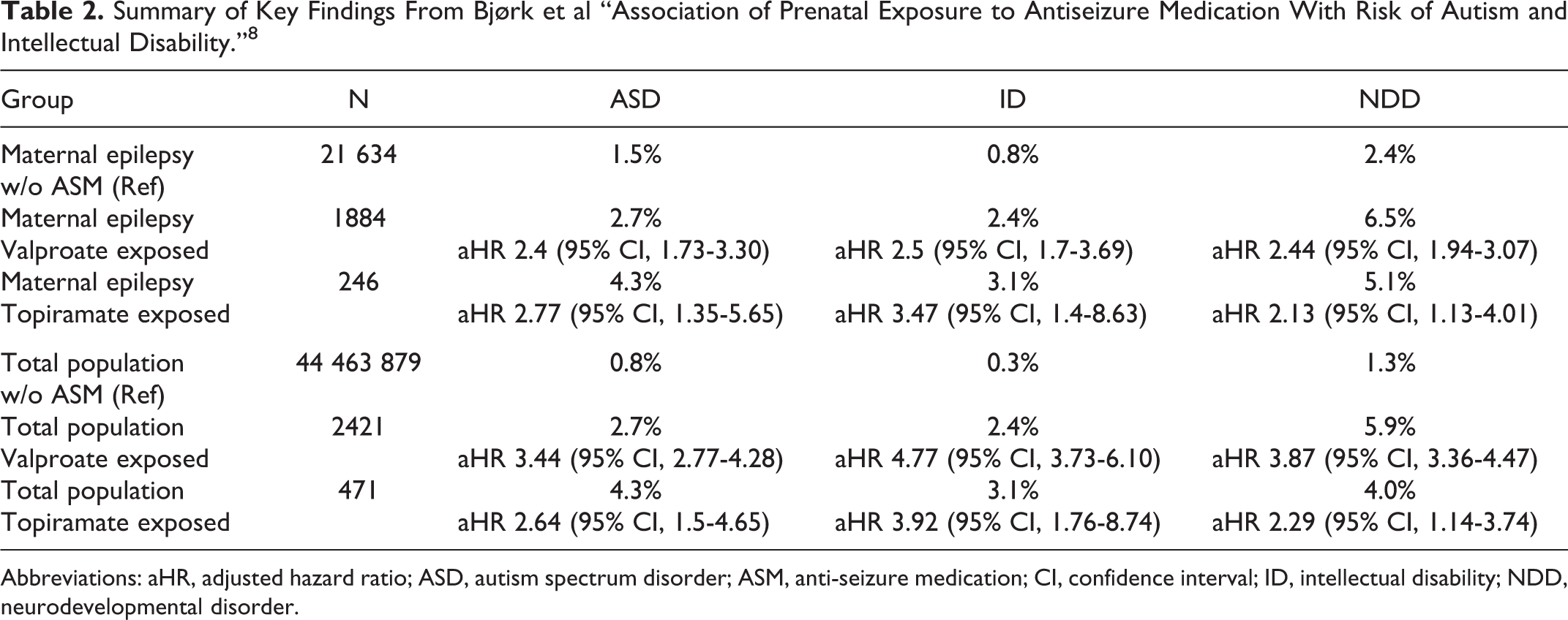
Abbreviations: aHR, adjusted hazard ratio; ASD, autism spectrum disorder; ASM, anti-seizure medication; CI, confidence interval; ID, intellectual disability; NDD, neurodevelopmental disorder.
Despite the relatively small sample size, this data suggests that topiramate exposure is associated with a significant risk of cognitive teratogenesis. The authors also looked at the risk of receiving any diagnosis fitting under the umbrella of a neurodevelopmental disorder (NDD). In both the epilepsy and total population groups, the risk of an NDD was also significantly elevated for topiramate and valproate (Table 2). Furthermore, within the total population group, the authors demonstrated a dose–response effect of valproate and topiramate. For topiramate, the risk of a NDD was greater with doses greater than or equal to 100 mg/d (aHR 2.93 [95% CI, 1.32-6.55]) than with exposure to less than 100 mg/d (aHR 1.71 [95% CI, 1.04-2.79]). There was, however, still a significantly elevated risk of NDD at the lower dose range.
This study should serve as a reminder to clinicians to be direct about the absence of data when counseling prospective mothers taking ASMs that are understudied. Currently, many ASMs that are “not valproic acid” have not been studied at all in terms of neurodevelopmental outcomes. These include, but are not limited to, cenobamate, clobazam, brivaracetam, eslicarbazepine, ethosuximide, lacosamide, perampanel, rufinamide, and zonisamide. It is common to conflate the risk of structural teratogenesis and the risk of cognitive teratogenesis but they are not the same. Structural and cognitive teratogenic risk should be discussed separately with transparency about insufficient information in one or both domains.
The present study found small but significantly elevated risks of ID among children in the total population cohort that had been exposed to carbamazepine, oxcarbazepine, or clonazepam. They found an increased risk of NDD in the same cohort for these 3 medications and for lamotrigine. This increased risk was not seen when the analysis was restricted to children born to mothers with epilepsy, which the authors concluded may imply that the risk has more to do with the indication for which the meds were prescribed. Levetiracetam was not associated with an increased risk of ASD, ID, or NDD in comparisons within the epilepsy group or the total cohort.
Bjørk et al. also report that polytherapy combinations were associated with an increased risk of NDD. In the total population cohort, exposure to the combination of lamotrigine with either valproate, oxcarbazepine, or topiramate as well as the combination of levetiracetam and carbamazepine were associated with an increased risk of NDD compared to the reference group. When analysis was restricted to the epilepsy group, only lamotrigine plus topiramate and levetiracetam plus carbamazepine carried a significantly increased risk of NDD. The combination of levetiracetam and lamotrigine was the only polytherapy studied that did not confer a significantly increased risk in either analysis. Interpretation of polytherapy data should be cautious, however, as patients taking polytherapy may have more severe disease and may be taking higher doses of each of the medications in the combination. Pregnancy registry data has shown that for structural teratogenesis related to polytherapy that includes valproic acid or topiramate, the dose of valproic acid or topiramate in the combination is still the major driver of teratogenic risk. 9,10
Despite significant progress in the past 2 decades, the pressing need for further research on ASM-related cognitive teratogenesis is far from satisfied. Settling on ASMs that are “not valproic acid” is not sufficient. We need to know more about how other ASMs and ASM combinations affect cognitive development both through further epidemiological studies such as this one, but also through prospective studies which can control for the impact of seizures during pregnancy, medication doses, and, perhaps more importantly, medication levels. Furthermore, the average age of diagnosis of NDD in this study was between 6.1 and 7.9 years which emphasizes the importance of studying these children in grade school and beyond.
